Abstract
Compared with traditional water-based dyeing, non-aqueous medium dyeing technology has been paid more and more attention in the textile dyeing industry because of its advantages of saving water and chemicals. In order to distinguish and identify water bath dyeing products and non-aqueous medium dyeing products, a traceability technology method for non-aqueous medium dyeing products was established with rare earth elements. First, traceable materials for two typical fibers, polyester and cotton, were selected from five rare earth elements: La, Ce, Nd, Eu, and Sm. Rare earth elements were added to mark products during the non-aqueous dyeing process. Then the rare earth elements content of marked products was detected by inductively coupled plasma–optical emission spectrometry, which was used to identify non-aqueous media products. The influence of this traceability method on the basic properties of dyed fabrics was studied. Compared with traditional anti-counterfeiting technologies such as label anti-counterfeiting and query label anti-counterfeiting, the method of inductively coupled plasma–optical emission spectrometry has a low method detection limit in the range of 0.0204–0.036 mg/kg, and a good relative standard deviation values in the range of 0.69–2.43%. The content of rare earth elements in dyed fabrics was detected by inductively coupled plasma–optical emission spectrometry at 6–7 mg/kg when 10 mg/kg of rare earth elements were employed during dyeing. Therefore, a detection and certification method for non-aqueous dyeing products has been established, which is of great significance to the anti-counterfeiting traceability of non-aqueous medium dyeing technology.
Keywords
Introduction
The consumption of water and energy in the textile dyeing industry has become an important problem which restricts the development of the textile industry. 1 Therefore, the research and development of textile dyeing in recent years has focused attention on saving water and energy. 2 For example, small liquor ratio dyeing, sewage recycling, 3 and modified textile pretreatment 4 were used to reduce wastewater discharge. But only focusing on a certain part of the production process limits its application in industrial production. 5 The non-aqueous medium dyeing system is a new dyeing technology with ecological and environmental qualities that has attracted more and more attention. Pei et al.6,7 employed an ecological environmental solvent instead of water as the dyeing medium, and the research has achieved noteworthy achievements. Developing traceability strategies for non-aqueous medium dyeing technology has become an urgent challenge, which has a positive effect on the development of new technology. 8 Currently, anti-counterfeiting technologies are employed in textiles such as barcode anti-counterfeiting, telephone anti-counterfeiting, thermochromic, photochromic anti-counterfeiting technology, and so on.9–11 These technologies are applied to textiles as complete commodities, such as hats, clothes, and shoes. Kong et al. 12 believed that the development of textile anti-counterfeiting technology should proceed from the characteristics of textiles. Adding some substances with anti-counterfeiting characteristics to fiber materials is a method that is more in line with textile anti-counterfeiting.
Rare earth elements (REEs) are an extremely coherent group of elements, and their accurate determination is very important in chemical research. REEs were discovered in 1794 by Finnish scholar John Gadolin.13,14 The International Union of Pure and Applied Chemistry (IUPAC) defined REEs as 15 lanthanides with atomic numbers 57–71 and two transition elements with atomic numbers 21 and 39, namely lanthanum (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb), lutetium (Lu), scandium (Sc), and yttrium (Y). Toxicological and ecotoxicological data have been associated with many REEs applications, 15 indicating that their application is safe to a certain extent. There were no safety problems or residues in non-ruminant animals fed <100 mg/kg of Ce or La. 16 Supplementation of cerium oxide (CeO) at 200 mg/kg has been found to improve average daily feed intake in female rabbits without any negative effect on serum. 17 With the expansion of the application range of REEs, REEs have gradually been paid attention and application in the textile industry.18,19 At present, they have been applied to various natural fibers and chemical fibers such as wool, linen, pure cotton, silk, viscose, rayon, acrylic, and other products.
Zheng et al. 20 proved that REEs would be a promising environment friendly mordant in natural dyeing. Various REEs have been used for formulating fluorescent functional inks and further printing designable patterns on different substrates to encrypt information.21,22 These applications show the preliminary application of REEs in anti-counterfeiting technology. Jin and Xu 23 prepared a modified infrared anti-counterfeiting material with nano-scale inorganic non-metallic particles doped with single or co-doped rare earth Yb (ytterbium) and invented an infrared anti-counterfeiting technology and method to realize the traceability certification of down fiber raw materials. Therefore, several REEs commonly used in the textile industry were selected for traceability marks in the traceability of non-aqueous medium dyeing products.
Inductively coupled plasma–optical emission spectrometry (ICP-OES) is a commonly used analytical tool for the determination of REEs. 24 The method has high accuracy, wide linear range, less matrix interference, and extremely high sensitivity for the determination of low content of REEs, which can fully meet the needs of fabrics for simultaneous determination of multiple REEs. 25 REEs in coal samples were determined by ICP–OES 26 with low detection limits (1.4 to 105 µg/g) and good precision (<5%), which was useful for quantitatively understanding REEs in various resources for developing effective recovery methods. Therefore, ICP–OES can be employed to detect traceable REEs added to non-aqueous medium dyeing products.
In this investigation, REEs were selected as traceability materials for non-aqueous medium dyeing products, and ICP–OES was employed to measure the content of REEs in non-aqueous medium dyeing products to distinguish them from water-bathed dyeing products. Taking cotton fabrics dyed with reactive dyes and polyester fabrics dyed with disperse dyes as examples, a detection method was established. Furthermore, the appropriate tracer elements were screened, and the long-term effect of REEs as tracer elements was verified. The effect of adding rare earth dyeing on the basic properties of fabrics was also discussed. Finally, the mechanism of rare earth dyeing was also studied.
