Abstract
Cigarette butts are one of the most discarded and littered items in the world, with a total amount of around 5.6 trillion, posing a grave threat to the environment and human health. This research seeks to recover cellulose acetate fibers from used cigarette filters and to evaluate the yarn-making capabilities of these fibers. The cigarette butts were gathered from various locations, and the fibers and coating were separated. The fibers were then treated with hydrogen peroxide and sodium hydroxide to eliminate the burned color and odor. Acetone and distilled water were used to filter and distill the material properly. The color coordinates of the chemically altered fibers, including reflectance value and color strength, were measured afterward. To assess the chemical, thermal, physical, morphological, and mechanical characteristics of the modified cigarette butt filter fiber, atomic absorption spectroscopy, Fourier transform infrared spectrometer (FTIR), thermogravimetric analysis, X-ray diffraction, scanning electron microscopy, and tensile properties were examined. Significant and appealing findings were obtained for the mechanical parameters, particularly the tensile strength of 1.2 gm/den for a single fiber, the fiber density of 1.2 dtex, and the effective length of 25 mm, all of which indicate the fiber’s suitability for yarn production. In addition, FTIR, thermogravimetric analysis, and X-ray diffraction measurements revealed that the cigarette butt filter fiber possessed the same characteristics as commercial cellulose acetate fiber. The preliminary results obtained on the recovered cellulose acetate fiber are encouraging for the application of this recovery material from cigarette butts to create a high-demand and value-added product, such as yarn for garment production.
Introduction
Cigarette smoking is the most widely recognized type of tobacco utilized around the world. One of the most significant categories of global waste is cigarette butts, which are primarily the filters from the millions of cigarettes that are consumed. Cigarette smoking use is profoundly habit-forming and harmful to well-being. All smoked cigarettes contain numerous malignant growth-causing poisons and their utilization expands the risk of tumors of the head, neck, throat, throat, and oral depression (counting disease of the mouth, tongue, lip, and gums) as well as different dental illnesses. 1 An expected 5.6 trillion cigarettes are smoked every year, out of which 66% are inappropriately discarded, and these cigarette butts contain approximately 7000 chemicals in them.2–4 By 2025, this amount may potentially rise by more than 50% as a result of rising tobacco production and global population numbers. 5 Cigarettes typically measure 85 or 100 mm in length and 8 mm in diameter. Filter, tobacco, additives, and cigarette wrapper make up each cigarette. The typical length of a cigarette’s tobacco is between 55 and 80 mm due to the 20–30 mm length of cigarette butt filters (CBFs).6–8 The chemical makeup of cigarette butt trash has an impact on the environment as it is made from cellulose acetate fiber. 9 CBFs contain many heavy metals such as manganese, strontium, copper, iron, chromium, cadmium, aluminum, lead, and so on, and many aromatic volatile solvents which are very highly toxic to the human body and to the environment.10,11
The cellulose acetate fiber used in the cigarette is not easily degradable in the environment. Although it is photodegradable, it takes almost 10 years to degrade into the environment. 12 Butt disintegration was slowed down and made more challenging by CBFs, a densely entangled network of fibers combined with plasticizers. 13 For these reasons, cigarette butts constitute a serious problem for human health and the environment as well. 14 However, in recent years, many researchers have attempted to investigate various beneficial uses of waste fibers from cigarette butts, which is a terrific endeavor to reduce waste and may also lessen the adverse effects on the environment. Researchers nowadays are trying to develop sustainable products from waste and abundant materials such as composites 15 and sustainable natural materials.16,17 It is challenging to recycle CBFs since there do not appear to be any simple methods or procedures that guarantee an efficient and cost-effective separation of the butts or a suitable treatment of the entrapped chemicals and appropriate uses of those filter fibers. The majority of researchers attempted to manually break up the cigarette butts and separate the fibers using the crushing method. 18 Various washing and chemical treatments were also carried out depending on the desired products. But different researchers tried to develop various applications of CBFs in different recycled materials for nonwoven production, 19 sound absorbing material, 20 nanocrystal cellulose, 21 supporting material for bricks and steel manufacturing, 22 Asphalt concrete, 23 storage for energy in supercapacitor electrode, 24 and acetate fiber extraction from cigarette butts and characterization of their properties. 25 It has been observed that recently researchers tried to recycle and reuse the CBFs in different applications, and a considerable recycling rate of recent years was found for the production of carbon-based materials of around 27.27%, for building/construction materials, hydrophobic fibers 9.09%, supercapacitors 6.06%, paper 3.03%, corrosion control 6.06%, insect control 9.09%, sound absorber 6.06%, and biofilm carrier 6.06%. 26 Researchers tried to recover cellulose acetate fiber from waste butts, and they also analyzed the mechanical and chemical characteristics of recovered fiber through Fourier transform infrared spectrometer (FTIR), scanning electron microscopy (SEM), thermogravimetric analysis (TGA), X-ray diffraction (XRD), and so on.
