Abstract
Per- and polyfluoroalkyl substances are a growing concern in the paper and nonwoven industry, as current research provides alarming results regarding the abundance and persistence of these substances from food packaging producers. It was reported that per- and polyfluoroalkyl substances have been detected in blood serum of thousands of individuals originating from food packaging that contains per- and polyfluoroalkyl substances. In addition, 20–56% of all products tested had detectable levels that immensely contribute to dietary exposure. Paper mills are also a significant source of exposure due to environmental contamination surpassing emission regulations set by the Environmental Protection Agency by over 100,000 parts per trillion. These studies exemplify the necessity for paper-based product producers to not only abide by regulations but also demonstrate the implementation of remediation efforts. With current research on novel remediation techniques and increased restrictions surrounding per- and polyfluoroalkyl substance usage for water repellency, decreased contamination and exposure will be achieved throughout the world. This article serves as a brief review that focuses on the nonwoven manufacturing sector, which includes pulp, paper, and tissue related to per- and polyfluoroalkyl substance contamination in the environment and drinking water sources. This review addresses the statistical evaluation of per- and polyfluoroalkyl substance levels throughout the world from paper and nonwoven product manufacturing, along with remediation methods and legislation to reduce contamination and exposure. To the best of our ability, this important and sensitive subject area has been researched extensively. However, many data gaps are present within the field, and limited studies have been conducted contributing to this brief review, which highlights the importance of further studies surrounding per- and polyfluoroalkyl substances in the paper and nonwoven sectors. This article encompasses all available information in the paper and nonwoven sector regarding per- and polyfluoroalkyl substances that can be used for reference by the general public and by manufacturers in the nonwoven production industry.
Introduction
Perfluoroalkyl and polyfluoroalkyl substances (PFAS) are a growing concern around the world due to their abundance within the environment and consumer products. These compounds are known as persistent organic pollutants as the carbon–fluorine bonds require high temperatures not commonly observed in nature. 1 Due to the conditions needed for degradation, PFAS are often referred to as forever chemicals and have the potential to enter groundwater and contaminate drinking water and food sources. 2 Bioaccumulation of PFAS in wildlife and humans from oral consumption poses a major health threat, as current research provides insight into toxicity and effects from repeated exposure.
Not only is the food web a source of bioaccumulation but the consumer products provide dermal routes for PFAS to accumulate within the body. Numerous products used daily by the population are commonly treated with PFAS, including household cleaners, clothing, cookware, food packaging, furniture, personal protective equipment, bedding, toiletries, shoes, leather, and many other products that provide water resistance.3,4 Some individuals are exposed more than the others due to their industrial occupations in packaging and textile production. Water-repellent applications of PFAS in many consumables, food sources, and textiles, and persistence in high-risk areas near industrial factories, have led to the presence of these substances in 50 states, along with all areas tested throughout the world. These locations include those as remote as the Artic, where industrial production and high population are not observed.5–7 Asia has immense industrial output, and it was discovered that this can increase exposure for travelers and immigrants, as Asian Americans were found to have far greater average levels of PFAS than any other US ethnicity. 8 This finding highlights the concerns about PFAS, as degradation does not occur naturally and they are able to travel throughout the environment from emissions during factory production.
These studies serve to highlight the importance surrounding PFAS emissions and contamination from the paper and nonwoven sectors addressed throughout this mini-review. Data gaps within these industries provide insight into which topics may be of interest to researchers as current remediation methods for combating PFAS emissions and contamination still pose a challenge for producers. PFAS have been detected in rural and unpopulated areas, demonstrating the need for stricter policies in countries that do not participate in PFAS regulations according to the Stockholm Convention that contributes to worldwide emission and exposure of humans and wildlife. Although significant improvements are necessary for the paper and nonwoven sectors, current remediation efforts and legislation have the potential to greatly reduce dietary and oral exposure in the environment and through paper-based products.
