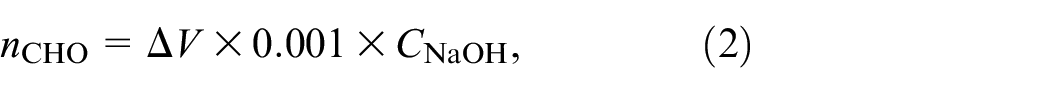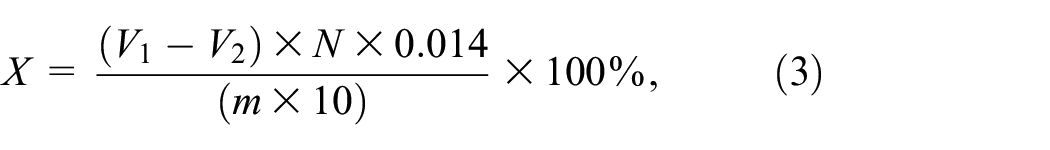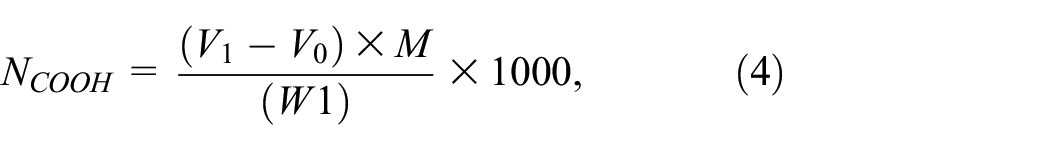Abstract
In delayed-curing and non-iron finishing, the sensitized fabric after low-temperature drying should have good surface planeness before curing at high temperatures, which implies that the finishing agent has not reacted with the cellulose fibers during storage; therefore, a suitable non-iron finishing agent is essential for achieving the above objectives. Meanwhile, there are currently no mature and efficient evaluation methods for the storage stability of sensitized fabrics. In this article, the content of characteristic groups of the sensitized fabrics with different storage conditions was measured. The storage time can be shortened from 30 to 6 days with high-temperature storage (40–60°C). The reaction kinetics equation was established under high-temperature storage; then using this equation to predict the cross-linking degree of sensitized fabrics during long-term storage at low temperature, the storage stability of sensitized fabrics can also be evaluated. The relative error of 2.5% between the predicted value and the measured value of the reaction degree showed the accuracy and effectiveness of the established method. This article provided a basis for the stability evaluation of the sensitized fabric and the selection of the finishing agent, finishing process, and storage conditions for the delayed-curing and non-ironing finishing process.
Keywords
Introduction
Cotton fabrics have excellent wearing properties; however, wrinkle will occur during wearing and washing, which reduces the application value of the fabric. Anti-wrinkle (non-iron) finishing is necessary for the reduction of the sliding between cotton fiber molecular chains, improvement of the fiber resilience, and the shape and size stabilization of the cotton fabric during washing and wearing.1–3 This process includes the impregnation of cotton fabric with a finishing solution composed of cross-linking agents (non-iron finishing agents), catalysts, and so on, and cross-linking occurs between cellulose and the finishing agent under high-temperature curing. However, the flat planeness of the cotton fabric from conventional non-iron finishing has an effect on its actual application. To avoid this short coming, the delayed-curing non-iron process was invented including the dry procedure of the fabric (called a sensitized fabric) without cross-linking reaction by padding the non-iron finishing solution at low temperature, and high-temperature curing and cross-linking when the garment is made for the ordered arrangement of the pleats and creases within the garment so that they are not easily deformed during wearing and washing.
Therefore, the main difference between the delayed-curing non-iron finishing and the conventional wrinkle finishing was the drying and curing finishing from the former.4–7 In detail, the fabric was first padded with non-iron finishing agents, dried at low temperature, and then made into garments. The cross-linking reaction between finishing agents and garment was carried out under the high-temperature curing; the washable folds, creases, and surface smoothness were obtained; and the shape and ease of care properties of the garment improved. However, the sensitized fabrics with the traditional non-iron finishing agent and catalytic system had poor storage stability due to the cross-linking reaction before the garment was cured, which affected the shape and surface smoothness of ready-made garments, and made it difficult to promote this technology.8,9
Therefore, the high storage stability of sensitized fabrics was the key to the delayed-curing finishing process, and quick and accurate evaluation of the storage stability of sensitized fabrics had great significance to realize the industrialization of the delayed-curing finishing. At present, the storage stability of the sensitized fabrics was evaluated by detecting the degree of cross-linking between the finishing agent and cotton fibers, which mainly included enzymatic degradation method, sulfuric acid hydrolysis method, dyeing method, infrared spectroscopy, high-performance liquid chromatography, and so on. These methods were mostly qualitative or semi-quantitative when evaluating the degree of cross-linking between finishing agents and cotton fibers,10–13 which resulted in low accuracy and efficiency. In addition, storage stability evaluation was performed by measuring changes in mechanical properties such as strength and wrinkle recovery angle (WRA) of the sensitized fabrics before and after storage;14,15 the changes in of properties such as WRA and the strength of the fabric before and after storage were measured after storage for 2–3 months.
In view of this situation, this article proposed a direct measure to evaluate the reaction degree (the storage stability) between the finishing agent and cotton fibers during the storage of sensitized fabrics at different temperatures and times (the higher storage temperature (40–60°C) can reduce the storage time from 30 to 6 days) by establishing the reaction kinetic equation between the finishing agent and the cotton fiber, testing the reaction rate constant (k) under different storage temperatures, and further proposing the Arrhenius formula of the relationship between the temperature (T) and the reaction rate constant on this basis. Then the reaction rate constant was calculated at the conventional storage temperature (20°C) for evaluating the stability of the sensitized fabrics prepared by the non-iron finishing agent under the storage conditions. This work provided a basis for the selection of non-iron finishing agents, the finishing process, and storage conditions for the delayed-curing process.
Experimental Section
Materials and Chemicals
Cotton fabric (100%) with 133 warp yarns per 2.54 cm and 100 wefts per 2.54 cm, weighing 133 g/m2, was desized and scoured by the supplier. Two-dimensional (2D) resin (DM-3511, DMeDHEU) and silicone smoothing agent (JF-5097) were purchased from Guangdong Demei Fine Chemical Co., Ltd. Trehalose·2H2O (Tr), catalysis agent (MgCl2·6H2O), concentrated sulfuric acid, copper sulfate, potassium sulfate, boric acid, methyl red, bromocresol green, ethanol, hydrochloric acid (HCl), sodium hypophosphite (NaH2PO2), hydroxylamine hydrochloride, calcium acetate (Ca(CH3COO)2·H2O), and sodium hydroxide (NaOH) were purchased from Sinopharm Chemical Reagent Co., Ltd.
