Abstract
In order to impart good application properties to locust bean gum size for all-polyester yarn, a series of acrylate monomers including methyl acrylate, ethyl acrylate, butyl acrylate, 2-ethylhexyl acrylate, and common hydrophilic monomer–acrylic acid were grafted onto molecular chains of native locust bean gum to obtain locust bean gum-g-P(AA-co-acrylate) products with similar grafting ratios through Fenton’s reagent, respectively. The effects of carbon chain length of the series of acrylates on sizing properties of the grafted locust bean gum for polyester warp were studied. It was found that by grafting acrylates onto the molecular chains of locust bean gum the sizing properties of the locust bean gum were improved markedly. Under the condition of similar grafting ratios, locust bean gum-g-poly(AA-co-methyl acrylate) showed the highest apparent viscosity, water solubility, film strength, wettability, and adhesion to polyester fiber in all the grafted locust bean gum prepared. Meanwhile, locust bean gum-g-poly(AA-co-butyl acrylate) and locust bean gum-g-poly(AA-co-2-ethylhexyl acrylate) had the highest wear resistance and elongation of sizing film, respectively. The modified locust bean gum grafted with acrylate monomers with different carbon chain lengths was able to meet various requirements in the sizing process for polyester yarn.
Warp sizing is the most important preparation procedure before weaving and affects weaving efficiency substantially. Sizing agents are basic polymer materials in the procedure, and their consumption is nearly 1 million tons annually in textile mills worldwide nowadays. 1 According to the proportion of consumption, the three major textile sizes are starch (~70%), polyvinyl alcohol (PVA; ~20%), and polyacrylate (~8%). 2 Under the double impacts of the COVID-19 pandemic and the locust plague in East Africa and South Asia, the main grain-producing countries, such as Egypt, India, Thailand, and Vietnam, have restricted grain exports successively.3,4 The prices of starch and its derivatives show a rising trend, which makes the demand for “sizing without grain” more urgent. Due to the non-renewability of petroleum resources and the poor biodegradability of petroleum-based polymers, the production and utilization of synthetic sizing agents are increasingly limited. In particular, PVA has been listed as an “environmentally unfriendly sizing agent” and banned in textile mills in many European countries.5,6 Consequently, a growing number of researchers are starting to seek non-edible bio-based polymer materials from agricultural and forestry by-products and chemically modify them to develop inexpensive and renewable sizing agents with high performance.
The locust tree is a common tree species in the North Temperate Zone and is popular for its good adaptability, fast growth, easy reproduction, and wide use. It has become the broadleaf tree species second only to eucalyptus in its artificial afforestation area and widely distributed in North American, European, and Asian countries, such as the United States, Hungary, and China.7–9 Wang’s 9 investigation showed that the planting area of locust trees in China alone had reached 10 million hm 2 . Locust bean gum (LBG) is extracted from the endosperm of the fruit of the locust tree and is mainly a galactomannan with mannose as the main chain. LBG has high yield and low price in most temperate countries and possesses similar physical and chemical properties to starch, but it is inedible and has low additional value of the product. Therefore, LBG has been taken into account by textile chemists as a substitute for starch size. LBG is a kind of vegetable gelatin with lots of hydroxyl in its molecular chain and belongs to a strong polar polymer.10,11 It has good sizing properties for cotton yarns, especially for the low-count and medium-count ones.12,13 However, LBG exhibits poor affinity to the most widely used synthetic fiber, polyester, which possesses numerous phenyl and ester groups and thus shows very weak polarity.2,14 Consequently, the poor affinity seriously limits the range of the LBG’s sizing objects.
Nowadays, polyacrylate, partial alcoholysis PVA, and water-soluble polyester are the main kinds of sizing agents for polyester warp yarns.15–17 All the sizing agents mentioned are petroleum-based polymers with poor biodegradability, especially PVA. Few textile mills carry out sizing for pure polyester filaments or yarns but polyester staple yarns are generally sized after blending with cotton (e.g. T/C65/35 yarn). Therefore, the amount of the sizing agents for polyester yarns is still quite large. For example, the third-largest sizing agent, polyacrylate, accounts for about 8% of the total consumption of sizing agents and is mainly used for polyester warp yarn. According to incomplete statistics, nearly 150,000 tons of sizing agents are consumed for pure and high-content polyester yarns annually.17,18
In order to expand LBG’s sizing objects to all-polyester and high polyester content yarns, acrylate monomers with various carbon chain lengths, that is methyl acrylate (MA), ethyl acrylate (EA), butyl acrylate (BA), and 2-ethylhexyl acrylate (EHA), are grafted onto native LBG through Fenton’s initiator in this study in order to decrease the macromolecular polarity and enhance the polarity similarity between LBG and polyester fiber. The sizing operation is carried out in water and thus requires the sizing agent to be water-soluble or water-dispersible at least. Native LBG cannot dissolve in water well in itself due to strong hydrogen-bond interactions and high average molecular weights in the range of 960–1100 kg/mol. On this basis, the introduction of hydrophobic grafted branches (e.g. polyacrylate) will decrease the water solubility of the LBG further. Therefore, a constant-feed molar ratio of hydrophilic monomer–acrylic acid (AA) to the acrylate was grafted onto the LBG simultaneously to maintain water solubility of the grafted product.
After the preparation of a series of LBG-g-P(AA-co-acrylate) with similar grafting ratios (GRs) but different molecular structures of polyacrylate branches, the main sizing properties of the LBG, that is apparent viscosity, contact angles of sizing paste on polyester fibers, water solubility and mechanical properties of sizing film, the adhesion to polyester fiber, and sizing performance for polyester/cotton 65/35 blended yarn (i.e. T/C65/35 yarn) were evaluated systematically. Then, the effects of the carbon chain length of the acrylate monomer on the sizing properties were revealed. Finally, the acrylate monomer with an appropriate carbon chain length is recommended for the preparation of LBG-g-P(AA-co-acrylate) to be able to meet various requirements in the sizing process for polyester yarn.
Experimental
Materials
In the graft polymerization, LBG powder was the substrate; AA was a hydrophilic monomer; MA, EA, BA, and EHA were acrylate monomers; H2O2 (oxidant) and (NH4)2Fe(SO4)2•6H2O (reductant) were initiators, that is, catalysts for the grafting; paradioxybenzene was the terminator for the graft polymerization. Acetone was used as the solvent to remove homopolymers (i.e. polyacrylates) from the LBG-g-P(AA-co-acrylate) product during the purification. LBG was kindly provided by Xi’an Quanao Biotechnology Co., Ltd (Xi’an, China) and technically pure. The reagents AA, MA, EA, BA, EHA, H2O2, (NH4)2Fe(SO4)2•6H2O, paradioxybenzene, and acetone were all purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China) and chemically pure. The all-polyester rovings (483 tex) offered by Yizheng Co., Ltd of Chemical Fiber (Yangzhou, China) were used for evaluating the adhesion of the LBG sizes. The length and fineness of the polyester fibers of the rovings were 45 mm and 1.80 dtex, respectively. The T/C65/35 warp yarns (12.8 tex) used for the sizing experiment were obtained from Weifang Runfeng Textile Co., Ltd (Weifang, China).