Experimental
Materials and Chemicals
Materials and chemicals are described in the Supporting Information.
Dyeing Process
Cotton fabric dyeing: Dyeing was carried out on a DYE-24 dyeing machine (Shang Hai Chain-Lih Automation Equipment Co., Ltd). The amount of water was 150% (o.w.f). The amounts of REEs were 1 mg/kg and 10 mg/kg (mass ratio of REEs to fabric). Either 1 mg/kg or 10 mg/kg of REEs was weighed and diluted in 60% (o.w.f) water and then added to dyeing system. About 4 g of cotton fabric was dyed with 2% (o.w.f, on amount of fabric) of reactive red 120. The bath ratio of fabric to D5 was 1:10. The amount of sodium carbonate was 5.75% (o.w.f). Reactive dye and sodium carbonate were diluted in 90% (o.w.f) fabric. After the REE solution, reactive dye and sodium carbonate solution were added to D5 medium, the initial dyeing temperature was 25°C maintained for 10 min, then it was raised to 90°C at a rate of 2°C/min, and maintained for 30 min. After dyeing, the temperature was dropped to 40°C at a rate of 2°C/min. The dyed fabric was washed twice using aqueous solution at a liquor ratio of 1:10 at 80°C for 15 min. After washing, the dyed fabric samples were dried for 1 h at 80°C.
Polyester fabric dyeing: 4 g of polyester fabric (gram weight: 89.6 g/m2) was dyed with pure disperse red 177 (0.5%, o.w.f). Liquid disperse red 177 with 25% (o.w.f) accelerant was used during dyeing. The bath ratio of fabric to D5 was 1:10. The amounts of REEs were 1 and 10 mg/kg. Dyeing was started at 25°C, and the temperature was raised to 80°C at the rate of 6°C/min, then the temperature was raised to 140°C at a rate of 3°C/min and kept for 60 min. After dyeing, the dyed fabric was washed twice using D5 solvent at a liquor ratio of 1:10 at 80°C for 15 min. At the same time, the fibers without adding rare earth were dyed as control samples under the same dyeing conditions.
After dyeing, part of the dyed fabric was immersed in 30 g/L of non-formaldehyde fixing agent DM-2514 solution for 3 min. Then it was pre-dried at 80°C for 5 min, and finally the fabric was baked at 160°C for 2 min.
ICP-OES Measurement
ICP-OES (Avio200, PerkinElmer, Co., United States) was employed to determine the content of REEs on the fiber. First, the instrument was turned on and stabilized, then it was tuned to obtain the best sensitivity, the lowest oxide, double charge interference, and better resolution. The sample tube was inserted into the blank solution and the sample solution. The plasma gas (argon) flow rate was 8 L/min, and the RF generator power was 1400W. The auxiliary gas and nebulizer flow rates were 0.3 and 0.7 L/min, respectively. The sample volume was 1.2 mL/min. The pump was peristaltic at 1.5 mL/min. The integration time was 0.5–2 s.
About 0.2 g of dyed sample was cut into pieces and placed in a digestion tube. Around 3 mL of nitric acid was added into the tube and digested for 2 h by graphite digestion instrument (SPB 50-48, PerkinElmer, Co., United States). The sample was digested and then it was taken out of the digestion tube and diluted to 25 mL. The samples were transferred to a 50 mL centrifuge tube. After centrifugation for 15 min at 10,000 r/min by a high-speed desktop centrifuge (TGL-15B, Shang Hai Anting Scientific Instrument Factory Co., Ltd, China).
Method Detection Limits and Accuracy
According to 10 times the standard deviation of the blank signals, the method detection limit (MDL) was calculated. At the same time, the relative standard deviation (RSD) was calculated. For the recovery experiments, the sample solution was first measured by ICP as the sample background value. About 0.1 mg/L multi-element stock standard solution was added to the sample solutions for ICP measurements to get the recovery ratio.
Screening of REEs
The screening contains the type and the concentration of REEs. According to the previous investigation, five kinds of REEs (La, Ce, Eu, Nd, and Sm) commonly used in textiles were selected during dyeing. At the same time, the REEs were prepared in different concentrations (1 and 10 mg/kg) for fabric dyeing. The content of REEs on the fabric was determined by ICP-OES.
Method Validation
Long-term Effect Verification
A washing fastness tester, Washing Color Fastness Tester (SW-24C, Ningbo Textile Instrument Factory, China), was used to simulate the effect of multiple washings. A dyeing fastness tester (Y571, Laizhou Yuanmao Instrument Co., Ltd, China) was employed to simulate the effect of multiple rubbing fastness during textile processing. The long-term effect verification standard of washing fastness was determined according to the method of ISO 105-C04:2010, and the rubbing fastness was determined according to the method of ISO 105-X12:2001. The fabrics treated with REEs were subjected to water washing and friction experiments, and then the content of REEs was detected by ICP-OES to verify whether they were still distinguishable from blank samples.
Method Uniqueness Verification
This method is focusing on the whole process of non-aqueous medium dyeing. If the traditional dyeing method could not achieve such an experimental result by adding REEs at the end of production, pad dyeing was simulated as an imitation method to verify the imitation effect. The pad dyeing process is as follows. About 1 g of cotton fabric was immersed in a solution of 10 mg/kg REEs for 10 min at a liquor ratio of 20:1. Then it was padded using a device (P-Al Vertical Pressure Dyeing Machine, Xiamen Rapid Precion Machinery Co., Ltd.). Finally, the dyed fabric was dried in an oven at 60°C for 10 min. The rare earth contents of the pad dyed products and the dyed products were detected by ICP-OES, respectively.