Synthesis of Carboxylated Polyaldehyde Trehalose
Carboxylated Polyaldehyde Trehalose (CPTr) was synthesized by two steps of selective oxidation. (1) The first step was the TEMPO-mediated oxidation of cellulose as previously reported. 16 Briefly, Tr (0.91 g, 4 mmol) was dissolved in acetic acid–sodium acetate buffer (200 mL, pH 4.5), TEMPO (8 mg, 0.052 mmol) was added to the reaction solution at 35°C, and the reaction system was placed in a constant temperature magnetic water bath and stirred for 5 h. After the reaction, 95% ethanol was added to stop the reaction, and the pH value was adjusted to 7.0 with 0.5 N HCl. The 6-carboxy trehalose (6-CTr) was kept at −20°C, and then dried by lyophilization to obtain the powder.
(2) The second step was based on the NaIO4 selective oxidation procedure in aqueous as reported with some modifications. 6-CTr (5.4 g) was dissolved in water (200 mL) and stirred with nitrogen (N2) for 0.5 h to remove the excess O2. At 15°C, 6.7 g NaIO4 was added to the dissolved 6-CTr, which was stirred for an additional 8 h. Barium dichloride (3.9 g) was added to the solution, and stirred for 0.5 h at 10°C. Then, CPTr was obtained in the filtrate after filtering the precipitation. The CPTr powder was prepared by the freeze-drying method.
Structure Characterization of CPTr
Fourier transform infrared (FTIR) spectroscopic analyses of Tr, 6-CTr, and CPTr were measured via Iraffinity-1S (Shimadzu, Japan) using the KBr method performed with 32 scans per sample with a scan range of 4000–400 cm−1 with 4 cm−1 resolution.
13C NMR (nuclear magnetic resonance) spectra of Tr, 6-CTr, and CPTr were characterized and analyzed on a BrukerAM-400 spectrometer; 0.05 g of the sample was dissolved in 0.5 mL of deuterium water (D2O) before the test.
Finishing Process of Delayed-Cure Sensitized Fabrics
Finishing solution of CPTr. CPTr (100 g/L), MgCl2·6H2O (20 g/L), and NaH2PO2 (10 g/L) were combined to form the finishing solution.
Finishing solution of DMeDHEU. DMeDHEU (150 g/L) and MgCl2·6H2O (30 g/L) were combined to form the finishing solution, and silicone smoothing agent (JF-5097) 5 g/L added.
Finishing solution of 1,2,3,4- butane tetracarboxylic acid (BTCA). BTCA (150 g/L) and NaH2PO2 (10 g/L) were combined to form the finishing solution.
Finishing process. The fabric was first padded using a two-bowl horizontal laboratory padder to attain a wet pickup of 90–100%. Then, impregnated fabrics were dried at 60°C (the wet pickup was controlled to 5–8%). Finally, the fabrics were stored in the sealed bag at the corresponding temperature (CPTr was 150°C, 2 min, DMeDHEU was 120°C, 2 min, and BTCA was 180°C, 2 min).
Properties of Treated Fabrics
According to the test standard, treated fabrics were stored under conditions at a relative temperature of 25 ± 1°C and a relative humidity of 65 ± 2% for 4 h before the measurement. The WRA of the treated fabrics was measured according to AATCC Test Method 66-2003. Each sample was measured five times in the weft (f) and warp (w), and the average value of each group was taken. The crease retention was measured according to AATCC Test Method 88C. The whiteness index (WI) of the treated fabrics was determined by AATCC Test Method 110-2005. The Datacolor 650® benchtop spectrophotometer was used to test four different positions, and the average of every sample was calculated. Tensile strength (TS) of the treated fabrics was determined with ASTM Testing Method D-1424-1996, and calculated using equation (1):
where Tt is the TS (cN) of treated fabric and Tu Is the TS (cN) of untreated fabric.
Analysis of the Cross-linking Degree (Characteristic Group Method)
Aldehyde Group Content of Sensitized Fabric (Hydroxylamine Hydrochloride Method)
The fabric sample was cut into pieces, cured in an oven at 80–90°C to constant quality, and transferred to a desiccator for cooling before use; 0.10 g of the sample was accurately weighed and mixed with 25 mL 0.25 N hydroxylamine hydrochloride-methyl/orange solution. After standing for 2 h, the mixed solution was titrated with a 0.1 N sodium hydroxide solution until the solution turned from red to yellow, and the titration was continued until the pH value of the solution was 4. The untreated sample was used as a blank test, and the experiment was repeated three times. The aldehyde group content (mmol/g fabric) of untreated cotton fabrics was negligible and was calculated according to equation (2):
where ΔV is the volume of NaOH consumed (mL), CNaOH is the concentration of NaOH (mol/L), and nCHO is the aldehyde group content in the sample (mmol/g).
Nitrogen Content of Sensitized Fabric (Kjeldahl Method)
The samples were ultrasonically washed with deionized water of pH = 10 for 20 min (to remove the unreacted 2D resin), dried naturally, and equilibrated in a standard environment for 24 h. 2
The nitrogen content of the finished fabric was determined according to the procedure of Ma et al. with slight modification; 0.5 g sample was accurately weighed, cut into pieces, and transferred into 500 mL nitrogen bottle; 0.2 g copper sulfate, 6 g potassium sulfate, and 20 mL concentrated sulfuric acid were added to the bottle and transferred to the nitrification furnace for nitration. After the sample was completely nitrated, the solution was blue-green and continued heated for 0.5 h. The nitrogen bottle was removed, transferred to the Kjeldahl nitrogen analyzer for distillation after cooling, and the produced gas was absorbed with 0.2% boric acid. After the distillation was complete, the solution was titrated with a 0.1 mol/L hydrochloric acid standard solution (the indicator was 1 part of 0.1% methyl red ethanol solution and 5 parts of 0.1% bromocresol green ethanol solution for temporary use), and the nitrogen content of the sample was calculated according to equation (3). The result was the average value from three parallel experiments, the calculation formula of nitrogen content (equation (3)):
where X is the combined nitrogen content of the sample (%); V1 is the consumed volume of the hydrochloric acid standard solution of the sample (mL); V2 is the volume of sulfuric acid or hydrochloric acid standard solution consumed by the reagent blank (mL). N is the equivalent concentration of the standard hydrochloric acid solution. 0.014 is the ratio of 1 mL 1N sulfuric acid. M is the mass of the sample (g).