In this research, we tried to recover the fiber from waste CBFs, by treating them with aqueous NaOH solution and filtering by acetone solution to use the cellulose acetate fiber in different textile applications such as producing yarn by mixing with cotton fiber. Until now, to our best knowledge, no research has been done to make textile yarn by using waste cigarette filter fiber for textile applications. To fulfill the research aim and analyze the possibility of the CBFs (Gold Leaf, B&H brand) being used to produce yarn, various mechanical, tensile, chemical, thermal, and morphological tests have been done to determine if any differences were present on the recovered cellulose acetate with respect to the unused specimen.
Materials and Methods
Fiber Extraction Process
Since the goal of the study was to examine the potential of CBFs to be made into textile yarn, two different cigarette brand waste filters were gathered from street bins, cigarette store trash cans, and nearly everywhere else they could be discovered. These butts contained unburned tobacco and ash. These items were cleaned by hand. With the use of a blade, the paper coating covering the cigarette butts was also removed, and the cellulose acetate filter fiber was then gathered. The fiber was then hand-shredded to open it up. The cellulose acetate fiber was washed with hot water for 30 min at 70° Celsius, followed by scouring and bleaching using laboratory-grade aqueous NaOH (99.9% w/w purity) and hydrogen peroxide (50% concentrated) for 1 h at 95° Celsius. Then, the butts were immersed in 0.02% H2SO4 solution of 100 mL for 30 min at room temperature according to Benavente and Caballero. The cellulose acetate fiber was rinsed multiple times with cold water following the process. The cigarette fibers were washed with acetone (99% w/w purity) for filtration purposes to remove heavy metals. The cleaned fibers are next dried in an oven dryer at a temperature of 60° for 60 min. Several characterization experiments, such as mechanical, FTIR, thermal, morphological, and so on, were conducted to evaluate the waste cellulose acetate fiber’s mechanical, FTIR, and other properties. Both untreated and chemically altered fibers were tested.
Heavy Metals Amount Analysis by Atomic Absorption Spectroscopy
Atomic absorption spectroscopy (AAS, AA-7000, Shimadzu) from Dhaka University was used to measure the concentration of heavy metals of Pb, Cr, Cu, and Zn in the solid form of the extracted cigarette butts before and after chemical modification. Both Gold Leaf CBFs and B&H CBF fibers went through the AAS test to see the amounts of metals in the research.
Discoloration Process
After the bleaching process, the color coordinates of the CBFs were determined using a Premier Colorscan SS5100H (India) spectrophotometer, and the CIE L*a*b* technique was used to determine the reflectance values and K/S values of all bleached samples. Here, both untreated and treated branded samples were assessed for their fiber’s bleaching and whiteness.
Fiber Characterization by FTIR
The functional groups of the materials were determined using an FTIR spectrophotometer (Model-Shimadzu 8400s, Japan) at the Dhaka University Centre for Advanced Research in the Sciences (CARS). It was employed to record the functional groups of all samples between 4000 and 400 cm–1. The samples were dried in an oven at 60° C. To create pellets, 0.2 g of powder sample was combined with 2 g of KBr.
Analysis of Crystallinity Characteristics
The crystalline and amorphous structures of the CBF sample were identified using XRD analysis. This experiment was carried out at room temperature using an X-ray diffractometer (model: D/teX Ultra 250 (H), Bangladesh atomic energy commission). We employed Cu-K radiation that scanned at a rate of 0.25° min–1 throughout a range of 2 = 0–80°. Isolated cellulose XRD patterns were acquired using a Theta/Theta STOE Jeol, Germany, X-ray diffractometer. The granules were pressed into a thin sheet between two glass slides for use as a sample. The X-ray images were captured using a Cu-K radiation source powered by 40 kV and 40 mA. From 20° to 80°, the patterns were recorded at a resolution of 2.
Thermal Gravimetric Analysis
A TGA was used to study the sample’s thermal characteristics (model: NETZSCH STA 449F3, Bangladesh Atomic energy commission). The samples were heated to a temperature of 700° Celsius in a nitrogen environment at a steady rate of 1.5° Celsius per minute.
Morphological Analysis
Non-modified and modified CBFs was examined by using a SEM (model: FESEM, MIRA3TESCAN-XMU). Drying the material and then applying a gold coating allowed for easier examination and for the prevention of charge. The intensity of the current was 15,000 volts.
Along with the above tests, some other tests were performed to evaluate the fibers such as water-bearing capacity (WBC); 1 gm of dried CBFs sample was kept in a beaker and then 25 mL of distilled water was added to the beaker. The sample was then incubated at 40° Celsius for an hour while being continuously stirred. The leftovers were centrifuged, and the WBC was determined. The moisture content and regain analysis of the samples were tested under the AATCC test method 20A-2017.
Effective Length Calculation
To analyze the probability of the modified cellulose acetate fiber for making yarn, the effective length of the CBFs was calculated through the Array Method following the ASTM D 1440-96 in BGMEA University of Fashion & Technology TTQC lab, where the representative sample of CBFs is parallelized, and the fibers are, respectively, straightened with the right and left hands. Numerous length groups of fibers are created. Using a Shirley comb sorter, groupings of fibers of varying lengths are identified. On a velvet pad, these sets of fiber lengths are organized in order of decreasing fiber length. One end of the bundle of fiber lengths is placed such that it hits the baseline. On the baseline, all groupings of fiber length are precisely arranged. When organizing groups with extremely short fiber lengths, more attention is necessary.
