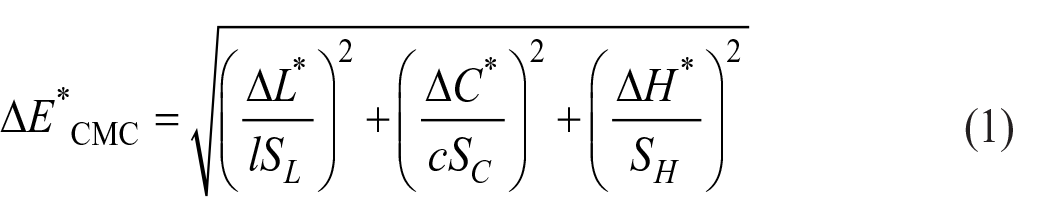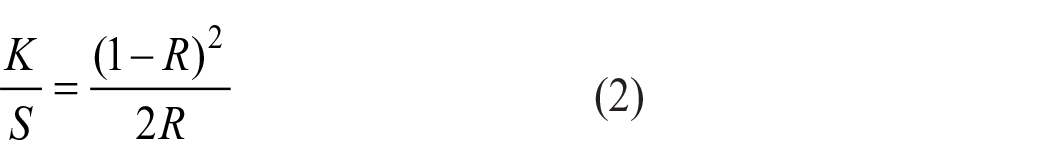Abstract
In this study, we used the new nanobubbles technology to create a green reactive washing process for the textile industry, as one of the most polluting sectors where environmentally friendly process designs are indispensable to protect the environment. With this technology, the possibility of eliminating the soaping-off step from the reactive washing sequence was investigated. For the design of an environmentally friendly reactive washing process, the effects of increasing the process temperature of the soaping-off step as well as the use of oxygen-enriched NBs in all washing steps after reactive dyeing with three different dyes were investigated. The results were evaluated by comparing the color coordinates, strength, and fastness of dyed cotton towels washed according to the conventional and alternative processes as well as examining the absorbance and chemical oxygen demand values of the washing baths. Alternative washings did not cause significant differences in color coordinates, while the lowest color strength and highest fastness values were obtained after washing with nanobubbles. The absorbance graphs showed that the most colorful baths belonged to the first bath of the nanobubble washing regime. Chemical oxygen demand measurements revealed that the alternative washing systems were more environmentally friendly than the conventional ones. Based on the results of this study, we concluded that it was possible to implement more eco-friendly washing methods by eliminating the use of soap.
Introduction
The idea of in-depth investigation of the state of liquid and colloid systems at the nanoscale has taken a new place in the field of physicochemicals. After this new field was introduced by the bubble dissolution and growth theory in the 1950s, the existence of attractive forces between the hydrophobic surfaces of nanobubbles (NBs) was demonstrated in 1994. Since then, empirical studies have been carried out and NB generators have been commercialized rapidly. 1 NBs, tiny gas bubbles with a respective diameter of <200 nm 2 , can form freely. Under the right conditions, NBs remain stable for long periods because of their high contact angle.3,4 The use of NBs in different fields to increase the efficiency of gas–liquid phase processes has been studied by many researchers in recent years. 5 NB applications have been accelerated in many sectors, including wastewater treatment,6 –10 surface cleaning,11 –14 agriculture, 1 the mining industry, 15 oil recovery,16,17 and medical applications. 18 In the textile industry, e-Flow is a new technology based on NBs developed and patented by Jeanologia. 19 With e-Flow, softening, enzyme flow and bleaching processes are performed with the use of a minimum amount of water. 20
Cotton, which constituted 24% of the world fiber production between 2018 and 2019, increased its percentage to 30% between 2019 and 2020.21,22 The reasons for the preference for cotton, which has the largest production share among natural fibers, 23 are its high strength, softness, biodegradability, biocompatibility, hydrophilicity, breathability, and lack of static electricity.24,25