Carboxyl Group Content of Sensitized Fabric (Calcium Acetate Titration Method)
The fabric sample was cut into pieces, cured in an oven at 80–90°C to constant quality, and transferred to a desiccator for cooling before use.17,18
The dried sample(0.6 g) was accurately weighed, and put into a 250 mL iodine volumetric flask, 50 mL of the freshly prepared 0.1 mol/L calcium acetate solution were added, and the mixture was left at room temperature for 2 h with an indicator of 0.02 mol/L phenolphthalein. The replaced hydrogen ions were titrated with
where V1 is the volume of sodium hydroxide standard solution consumed by titration (mL), V0 is the volume of sodium hydroxide standard solution consumed by 50 mL of calcium acetate solution (mL), M is the concentration of sodium hydroxide standard solution (mol/L), and W1 is the mass of the sample (g).
Results and Discussion
Preparation and Characterization of CPTr
The oxidation mechanism of the TEMPO-laccase system and NaIO4 system, and the reaction mechanism of the two-step oxidation of trehalose to prepare CPTr are shown in Figure 1(a). In the first oxidation reaction, TEMPO was generated to the corresponding TEMPO+ under the oxidation of laccase and oxidized the primary hydroxyl group in the trehalose structure to an aldehyde group, which was then oxidized to the carboxyl group to obtain carboxylated trehalose (6-CTr). In the second oxidation reaction, periodate was formed by the cyclic complex with the adjacent hydroxyl (-OH) of trehalose, then the epoxidation of glucose cracked the glucose ring to form two aldehyde groups. After selective oxidation reaction, the trehalose polyuronic acid (CPTr) was prepared.
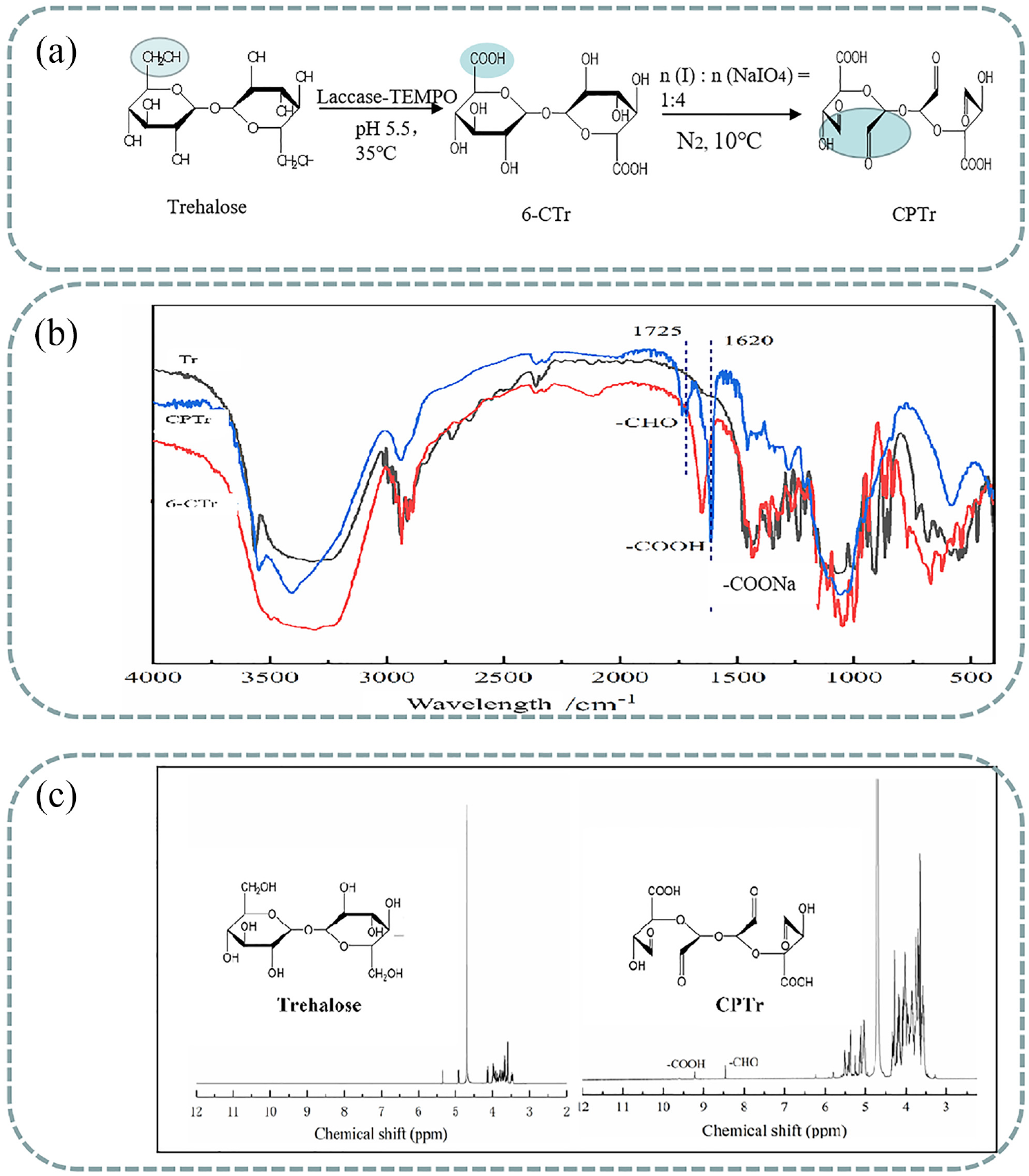
(a) Scheme of the TEMPO-NaIO4 system oxidation of trehalose to prepare CPTr; (b, c) FTIR, 1H NMR spectra of trehalose and CPTr.
In Figure 1(b), the characteristic absorption peak at 1620 cm−1 was attributed to the symmetrical and asymmetric stretching vibrations of the carboxyl group, generated by the primary hydroxyl group in trehalose after carboxylation. The trehalose had no absorption peak at this band, indicating that the primary hydroxyl groups of the trehalose molecule were selectively oxidized to carboxyl groups. The aldehyde group absorption band of CPTr appeared at 1725 cm−1, indicating that the hydroxyl group in the glucose of 6-CTr was selectively oxidized to the aldehyde group by NaIO4. 17 Those changes of characteristic peaks indicated that CPTr was prepared via the two step selective oxidation with the TEMPO-laccase system and NaIO4 system.