Tensile Strength Test
The waste CBF’s tensile properties such as tensile strength, elongation, and fiber density were calculated by using testometric materials testing machines (HVI, M/C No: 0250-07244), WinTest™. The testing machine gives very correct and precise data where four samples were tested and analyzed.
Results and Discussion
Heavy Metals Analysis in CBFs in the Extraction Process
Cigarette butts contain cellulose acetate, which is usable with proper treatment. Usually, cigarettes are composed of around 5000 chemicals that are directly harmful to the human body and environment. 27 Cigarette butts mainly contain different heavy metals, aromatic compounds, carbonylated compounds, and many more. To analyze the concentration of the heavy metals extracted from CBFs, we immersed the samples in 0.02% of H2SO4 (w/v) acidic solution which is preferable according to Benavente et al. 28 From the result of the AAS, it is shown that the amount of heavy metals extracted from both filter fibers before chemical modification for Pb is 7.8 μg/g, Cr is 14 μg/g, Cu is 0.6 μg/g, and Zn is 6.2 μg/g for Gold Leaf CBFs and Pb is 7.2 μg/g, Cr is 13.1 μg/g, Cu is 0.5 μg/g, and Zn is 5.8 μg/g for B&H CBFs. After chemical modifications with caustic soda and filtration with acetone, the modified CBF fibers are tested in AAS, and the results are Pb is 0.02105 μg/g, Cr is 0.0958 μg/g, Cu is 0.0115 μg/g, and Zn is the below detection limit for Gold Leaf CBFs and Pb is 0.0317 μg/g, Cr is below the detection limit, Cu is 0.01013 μg/g, and Zn is below the detection limit for B&H CBFs. There is evidence of the presence of some heavy metals, whose concentration is extremely low in terms of parts per million (PPM), necessitating careful management of the time required to purify CBF fibers.
Discoloration of Used Filter Fiber
The separated fibers from cigarette butts were cleaned, and 10 gm of fiber for each sample (Gold Leaf and B&H) was weighed in the electronic balance. The samples were properly cleaned with distilled water and soaked in the water for 1 h.
Several washes were done to remove the dust materials from the fiber. After that the samples were chemically treated with NaOH and H2O2 at 80°C for 60 min. After several cold and hot washes, the samples were shocked in organic solvent ether for 30 min with magnetic stirring. Then the cleaned white cigarette butt fibers were shocked and washed in acetone (CH3COCH3) and distilled water until the fibers were fully odorless and cleaned white. 