Analysis of REEs Dyeing Mechanism
The Fourier transform infrared spectroscopy (FTIR), ultraviolet (UV), scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS), and particle size measurements are described in the Supporting Information.
Results and Discussion
Establishment of ICP-OES Method
According to the national standard, three times the standard deviation of 11 consecutive samples was taken as the limit of detection (LOD), and 10 times as the limit of quantification (LOQ). As shown in Table 1, ICP-OES method had low detection limits in the range of 0.0204–0.036 mg/kg, and the content could be quantitatively analyzed when the content was greater than 0.1120 mg/kg. The good RSD values were in the range of 0.69–2.43% (≤5%), indicating that the error of this method was small. Therefore, it was feasible to use the ICP-OES method to test the REEs.
Analytical parameters of the proposed method.
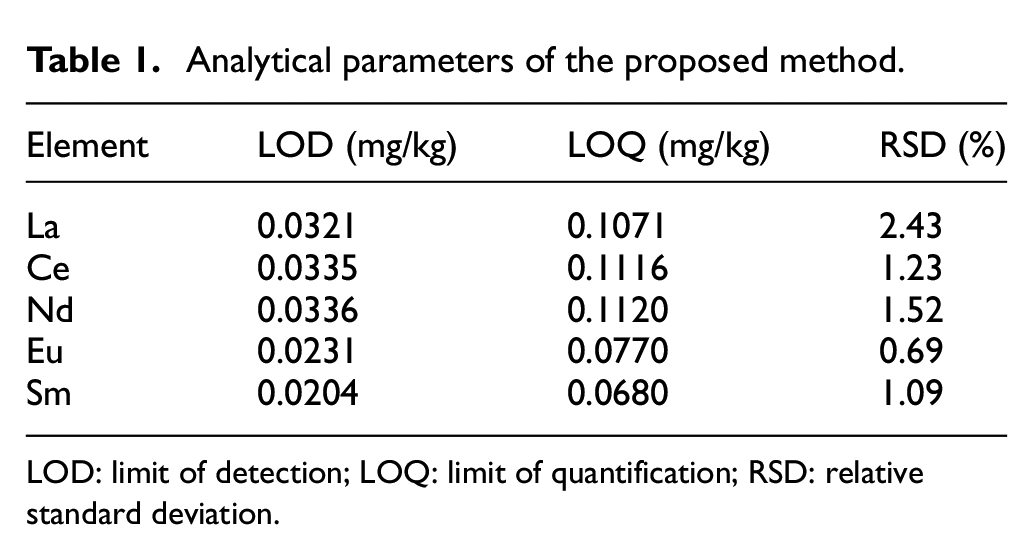
LOD: limit of detection; LOQ: limit of quantification; RSD: relative standard deviation.
The recovery ratios are shown in Table 2. About 0.1 mg/L of multi-element stock standard solution was added to the sample solutions to calculate the recovery rate. Because the contents of La and Ce elements in natural fibers were unstable, they were discarded during the experiment. The recovery ratios of Sm, Nd, and Eu were 95.51%, 105.4%, and 95.60% respectively. When the content of the tested component is less than 1 mg/kg, the allowable limit of the recovery rate is 80–120%. From the recovered test results, the proposed method is suitable for the determination of REEs in actual samples.
Analytical results for determination of REEs in actual samples.
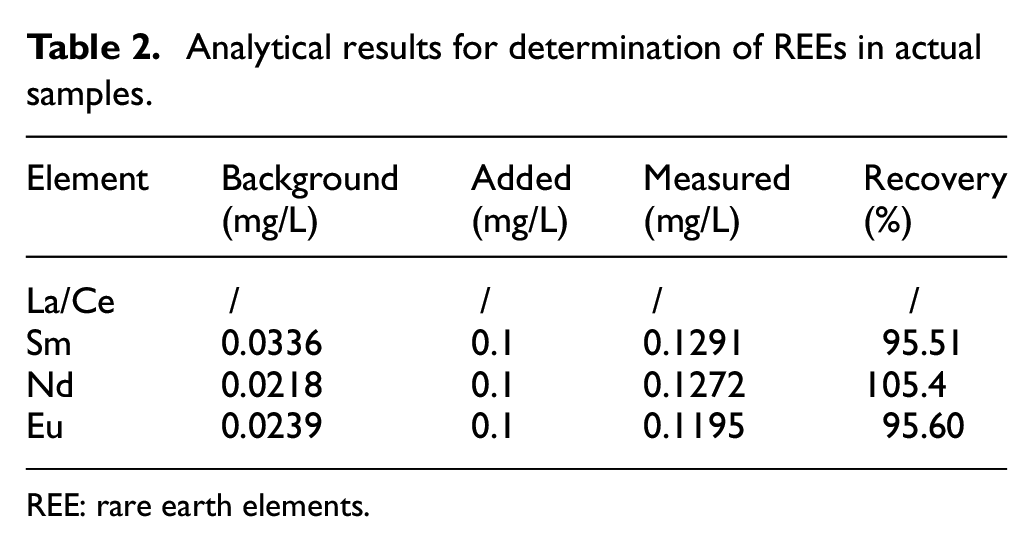
REE: rare earth elements.