Based on the FT-IR results of Tr, 6-CTr, and CPTr, in order to further analyze the hydroxyl structure changes of Tr with two-step selective oxidation, the 1H NMR spectra of Tr, 6-CTr, and CPTr were evaluated and are shown in Figure 1(c). Compared with the 1H NMR spectra of trehalose and CPTr, the characteristic peak at 8.45 ppm was attributed to the characteristic chemical shift of the aldehyde group (-CHO) after selective oxidation of the NaIO4 system, and the characteristic peak at 9.15 ppm was assigned to the characteristic chemical shifts of the carboxyl group (-COONa) generated by the selective oxidation of the TEMPO system, which also indicated that the TEMPO-sodium periodate system was feasible to prepare CPTr.
Systematic Analysis of the Storage Stability of Sensitized Fabrics
Formaldehyde-free finishing agents suitable for delayed-curing non-iron finishing should meet the following requirements: (1) the storage stability of sensitized fabrics was high, and the finishing agents were not prone to cross-linking with cotton fabrics during storage; and (2) high-temperature curing of the non-iron finishing agent had high reactivity with cotton fabrics, and the cross-linking reaction was sufficient, giving the fabric excellent anti-wrinkle performance. Therefore, to evaluate whether CPTr was used for delayed-curing non-iron finishing of cotton fabrics, experiments and analysis were carried out from the above two aspects.
The sensitized fabrics finished with CPTr were stored at different temperatures (40–60°C) for different times. The stored fabric was cured at corresponding temperature (CPTR was 150°C, 2 min, DMeDHEU was 120°C, 2 min, and BTCA was 180°C, 2 min). The cloth surface planeness, pleat planeness, the WRA, strength retention rate, and whiteness of the sensitized fabrics were tested, and are shown in Table 1.
The properties of sensitized fabrics before and after cuing.
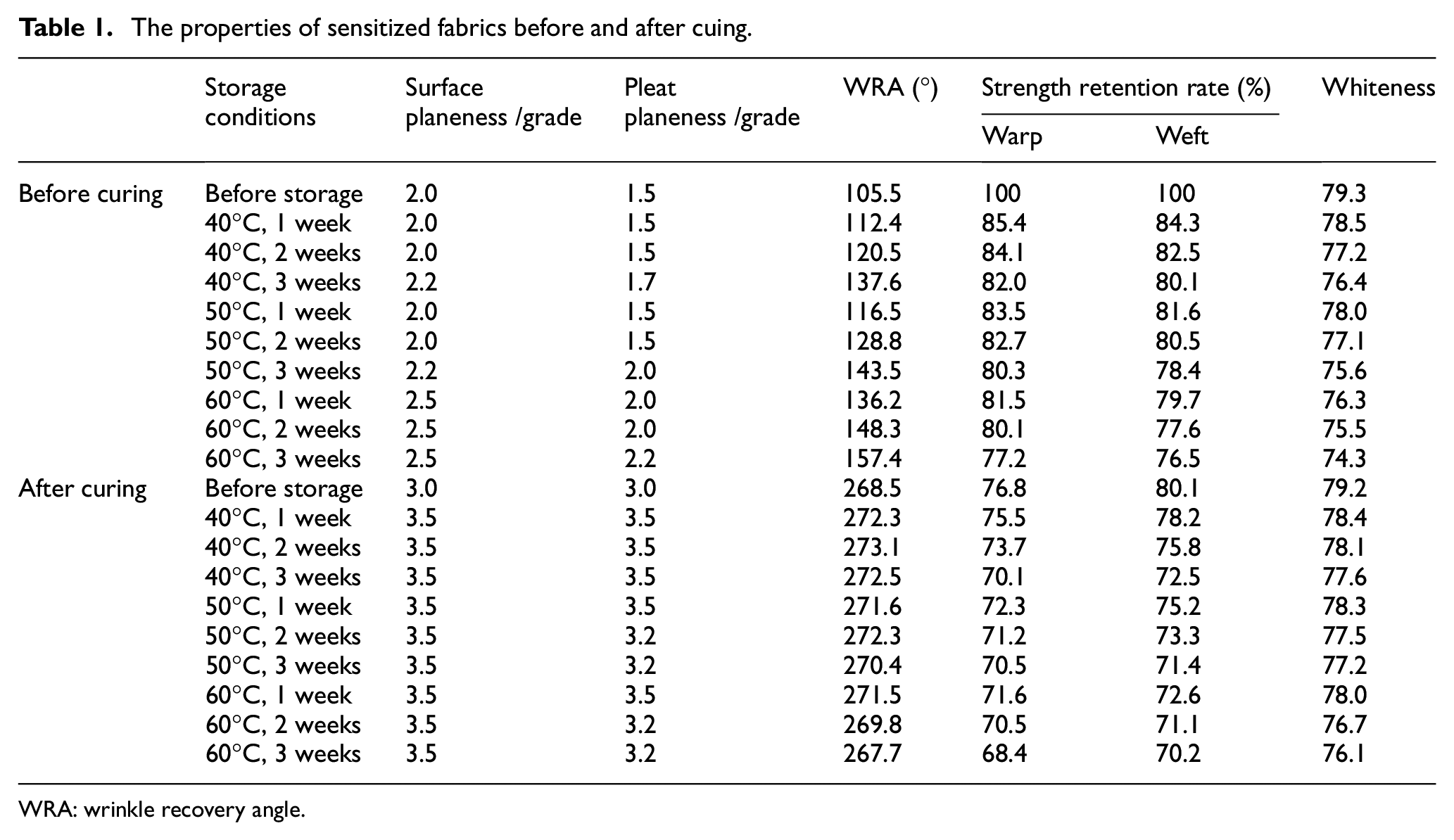
WRA: wrinkle recovery angle.
Before curing, with the prolongation of storage temperature and time, the WRA of the sensitized fabric was increased, the change of whiteness was not obvious, the breaking strength retention rate of the fabric was reduced, while the strength retention rate of the finished fabric was still maintained at about 75% (not exceeding the range required by this experiment, and the strength damage was less than or equal to 35%). When CPTr-treated fabrics were stored at 40°C, 50°C, and 60°C, the WRA of the fabric increased with the storage time, and increased by 32.1°, 38°, and 51.9° after storage 3 weeks, indicating that the aldehyde group of CPTr had a partial cross-linking reaction with the hydroxyl group of cellulose during the storage. The overall analysis showed that when CPTr-sensitized fabrics were stored at storage temperatures of 40°C, 50°C, and 60°C for 1–2 weeks, the effect of WRA and strength retention rate of the fabric had a small change, which accorded with the finishing requirements of delayed-curing and non-iron finishing cotton fabrics.