Graft Copolymerization
Before the graft polymerization, 60 g of native LBG powders were dispersed in distilled water and then transferred into a 500-mL four-neck flask in a thermostat water bath. The flask was maintained at 40°C in the water bath, and the flask was deoxygenated by passing pure N2 for at least 30 min before adding reactants. Fenton’s reagent including 25.2 g of oxidant (H2O2) and 5.8 g of reductant [(NH4)2Fe(SO4)2·6H2O] was dissolved in distilled water. The molar ratio of H2O2/(NH4)2Fe(SO4)2·6H2O was kept at 50/1. The AA and acrylate monomers (i.e. MA, EA, BA, or EHA), oxidant and reductant solutions were added into the flask simultaneously. In order to eliminate the influence of the GR of the modified LBG on the sizing properties, the GR of all the LBG-g-P(AA-co-acrylate) was controlled at similar values. According to Li et al.’s 19 report on sesbania gum-g-P(AA-co-MA), sizing properties of the grafted vegetable gum for polyester yarns were satisfactory when the GR was around 19%. Therefore, the GR was controlled within a fixed range (19% ± 2.5%) after many tentative graft copolymerization experiments in this study. The feed molar ratio of AA/acrylate was maintained at a high constant value (7/3) to keep the grafted product water soluble. The sums of feed weight of AA and MA, EA, BA, and EHA monomers were 15.0, 16.8, 21.0, and 22.8 g, respectively. The addition was completed in 10–20 min, and the final bath ratio of LBG to distilled water was 1/7. The graft polymerization was carried out under mechanical stirring using a stirrer at 400 r/min under pure N2 protection for 6 h. About 2% of paradioxybenzene solution was added to terminate the polymerization. The product was filtered, washed thoroughly with ethanol, dried in a vacuum oven, ground to a fine powder and stored in a desiccator. Each graft polymerization was repeated three times. The graft copolymerization of the LBG with AA and acrylate is shown in Figure 1.
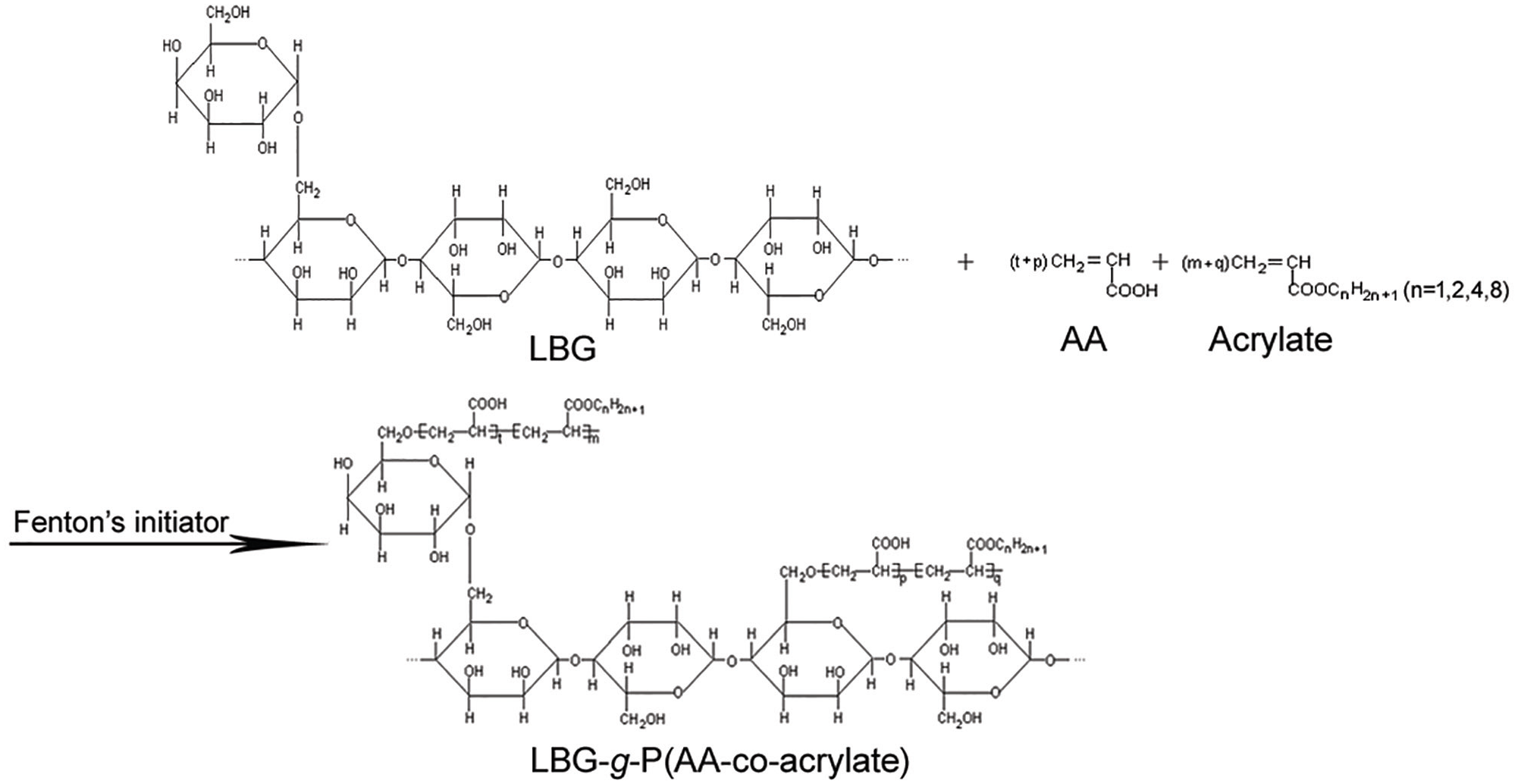
Graft copolymerization diagram of native LBG with AA and acrylate through Fenton’s reagent.
Measurement of Grafting Parameters
The grafting parameters of the LBG-g-P(AA-co-acrylate), that is, the conversion of monomers to polymer (MC), GR and grafting efficiency (GE), were measured in the study. The conversion of monomers to polymer is the weight percentage of the polymers formed (i.e. grafted branches and homopolymers) to the total monomers added. The GR is the weight percentage of polyacrylic acid (PAA) and polyacrylate branches grafted onto the LBG to the LBG. The GE is the weight percentage of PAA and polyacrylate branches grafted onto the LBG to the polymers formed (i.e. grafted branches and homopolymers). The detailed measurement processes are described in Supporting Information.
FTIR Characterization
A Fourier transform infrared spectrometer (FTIR) was used to verify the grafting of AA and acrylate onto the LBG. Previous reports indicated that water and acetone were good solvents for PAA and polyacrylate, respectively.2,14 In order to purify the grafted LBG, the sample was successively extracted by water and acetone for 48 h (water 24 h + acetone 24 h) to ensure the thorough removal of the homopolymers (i.e. PAA and polyacrylate). Measurements of the native LBG and the purified LBG-g-P(AA-co-acrylate) were taken on a Thermo Nicolet (Avatar 380) spectrophotometer through the diffuse reflectance technique with a spectral resolution of 2 cm−1 for 64 scans.