PFAS in the Paper Industry
Toxicity
As bioaccumulation is prevalent from dietary and dermal exposure, health risks are of concern for consumers and populations residing near industrial sites. Studies have shown that repeated exposure is associated with increased cholesterol and blood pressure along with an increased risk for kidney disease.9–11 PFAS are also classified as endocrine-disrupting chemicals, which have been associated with reproductive and thyroid dysfunction, birth defects, and delayed development. Although PFAS have been suggested to cause such health aliments, it is difficult to pinpoint the exact causes of these effects as a result of PFAS. However, researchers have concluded that PFAS bind to albumin proteins and travel to target organs, such as the kidney, liver, and brain. Neurotoxic and carcinogenic effects have also been reported as a result of repeated exposure.11,12 Due to bioaccumulation, PFAS are also passed to offspring from pregnant and nursing mothers, which can lead to early neurotoxic effects, including delayed development as children are more susceptible to health effects than adults. 12
Paper-based Food Packaging
It has been discovered that pulp and paper production is a major source of PFAS emissions and contamination, as application of these substances is used to treat items, such as parchment paper, food packaging, paper materials, and cardboard. One study analyzed 400 paperboard substrates using particle-induced gamma ray emission and found that PFAS were present in food contact papers, including 56% of dessert wrappers, 38% of sandwich wrappers, and 20% of cardboard, while their presence was 0% in paper cups that were not in contact with food. 13 Food packaging treated with PFAS poses a detrimental health risk due to migration into food. A study on popcorn bags determined that after cooking, microwaved popcorn contained 3.2 mg/kg of fluorinated compounds, and PFAS contaminated the butter inside as well at 0.1 mg/kg. These compounds were still present after 40 days at 4°C. It was also discovered that oil has the potential to enhance PFAS migration, and this increase is also facilitated by emulsifiers. 14 Another study focused on a wide variety of food products, such as pizza, hamburgers, potatoes, bread, cheese, and desserts. It was found that dietary consumption of PFAS was present at up to 3.52 mg/kg per day in children and up to 1.76 mg/kg per day in adults. This research focused on migration of PFAS from paper-based food packaging and could play a major role in these results because PFAS were present in every item tested. 15 Individuals who consume foods that contain higher levels of oil from fast food, pizza, and popcorn often have an increased risk of dietary exposure, as noted in a previous study reporting that oil increases the mobility of PFAS from paper substrates. 14 These individuals also eat foods packaged with PFAS-treated materials more often than others eating fresh foods. The National Health and Nutrition Examination survey collected blood samples and dietary data from more than 10,000 people between 2003 and 2014. It was discovered that individuals who cooked at home averaged lower PFAS levels in blood serum. 16 These findings suggest that a healthier lifestyle can limit PFAS exposure and help prevent fluorinated compound-associated illnesses due to a decrease in paper-based packaging contact.
Paper Mill PFAS Emissions
Paper mill effluents enter waterways by release and spills, and they have been shown to be toxic to the environment. 17 It has been reported that paper mills produce 825 tons of PFAS-coated paper daily for various applications, such as food packaging, toiletries, and cardboard. It has also been reported that 26 million gallons of water are released into the environment from these facilities per day. More specifically, 269 pulp and paper mills were evaluated by the Environmental Defense Fund (EDF), and it was determined that 42 sites release waste directly into rivers, which is harmful to aquatic life and humans because PFAS travel into groundwater, drinking water, and soil. According to data analysis conducted by EDF, PFAS levels exceeded 70 parts per trillion (ppt), which is the recommended maximum PFAS level in waterways regulated by the EPA in 2018. Chemours and Daikin facilities were among the top companies causing PFAS contamination at concentrations of 83,000 and 103,000 ppt daily. 18 These levels are of great concern due to the toxicity of paper mill sludge to aquatic organisms and potential health effects observed from nearby populations that use these water sources for drinking water and agriculture. One study conducted on PFAS at Tyrifjorden lake in Norway also reported paper mill sludge as a major PFAS contamination source. This research analyzed paper mill effluent entering the lake at 189 tons and then exiting the lake at 154 tons to other environmental compartments. Sediment values averaged 40 tons of effluent, and it was predicted that PFAS concentrations would still be present at 2 × 10−3 to 8 × 10−3 ng/L. 19 At the time of measurement in 2018, PFAS levels ranged from 25 to 68 ng/L and indicated bioaccumulation in biota. This research provides significant data to compare with values reported by the EDF, as paper mills in this region resulted in PFAS emissions on a smaller scale but PFAS were still detected in aquatic organisms. It can be assumed that the 42 release sites analyzed in the United States led to far greater complications for aquatic life. Further research should be conducted to determine the health effects in fish, as PFAS have been shown to accumulate in fish livers and further negatively impact the food web.