Screening of REEs
According to the experimental conditions of dyeing, the dyed samples with La, Ce, Nd, Eu, and Sm, and blank dyed samples without REEs were prepared. At the same time, different concentrations of REEs were selected as 1 and 10 mg/kg. The results were analyzed by ICP-OES as shown in Table 3 and Supplemental Table SIV.
The content of REEs in the fiber after dyeing with 1mg/kg rare earth.
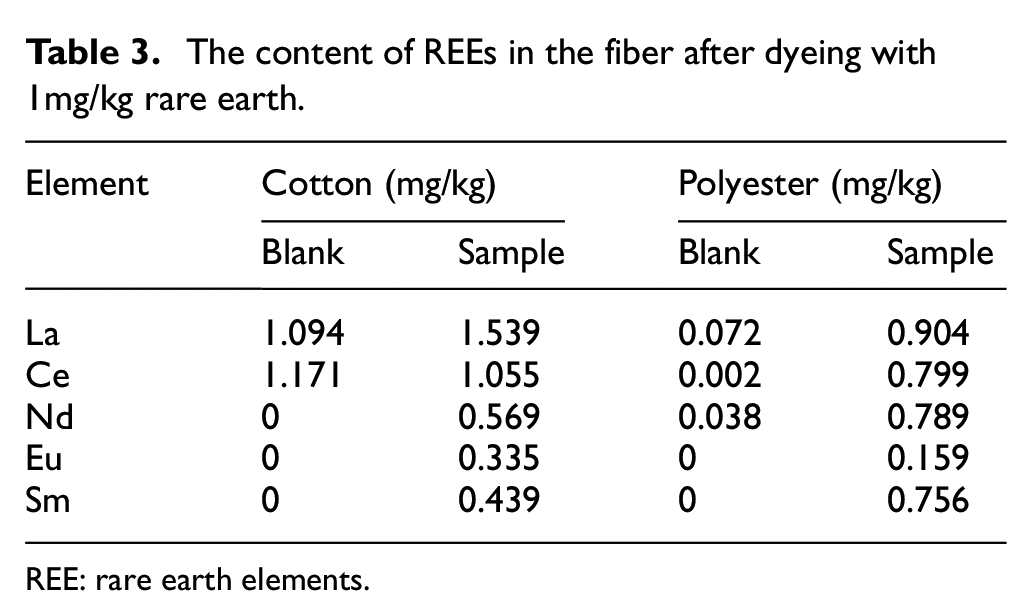
REE: rare earth elements.
From the contents of La and Ce elements in Table 3 and Supplemental Table 4, the elements contents of cotton without REEs dyeing were abnormal because the amounts of La and Ce exceed the total amount of addition for cotton with REEs dyeing. The reason may be that these two elements (La and Ce) are widely distributed in the natural environment and cotton fabrics contain these two elements.27,28 For the elements of Nd, Eu, and Sm, the cotton blank sample does not contain these three elements because these three elements are not common in nature, and the probability of these three elements being contained in natural fabrics is very small. Therefore, Nd, Eu, and Sm elements are more suitable as trace elements for non-aqueous dyeing. The amount of REEs was added to 10 mg/kg during dyeing (Table 4). For cotton fiber dyeing, the dyeing amount of Nd element was 57.49%, and the dyeing amount of Eu element was 72.54%. The dyeing amount of Sm element was 71.85%. For polyester, the dyeing amount of Nd element was 70.80–2.89%, and the dyeing amount of Eu element was 62.07%. The dyeing amount of Sm element was 72.89%. Therefore, Nd, Eu, and Sm were selected as traceable elements for cotton and polyester dyeing in the non-aqueous dyeing system.
The content of REEs in the fiber after dyeing with 10 mg/kg rare earth.
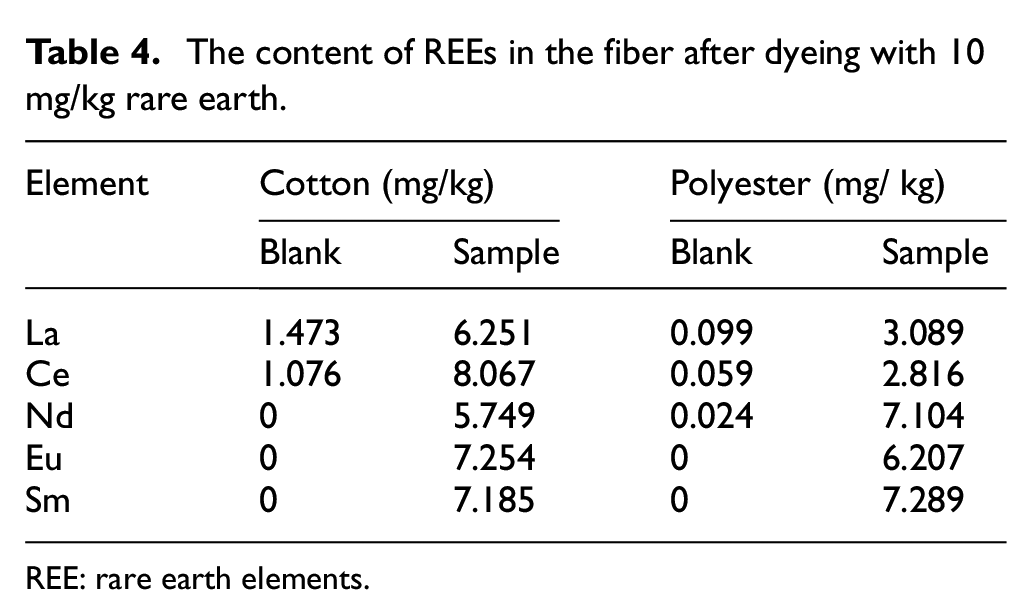
REE: rare earth elements.