1H-NMR Characterization
Hydrogen Nuclear Magnetic Resonance (1H-NMR) characterization was employed to confirm the grafting of AA and acrylate onto the LBG. The native LBG and the purified LBG-g-P(AA-co-acrylate) were dissolved in dimethyl sulfoxide (DMSO) solvent and characterized by an AVAMCE III 400 MHz Digital NMR spectrometer (Bruker Co., Ltd. Switzerland). The concentration of LBG solution was about 5% (w/w).
Measurement of Application Properties of Sizing Paste
The main application properties of sizing paste of the native LBG and LBG-g-P(AA-co-acrylate), such as apparent viscosity, surface tension, and contact angle on polyester fiber, were measured in the study. The detailed measurement processes are described in the Supporting Information.
Preparation of Sizing Films and Measurement of Their Application Properties
Sizing film was cast with 400 g of the LBG paste (6%, w/w), and the surface morphology of the film was observed by scanning electron microscope (SEM). The main mechanical properties (e.g. tensile strength, elongation, and wear resistance) and water solubility of sizing film were evaluated in the study. The preparation method of the sizing film, SEM observation, and the detailed measurement processes of the mechanical properties and water solubility of the sizing film are described in the Supporting Information.
Measurement of Adhesion to Polyester Fibers
A roving-sizing method was employed to measure adhesion of the LBG to polyester fibers. The roving method is a standard to estimate adhesion of a sizing agent to fibers in China (FZ/T 15001-2008, a criterion regulated by Textile Association of China). In the measurement, a slightly sized roving is drawn to tensile failure. The failure loads were adopted to exhibit the adhesion. The preparation of the slightly sized roving and the measurement of the adhesion are described in the Supporting Information.
Sizing Experiment
The cooking process of the LBG sizing paste was the same as the one described in the measurement of apparent viscosity except that the concentration of the sizing paste was 12% (w/w). The sizing experiment for T/C65/35 warp yarns was carried out using a GA392 laboratory single-yarn sizing machine purchased from Jiangyin Tongyuan Textile Machinery Co., Ltd (Jiangyin, China). The sizing style of the machine was single-dip-single-nip. The warp yarns were wound in the machine and the cooked sizing paste was poured into the size box, where it was adjusted to 95°C. The sizing tension was 1.0 N, and the running speed of the sizing machine was 30 m/min. A hot air and cylinder allied drying style was employed in drying chamber, where it was adjusted to 110°C. The sized yarns were dried in the chamber for 3 min and finally conditioned at 65% relative humidity and 20°C for at least 48 h before the performance tests.
Measurement of Tensile Properties of Sized Yarns
The tensile strength and elongation of the sized T/C yarns were determined on a model YG023A electric strength tester purchased from Laizhou Electronic Instrument Factory (Laizhou, China). The initial chuck-distance and drawing speed were 500 mm and 500 mm/min, respectively. For every sample, fifty replications were taken, and their mean values were obtained.
Measurement of Abrasion Resistance of Sized Yarns
The sized T/C yarns were abraded reciprocatively on an LFY109B electric yarn abrader purchased from the Textile Science Research Institute of Shandong Province (Qingdao, China). The resistance to reciprocating friction of the sized yarns was evaluated in the reciprocal motion times of the yarns until breaking. The tension exerted on the yarns was 9.8 cN. The values reported represent the mean value of 50 tests for each case. The abrasive material used was W5(06) abrasive paper manufactured by Shanghai Emery Wheel Company (Shanghai, China).
Statistical Analysis
The data were analyzed using SAS software (SAS Institute, Inc., Cary, NC). Tukey’s HSD test was used to compare the multiple means in the investigation. The confidence interval was set at 95% and a “p” value smaller than 0.05 was considered to be a statistically significant difference by Tukey’s HSD test. According to the operation results of the analysis software, small letters, such as a, b, and c, were inserted above or below the data points in Figures 4, 5, 7, and 8. The data points with the same small letter were not statistically significantly different from each other.
Results and Discussion
FTIR Analysis of Native and Grafted LBG
The FTIR spectrum of the native and grafted LBG is shown in Figure 2. The major characteristic peaks, such as the ones at ~3360 and ~2920 cm−1 due to stretching vibrations of hydroxyl and methyl/methylene respectively,19,20 could be observed in the curve of the native LBG. As for the LBG-g-P(AA-co-acrylate), a new characteristic absorption band of carbonyl of carboxyl and ester groups at ~1738 cm−1 appeared in each grafted sample’s curve. The peak proved the existence of carboxyl and various acrylic esters (i.e. the methyl, ethyl, butyl, or ethylhexyl ester)19,20 in the grafted branches of the modified LBG and was regarded as the evidence for the grafting of AA and acrylate onto the molecular chains of LBG.
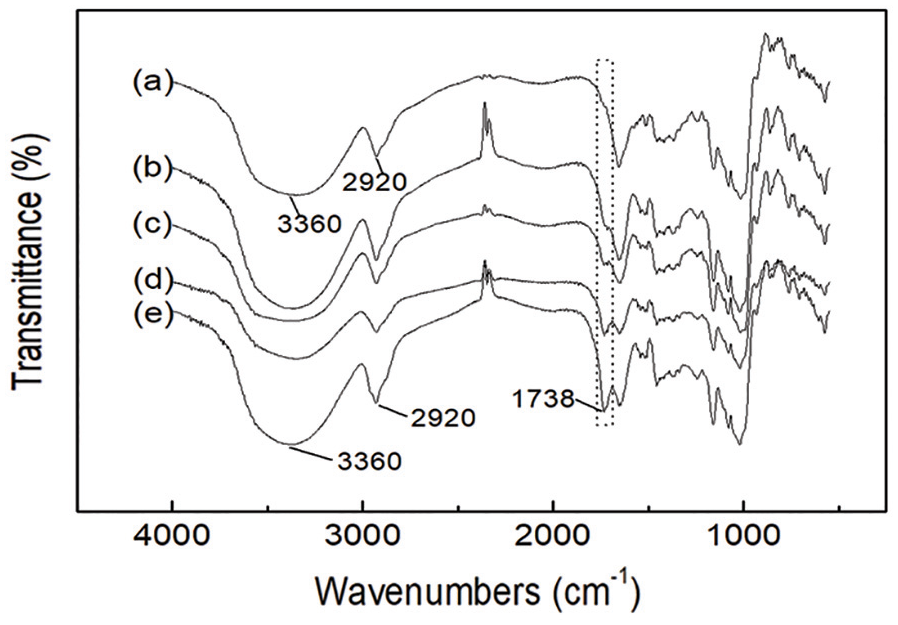
FTIR spectrum of native LBG (a), LBG-g-P(AA-co-EHA) (b), LBG-g-P(AA-co-BA) (c), LBG-g-P(AA-co-EA) (d), and LBG-g-P(AA-co-MA) (e).