Paper mill sludge is composed of cellulose fibers, calcium carbonate, kaolinite, and talc. 20 Trace elements of metals have been detected, such as cadmium, copper, lead, chromium, zinc, iron, manganese, nickel, cobalt, arsenic, vanadium, barium, and titanium, that have the potential to be toxic to wildlife when large amounts of sludge are repeatedly released at waste sites. 21 Synthetic compound emission from the bleaching phase of pulp and paper is of concern, as these substances are known to be toxic as well. Phenols, furans, dioxins, benzenes, and fluorinated compounds are among some of the chemicals produced during bleaching that enter waterways and leach through soil. 22 Perfluorooctanoic sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) are among the most abundant PFAS that have been detected in paper mill sludge, with lower levels of perfluoroalkyl carboxylic acid (PFCA), perfluorooctane sulfonamide (FOSA), perfluorooctanesulfonamidoacetic acid (FOSAA), 8:2 fluorotelomer sulfonic acid (FTS), 10:2 FTS, 12:2 FTS, 14:2 FTS, N-ethyl perfluorooctane sulfonamido acetic acid (N-EtFOSAA), and N-ethyl perfluorooctane sulfonamido ethanol (N-EtFOSE) as shown in Table 1. Table 1 also shows the environmental compartments in which these PFAS were detected. 20
PFAS in paper mill sludge. Sediment and water samples taken near paper mill release site show specific PFAS in each compartment. 20
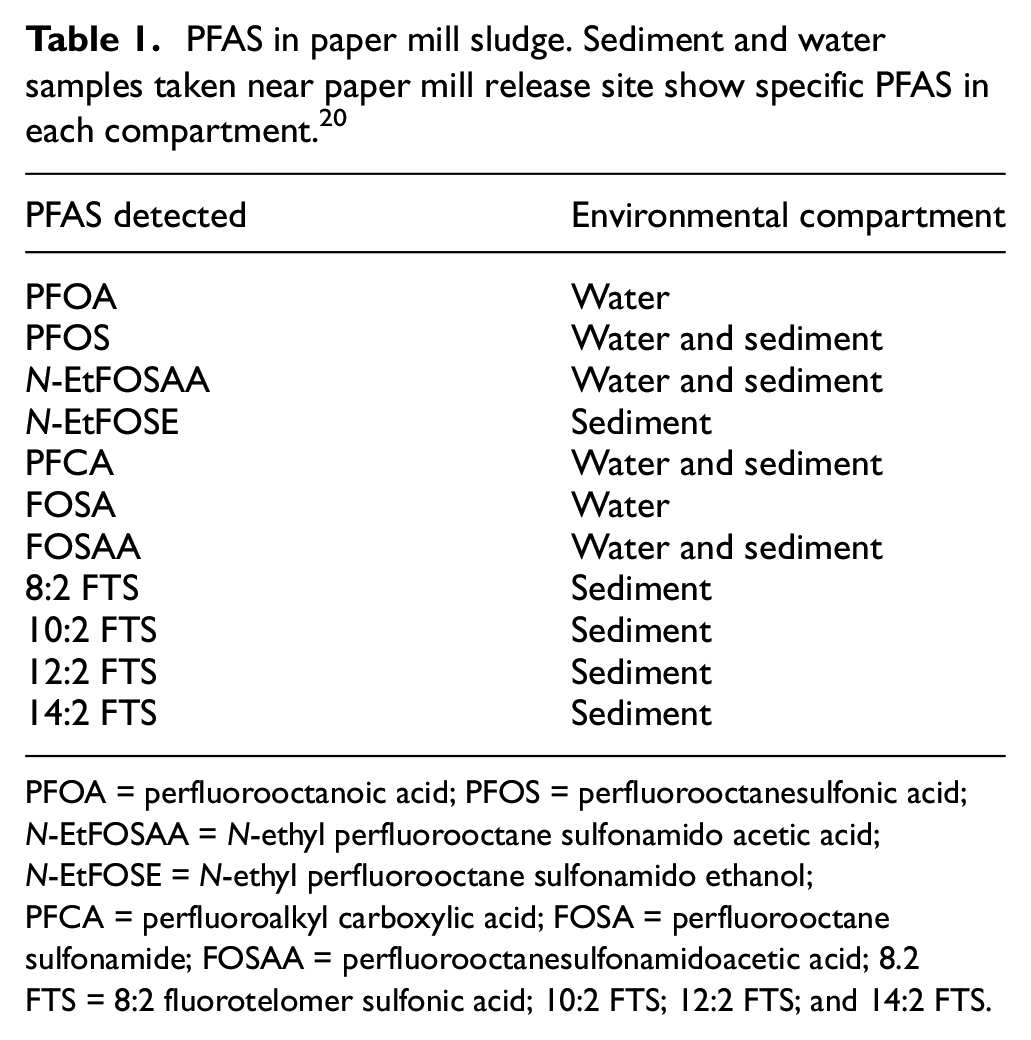
PFOA = perfluorooctanoic acid; PFOS = perfluorooctanesulfonic acid; N-EtFOSAA = N-ethyl perfluorooctane sulfonamido acetic acid; N-EtFOSE = N-ethyl perfluorooctane sulfonamido ethanol; PFCA = perfluoroalkyl carboxylic acid; FOSA = perfluorooctane sulfonamide; FOSAA = perfluorooctanesulfonamidoacetic acid; 8.2 FTS = 8:2 fluorotelomer sulfonic acid; 10:2 FTS; 12:2 FTS; and 14:2 FTS.
