Abstract
Fluoroalkyl end-capped vinyltrimethoxysilane oligomer [RF-(CH2-CHSi(OMe)3)
n
-RF: n = 2, 3; RF = CF(CF3)OC3F7: RF-(VM)
n
-RF] was found to undergo the sol-gel reaction under alkaline conditions in the presence of chemically modified cellulose fibers treated with N-methylglucamine units (
Keywords

Introduction
Much attention has been focused on the use of the cellulose fibers in the fields of wastewater treatment and chemical industries as an inexpensive alternative to the adsorbents such as activated carbon due to their large surface area around 200 m2/g.
1
Chemically modified cellulose fiber derivatives treated with polyethyleneimine have been already applied to the mercury removal from wastewater.
2
N-methylglucamine-type cellulose fiber derivatives were also applied to the removal of boron (III) from wastewater with a higher removal rate.
3
From the increased applicable viewpoint of these chemically modified cellulose fiber derivatives into a wide variety of fields, it is of particular interest to explore the novel composite materials based on these cellulose fiber derivatives and organic polymers, especially fluorinated polymers possessing a unique surface active characteristic related to longer fluoroalkyl groups, which cannot be achieved by the corresponding non-fluorinated polymers. However, to our knowledge, such studies have been very limited so far. During our comprehensive studies on the fluorinated polymers possessing a surface active characteristic,4-9 we have already found that two fluoroalkyl end-capped vinyltrimethoxysilane oligomeric silica nanocomposites [RF-(CH2CHSiO3/2)
n
-RF; RF = CF(CF3)OC3F7: RF-(VM-SiO3/2)
n
-RF], which were prepared by the sol-gel reaction of the corresponding oligomer [RF-(CH2CHSi(OMe)3)
n
-RF: RF-(VM)
n
-RF] under alkaline conditions, can exhibit an oleophobic/superhydrophobic characteristic on their modified glass surface.
10
RF-(VM)
n
-RF oligomer can also undergo the sol-gel reaction in the presence of alkyl-modified cellulose (AM-Cellu) under alkaline conditions to supply the corresponding fluorinated oligomeric silica/AM-Cellu nanocomposites [RF-(VM-SiO3/2)
n
-RF/AM-Cellu].
11
These obtained nanocomposites have been applied to the surface modification of filter paper to provide a superoleophilic/superhydrophobic property on the modified surface, demonstrating that the modified filter paper is effective for the separation membrane for W/O emulsion to isolate the transparent colorless oil.
11
Here we report that RF-(VM)
n
-RF oligomer can undergo the sol-gel reaction under alkaline conditions in the presence of chemically modified cellulose fibers treated with N-methylglucamine units (
Experimental
Measurements
Field emission scanning electron micrograph (FE-SEM) was recorded by using JEOL JSM-7000F (Tokyo, Japan). Energy dispersive X-ray (EDX) spectra were obtained using JEOL JSM-7000F (Tokyo, Japan). The contact angles were measured by the use of Kyowa Interface Science Drop Master 300 (Saitama, Japan). Ultraviolet–visible (UV–vis) spectra were measured using Shimadzu UV-1600 UV–vis spectrophotometer (Kyoto, Japan).
Materials
Chemically modified cellulose fibers treated with N-methylglucamine units (
Preparation of the fluoroalkyl end-capped vinyltrimethoxysilane oligomeric silica/Cell-fiber-MeGlu composites [RF-(VM-SiO3/2) n -RF/Cellu-fiber-MeGlu]
A typical procedure for the preparation of RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Preparation of RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

a Yield was based on oligomer and Cellu-fiber-
Contact angle measurements of dodecane and water on the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-MeGlu composite powders surface
The RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Removal of aromatic compounds from aqueous methanol solution by using the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-ImDia composites
RF-(VM-SiO3/2)
n
-RF/Cellu-fiber
Preparation of liquid marbles
Liquid marbles were prepared by rolling the liquid drops on the composite powders layer. The amount of all liquid drops was adjusted to 10 μL, and the liquid marbles thus obtained were transferred onto another substrate by careful manipulation with a micro spatula [see Figure 10-
Results and discussion
Preparation and surface wettability of fluoroalkyl end-capped vinyltrimethoxysilane oligomeric silica/Cellu-fiber-
Sol-gel reaction of fluoroalkyl end-capped vinyltrimethoxysilane oligomer: RF-(CH2-CHSi(OMe)3)
n
-RF: n = 2, 3; RF = CF(CF3)OC3F7: RF-(VM)
n
-RF] proceeded under alkaline conditions in the presence of chemically modified cellulose fibers treated with N-methylglucamine units (