1H-NMR Analysis of Native and Grafted LBG
The 1H-NMR spectra of the native and grafted LBG are shown in Figure 3. Chemical shift peaks at ~2.5 and ~3.3 ppm in all the spectra corresponded to proton peaks of the solvent (DMSO) and residual H2O, respectively. In addition to all the chemical shift peaks of native LBG, such as the proton ones of the mannose and galactose rings in the range of 4.2–0.35 ppm,21–23 new chemical shift peaks at ~3.5 and ~12.9 ppm were found in the spectra of the grafted samples and could be considered as the proton peaks of methyl/methylene directly linked to the carbonyl of acrylic esters and carboxyl of AA, respectively.2,24 The two new peaks confirmed the existence of carboxyl and various acrylic esters in the grafted branches of the modified LBG. Moreover, the increases in the intensities of alkyl proton peaks (e.g. –CH2– and >CH–) appearing in range of 0.9–2.0 ppm in the spectra of the grafted samples could be considered as another proof of the presence of PAA and polyacrylate branches on the LBG-g-P(AA-co-acrylate).
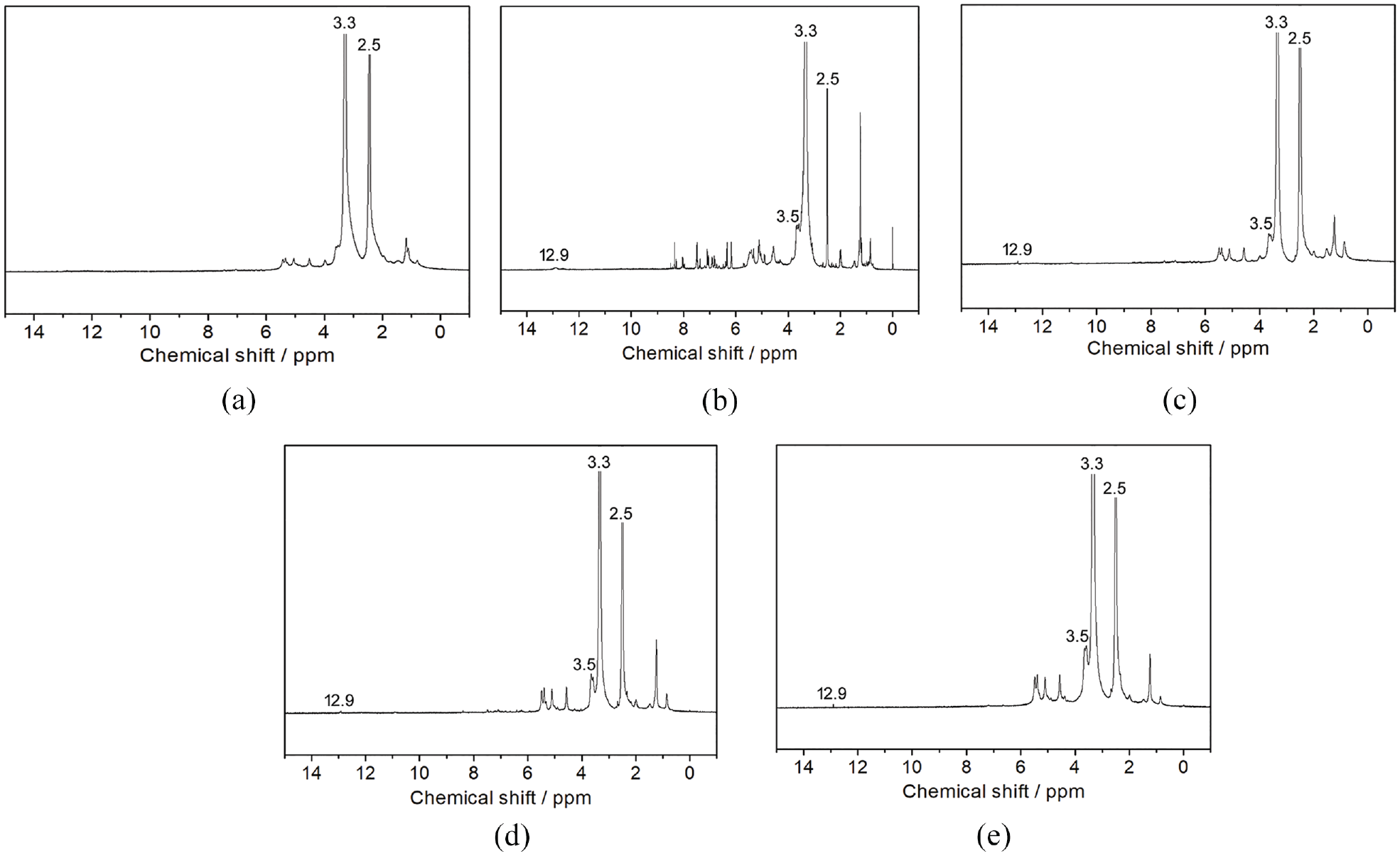
1H-NMR spectra of native LBG (a), LBG-g-P(AA-co-EHA) (b), LBG-g-P(AA-co-BA) (c), LBG-g-P(AA-co-EA) (d), and LBG-g-P(AA-co-MA) (e).
Effects of Carbon Chain Length of Acrylate Monomer on Grafting Parameters
Figure 4 depicts the effects of the carbon chain length of the acrylate monomer on the conversion of monomers to polymer, GR, and the efficiency of the grafted LBG. With the increase in the carbon chain length, all the conversions of monomers to polymer were around 98.3% and did not change significantly. In order to eliminate the impact of the GR of the grafted LBG sizes on their properties, the GR of all the grafted LBG synthesized by acrylate monomers with various carbon chain lengths were controlled within an appropriate range (19 ± 2.5%) according to Li et al.’s 19 report. As for the GE, it decreased in a range of 66.4–45.6% stepwise with the increase in the carbon chain length.
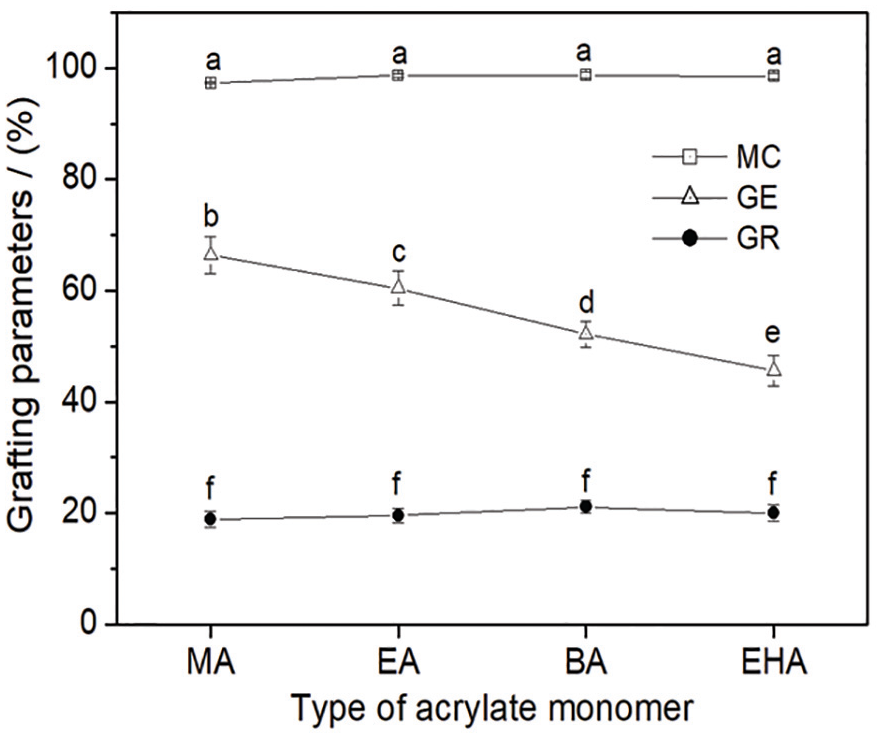
Grafting parameters of LBG-g-P(AA-co-acrylate).