Although paper mills produce paper-based products containing detectable levels of PFAS and are a major contributor to PFAS emission from effluent as manufacturers have used PFAS for water repellency in the past, continuous efforts are currently being made to remediate and prevent further human and wildlife exposure. Experimentation and optimal substitution determination will greatly reduce PFAS output in corroboration of persistent remediation actions that paper mill processes currently exhibit. Microbial treatments and filtration systems extensively decrease PFAS contamination from paper manufacturers’ waste providing a positive outlook for the future of paper mill contribution to PFAS contamination.
PFAS in Toilet Tissue and Wastewater
Wastewater testing is a common method to determine chemical exposure, diseases, and drug use among populations, as these substances are recycled into water. PFAS have been detected at water treatment facilities, and scientists tested toilet tissue to determine if this was a possible contaminant. After analysis of water sludge and toilet paper brands in North America, Central America, South America, Western Europe, and Africa, it was found that toilet paper is a source of PFAS contamination. This study also determined the difference between recycled and non-recycled toilet paper brands. It was found that recycled toilet paper had less PFAS concentration than non-recycled. More specifically, 6:2 polyfluoroalkyl phosphate diester was the abundant substance detected in non-recycled toilet tissue, which could be due to higher concentrations of PFAS still present from the factory production compared to reprocessed paper. In North America, PFAS concentrations were highest across brands tested, and Africa had the lowest. 23 This study implicates that exposure from toilet tissue is a major concern, as PFAS can be absorbed dermally and enter waterways from water treatment facilities.
It has been reported that PFAS are present in nearly all wastewater treatment plants, not only from toilet paper but also from upstream sources, such as manufacturing plant emissions, household products, and human waste. In 2021, the Washington State Department of Ecology conducted a study on PFAS contamination in three wastewater treatment plants. Analysis of influent, effluent, sludge, and biosolids showed that most concentrations were below the recommended levels of 10 ng/L in drinking water after treatment. However, PFOA exceeded these levels at 11.7–13.5 ng/L, which poses a health threat to nearby individuals who consume this water. 24 The US Geological Survey published in August of 2023 reports that 45% of tap water in the United States is estimated to contain at least one type of PFAS, which affects 18–80 million people. 25 Another study focused on averages of PFAS concentrations in wastewater throughout the United States, and it was determined that PFOA had increased 6.0 ± 1.6 ng/L from influents to effluents over time. The average FOA presence has generally decreased by 13% from 1999 to 2020, and PFOS levels have not significantly changed in wastewater. Levels of perfluorobutanoic acid (PFBA) from 2005 to 2015 showed an average increase due to its short-chain replacement to PFOA and PFOS, as it was discovered that bioaccumulation is 0.12 compared to 2.9 years for PFOS. Although PFBA is a healthier replacement, this fluorinated compound can still cause PFAS-induced illness over time. 26
PFAS in Nonwovens Production
Nonwoven materials use hydroentanglement or spunlacing to bond fibers together. This process implements highly pressurized water to tangle fibers and forms fabric as the end result. This method can produce fragments of fibers known as microfibers and nanofibers that lead to stronger nonwoven fabric. 27 The wet-laid process is used to produce pulp and paper products, such as teabags, coffee filters, wallpaper, and wipes. 28 A report published by the United Nations Environment Program has shown three major types of PFAS that have been used during the production of paper-based packaging consisting of perfluoroalkane sulfonyl fluoride, fluorotelomer-based alcohols, or acrylate and methacrylate esters attached to side chains as one group of PFAS. Another type of PFAS implemented is phosphate ester salts synthesized from perfluoroalkane sulfonyl fluoride or fluorotelomer-based alcohols with phosphoric acid. The last major type of PFAS used in production is perfluoropolyether. However, these substances have been phased out due to potential health conditions related to fluorinated substances.27,28 As PFAS become less involved within the paper production industry, it has not been explored whether water sources used in manufacturing processes contain PFAS, but it may be an unexpected route of contamination that needs further research along with contaminated raw materials unknowingly used in production.
Chemicals used in the hydroentanglement process are dependent on the substrate that is produced. Polyester- and propylene-based products are among the most popular nonwovens used by consumers. These include disposable hygiene products, such as wipes, diapers, and medical dressings. Synthetic chemical byproducts may be observed from the hydroentanglement process of these nonwoven materials. Cotton-based disposable products often have water repellency removed with scouring and bleaching with hydrogen peroxide, sodium hypochlorite, sulfuric acid, and hydrochloric acid. 29 Given that water resistance is not desired in cotton consumables, such as diapers, wipes, toilet paper, and other products that contribute to pulp and paper mill PFAS emissions, it is unclear where this contamination in toilet paper originates, as suggested in the work by Thompson et al. 23 In summary, Figure 1 demonstrates all of the pathways of common exposure to PFAS through the environment and food web from paper mill waste entering waterways and paper-based product use contributing to global PFAS exposure.