Method Validation
Long-term effect verification
To simulate the influence of cloth manufacturing and household washing, the dyed fabric was rubbed 50 times and washed 20 times. It can be seen from Figure 1(a) and (b) that, before treating with fixing agent, the contents of REEs on cotton fabric and polyester fabric were 4.19 and 6.10 mg/kg, respectively. Before treating with fixing agent, the REE content on the cotton fabric decreased from 4.19 to 3.29 mg/kg after washing 20 times. The REE content of the polyester fabric decreased from 6.09 to 2.62 mg/kg. After treating with fixing agent, the REE content on the cotton fabric and polyester fabric decreased from 4.83 to 3.61 mg/kg and 6.52 to 3.21 mg/kg, respectively. Therefore, the samples dyed with REEs were still distinguishable from the blank samples when the number of washes was 20 times.
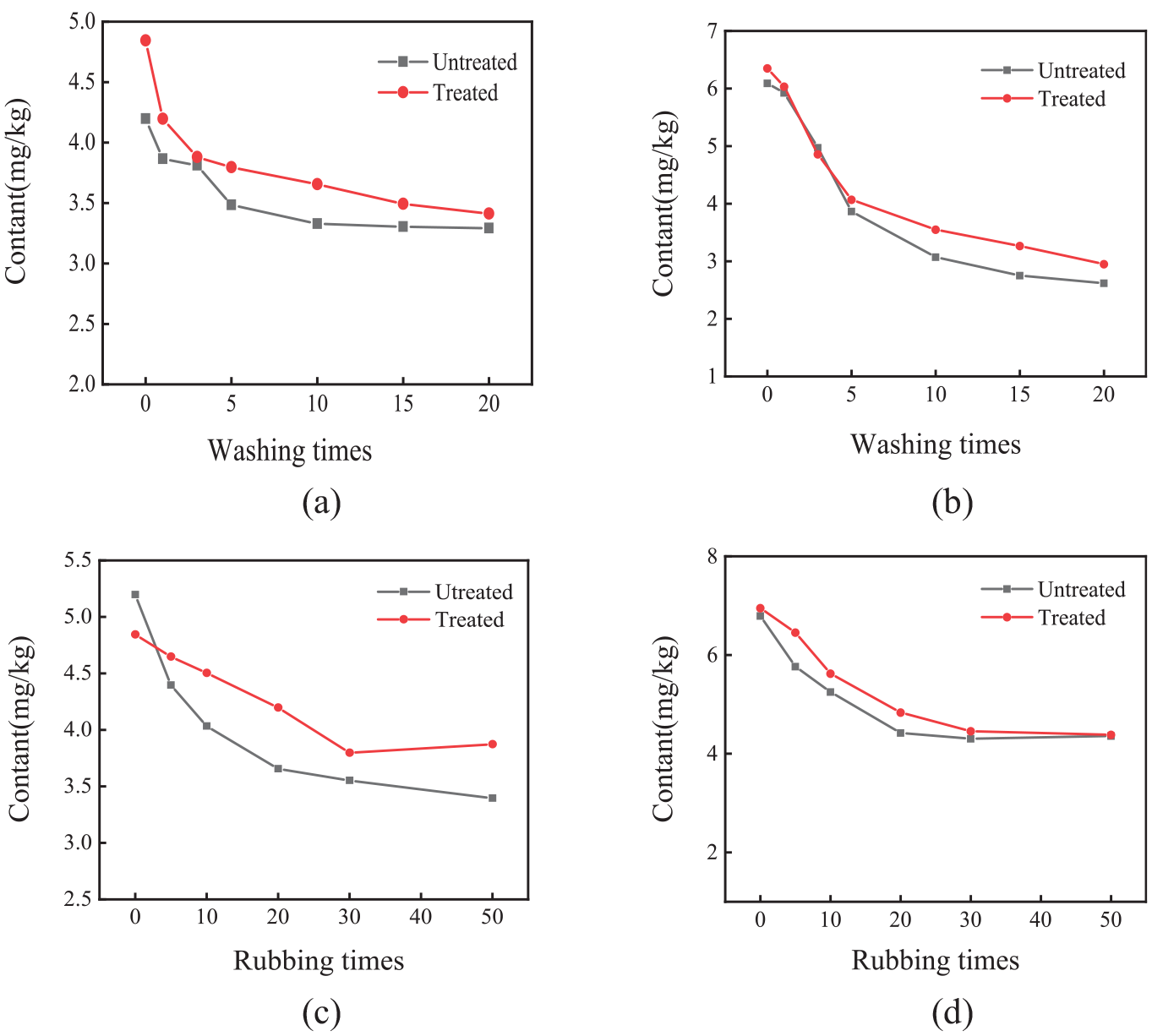
Effect of fixing agent treatment on rare earth content: cotton (a and c); polyester (b and d).