Conversion of monomers to polymer does not relate to the substrate, that is, LBG in this study. The conversion of monomers to polymer has a close relationship to the molecular structure of grafting monomer and is an important parameter to mark whether a graft polymerization process is reasonable or not. If a large amount of graft monomer (e.g. acrylate) joins in neither graft polymerization nor homopolymerization, the preparation cost of graft copolymer will be increased substantially. Furthermore, acrylate monomers are usually allergenous or even carcinogenic and the residual monomers will pollute the environment unavoidably. Therefore, it is necessary to investigate the carbon chain length of acrylate monomers on conversion of monomers to polymer first. Under appropriate free-radical polymerization conditions (e.g. adequate oxidant concentration, proper molar ratio of oxidant/reductant, and polymerization temperature), an adequately long polymerization time is a sufficient condition for the conversion of nearly all the monomers to polymers. Obviously, 6 h is long enough for the graft copolymerization in the study due to the high conversion of monomers to polymer (~98.3%). In the copolymerization, the formation of grafted branches (i.e. PAA and polyacrylate) was based on the bonding of LBG macromolecule free radicals with AA and acrylate monomers. The acrylate monomer with long carbon chain occupies a larger space than the one with a short carbon chain. The space volumes that methyl, ethyl, butyl, and ethylhexyl of MA, EA, BA, and EHA monomers occupy increase in turn. Thus, the difficulty for the four acrylates to take part in the graft copolymerization keeps increasing when they are in the vicinity of the LBG free radicals due to the increasing steric hindrance. As a result, with the increase in the carbon chain length, more acrylate monomers have to be fed into the reaction system in order to obtain similar GR. In other words, the longer the carbon chain, the lower is the efficiency (GE) for the acrylate monomers to take part in the graft copolymerization.
Effect of Carbon Chain Length of Acrylate Monomer on Apparent Viscosity
The effect of the monomer structure of the acrylates on the apparent viscosity of the LBG sizing paste is described in Table 1. All the grafted LBG pastes possessed much lower viscosity than the native LBG. In addition, the viscosity of the grafted LBG paste decreased with the increase in carbon chain length of the acrylate monomer slightly.
Apparent viscosity of LBG sizing paste and water-dissolution time of LBG sizing film.
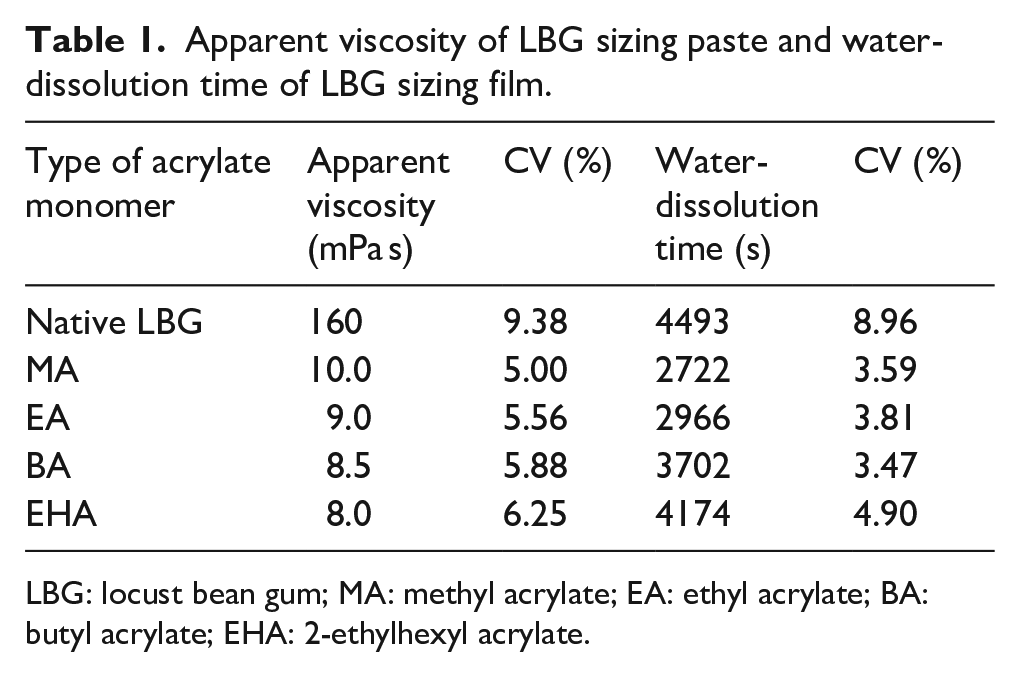
LBG: locust bean gum; MA: methyl acrylate; EA: ethyl acrylate; BA: butyl acrylate; EHA: 2-ethylhexyl acrylate.
Some investigations on the graft copolymerization of polysaccharide (e.g. sesbania gum and starch) with acrylic monomers initiated by Fenton’s reagent have been completed.19,20 On the basis of these investigations, when the molar ratio of H2O2/(NH4)2Fe(SO4)2·6H2O is 20/1, enough H2O2 can join in the redox reaction with (NH4)2Fe(SO4)2·6H2O to generate primary free radicals and initiate graft copolymerization. The molar ratio of H2O2/(NH4)2Fe(SO4)2·6H2O was increased to a higher value (50/1) in the study. There were two main reasons for the increase in the molar ratio. On one hand, enough H2O2 could be guaranteed to participate in the redox reaction and initiate the graft copolymerization of LBG with AA and acrylate monomers. On the other hand, excessive H2O2 could oxidize the LBG macromolecule, cut off its molecular chains, and thus reduce the apparent viscosity of its paste to the appropriate range (8–15 mPa s) for warp sizing.25,26
As for the grafted LBG samples, with the increase in carbon chain length of the acrylate monomer, side chains of the graft copolymers were easier to extend and their hydrophobicity kept increasing. Longer hydrophobic side chains tended to tangle or aggregate in water. 6 The hydrophobicity could reduce the intermolecular forces between the grafted LBG and water. Consequently, the apparent viscosity of the grafted LBG paste decreased therewith.
Effect of Carbon Chain Length of Acrylate Monomer on Water Solubility
The effect of the monomer structure of the acrylates on the water-solubility of LBG sizing film is also described in Table 1. All the grafted LBG films required less time to dissolve in water than the native LBG. Moreover, the water-dissolution time of the grafted LBG paste increased gradually with the increase in carbon chain length of the acrylate monomer.