29 The used brownish CBFs were converted into the white cleaned fiber without any bad smell, as shown in Figure 1(a). NaOH is a well-known chemical for extracting nicotine from cigarette butt fiber and ether, acetone solvents are used for the removal of bad odor from CBFs. 27 The CBF color characteristics were determined through spectrophotometric analysis.
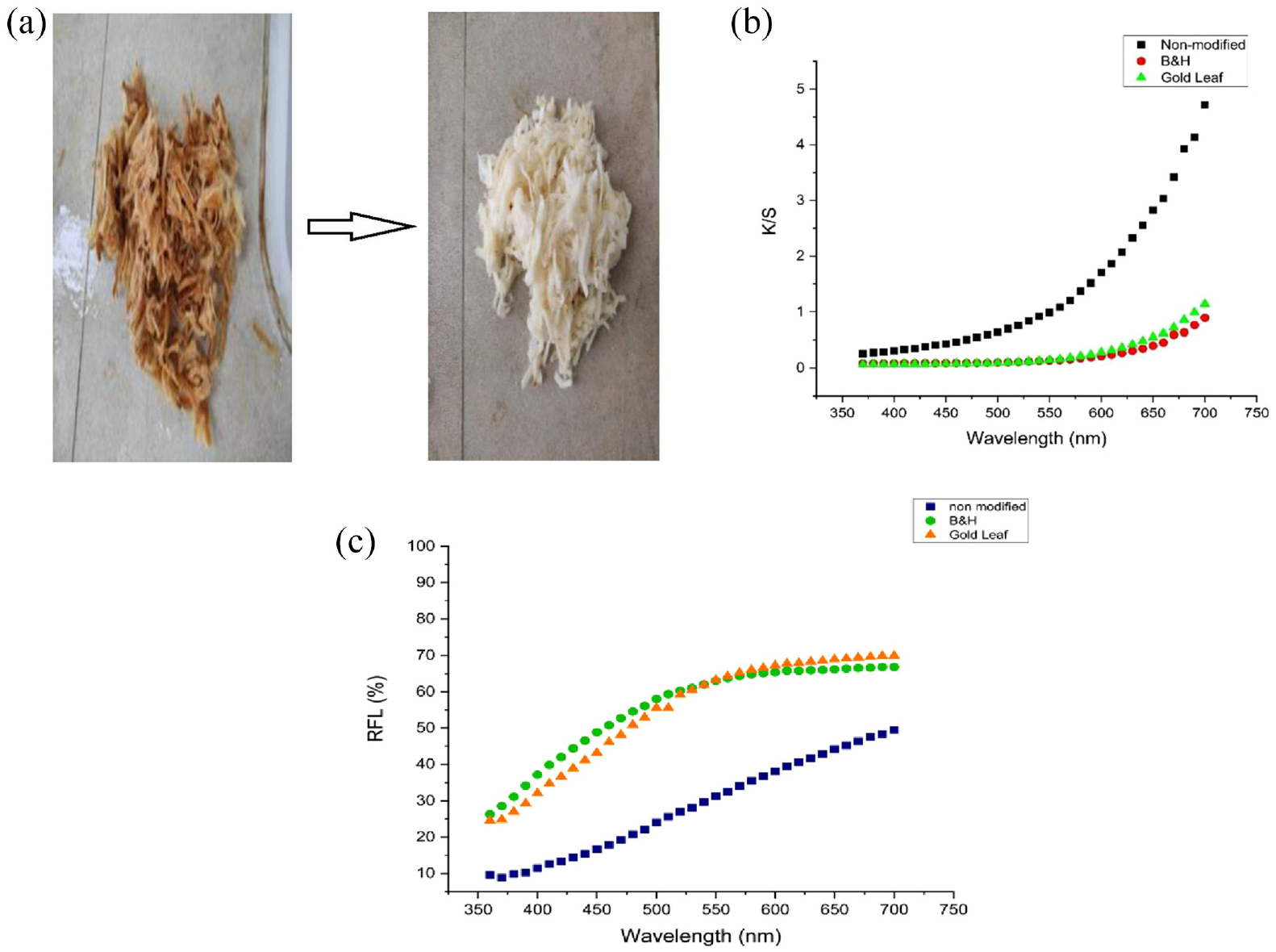
(a) Chemically modified used CBFs before modification and after modification. (b) The color strength value versus wavelength (nm) of the non-modified cigarette filter fiber and chemically modified cigarette filter fiber: (i) B&H and (ii) Gold Leaf. (c) The color reflectance value versus wavelength (nm) of the non-modified cigarette filter fiber and chemically modified cigarette filter fiber (i) B&H and (ii) Gold Leaf.
Spectrophotometric Analysis
A comparatively much whiter fiber was obtained after the chemical modification of the CBFs in NaOH and H2O2. Hydrogen peroxide is a very useful bleaching agent for cellulosic materials. 30 Perhydroxyl ions work to remove the brown colors produced from burning cigarette butts and made the fibers whiter, 31 but some strength, as well as weight, was lost in this process: about 48% from the weight loss test, because NaOH and hydrogen peroxide remove lignin, residues, and other toxic chemicals in cigarette butts, and also break the polymeric chain of cellulose materials. 32 From the spectrophotometric analysis, it is shown that the untreated CBFs have high color strength in the reddish region.
After smoking, the cigarette filter turned a reddish-brownish color. On the contrary, the chemically treated CBFs showed a much lesser color strength which means hydrogen peroxide ruins the maximum portion of the reddish color from CBFs. For the three samples, there is a big difference between the untreated cigarette filter and the other two chemically treated samples.
The color yield (K/S) value of untreated cigarette butts is 4.719, which is greater than those of the other two chemically treated samples. Similarly, the lightness value (L*) of the treated sample increased gradually from 62.668 to 82.886 and 82.829, which means the brightness of the chemically treated samples increased after the chemical treatment. At the same time, the difference between the red/green axis (a*) also changed. The reddish value 5.813 changed to greener for samples 2 and 3: −0.960 and −0.321. The difference between the yellow/blue axis (b*) changed a little for the chemically treated sample. However, the samples B&H and Gold Leaf showed a big difference compared with the untreated samples, which means the cigarette filter color had changed after chemical treatment, 33 as shown in Figure 1(b) and (c).