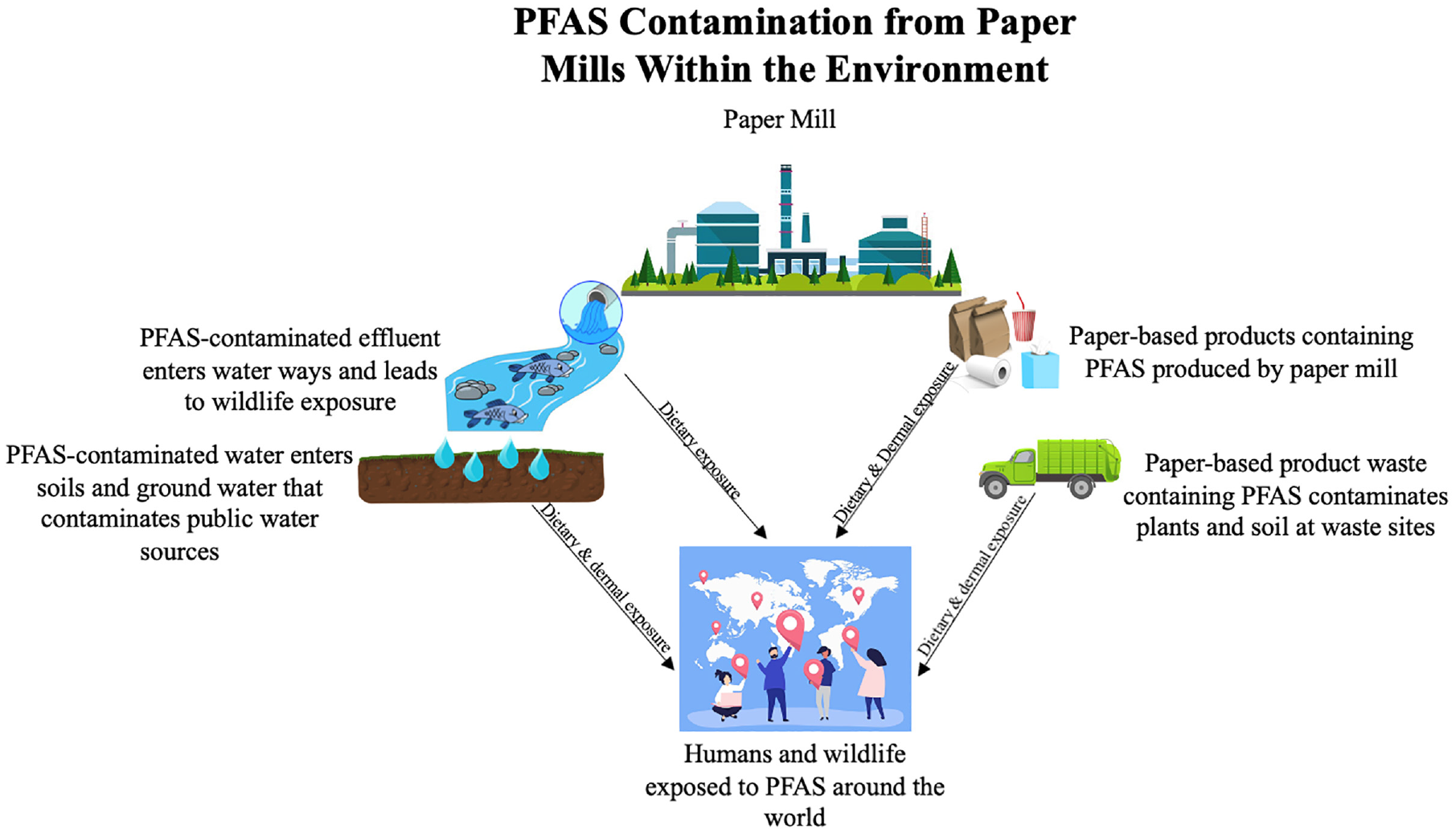
PFAS contamination from paper mills within the environment. This scheme shows human and wildlife exposure as a result of paper mill waste and paper-based product use.