Reactive dyes have become the most suitable dyes for dyeing cotton products due to their brilliancy, wide range of hues, excellent wet-fastness, ease of application, and low price.25 –27 For the dyeing of the cotton product, electrolytes (30–100 g L−1), such as sodium sulfate or sodium chloride, are used for the adsorption of the dye28,29 according to the desired color strength, while alkali (pH 11) is used for the formation of covalent bonds between the fiber and the dye.30,31 Meanwhile, the dyes remaining in the bath and not fixed on the fibers are hydrolyzed. 32 That is why, to obtain good wet fastness after dyeing, a washing process consisting of several steps is carried out causing intense water consumption. These washing steps include one or more soaping-off stages depending on the color depths.33 –35
From the sustainable and green production perspective, unfortunately, this class of dye is not environmentally friendly because of the discharge of alkaline wastewater, high concentrations of electrolytes, and unfixed/hydrolyzed dyes.25,36,37 The reactive dyes, of which up to 40% can be hydrolyzed in the dyeing process, have high affinity for fiber through van der Waals, hydrogen bonding, and ion-dipole forces. For this reason, hydrolyzed dyes are removed by a multistep washing process after dyeing and high washing fastness, which is a characteristic of reactive dyeing, is obtained. The washing process, which includes various steps such as rinsing, washing, and soaping-off, and the treatment of wastewater produced by these processes, causes high water and energy consumption, and may constitute 50% of the total cost of reactive dyeing. Accordingly, indisputably one of the biggest problems in dyeing is water and energy consumption.32,38 –40
In the literature, there are many articles on water saving,41,42 reducing the use of chemicals,43,44 treating wastewater with different methods and removing color in wastewater,45 –47 etc., within the scope of environmental production in many areas of textile. However, there are hardly any studies on the use of nanobubbles, a new technology, in the development of environmentally friendly textile processes. For this purpose, the effect of NBs, as a new technology, in reactive washing and reducing the load of wastewater was investigated. In addition, the elimination of soap usage by the temperature increase method was investigated. The success of these two methods in developing an environmentally friendly washing process was analyzed by measuring the color coordinates, strength and fastness of dyed towel fabrics, and determining the absorbance and chemical oxygen demand (COD) values of the washing baths.
Experimental
Material
In this study, pre-treated 100% cotton towel (weight 370 g m−2 produced from the warp and weft yarn count of Ne 20/2 and Ne 16/1, respectively) was used. Bifunctional reactive dyes Synozol Blue K-BR (CI. Reactive Blue 221) and Synozol Red K-HL (mixed) were supplied from EKSOY Chemicals. Another dye, monofunctional Itofix Turquoise Blue GN 266% (mixed), was supplied from Ilteks Dye and Chemicals. Sodium chloride (NaCl) and sodium carbonate (Na2CO3) were of technical grade. Acetic acid (80%) (CH3COOH), technical quality, and Isopon HDS-T (Bozzetto Group) were used in the washing processes after reactive dyeing. Dyeing and washing processes were carried out by ATAÇ Lab-Dye HT. Water with nano-sized bubbles was obtained using the nanobubble generator provided by BST Water.
Method
The pre-treated cotton towel was dyed with reactive dyes in one depth (1%) by the exhaustion method. The towel was first treated with dye (1% o.w.f.), salt (30 g L−1), and alkali (15 g L−1) at 25°C for 10 min and later the dye bath temperature was raised to 60°C (at 1–2°C/min). Dyeing was conducted at this temperature for 60 min. The liquor ratio was 10:1 in wet treatments. All treatments were repeated three times. The diagram of the reactive dyeing is given in Figure 1.