FTIR Analysis of CBFs
First, Figure 2(a) indicates the FTIR data of the commercial cellulose acetate fiber, which was brought from the local fiber market of Bangladesh. The FTIR data of commercial CA indicate broad stretching peaks at 3400 cm–1, which indicate the O-H bonds which mean hydrogen bonds in the fiber. Other peaks at 2900–3000 cm–1 stretching indicate C-H functional groups which indicate alkane group. 34 At 1754 cm–1, the carbonyl functional group (C=O) appeared, which represents the acetate functional groups in cellulose. 19 Also at 892 cm–1, another peak appeared which indicates the –O-H stretching bond represents the water-absorbing group of cellulosic materials.35,36
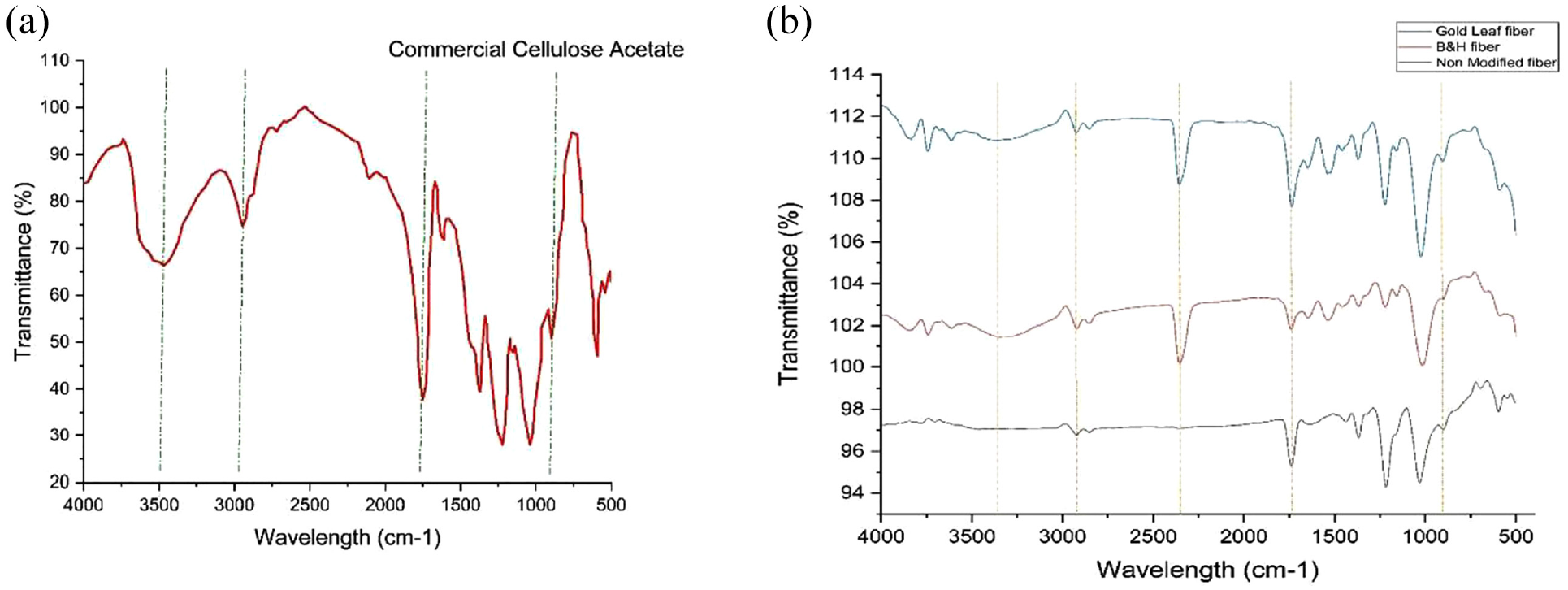
(a) FTIR spectra of commercial cellulose acetate fiber. (b) FTIR spectra of non-modified CBFs, chemically modified B&H CBFs, and chemically modified Gold Leaf CBFs.
Second, untreated CBFs and chemically modified CBFs (B&H, Gold Leaf) both underwent FTIR analysis. Figure 2(b) shows the non-modified cigarette filter fiber has a wider peak in 3300–3400 cm–1; on the other side, after chemical modification, both CBF samples showed clear broad stretching peaks O-H at 3200–3500 cm–1, which indicates that after chemical modification the large peak of water group is regenerated which was not present in non-modified CBFs; it also indicates the proper removal of nicotine and other metallic compounds from CBFs. At 2920 cm–1, B&H and Gold Leaf CBF samples show comparatively sharp strong peaks which indicate C-H rather than the non-modified CBFs. At 1740 cm–1, the rising band indicates the C=O functional group which indicates acetyl groups in acetate fiber. 37 After chemical modification, the peaks at 1740 showed a very sharp band. At 895 cm–1, it shows the water-absorbing group O-H.38,39 So, this spectral observation shows the proper chemical modification of cigarette filter fiber after use.
XRD Analysis of CBFs
XRD is an analytical technique that is the diffraction of X-rays by matter, especially for crystalline materials. To get more information about the chemically modified CBF fibers with sodium hydroxide and hydrogen peroxide, an XRD test was done.