As shown in Figure 1(c) and (d), before treating with fixing agent, the REE content of the cotton fabric decreased from 5.51 to 3.59 mg/kg, and the REE content of the polyester fabric decreased from 6.05 to 2.54 mg/kg after rubbing 50 times. After treating with fixing agent, the REE content on cotton and polyester fabric changed little. Therefore, the friction effect of the fabric was slightly improved by the fixing agent. After simulating washing (20 times) and rubbing (50 times) experiments during textile processing, it was found that the tracked samples were still distinguishable from the blank samples. The effect of color-fixing agent on the fastness of rare earth was not obvious.
Effect of REEs on the Basic Properties of Fabrics
As shown in Figure 2(a), the fixation of reactive red 120 without REEs was 83.17% while it was 83.60% after adding REEs. The fixation of disperse red 177 without REEs was 88.01% while it was 88.35% after adding REEs, indicating that REEs had little effect on the permeability of dye. After treating with REEs, the lightness (L*) values, redness/greenness (a*) values, and yellowness/blueness (b*) values of fabrics were slightly higher than that of fabric without using REEs, which proved that REEs finishing had little effect on the color depth of the fabric. Therefore, REEs had almost no effect on the dye uptake and the fixation of the dye.
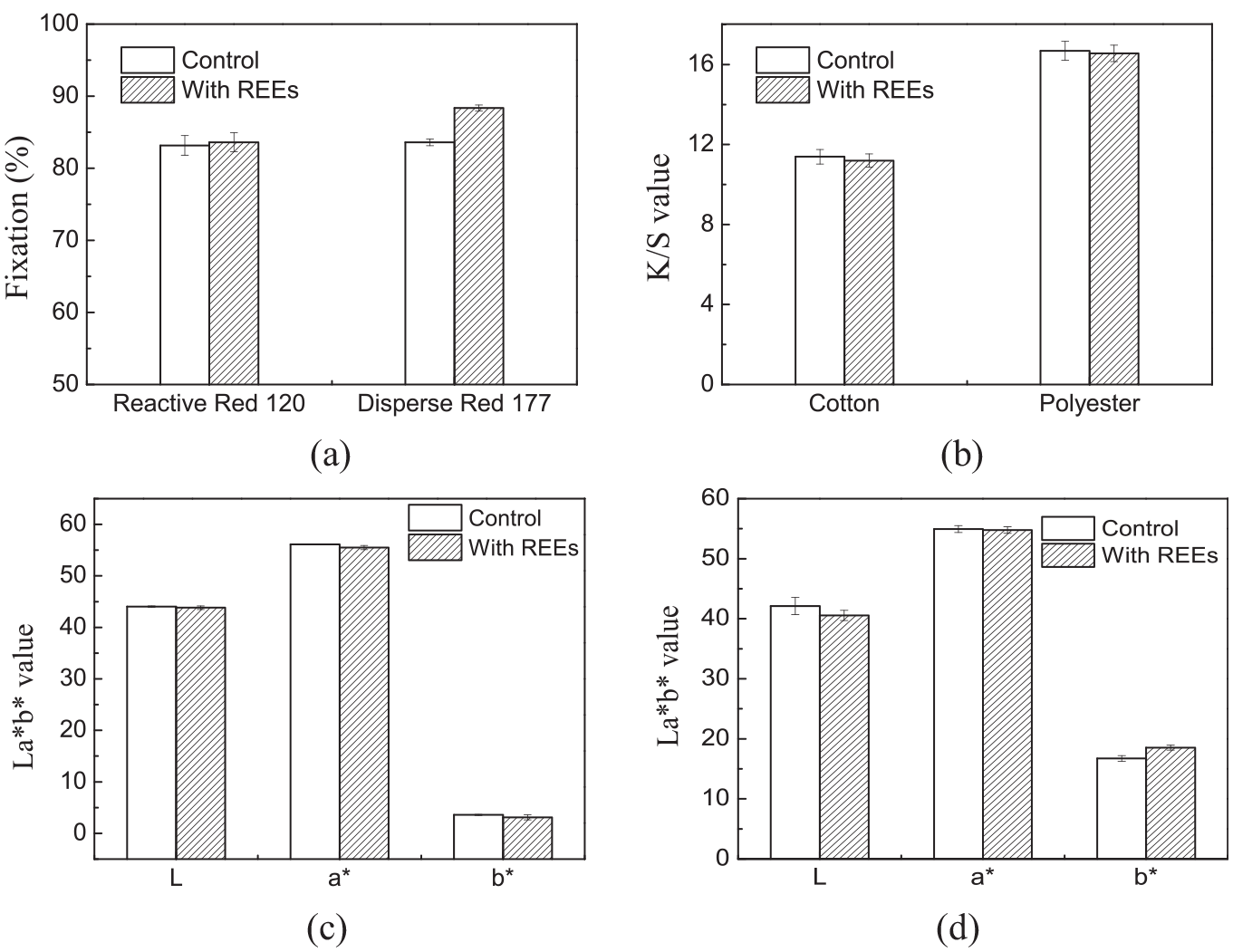
Effect of REE finishing on (a) fixation of dye; (b) K/S value (b); (c) La*b* of cotton; and (d) La*b* of polyester.
Supplemental Tables SII and SIII show the rubbing and washing fastness of the dyed fabrics before and after treating with REEs. Taking cotton fabric as an example, the rubbing and washing fastness of the dyed fabric was increased from 4–5 to 5 after rare earth treatment, which proved that the fabric fastness was slightly improved after treating with an REE. For polyester fabric, its dry rubbing fastness was not changed, but the wet rubbing fastness was improved by half a grade, and the washing fastness was also improved by half a grade. Therefore, REEs had no obvious effect on the fastness and strength of the fabric.