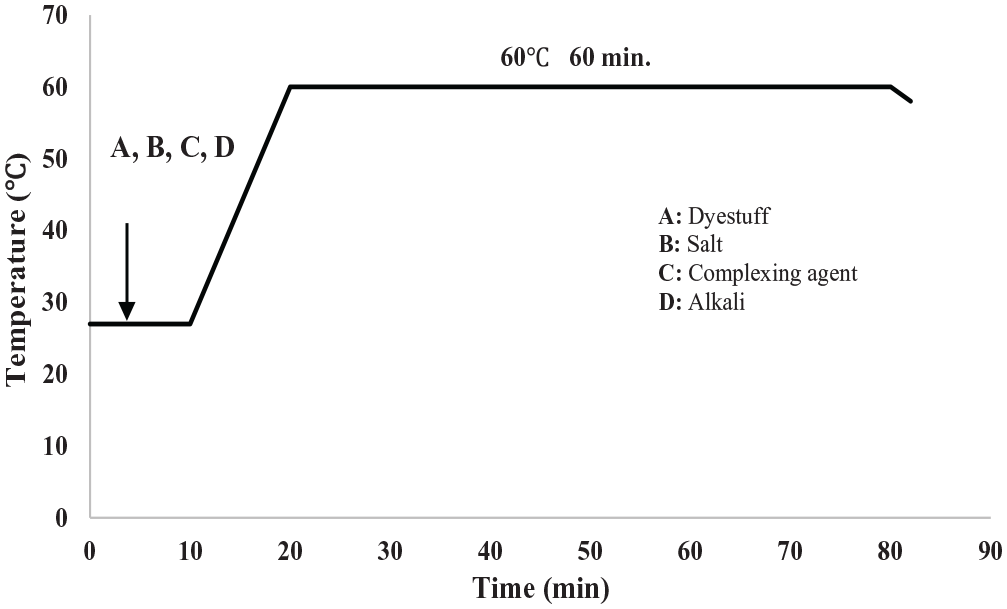
Time–temperature diagram of conventional reactive dyeing.
The reactive washing process consisted of six stages. After rinsing at medium temperature (50°C, 10 min), a second wash with acetic acid (50°C, 10 min) and soaping with 1 g L−1 soap at 85°C for 10 min were performed. Two 10-min rinsings were run at 80°C and 70°C, respectively, before a final rinse at 50°C for 10 min.
Two methods have been developed as alternatives to conventional washing (C). In the increased temperature (T) method, unlike conventional washing, the temperature of the soaping-off step was 10°C higher. In the other method, the NBs method, the use of soap was eliminated by using nanobubbles in all steps of washing. Figure 2 shows the details of the washing processes.
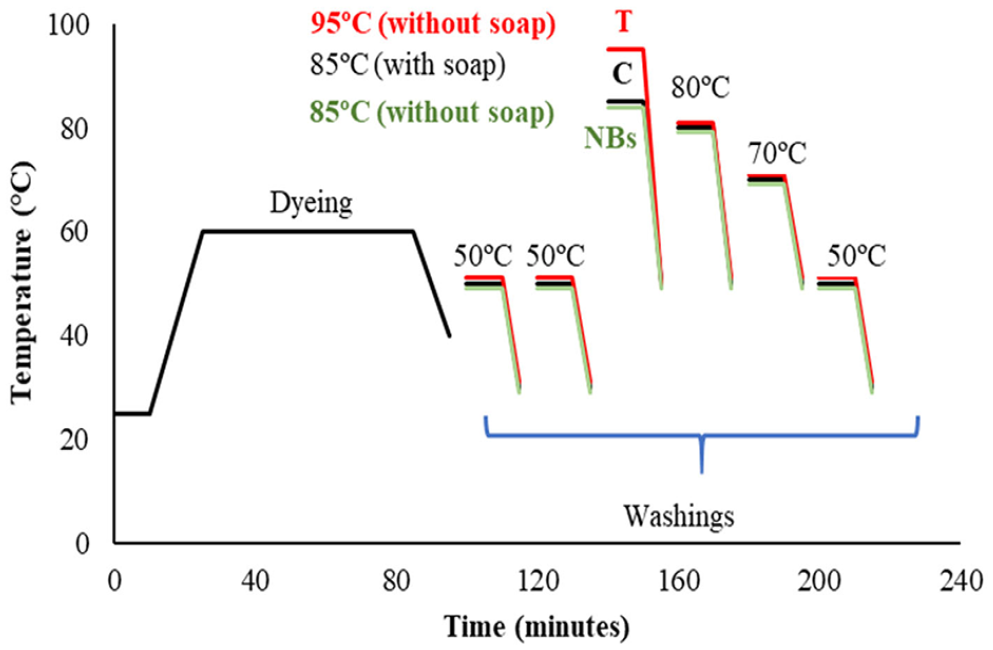
Time–temperature diagram of conventional and alternative washings.