The chemically modified CBF crystal structure was investigated through a 2-theta angle with XRD. From the test, the chemically modified sample was confirmed as having a cellulose-II structure from the XRD pattern and characteristics. Figure 3(i) shows that three broad peaks shown in the Bragg diffraction 2-theta angles at 17.02°, 22.81°, and 35.04° for B&H CBFs and 16.98°, 22.7°, and 34.97° for Gold Leaf CBFs, which are mostly similar to the chemically modified cellulose acetate, respectively, from the references of previous articles.40–42 Cellulose peaks at 17.02° & 16.98°, 22.81° & 22.7°, and 35.04° & 34.97° corresponding to (101), (020), and (212) planes were assigned to the cellulose-II as similar.43,44
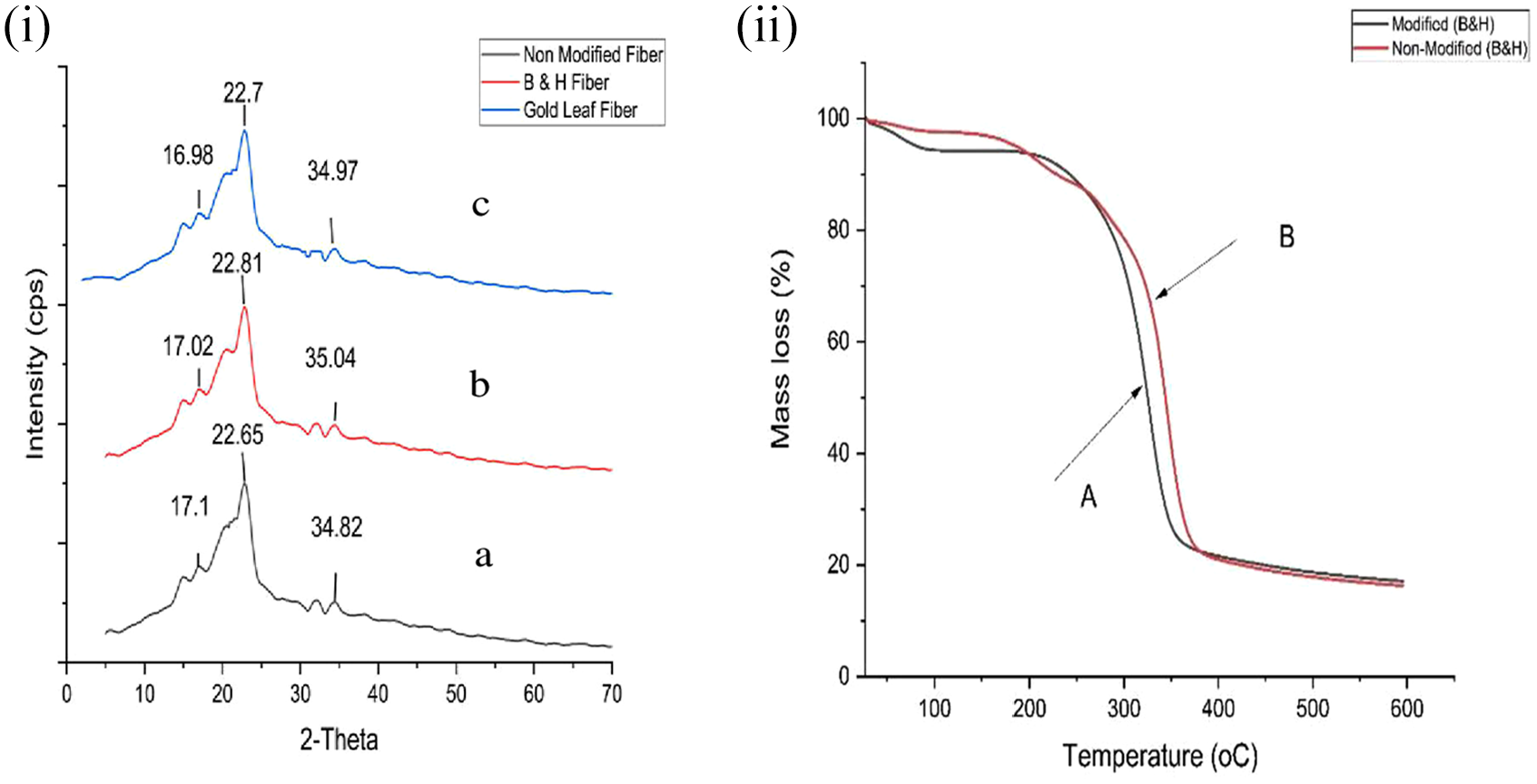
(i) The XRD graph design of cellulose acetate fiber from CBFs: (a) non-modified fiber, (b) B&H fiber, and (c) Gold Leaf fiber. (ii) Thermogravimetric analysis of non-modified CBF (B) and chemically modified CBF (A) samples.
Thermal Degradation Analysis of CBFs
Thermogravimetric analysis (TGA) is an analytical technique that analyzes the weight change that occurs when a sample is heated at a consistent rate to determine the thermal stability and proportion of volatile components of a substance. Figure 3(ii) shows that the initial weight change started for the chemically modified sample (B&H) from 85°C which showed little weight loss happened at this temperature, which also represents the evaporation of water absorbed in the fiber. On the contrary, the non-modified CBF’s thermal change in the same temperature is very low, just 1.9% approximately which is the cause of heavy metals existing in the non-modified CBFs. Major degradation in both samples was observed from the point 260°C approximately and the highest degradation of both samples A and B occurred at the point 384°C which indicates the maximum degradation. 45 The TGA curve stipulates that even after chemical modification in the CBFs, it may change the polymorph a little but both cellulose acetate samples of cigarette butts show similar thermal behaviors. 46
Morphological Analysis of CBFs
The surface characteristics and the surface morphology of the used cigarette filter fibers and chemically modified CBFs were investigated under JSM-7610F which is an ultrahigh-resolution Scotty Field Emission SEM. The SEM in Figure 4 shows that the sample in Figure 4(a) is the non-modified cigarette butt fibers and the sample in Figure 4(b) is chemically modified cigarette butt fibers.