After curing, the surface planeness grade of the cured fabrics reached 3.5, and the pleat planeness grade of the fabrics also reached more than grade 3.2. The WRA of the fabric after curing exceeded 267°, and the breaking strength retention rate of fabrics with different storage conditions changed greatly. Under the same storage time, the strength of the fabric was changed greatly with the storage temperature increased, especially when the finished fabric was stored at 60°C for 3 weeks. Under the same storage conditions, the warp strength of the fabric was less reduced than the weft. In addition, the whiteness of the sensitized fabric was decreased from 79.3 to 76.1 after curing, which was a small change. Therefore, CPTr could be fully cross-linked with cellulose, given the fabric’s better anti-wrinkle properties after the curing process, and was suitable for delayed-curing and non-iron finishing of cotton fabrics.
Evaluation Method of Cross-linking Degree (Content Change of Characteristic Group)
The Arrhenius equation was an empirical formula for the relationship between the chemical reaction rate constants and the temperature established by Arrhenius of Sweden. The premise of the Arrhenius empirical formula was that the activation energy Ea was a constant, which was independent of temperature consistent with the experimental results within a certain temperature range.19–22 The cross-linking methods of CPTr, DMeDHEU, BTCA, and cellulose at different temperatures were consistent (the cross-linking mechanisms of three finishing agents are shown in Figure 2), so the Arrhenius formula was suitable for this cross-linking reaction.
(a) Calculation of the Rate Constant of Reaction. The cross-linking reaction between the finishing agents and cotton fabrics was characterized as a pseudo-first-order reaction. The reaction rate was dependent on the reactant concentration and temperature. Under constant-temperature conditions, the equation was as follows:

where r is the reaction rate; k is the reaction rate constant; b1 is the concentration of DMeDHEU, CPTr, or BTCA; b2 is the catalyst concentration; b3 is the concentration of hydroxyl groups in the fiber; and a, c, and d are the reaction order. During delayed curing, the catalyst concentration remained unchanged before and after finishing, the fiber hydroxyl concentration remained almost unchanged, the reaction order was 1, and equation (5) was simplified to equation (6):

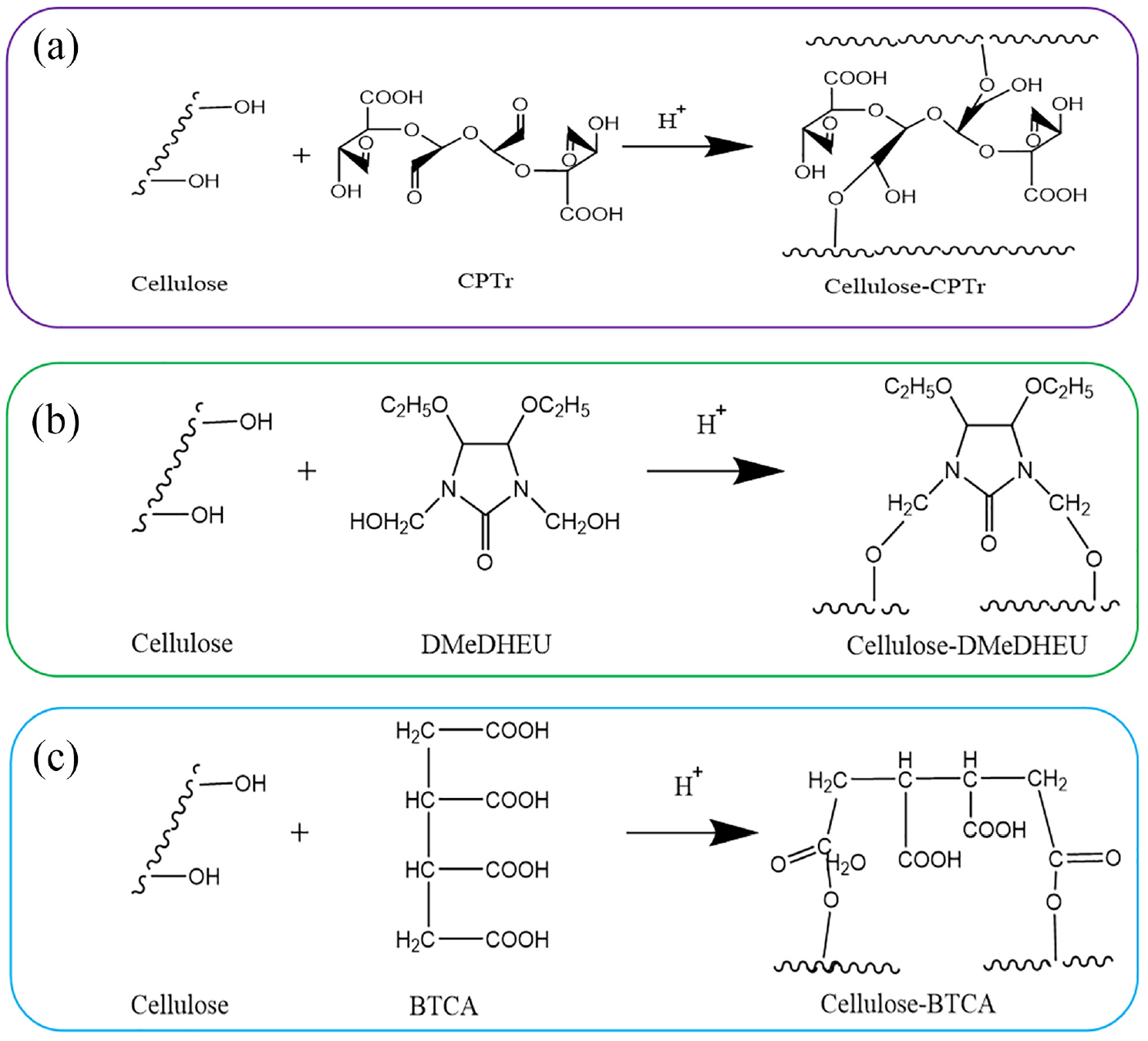
Cross-linking mechanisms of CPTr (a), DMeDHEU (b), and BTCA (c) with cellulose.