PFAS Substitutions and Remediation
Policymakers are currently making efforts to replace these chemicals with non-toxic alternatives for water resistance, as dietary and dermal PFAS exposure can lead to health complications, such as increased cholesterol, high blood pressure, suppressed immune system, altered liver and kidney function, delayed development, and health conditions as a result of endocrine disruption.9–11,30 As current research develops solutions to reduce PFAS usage, many issues still reside in finding a proper replacement to alleviate exposure while still providing water and oil resistance compatible with paper substrates. Waxes, polymer films, and laminates made from polyethylene can be used as a film for paper-based products and are not known to pose a health threat to the public or environment. Current research suggests that polyester-based treatments, such as polylactic acid, polybutylene adipate terephthalate, polybutylene succinate, and polyhydroxyalkanoates, may also be used, as they are biodegradable. In addition, plant proteins have also been tested, and the optimal treatment was determined to be soy protein. 31
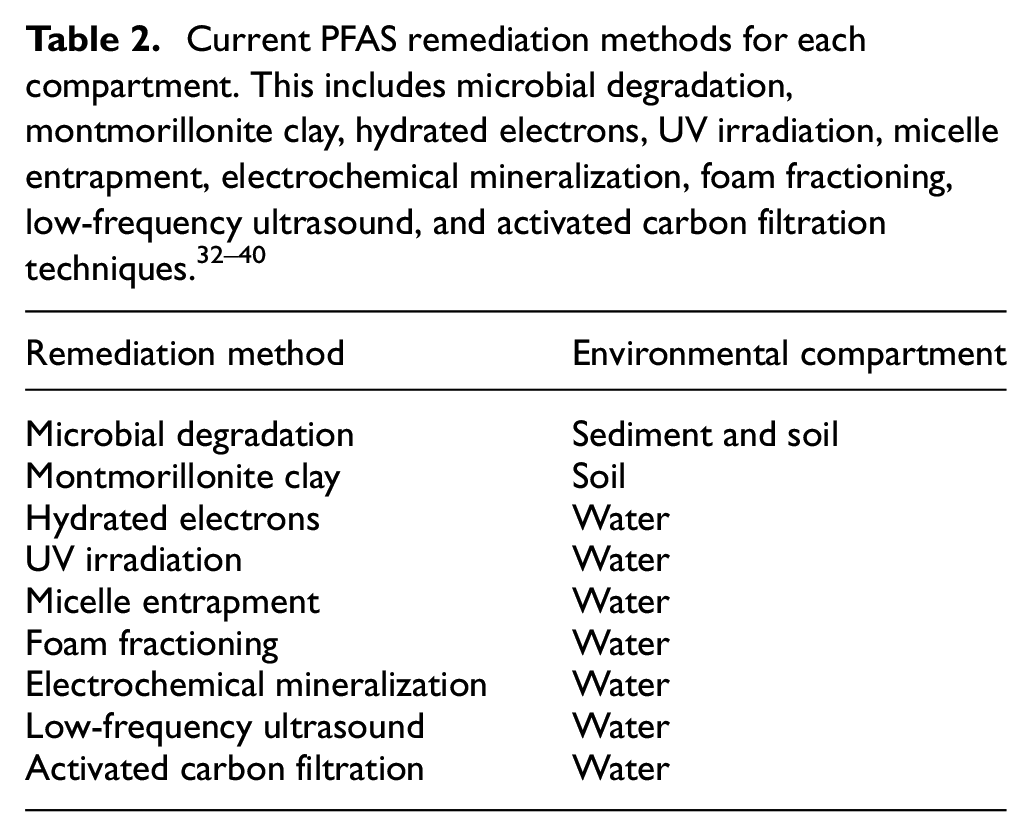
Bacterial remediation is used to prevent PFAS contamination from entering other environmental compartments. A mixture of Klebsiella sp., Alcaligenes sp., and Cronobacter sp. has been shown to reduce biochemical oxygen demand by up to 91% and to reduce organic halides by 45.4%. 32 Paper mill waste is also commonly burned before it is released at sites, but this leads to further emission of chemicals into the air. Scientists have experimented with montmorillonite clay as a remediation technique, and it was found that its presence during pyrolysis reduced residual weight. This can help decrease PFAS contamination, as the clay facilitates thermal degradation. 33
Studies focusing on the use of hydrated electrons suggest that these molecules have the potential to be implemented for PFAS degradation. Molecular interactions of hydrated electrons with PFAS were determined from ab initio molecular dynamics (AIMD) simulation and demonstrated defluorination of PFOA and PFOS. However, the AIMD simulation did show greater potential for the degradation of PFOA compared to PFOS in the presence of hydrated electrons, which suggests PFOS may be more resistant when this method is implemented for remediation. This is expected due to the activation barrier for C–F bond dissociation in PFOS, which is measured to be three times greater than the barrier for defluorination of C–F bonds in PFOA. As this study focused only on the two most prominent PFAS in the environment, PFOA and PFOS, the molecular dynamics studied via AIMD simulation demonstrates the ability for defluorination of other PFAS. The use of hydrated electrons is particularly of use for wastewater treatment due to the polarization of surrounding water molecules resulting in initiation of defluorination from reaction with the surface of the carbon and fluorine at various positions within the compound. 34 Seven hydrated electron systems were created to determine the rate of PFAS degradation using 3-indole acetic acid, indol-1-yl-acetic acid, indole-3-carboxylic acid, indole, skatole, tryptamine, and donaxine. Within an 8 h interval of PFOA and PFOS exposure to these systems, defluorination for PFOA increased by 9–13% in solution with 3-indole acetic acid, indol-1-yl-acetic acid, and indole-3-carboxylic acid when compared to direct ultraviolet light (UV) exposure only. In the presence of indole, skatole, and donaxine, PFOA degradation and defluorination occurred within 2.5 h and was undetectable after 8 h of incubation. PFOS degradation and defluorination were less affected by the presence of these systems coupled with UV irradiation. However, a slight increase in improvement was still observed that suggests these treatments may be implemented in addition to UV irradiation which has been studied for PFAS degradation previously. 35