SEM figure of non-modified CBF (a) and chemically modified CBF (b) samples (magnification ×500)
From the analysis, it is seen that from ×500 magnification, the non-modified samples contain many contaminants that were displaced on the surface of the fibers. The used CBFs contain many metals and chemical compounds on their surfaces, which make the surface rough. But after being treated with hydrogen peroxide and sodium hydroxide, and also treated with many organic solvents several times, the CBF morphological investigation showed that the fiber surface turned more smooth and fresh after the treatment from the analysis of SEM figures with ×500 magnification. But some roughness and pores are created on the surface after NaOH treatment. 47
WBC of CBFs
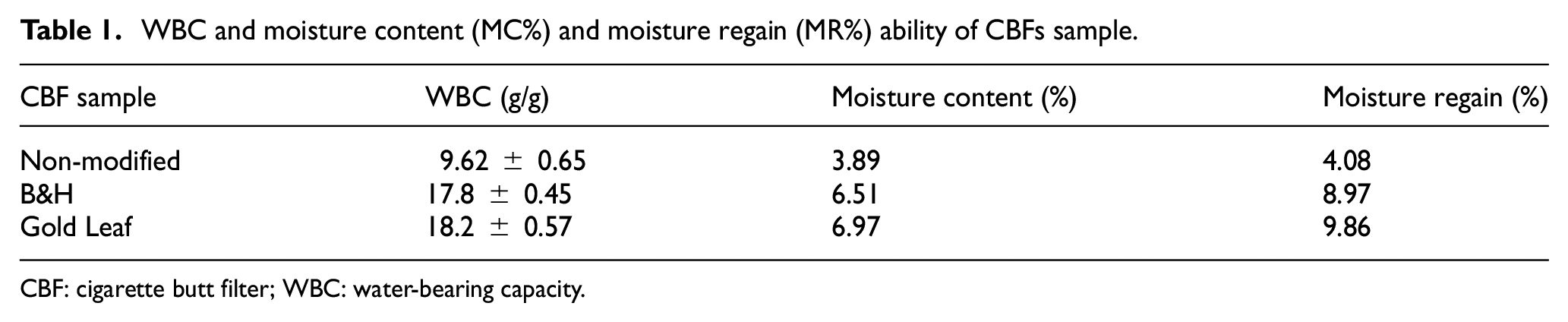
As the chemically modified cigarette filter fibers are treated with NaOH, it removes various chemical compounds and also nicotine from cigarette butts. The removal of different contaminant chemicals makes the cellulose acetate fiber more absorbent and more hydrophilic as hydroxyl groups increased with the treatment. The hydrophilic capacity of chemically modified CBFs was tested by WBC.36,48 The WBC of three samples of non-modified, chemically modified B&H, and Gold Leaf CBFs is shown in Table 1; the Gold Leaf and B&H CBF samples have more WBC than the non-modified CBFs.
WBC and moisture content (MC%) and moisture regain (MR%) ability of CBFs sample.
CBF: cigarette butt filter; WBC: water-bearing capacity.
Moisture Test of CBFs
Most of the properties and qualities of fiber depend on the temperature and humidity. Fibers are likely hygroscopic, meaning that depending on relative humidity, they absorb or release moisture from the surrounding air. If the environment gets dryer, the fiber will release moisture. If the surrounding air is too humid, fiber humidity will rise. AATCC test method 20A-2017 was used for the moisture test. 49 From the test, it is shown that the non-modified chemical sample’s moisture content (MC%) and moisture regain (MR%) are less than those of the chemically modified B&H CBFs and Gold Leaf CBFs. This is because chemical modification with NaOH and with other organic solvents removes many hydrophobic materials from the CBF samples. The results are shown in Table 1.
Fiber Length Calculation
CBF length was calculated by the array method. The fiber length of the waste cigarette filter fibers is an essential factor for making yarn by blending with other fibers. From the graphs, different lengths were determined and the effective length of the fiber is one of them.
Here,
OQ = 1/2 (OA),
OK = 1/4 (OP),
SK =1/2 (KK′),
OM = 3/4 OR,
OL= 1/4 (OR).
From Figure 5(i), according to the array approach, the effective length of the CBFs is (LL′) 2.5 cm and the maximum length (OA) of the fiber is 2.6 cm. The determined effective length is slightly shorter, yet it is suitable for yarn production when blended with suitable raw fibers such as cotton or polyester.