Equation (6) variables were separated and integrated into equation (7):

where b0 is the mass concentration at the beginning of the reaction (t = 0), and b1 is the concentration of the finishing agent after storage for time t.
The concentration change of the finishing agent during the reaction kinetics study was represented by the change in characteristic group content in the fabric (nitrogen content of DMeDHEU finished fabric, aldehyde content of CPTr-finished fabric, and carboxyl content of BTCA-finished fabric). b0 and b1 changed by Nm and Nm – N (Nm is the characteristic group content of the fabric after padding, drying, and curing; N is the characteristic group content after storage time t). Therefore, equation (7) was changed to equation (8):
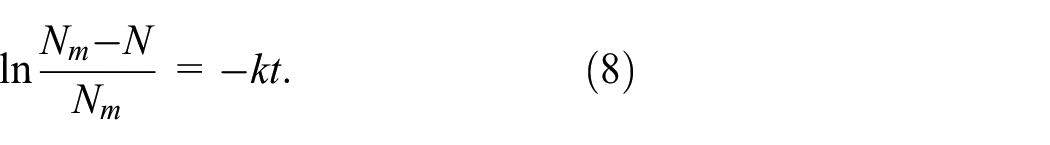
(b) Calculation of Activation Energy. The relationship between reaction rate and temperature was given by the Arrhenius formula, which was expressed as equation (9):
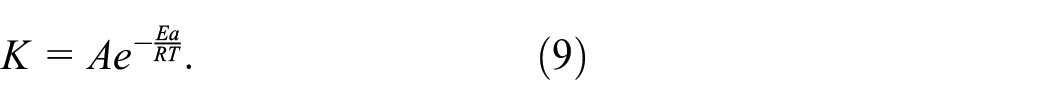
The integral equation of equation (9) was as follows:
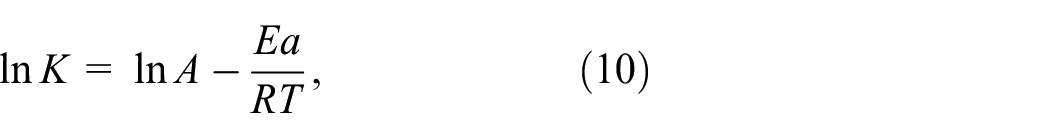
where K is the reaction rate constant, A is the pre-factor (which was also termed the frequency factor), Ea is the apparent activation energy, R is the molar gas constant (8.314 J/(mol K)), and T is the absolute temperature (K).
From equation (8), the reaction rate constant (k), the values of Ea and A were calculated by measuring the characteristic group content of the fabric, which was substituted into equation (9). The reaction rate constant of the characteristic group content at any temperature was calculated, and compared with the actual measured rate constant of characteristic group content, and the applicability of the Kjeldahl method to evaluate the delayed-cure stability was analyzed.
Kinetic Equation Analysis of Storage Time Reaction of Three Finishing Agents
From equation (7), the characteristic group contents at different storage conditions were measured, and are shown in Table 2. The relationship between ln(Nm – N)/Nm and the storage time was analyzed, and then a linear fitting was performed. The results are shown in Figure 3(a) –(c).
Characteristic group contents with different storage conditions.
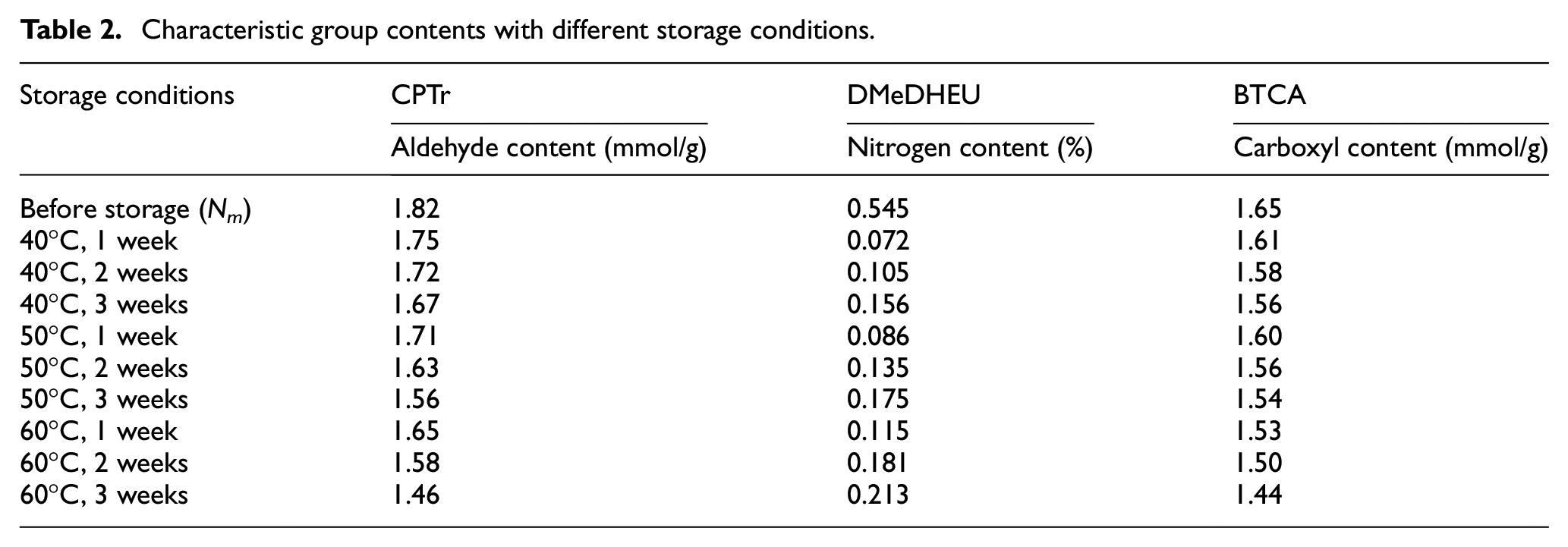
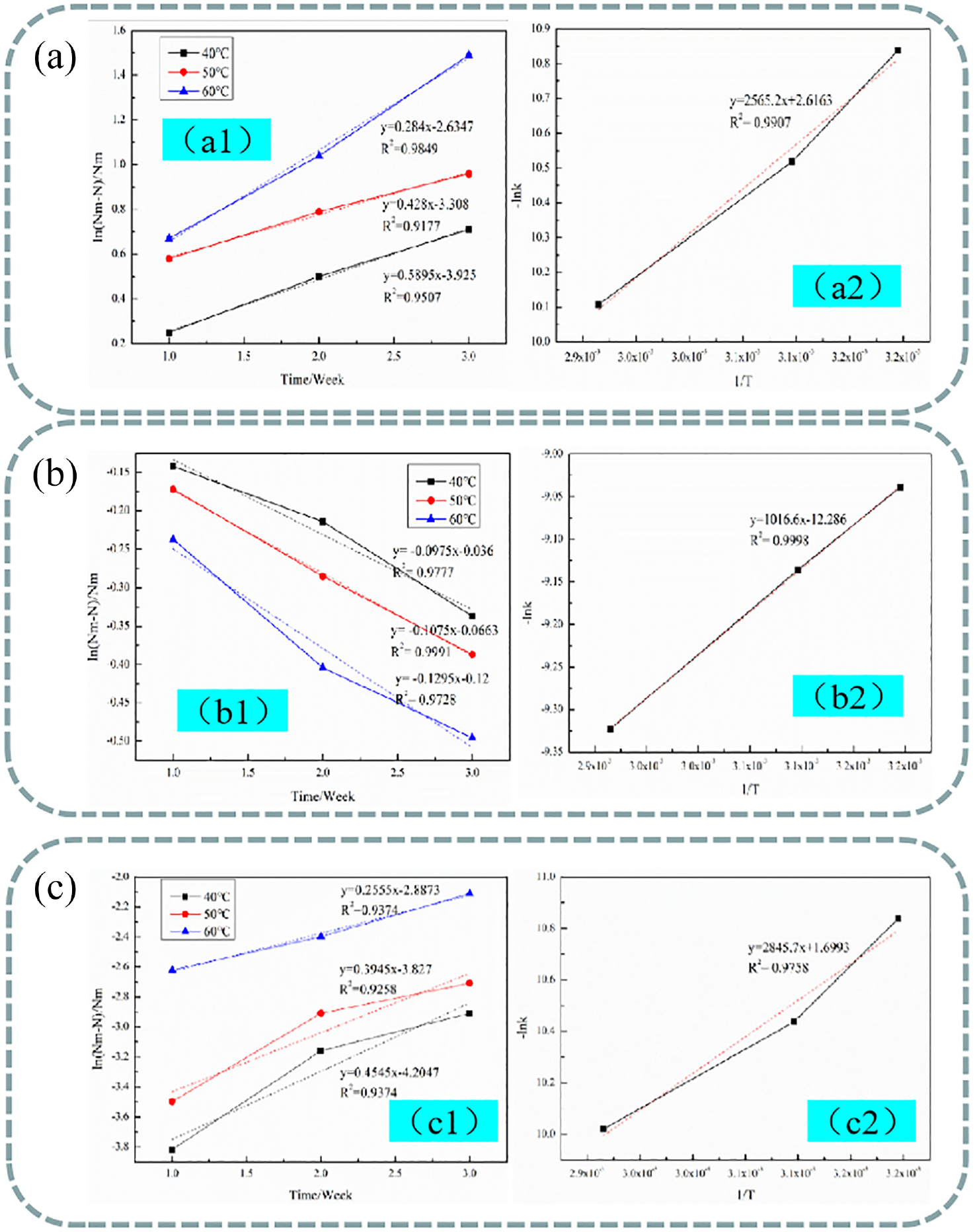
Cross-linking kinetic equation of CPTr (a), DMeDHEU (b), and BTCA (c).