Native LBG is a typical long-chain polysaccharide. Its long molecular chains arrange regularly and exhibit high rigidity. Strong intermolecular hydrogen-bond interaction makes the LBG difficult to dissolve in water. During the grafting, excessive H2O2 of Fenton’s reagent oxidized the LBG simultaneously. The oxidation reduced the degree of polymerization of the LBG remarkably. Meanwhile, the introduction of PAA and polyacrylate branches expanded the distance between the molecular chains of the LBG, broke their intermolecular hydrogen-bond interaction to some extent, and enhanced the degree of branching of the LBG. As a result, the grafting contributed to the improvement of the water solubility of the LBG film. As mentioned above, the increase in carbon chain length of acrylate monomer could enhance the hydrophobicity of the branches of LBG-g-P(AA-co-acrylate). Therefore, the longer the carbon chain of acrylate monomer, the poorer was the water solubility of the grafted LBG film.
Effects of Carbon Chain Length of Acrylate Monomer on Mechanical Properties of Sizing Film
The effects of the monomer structure of the acrylates on the tensile strength, elongation, and wear loss of LBG sizing film are depicted in Figure 5. The grafting is an efficient way to improve the mechanical properties of LBG film. With the increase in carbon chain length of the acrylate monomer, the tensile strength of the films kept decreasing while the tensile elongation increased stepwise. The LBG-g-P(AA-co-BA) sizing film exhibited the highest wear resistance. SEM images of sizing films of native LBG (a) and various LBG-g-P(AA-co-acrylate) (b–e) are shown in Figure 6.
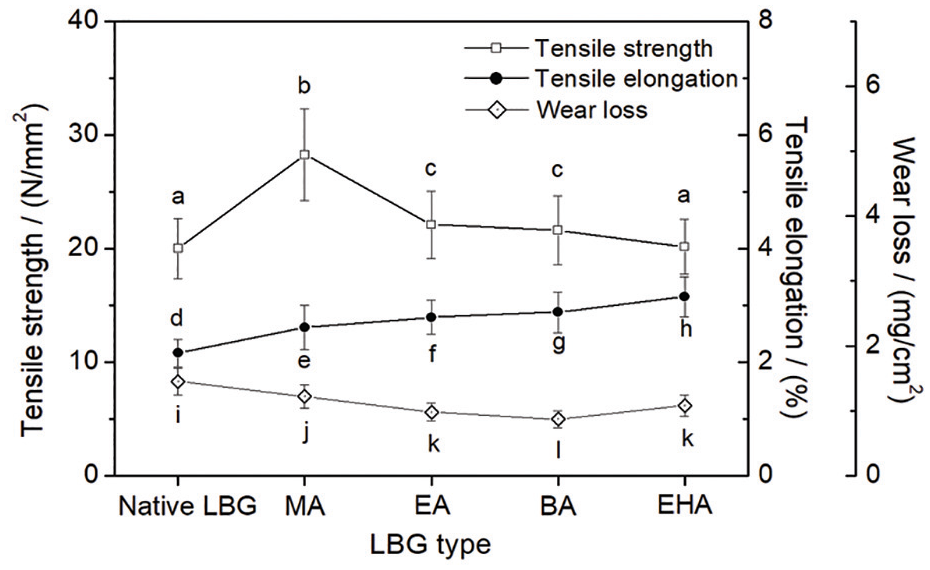
Mechanical properties of native LBG and LBG-g-P(AA-co-acrylate) sizing films.

SEM images of sizing films of native LBG (a), LBG-g-P(AA-co-MA) (b), LBG-g-P(AA-co-EA) (c), LBG-g-P(AA-co-BA) (d), and LBG-g-P(AA-co-EHA) (e).
LBG sizing film was prepared by aqueous casting method. As shown in Table 1, the improved water solubility of LBG and fluidity of LBG paste attributed to the grafting favored the formation of continuous and uniform LBG film with less water-insoluble impurity. During the mechanical evaluation, most fractures were found to occur at excessively thin or water-insoluble granule-containing parts of the native LBG film. The grafting greatly reduced the possibility of such mechanical failure. Thus, the grafted LBG films exhibited higher tensile properties and wear resistance than the native one. According to the surface morphology observation, the native LBG film had more holes and cracks than the grafted LBG ones. The former was much rougher than the latter as shown in Figure 6.
All the GRs of the LBG-g-P(AA-co-acrylate) arrived at ~19%. The introduction of such a great number of polyacrylate branches destroyed the original regularity of the LBG molecular chains and brought marked beneficial effects on the flexibility of the LBG. The flexibility of polyacrylate molecular chain is closely related to the carbon chain length of the side group of its structural unit. Several investigations showed that increasing the carbon chain length was helpful to enhance the flexibility of polyacrylate molecular chain.27,28 As a result, the longer the carbon chain of acrylate monomer, the more flexible was the molecular chain of the LBG-g-P(AA-co-acrylate) prepared. It can be speculated that the grafted LBG film will exhibit higher elongation at the expense of breaking strength by tensile stress when increasing the carbon chain length of the acrylate. The results in Figure 6 confirm the speculation well. The wear resistance of a polymer film is determined comprehensively by the strength and elongation of the film. The LBG-g-P(AA-co-BA) film possessed both moderate tensile strength and elongation in all the grafted samples. Therefore, the wear loss of the LBG-g-P(AA-co-BA) film was slightly lower than those of the other grafted LBG ones. In other words, the LBG-g-P(AA-co-BA) film showed the best wear resistance.
Effect of Carbon Chain Length of Acrylate Monomer on Contact Angle
The effects of the monomer structure of the acrylates on contact angle and surface tension of the LBG solution on polyester fibers are displayed in Figure 7. It can be observed from the figure that the grafted LBG solutions possessed much smaller contact angle and lower surface tension than the native one. The contact angle and surface tension of the grafted LBG solution increased slightly with the increase in carbon chain length of the acrylate monomer.
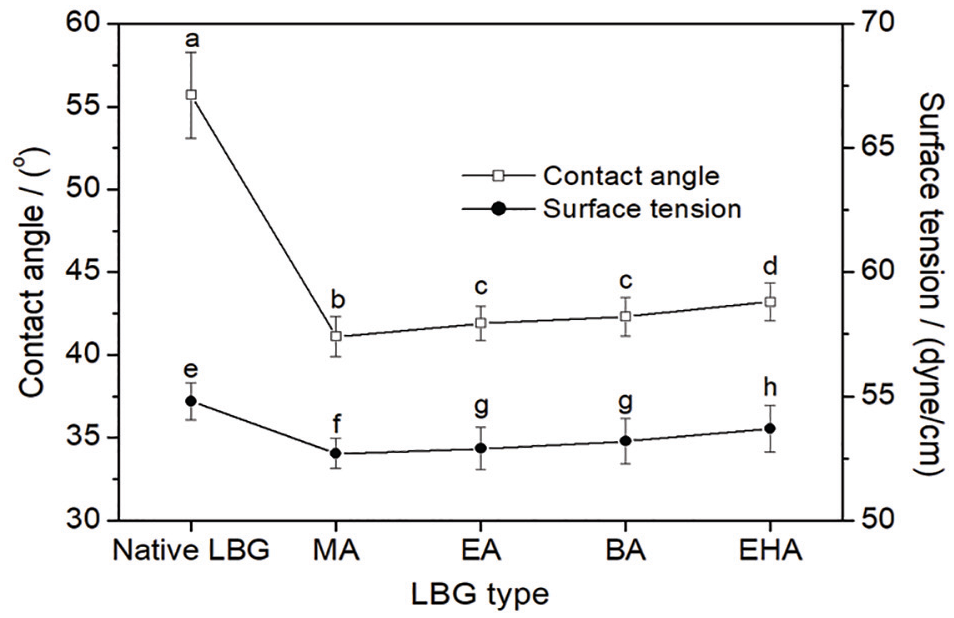
Contact angle and surface tension of LBG solution.