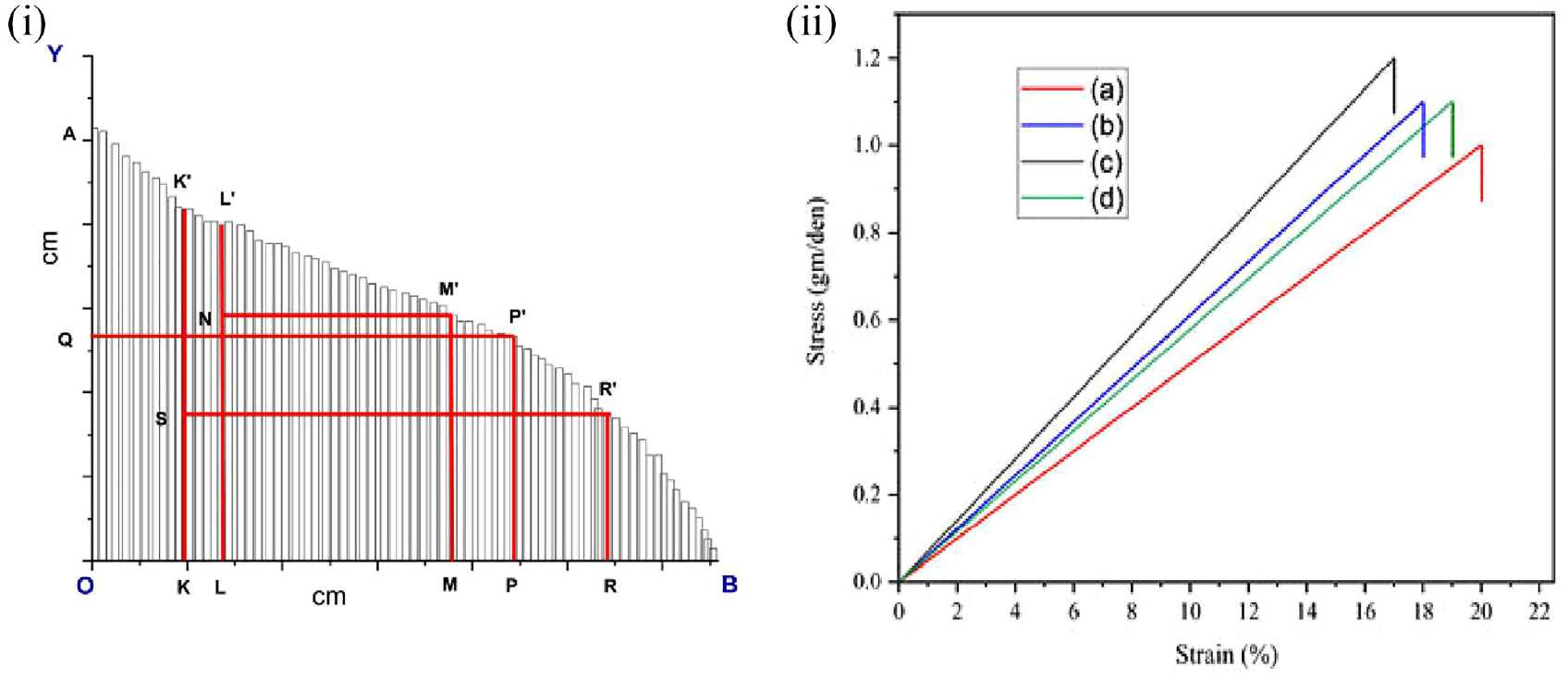
(i) Comb sorter diagram of waste CBFs. (ii) Tensile stress–strain curve for the CBFs. Here, (a) and (d) belong to non-modified B&H and Gold Leaf fiber, respectively, and (b) and (c) belong to modified B&H and Gold Leaf fiber, respectively.
Tensile Properties Analysis
The tensile properties of CBF samples are shown in Figure 5(ii). The stress–strain curve of the CBF indicated a straight line of stress which suddenly dropped in a certain stress value which is because of the regenerated cellulose acetate fiber’s fibrous and porous molecular structure. Because of the high porosity and fibrousness, the tenacity of the fiber is comparatively lower than that of the cotton fiber and the elongation to break (%) is higher. 50 The average tenacity and elongation of the CBFs were found to be 1.1 gm/den and 18.5%, respectively. The breaking tenacity of the CBFs was a little lower than the commercial cellulose acetate fiber 51 because of the chemical modification with NaOH. But the determined length is comparatively suitable because of its proper length, strength, and elongation.
Density Analysis
The linear density of the fiber is another crucial aspect of yarn production. This research also measured the fiber density of CBFs to assess the viability of manufacturing yarn from CBFs blended with cotton or other raw fibers. The average density of CBFs (B&H, Gold Leaf) was determined to be 1.2 dtex, which is acceptable and equivalent to commercial cellulose acetate fiber and cotton fiber.52,53 Therefore, the CBFs can be used to create blended yarn.
Conclusion
To overcome the significant disadvantage of cigarette filters, the filter fibers can be recycled and repurposed as alternative materials, such as the raw materials for blended yarn with other suitable fibers such as cotton, viscose, or polyester. The primary objective of this study is to recover the waste cellulose acetate fiber from cigarette butts, treat those fibers with the proper chemicals to make them clean and then examine the qualities of those fibers to determine whether they can be used to manufacture blended yarn. We present a strategy for purifying this polymer based on multiple solvent-based solid–liquid extraction processes. The use of a spectrophotometer to test the discoloration of the CBFs revealed outstanding reflectance values, indicating that the burned color was effectively removed from the utilized fiber. In addition, FTIR, TGA, and XRD tests were carried out to evaluate the fibers; they revealed that the employed cellulose acetate fiber had similar chemical, thermal, and crystal properties to those of commercially available cellulose acetate fiber. Furthermore, an AAS test attested to the fact that we can also remove heavy metals from CBFs using our extraction method. In addition, the morphological study showed that the modified cellulose acetate fiber had a rounded shape. Most importantly, it was discovered that the used cigarette filter fiber had an effective length of 25 mm, a tensile strength of 1.1 gm/den, a fiber fineness of 1.2 dtex, a moisture content of 7%, and a regain of 9%, respectively, all of which indicated that the used cigarette filter fiber was suitable for making yarn. However, due to its low strength, it was necessary to blend the used cigarette filter fiber with other raw materials, such as cotton. The recycling of CBFs and turning this waste into a resource like yarn for clothing can also be a remedy to the pollution caused by cigarette butts. The significant finding is that it is possible to produce useful yarn from used cigarette filters, which has enormous economic potential and may help to lessen the impact of discarded filters on the environment. Future studies will also focus on developing yarn from this chemically altered CBFs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