Preparation of fluoroalkyl end-capped vinyltrimethoxysilane oligomeric silica/Cellu-fiber-
As shown in Figure 1 and Table 1, the expected composites were obtained in 56–90% isolated yields. FE-SEM (Field Emission Scanning Electron Microscopy) photographs of the composites (Runs 1 and 5 in Table 1) have been recorded in order to clarify the formation of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

FE-SEM (Field Emission Scanning Electron Microscopy) pictures of pristine Cellu-fiber-

FE-SEM (Field Emission Scanning Electron Microscopy) pictures of pristine Cellu-fiber-ImDia
Figures 2-(a) and 3-(a) show that the pristine Cell-fiber-
In addition, we have measured the energy dispersive X-ray analyses (EDX) of these composites to verify the presence of the fluorinated oligomer in the composites. The results are shown in Figures 2-
EDX mapping micrographs on the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Next, we tried to study on the surface wettability of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Dodecane and water contact angle values on the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

a Each Run No. corresponds to that of Table 1.
As shown in Table 3, dodecane and water contact angle values of the pristine Cellu-

Photograph of the water and dodecane droplets on the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Heretofore, it is well-known that the fabrication of superamphiphobic surface can be realized by lowering the surface energy and enhancing the surface roughness.13-19 Thus, it is suggested that such surface wettability can be realized by enhancing the surface roughness, of whose surface would be easily prepared through the sol-gel process of the present RF-(VM)
n
-RF oligomer illustrated in Figure 1. Especially, the Cellu-fibers are very convenient for the architecture of such roughness surface owing to their micrometer-size controlled fibers, and the longer fluoroalkyl groups on the Cell-fiber composite surface [see EDX mapping illustrated in Figures 2-
Application of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
ImDia
composites to the separation of the mixture of hydrocarbon oil and fluorocarbon oil
In order to clarify such unique wettability (superamphiphobic property) related to the present fluorinated composites, we tried to apply the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

Separation of the mixture
As shown in Figure 5-
Application of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
ImDia
composites to the removal of organic aromatic compounds from aqueous methanol solutions
In this way, it was clarified that the present fluorinated composite powders can provide the superamphiphobic (superoleophobic and superhydrophobic) property on the composite surface. Such unique wettability would enable the present composites to interact predominately with fluorinated organic molecules in aqueous solutions. Thus, we tried to study on the removal ability of fluorinated aromatic compounds from aqueous methanol solutions by using the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