Analysis of REES Dyeing Mechanism
Effect of REEs on Dye Aggregation
Based on the particle size data displayed in Supplemental Figure S3, the particle sizes of reactive red 120 and disperse red 177 were 194 and 143 nm without REEs, respectively. If 10 mg/kg of REEs was added to the dye solution, the particle size of reactive red 120 was increased to 214 nm, but the particle size of disperse red 177 changed little (149 nm).
The FTIR spectra of reactive red 120 dye and color lakes derived from the interaction of REE with reactive red 120 are shown in Supplemental Figure S4a. After formation, the dye absorption of color lakes was shifted from 1036 to 1038 cm−1 and from 1180 to 1155 cm−1 respectively, indicating that the reaction between rare earth ions and sulfonate ions had been changed in the structure of sulfonate anions. The dye absorption at 1319 cm−1 was shifted to 1405 cm−1, which proved that the rare earth ion had a coordination effect with the –OH in the ortho position of azo group. 29 Furthermore, the dye absorption at 1612 cm−1 was shifted to 1589 cm−1, which explained that rare earth ions had an effect on the azo group on the dye molecular structure.
To investigate the influence of REEs on the aggregation of reactive dyes, the absorbance of dye solution was measured before and after adding REEs. As shown in Supplemental Figure S4b, the color depth of dye solution with REEs was clearer. The absorbance of reactive red 120 pure solution (0.52) was greater than that of dye solution (0.11) with REEs. Because the REE compounds were dissolved in water, they were ionized into rare earth ions with positive trivalent. The hydroxyl, amino, and other groups in the molecular structure of dyes contain lone electron pairs, which can form ionic bonds with rare earth ions. The electron cloud was deformed under the action of gravity, resulting in the formation of macromolecular complexes between rare earth ions and dyes. This complex enabled the electrons in the dye molecules to be excited as long as they received lower-energy radiation, increasing the wavelength of the absorbed light and the absorption coefficient, and the color became darker. 30
Elemental Analysis on Fabrics Dyed with REEs
In order to analyze the change in chemical environment before and after adding rare earth during dyeing, XPS was used to analyze the fabric chemical environment. As shown in Supplemental Figure S6, compared with the full XPS spectra of blank cellulose fabric, cotton fabric and polyester fabric showed different electronic peaks around 1072 eV after dyeing with REEs. According to the electron peak of the element, this peak is the electron peak of Sm. 31
Supplemental Table SIV showed the changes in element content on the fabric surface before and after dyeing with REEs. The XPS spectra of cotton without REEs contained C, O, and Cl, whose contents were 67.66%, 31.07%, and 0.27%, respectively. With the addition of REEs, C, O, Cl, and Sm content on the fabric surface were 73.72%, 24.52%, 1.54%, and 0.22%. The XPS spectra of polyester without REEs contained C, O, and Cl, which contents were 71.45%, 27.24%, and 1.31%, respectively. With the addition of REEs, C, O, Cl, and Sm content on the fabric surface were 73.96%, 23.73%, 1.84%, and 0.47%. After dyeing with REEs, the content of C element increased but the content of O element decreased. The reason may be that the positively charged rare earth ions easily bind to the negatively charged fabric. Rare earth cations can combine with hydroxyl groups of cotton fiber molecular structure, increasing the dye uptake and the content of C element on fabric surface.
The carbon atoms in the fabric can be divided into three binding forms (C1s1, C1s2, C1s3), and these three forms are represented by three corresponding forms in the XPS spectrum of C1s. The energy spectrum peak C1s1 is the connection between carbon atom and carbon atom, carbon atom, and hydrogen atom (C–C, C–H), and its electron binding energy is relatively low, about 284.60 eV. C1s2 is a carbon atom connected with an oxygen atom of a hydroxyl group(C–OH). The energy spectrum position is about 286.22 eV. C1s3 is a carbon atom connected with two oxygen atoms (O–C–O), which has a higher electron binding energy because of the higher oxidation state. The energy spectrum position of C1s3 is about 288.10 eV. As shown in Figure 3, the chromatographic peak height of the C–H or C–C of the fiber was increased after adding REEs during dyeing. The peak height of C–OH and O–C–O was decreased. For fabric dyed with REEs (Figure 3(b) and (d)), rare earth cations might combine with hydroxyl groups on the fiber, resulting in the content of C–OH being decreased. The content of C1s is relatively high in the molecular structure of dyes. The dye uptake was slightly increased after adding REEs. Therefore, the signal strength C1s2 and C1s3 peak became very weak, but the C1s1 was stronger after adding REEs.