In Table 2, with the storage temperature and storage time increased, the aldehyde group content of CPTr-finished fabrics showed a downward trend, and the carboxyl group contents of the BTCA-finished fabrics also gradually decreased. This was because aldehyde groups and the carboxyl groups were partially cross-linked with the cellulose during the storage process, resulting in the reduction of the contents of the finished fabric. During the storage process of DMeDHEU-finished fabrics, the nitrogen content of the fabrics tended to increase; because the DMeDHEU was gradually fixed in the fabrics with the progress of the cross-linking reaction, the DMeDHEU content of the fabrics was increased, which increased the amount the nitrogen content.
Figure 3(a1) shows the ln(Nm– N)/Nm was linear with storage time after storage at 40°C, 50°C, and 60°C. The fitting functions were: y = 0.284x – 2.6347, R2 = 0.9849; y = 0.428x – 3.308, R2 = 0.9177; y = 0.5895x – 3.925, R2 = 0.9507, where y is ln(Nm– N)/Nm and x is storage time. The slopes of the fitting functions were the reaction rates (k) under different storage conditions. Linear fitting verified that the cross-linking reaction between the CPTr and cotton fabric conformed to the pseudo-first-order reaction equation. The reaction rates (k) after storage at 40°C, 50°C, and 60°C were 0.284, 0.428, 0.5895, respectively. Because the units of k and t in equation (8) were (24 h)−1 and °C, when substituted into equation (10), the units needed to be converted to s−1 and thermodynamic temperature T. After the unit conversion, the reaction rate K and 1/T were analyzed, and the fitting equation in Figure 3(a2) was Y = 2565.2X+ 2.6163, R2 = 0.9907. Therefore, combined with the Arrhenius equation (9), the Ea of CPTr combined with cotton fabric under these process conditions was 21.327 kJ/mol.
Figure 3(b1) shows the ln(Nm– N)/Nm was linear with storage time after storage at 40°C, 50°C, and 60°C. The fitting functions were: y = −0.0975x – 0.036, R2 = 0.9777; y = −0.1075x – 0.0663, R2 = 0.9991; y = −0.1295x – 0.12, R2 = 0.9728. Linear fitting verified that the cross-linking reaction between the DMeDHEU and cotton fabric also conformed to the pseudo-first-order reaction equation. After the unit conversion, the reaction rate K and 1/T were analyzed, and the fitting equation in Figure 3(b2) was Y = 1016.6X – 12.286, R2 = 0.9998. Therefore, combined with the Arrhenius equation (9), the Ea of DMeDHEU combined with cotton fabric under these process conditions was 8.452 kJ/mol.