Another study showed that micelle structures comprising cetyltrimethylammonium bromide in the presence of indole acetic acid led to the confinement of PFAS when released into a solution containing PFOA within 2.5 h under UV irradiation. These micelle structures comprised hydrated electrons under anaerobic and alkaline conditions that provide a novel method for the use of these hydrated electrons. 36 PFAS photodegradation coupled with other techniques has proved to be successful for the cleavage of the C–F bonds as well. An experiment focusing on the reaction kinetics of PFOA when exposed to UV irradiation along with various catalysts, such as SiC/graphene, SiC, and nano-TiO2, have shown decomposition. Nano-TiO2 was determined to be the optimal catalyst in this study as degradation was up to 16.7% higher compared to Si/graphene and SiC. 37 This technique can be used for PFAS-contaminated water to enhance the rate of degradation and provide another option for remediation in addition to the indole derivative implementation.
Electrochemical mineralization is another technique that degrades PFAS and can be used for the purification of wastewater. It has been reported that PFAS can be mineralized by a system containing nanocrystalline PbO2 that is more efficient for short-chain PFAS, such as perfluoroalkyl carboxylic acids (PFCAs). It was also discovered during this experiment that hydroxyl radicals and water assisted in defluorination of both PFCAs and PFOA. 38 The use of low-frequency ultrasound has also been shown to assist the electrochemical oxidation process by generating hydroxyl radicals at higher concentrations than can be used for water treatment jointly with electrochemical processes. This study found that, in addition to low-frequency ultrasound, increased effects were observed in the presence of the Ti/SnO2–Sb/Ce–PbO2 anode. 39 Electrochemical oxidation with foam fractioning is another method for the degradation of PFAS. Foam fractioning accentuates the surfactant properties of PFAS and adsorbs the compounds from air bubbles. PFAS can then be collected and filtered out of water once gathered on these air bubbles. However, long-chain PFAS are harvested more efficiently than short-chain PFAS. Incorporation of electrochemistry with foam fractioning can be implemented for groundwater treatment and soil leachate. 40
Filters and adsorbent nanofibers are novel methods for remediation to remove short-chain PFAS from wastewater. Many companies replaced long-chain PFAS with short-chain PFAS due to their ability to degrade faster, but PFAS still accumulate in water regardless of the faster degradation period. One study assessed the removal of hexafluoropropylene oxide dimer acid and its ammonium salt (GenX), which is the mixture commonly implemented to replace long-chain PFAS in products. Two techniques for GenX removal were electrospun polyacrylonitrile nanofibers and amidoxime surface-functionalized electrospun polyacrylonitrile nanofibrous membranes. It was determined that the compound mixture was removed at approximately 0.6 mmol/g. 39 This study provides insight into water treatment methods and can lead to advanced water remediation processes that are currently not implemented for short-chain PFAS.
Another remediation method is the use of filtration systems using activated carbon at residences and factories due to the reported abundance of PFAS in nearly half of the US drinking water. 41 Six filtration methods can be implemented in the form of filtered pitchers and faucet-mounted, faucet-integrated, countertop, and under-the-sink filters. Homes may also have a filtration system that filters PFAS from all water sources, which can be used in commercial production facilities to prevent product contamination as well. 42 The Environmental Working Group tested 10 filter brands and found that most do not effectively remove these substances. Only 3 out of 10 brands tested, “Clearly Filtered,” “Zero Water,” and “Berkley,” reduced PFAS to non-detectable levels, so it is recommended for consumers to use water pitchers or reverse osmosis systems from these brands. The Berkely filtration system removed 98% of PFAS and Clearly Filtered and Zero Water removed 100% of PFAS in this study.42–44 Although these techniques have been shown to greatly reduce and completely eliminate PFAS from water, filters only serve as a short-term solution for contamination and exposure. However, filters can be used in commercial manufacturing during wet-laid processes to reduce potential contamination from water sources containing PFAS. As research on remediation and PFAS replacements continues, experimentation may lead to the correct treatment, whether that is a single compound, mixture of layered films, more effective filters, or plant-based solutions that are biodegradable and safe for use.