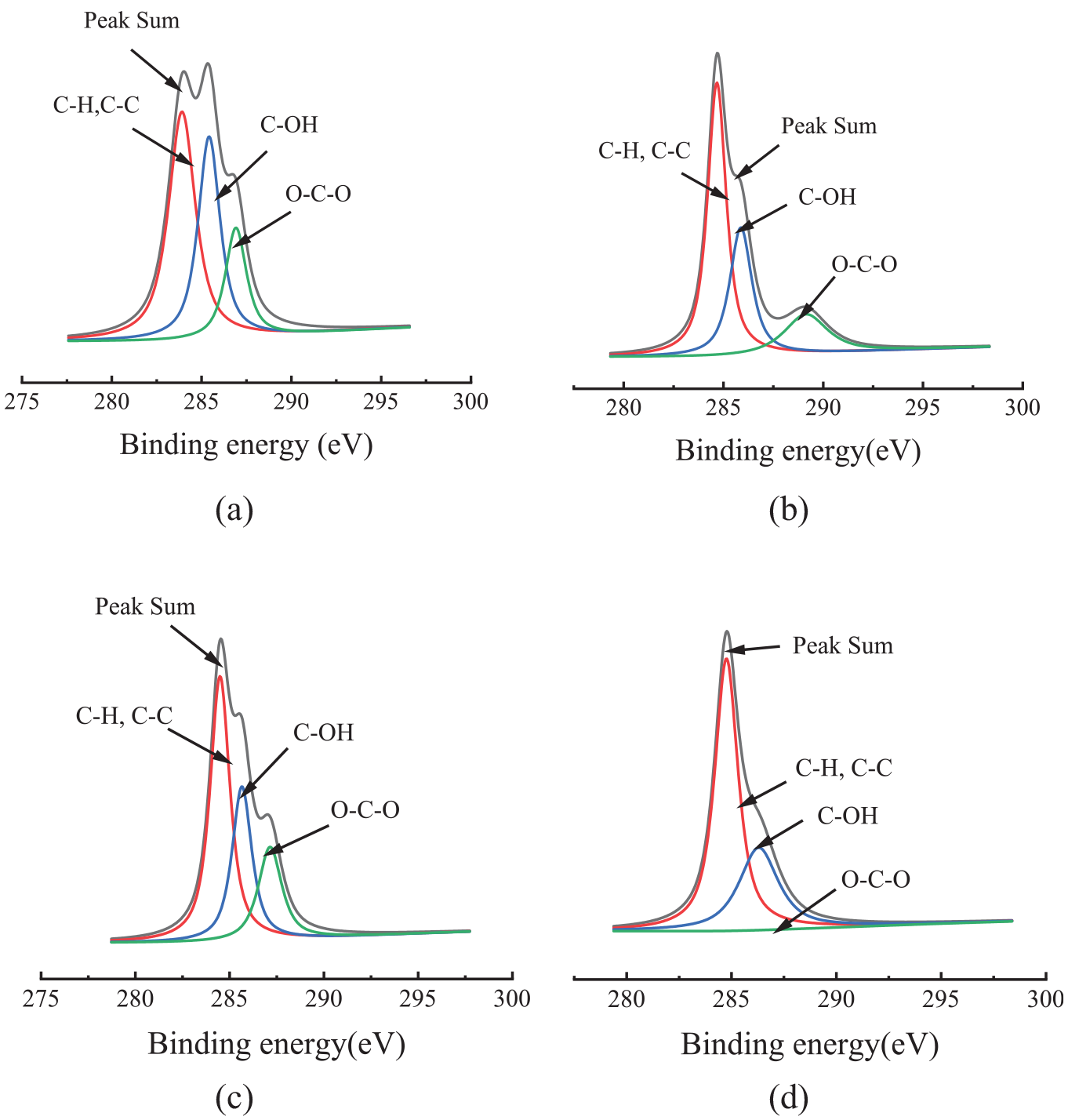
XPS peak spectrum of adding rare earth dyed fabrics: blank cotton (a); cotton dyed with red 120 and REEs (b); blank polyester (c); and polyester dyed with red 177 and REEs (d).
Conclusion
In this work, the traceability technology of rare earth-labeled non-aqueous medium products was studied. Nd, Eu, and Sm are suitable for tracing cotton fabrics. La, Ce, Nd, Eu, and Sm are suitable for tracing polyester fabrics. The marked products were detected by ICP-OES, which was clearly distinguishable from unmarked products. The method of ICP-OES had low MDL in the range of 0.0204–0.036 mg/kg, and good RSD values in the range of 0.69–2.43%. The content of REEs in dyed fabrics was detected by ICP-OES at 6–7 mg/kg when 10 mg/kg of REEs were employed during dyeing. After simulating multiple processes in textile processing, the rare earth content can be stabilized at 3 mg/kg, which was still significantly different from water bath products. From the washing, rubbing fastness, and breaking strength of dyed fabrics, the traceability method does not affect the basic properties of dyed fabrics. There was ionic bond between rare earth and dye to form macromolecular complex, which could influence the chemical environment of the dyed fabrics and the solubility of dyes. This paper studies the entire production process from the source, and a set of testing and certification methods for non-aqueous dyeing products was established. The anti-counterfeiting traceability of clean dyeing products has long-term significance for the development of dyeing industry.
Supplemental Material
sj-docx-1-aat-10.1177_24723444231206855 – Supplemental material for Application of Rare Earth Marking on Anti-counterfeiting Waterless/Less-Water Dyeing Technology
Supplemental material, sj-docx-1-aat-10.1177_24723444231206855 for Application of Rare Earth Marking on Anti-counterfeiting Waterless/Less-Water Dyeing Technology by Jingru Chen, Liujun Pei, Wenhua Shi, Muhammad Asad Saleem, Chenjia Jiao, Hongjuan Zhang and Jiping Wang in AATCC Journal of Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (22072089), the Key Technology Research and Development Project of Anhui Province (2023t07020001), the Industrial and Agricultural Project of Haining Science and Technology (2021003), and the Opening Project of Key Laboratory of Clean Dyeing and Finishing Technology of Zhejiang Province (QJRZ2301).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