Figure 3(c1) shows the ln(Nm– N)/Nm was linear with storage time after storage at 40°C, 50°C, and 60°C. The fitting functions were: y = 0.2555x – 2.8873, R2 = 0.9374; y = 0.3945x – 3.827, R2 = 0.9258; y = 0.4545x – 4.2047, R2 = 0.9374. Linear fitting verified that the cross-linking reaction between the BTCA and cotton fabric also conformed to the pseudo-first-order reaction equation. After the unit conversion, the reaction rate K and 1/T were analyzed, and the fitting equation in Figure 3(c2) was Y = 2845.7X+ 1.6993, R2 = 0.9758. Therefore, combined with the Arrhenius equation (9), the Ea of BTCA combined with cotton fabric under these process conditions was 23.659 kJ/mol.
Storage Stability Compared with Other Finishing Agents
The reaction kinetic equation and Arrhenius formula were calculated under the temperature of sensitized fabric at 20°C; the degree of cross-linking of finished fabrics during storage is shown in Table 3. According to the fitting equation between the temperature (1/T) and reaction rate constant (K) in Figure 3(a) –(c), the calculated reaction rates K of finished cotton fabric treated with CPTr, DMeDHEU, and BTCA that were stored at 20°C were 1.014 (24 h)−1, −0.0781 (24 h)−1, 1.046 (24 h)−1, respectively. The measured aldehydes, nitrogen, and carboxyl content of the fabric after storage at 20°C for 5 days were 1.78 mmol/g, 0.0485%, and 1.63 mmol/g, respectively. By combining equation (7), the rate equation of 20°C was y = 1.014x+ 1.056, y = −0.0781x+ 0.0073, and y = 1.046x+ 0.883, where y is ln(Nm– N)/Nm and x is storage time.
Calculated values (CV) and measured values (MV) of cross-linking degree.
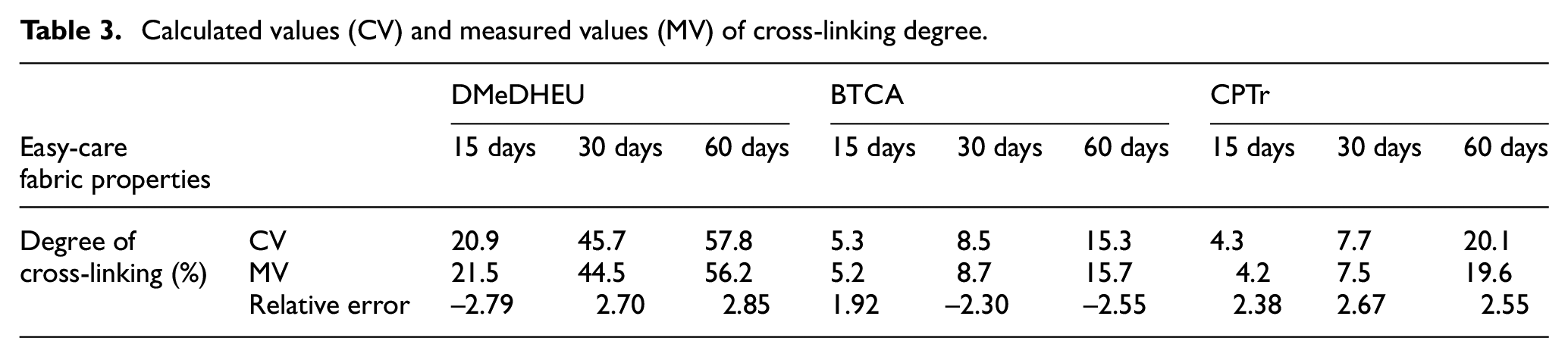
In Table 3, the Kjeldahl method for N-methylol resins (DMeDHEU) was used to determine the change of nitrogen content combined with the sensitized fabrics during storage, reflecting the degree of reaction between DMeDHEU and cotton fibers.15,23 According to the established kinetic equation of the reaction and the Arrhenius formula, the relative error between the predicted value and the measured value of the sensitized fabrics stored at 20°C from 15 to 60 days was less than ±2.9%, indicating that the kinetic equation had a high degree of good reliability. The sensitized fabric had been stored at 20°C for 30 days, and the reaction degree reached 44.5%. It was not a suitable delayed-curing and no-iron finishing agent because the active group of the finishing agent was N-methylol, which had strong reactivity with cotton fibers.
For the BTCA finishing agent, the change of the carboxylic acid group content of the sensitized fabric during storage used the calcium acetate titration method, reflecting the degree of reaction between BTCA and the cotton fibers.8,18,19 According to the established kinetic equation of the reaction, the relative error between the predicted value and the measured value of the reaction degree of sensitized fabrics stored at 20°C from 15 to 60 days was less than ±2.6%, indicating that the kinetic equation had a high degree of accuracy and good reliability. The reaction degree of sensitized fabric was 15.7% after being stored at 20°C for 60 days, which had good storage stability. It can be used as a delayed-curing and non-iron finishing agent because the activity of the active group of the finishing agent was low with the catalysis of sodium phosphate.
For the self-developed polyuronic acid finishing agent (CPTr), the change of the content of aldehyde groups during storage by the acetylacetone color development method can reflect the degree of reaction between CPTr and the cotton fibers. According to the established kinetic equation of the reaction between the finishing agent and cotton fiber, and the Arrhenius formula, the relative error between the predicted value and the measured value of the reaction degree of sensitized fabrics stored at 20°C from 15 to 60 days was <±2.7%, which indicated that the kinetic equation had a high degree of accuracy and good reliability. The sensitized fabric had good storage stability with the reaction degree of 19.6% after being stored at 20°C for 60 days. Then, CPTr was suitable for the delayed-curing and non-iron finishing; the active groups were the aldehyde group and carboxyl group.
Conclusion
In this article, the polyuronic acid finishing agent (CPTr) was prepared by selectively oxidizing trehalose with the TEMPO-laccase system and periodate system. The finished sensitized fabrics had high storage reactivity at room temperature, good stability, high reactivity, and excellent non-iron performance. The CPTr and multi-component composite catalytic system was also developed for this project technology. After the sensitized fabric was stored at 20°C for 60 days, the cross-linking degree of CPTr-finished fabrics was only 19.6%, far lower than 50% of the DMeDHEU-finished fabrics. The WRA of CPTr-finished fabrics after curing was 270°, and the strength retention rate was above 70%. After measuring the characteristic group content of the finishing agents under different storage conditions, the reaction kinetic equation and cross-linking degree of the sensitized fabric were established. The established method was accurate and efficient, and the error between the predicted value and the measured value of the reaction degree was about 2.7%, which provided a basis for the evaluation of the storage stability of sensitized fabrics and the selection of suitable delayed-curing non-iron finishing agents.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (21674043) and the China Postdoctoral Science Foundation (2021M701452).