Schematic outline for the analysis of the removal ratio of aromatic compounds by the use of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Firstly, we tried to study on the removal ability of bisphenol AF(BPAF) and bisphenol A (BPA) by the use of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
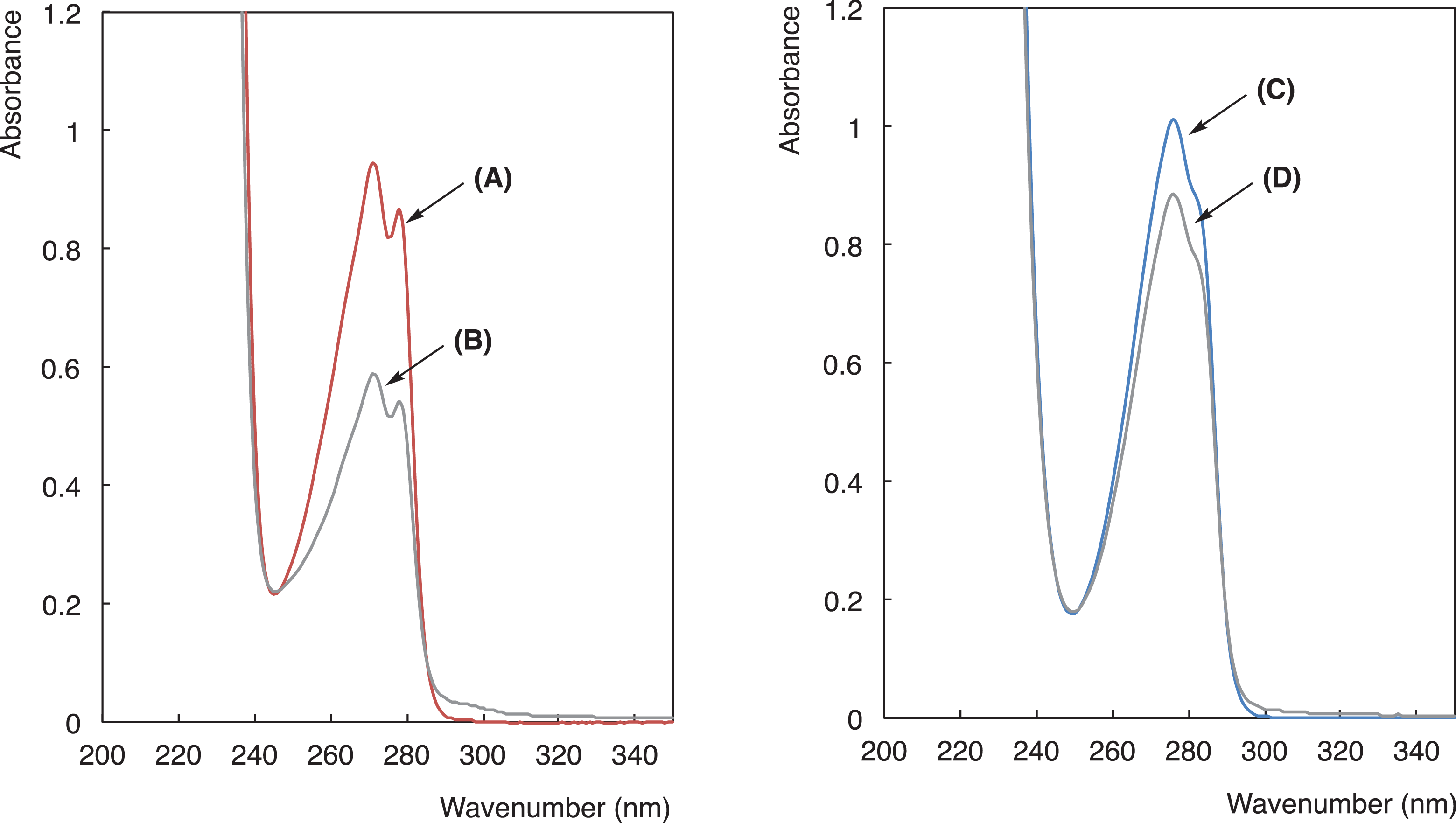
UV-vis spectra of BPAF (101 mg/dm3)[
As shown in Figure 7, UV-vis spectra show that the effective decrease of the absorbance related to BPAF and BPA can be observed by the addition of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

UV-vis spectra of PF-AP (10 mg/dm3) [
We have studied on the removal ability of these aromatic compounds by using some of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