Contact angle is often employed to indicate the wettability of a polymer solution to fibers. High wettability means that a liquid drop can spread widely on the fiber surface and form a small contact angle. Good wetting of sizing paste to the fibers lays a solid foundation for its full spread on the surface of the fibers and penetration into the fibers. According to Young’s equation as shown in equation (1), the contact angle of a polymer solution on fibers closely relates to their interfacial tension and surface tension of the polymer solution:

where θ is contact angle of a solution on a solid; γS and γL are the surface tension of the solid and the liquid, respectively; and γSL is the interfacial tension between the solid and the liquid.
The surface tension of a polymer solution has a close relationship to the polarity of the polymer. Generally speaking, the weaker the polarity of a polymer, the lower is the surface tension of its solution. The grafted LBG prepared in this study had similar GRs (~19%), which indicated that the weight percentage of the branches grafted onto the LBG to the LBG substrate was similar. The shorter the carbon chain of acrylate monomer, the lower is the molecular weight of the structural unit of polyacrylate branches on the grafted LBG. On the basis of the similar GRs, the lower the molecular weight of the structural unit of polyacrylate branches, the larger the number of ester groups introduced into the molecular chain of LBG. When the weights of the grafted LBG samples were all the same, the LBG grafted with the acrylate with the shortest carbon chain [i.e. LBG-g-P(AA-co-MA)] would possess the most ester, which was a kind of typical weak polar group. As shown in Figure 2, with the decrease in carbon chain length of the acrylate monomer, the stretching vibration of the carbonyl of ester group of the grafted LBG increased gradually, which was evidence of the increase in the ester number of the grafted LBG. Thus, it could be speculated that, with the decrease in carbon chain of the acrylate monomer, the polarity of the grafted LBG and the surface tension of its solution should both decrease. The speculation was consistent with the variation trend of the surface tension measured in the experiment as shown in Figure 7.
Polyester fiber contains a large number of ester groups. Based on the similar dissolve mutually theory, interfacial tension between the solution of the grafted LBG containing more ester and polyester fiber was also lower. Back to Young’s equation, with the decrease in carbon chain length of the acrylate monomer, the surface tension (γL) of the grafted LBG solution and the interfacial tension (γSL) between the grafted LBG solution and polyester fiber both decreased while the surface tension (γS) of the polyester fiber was constant. Consequently, the contact angle of the grafted LBG solution on polyester fiber kept decreasing therewith.
Effect of Carbon Chain Length of Acrylate Monomer on Adhesion to Polyester Fiber
Sizing operation demonstrates that the serviceability of a sizing agent is determined by the adhesion to fibers to a great extent.29–31 Good adhesion can increase the strength of sized yarn by binding fibers together and strengthening the connection between sizing film and yarn body. It is meaningful to develop more kinds of textile sizes to possess good adhesion to polyester, for which the sizing problem has disturbing textile researchers for a long time. The effect of the monomer structure of the acrylates on adhesion of the LBG to polyester fiber is also displayed in Figure 8. The grafted LBG exhibited much better adhesion to polyester fiber than the native LBG. The adhesion of the grafted LBG decreased with the increase in carbon chain length of the acrylate monomer.
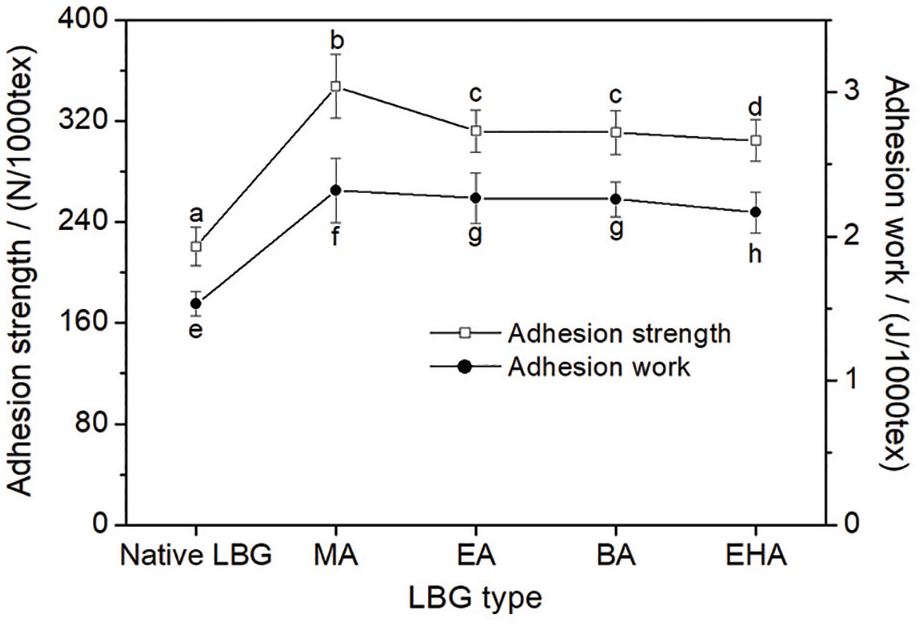
Adhesive properties of native LBG and LBG-g-P(AA-co-acrylate) to polyester fiber.
As elucidated in the above text, a large polar difference of native LBG and polyester fiber directly gives rise to weak bonding to each other and a polar–nonpolar pair can never form strong adhesion. It is infeasible to obtain sized yarns of high quality when using native LBG as a sizing agent for polyester warp. After the grafting of polyacrlate branches, a large amount of a weak polar group—ester—was introduced and thus the polarity of the LBG was decreased efficiently. The polarity similarity between the grafted LBG and polyester fiber was enhanced and thus the affinity of the LBG sizes to polyester fibers increased. Moreover, as shown in Figure 7, the grafting of acrylate monomer improved the wettability of the LBG solution to polyester fibers as well. Good wettability of sizing paste on the fiber surface is the basis of its penetration into yarns. The better affinity and higher wettability resulted in stronger adhesion of the grafted LBG to polyester fiber than that of the native one.
When the GRs of the LBG-g-P(AA-co-acrylate) were nearly the same, the number of the ester groups introduced into the grafted LBG increased gradually with the decrease in carbon chain length of acrylate monomer. Due to the improvement in the affinity and the wettability to polyester fiber, the adhesion of the LBG-g-P(AA-co-acrylate) increased with the decrease in the carbon chain length. As a result, the LBG-g-P(AA-co-MA) size exhibited the strongest adhesion to polyester fiber in all the grafted LBG samples.