Textile-producing manufacturers are taking steps to eliminate PFAS treatment and to implement other solutions for water and oil repellency. In 2022, Milliken & Company announced that PFAS would no longer be used in the finishing process by 2023. This company was the first to take industrial action to aid in the reduction of exposure and emissions. Research on other oil-repellent treatments led Milliken & Company to stop producing oil-resistant textiles due to the lack of studies for substitutions. 45 The action to halt this line of production serves as an example for other manufacturers to follow due to the company’s realization of detrimental effects from PFAS and the strict initiative that was taken to combat this issue.
Retailers have also announced the removal of PFAS in paper-based food packaging to reduce migration into food. McDonalds, Burger King, and Chick-fil-A pledged to implement other methods for water and oil resistance. According to the Food and Drug Administration (FDA), long-chain PFAS did not pose a threat in food packaging although they were phased out from other products in 2016. 46 Now that new research has shown migration of PFAS, companies are experimenting with alternatives. Bamboo, palm leaf, and polylactic acid are potential replacements for PFAS-treated paper plates, bowls, takeout containers, and food trays. 47 These alternatives may also be used in other textile products if deemed effective with future research.
In summary, many remediation methods have been studied and implemented for water treatment, such as electrochemical processes, hydrated electrons, low-frequency ultrasound, and filtration via activated carbon and adsorbent nanofibers. PFAS-contaminated soils and sediments can be treated with bacterial remediation and Montmorillonite clay. PFAS are not a cause of concern in the air as they persist in soil, sediment, and water, which are the main environmental focuses of remediation efforts summarized in Table 2. More remediation techniques for water treatment have been established in comparison to soil and sediment. As new research on PFAS degradation occurs, an increase in remediation techniques may be observed. However, bacterial remediation of PFAS-contaminated soils and sediment is well established and is the most prominent method of treatment.
Legislation
Strict regulations are being set in place by states to ensure limitations of PFAS exposure from food packaging and paper products. In January 2023, California prohibited individuals and businesses from distributing foods and food packaging that contain PFAS above 100 ppm, and some states have now started the ban completely. As of May 2023, the 12 states of the United States, such as California, Colorado, Connecticut, Hawaii, Maryland, Maine, Missouri, New York, Oregon, Rhode Island, Vermont, and Washington have begun the process of banning PFAS in food packaging. 48 The European Chemicals Agency has updated its regulations to implement a complete ban of PFAS use on all products with an 18-month transition period to replace current methods with safer alternatives. It has been reported that these substitutions are unknown at this time, and further research must be conducted during the 18 months to start production with new treatments. It is unknown what the United States is going to implement regarding paper-based products following the ban as well. 49 The National Health Commission of the People’s Republic of China reported in 2023 that the phase-out of production of food contact materials with PFAS has begun, and PFAS will be replaced with silicone-based films, rubber, and plastic food containers, leading to a decrease in paper-based substrates. 50 Other countries such as India, Indonesia, and Bangladesh, which are among the top-producing consumer product countries, have not passed legislation to regulate PFAS in paper products or food packaging, but this may change as research presents data highlighting the detrimental health risks.51–55 As the United States, European Union, and China begin banning PFAS, the abundance and persistence of these substances will still present a challenge but may be mitigated with these stricter policies in the future.
Conclusion
PFAS have been used since the 1940s in consumer products, and research has repeatedly shown over time that fluorinated treatments are detrimental to human health and wildlife. 56 PFAS contamination and emissions have driven policymakers and paper mills to conduct changes in paper, pulp, and food packaging production. Current research focusing on remediation techniques and PFAS contamination prevention provides solutions for paper mills to continue with safer alternatives during production, such as polymer-based materials, waxes, and plant protein treatments to mimic-water repellency effects. Effluent filtration, electrochemical mineralization, hydrated electrons, UV irradiations, micelles, foam fractioning, microbial degradation, and montmorillonite clay can be used to reduce PFAS emissions and serve as remediation methods for contaminated sites.32–40 With new substitutions and applications discovered through current and future experimentation, optimal remediation of contamination and exposure is possible.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.