Figure 9 shows that each RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Application of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-MeGlu composites and -I
mDia
composites to the fabrication of liquid marbles
Hitherto, it is well-known that the rolling of a hydrophilic liquid drop on a hydrophobic powder layer makes it possible to prepare a liquid marble, of whose powder can self-assemble at the liquid-air interface.20-23 Liquid marbles are in general millimeter-sized liquid drops, and there has been a great interest in liquid marbles, due to their high potential application into numerous fields such as sensors, microfluidics, and material delivery carriers.24-32 Therefore, it is suggested that our present fluorinated composite powders possessing a superamphiphobic property enwrapping the water drop or the traditional oil drops would allow the fabrication of not only water marble but also oil marbles. From the applicable point of view of the present fluorinated composites possessing a superamphiphobic characteristic, it is of particular interest to fabricate liquid marbles by using the present fluorinated composite powders. Firstly, we tried to study on the fabrication of water marble stabilized by the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
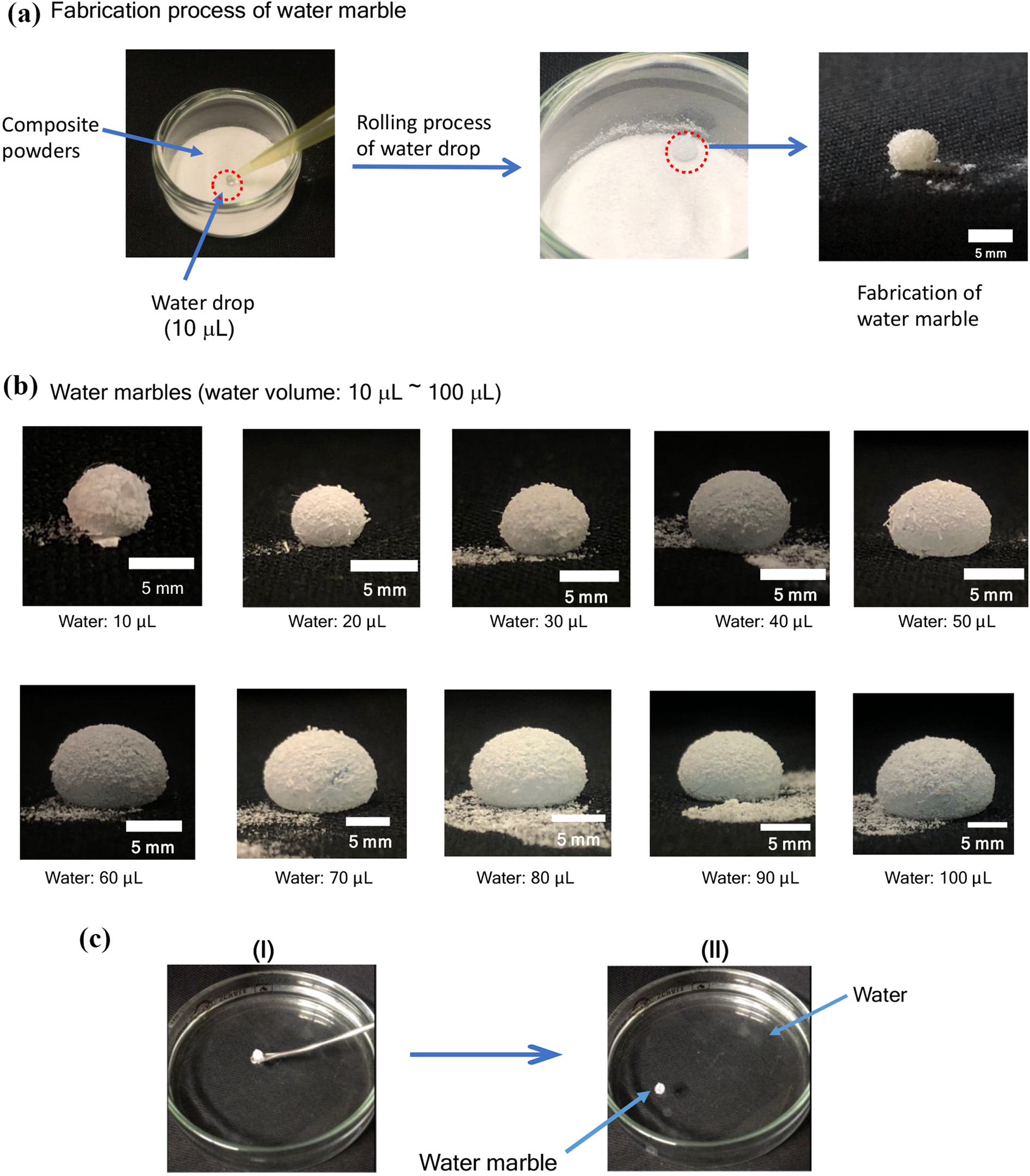
Fabrication process of water marble by using the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
As shown in Figure 10-
Similarly, the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

Photograph of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
As indicated above, our present composite powders can exhibit not only a superhydrophobic but also superoleophobic property on their surface. Thus, it is suggested that the present composite powders should be applicable to the fabrication of several type of oil marbles. In fact, we have succeeded in the fabrication of stable liquid marbles from oil drops such as glycerine, ethylene glycol and dodecane by using the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-

Photograph of the RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Conclusions
RF-(VM-SiO3/2)
n
-RF/Cellu-fiber-
Footnotes
Acknowledgment
The authors are sincerely grateful to Chubu Chelest Co., Ltd. (Mie, Japan) for supplying Cellu-fiber-
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by a Grant-in-Aid for Scientific Research 19K05027 from the Ministry of Education, Science, Sports, and Culture, Japan.