Effects of Carbon Chain Length of Acrylate Monomer on Tensile Properties and Abrasion Resistance of Sized Yarn
The effects of the monomer structure of the acrylates on tensile properties and abrasion resistance of the T/C65/35 warp yarn sized by the LBG are shown in Table 2. As mentioned above, native LBG was not suitable for sizing high-content polyester yarn due to its poor compatibility with polyester fiber, excessively high viscosity and low wettability to polyester fiber. It could be seen from Table 2 that the tensile strength and abrasion resistance of the polyester/cotton blended yarns sized by the grafted LBG were better than those of the yarns sized by native LBG. With the increase in carbon chain length of the acrylate monomer, the tensile strength of the sized yarns gradually increased at the expense of the elongation. In terms of the abrasion resistance, the warp yarns sized by LBG-g-P(AA-co-BA) could resist the most friction.
Tensile properties and abrasion resistance of raw T/C65/35 warp yarn and the yarns sized by LBG.
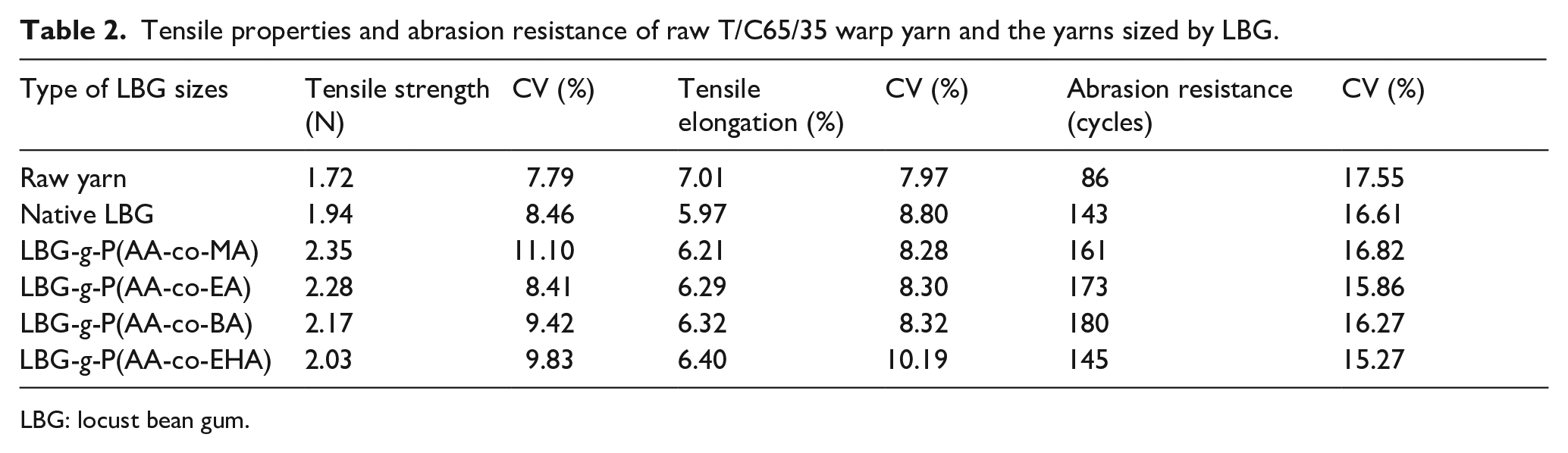
LBG: locust bean gum.
It can be observed from Figure 7 that the shorter the carbon chain of acrylate monomer, the smaller was the contact angle of the grafted LBG solution on polyester fiber. In other words, reducing the carbon chain length was more conducive to the wetting and spreading of the grafted LBG solution on the surface of polyester fiber and to the penetration of sizing paste into the T/C yarn. Therefore, the fibers in the yarn could adhere to each other much better. The cohesive force between fibers was enhanced and the tensile strength of the sized yarn was improved. Generally speaking, the shorter the carbon chain of the side group of the acrylate monomer, the lower is the flexibility of the polyacrylate chain formed. 14 As a result, decreasing carbon chain length of the acrylate could simultaneously deteriorate the elongation of both the grafted LBG sizing film coated on the yarn surface and the grafted LBG adhesive layer between the fibers in the yarn. Consequently, the tensile elongation of the sized yarn decreased continuously with the decrease in the carbon chain length.
During the weaving process, the warp yarn has severe friction with the weft yarn, dropper, heald, and reed. Thus, abrasion resistance is one of the most important mechanical properties of sized yarn and affects weaving efficiency markedly. The abrasion resistance of a sized yarn is the comprehensive embodiment of tensile strength and elongation of the yarn. The warp yarn sized by LBG-g-P(AA-co-BA) possessed both moderate tensile strength and elongation in all the sized ones and showed the best abrasion resistance.
Conclusion
The carbon chain length of acrylate monomer has marked effects on various sizing properties of LBG-g-P(AA-co-acrylate) with similar GRs. The GE, apparent viscosity, water-solubility, and mechanical properties of sizing film, wettability and adhesion to all-polyester fibers, tensile properties, and abrasion resistance of sized high-content polyester yarn are directly determined by the carbon chain length of acrylate monomer grafted onto the molecular chains of LBG. The decrease in the carbon chain length favors the increases in the GE of acrylate monomers onto LBG, the apparent viscosity of the grafted LBG paste, the water solubility and tensile strength of the grafted LBG sizing film, the wettability and adhesion of the grafted LBG paste to polyester fiber, and the tensile strength of the sized T/C65/35 yarn, but causes the tensile elongation of the grafted LBG sizing film and the sized yarn to decrease gradually. The sizing film of the LBG-g-P(AA-co-BA) exhibits the best wear resistance and the yarn sized by LBG-g-P(AA-co-BA) shows the best abrasion resistance. The modified LBG, which are grafted with acrylate monomers with different carbon chain lengths, are expected to meet various requirements in sizing process for pure and high-content polyester yarn. From the perspective of grain-saving, the graft copolymerization of LBG with AA and different acrylate monomers provides an innovative way to prepare bio-based sizing agent from forestry by-products.
Supplemental Material
sj-doc-1-aat-10.1177_24723444221103684 – Supplemental material for Effects of Carbon Chain Length of Acrylate Monomer on Sizing Properties of Locust Bean Gum-g-P(AA-co-Acrylate)
Supplemental material, sj-doc-1-aat-10.1177_24723444221103684 for Effects of Carbon Chain Length of Acrylate Monomer on Sizing Properties of Locust Bean Gum-g-P(AA-co-Acrylate) by Haojie Lu, Zhihao Ji, Manli Li, Enqi Jin and Jiu Zhou in AATCC Journal of Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported financially by the Technological Research Project for Public Welfare of Zhejiang Province (grant nos LGG21E030005 and LGG22E030002) and the Project of Key Laboratory of Clean Dyeing and Finishing Technology of Zhejiang Province (grant no. QJRZ1902). The financial sponsors do not endorse the views expressed in this publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