Measurement
The L*, a*, b*, C*, and h0 values were measured using reflectance measurements (under D65 illuminant and 10 standard observers), with the Konica Minolta CM-3600D spectrophotometer. Equation (1) was used for color difference (CMC (2:1)) measurements.
In Equation (1), L and C were constants defined by the user and weight the importance of lightness and chroma relative to the hue of the measured color. SL, SC, and SH are the main weighting factors for lightness, chroma, and hue. 48
The K/S values were calculated using the Kubelka–Munk equation. The color strength (K/S) formula is presented in Equation (2).
In Equation (2), K/S is the ratio of light absorption (K) to the light scattering (S) through the sample. R is the decimal fraction of the reflectance of fabric, K is the absorption coefficient, and S is the scattering coefficient. 49
The fastness properties of reactive dyed samples were determined according to standard methods of EN ISO 105-C06/A1M (color fastness to domestic and commercial laundering) and EN ISO 105-X12:2002-12 (color fastness to rubbing).
A UV–VIS spectrophotometer (RayLeigh VIS723 G) was used to determine the absorbance values of the washing baths at 400–700 nm.
COD measurements of soaping-off baths were conducted using the photoLab S12 photometer and Merck Millipore COD cell test.
Results and Discussion
Color Coordinates
Color coordinates of towels dyed with three different colors and washed with different methods are given in Table 1.
Color coordinates of dyed cotton towels.
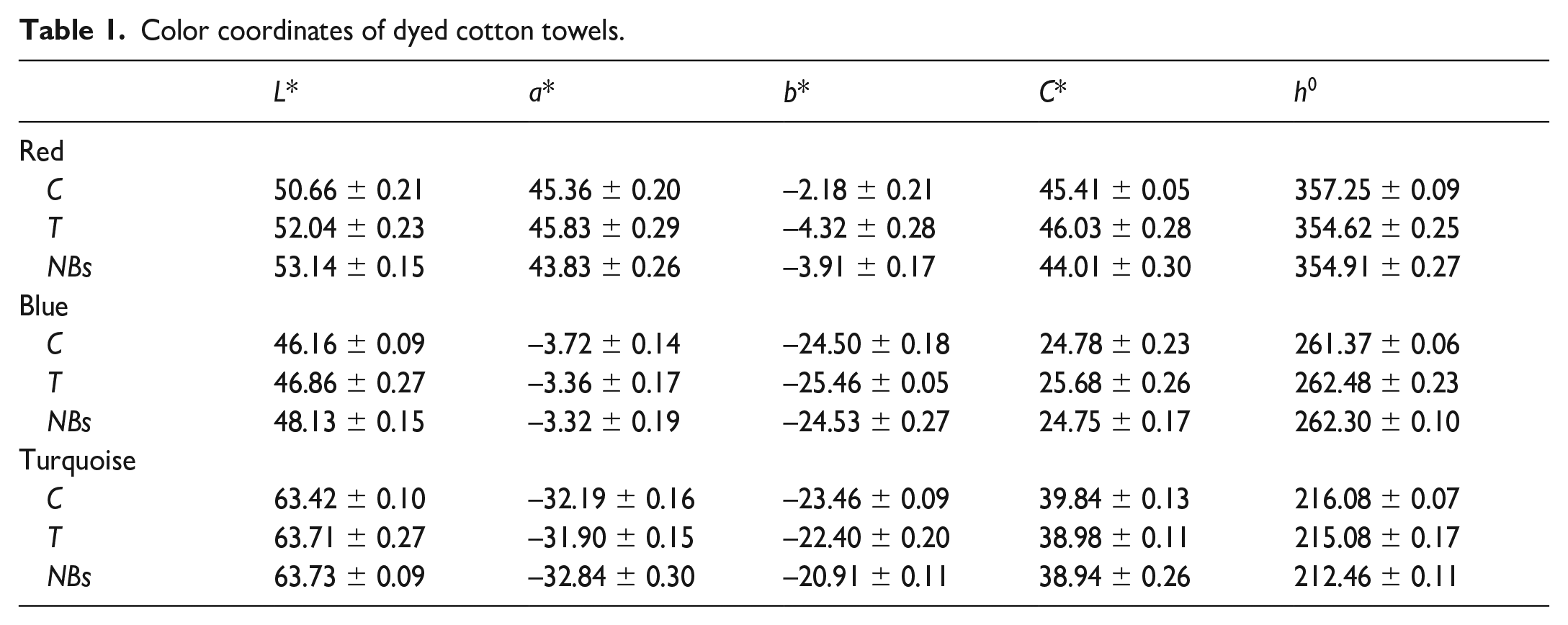
The coordinates of the dyeing using red, blue, and turquoise dyes were similar within their color groups. However, there were also minor differences. In red dyeing, the colors obtained from C and T washings were redder than that from NBs, while the blueness of T was higher than the others. In the NBs washing following the turquoise dyeing, a less blue color was obtained compared to the others, and less green color was obtained with T. The differences in the a* and b* coordinates of the blue color were negligible. These differences between washes are also visible in Figure 4.
Color Strengths and Differences
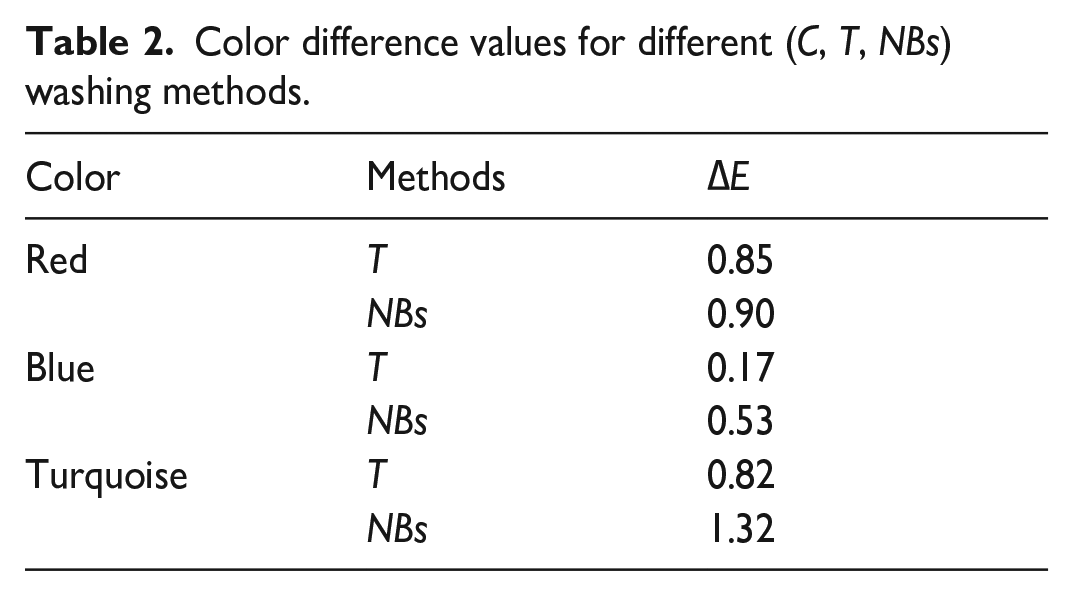
Figure 3 shows the color strengths of dyed cotton towels. The color differences caused by the washings performed with different methods after the dyeing compared to the conventional washing are given in Table 2. The reflectance values used in the color strength calculation are 530 nm for red, 620 nm for blue, and 680 nm for turquoise. In Figure 4, images of fabrics subjected to dyeing and different washing processes are given.
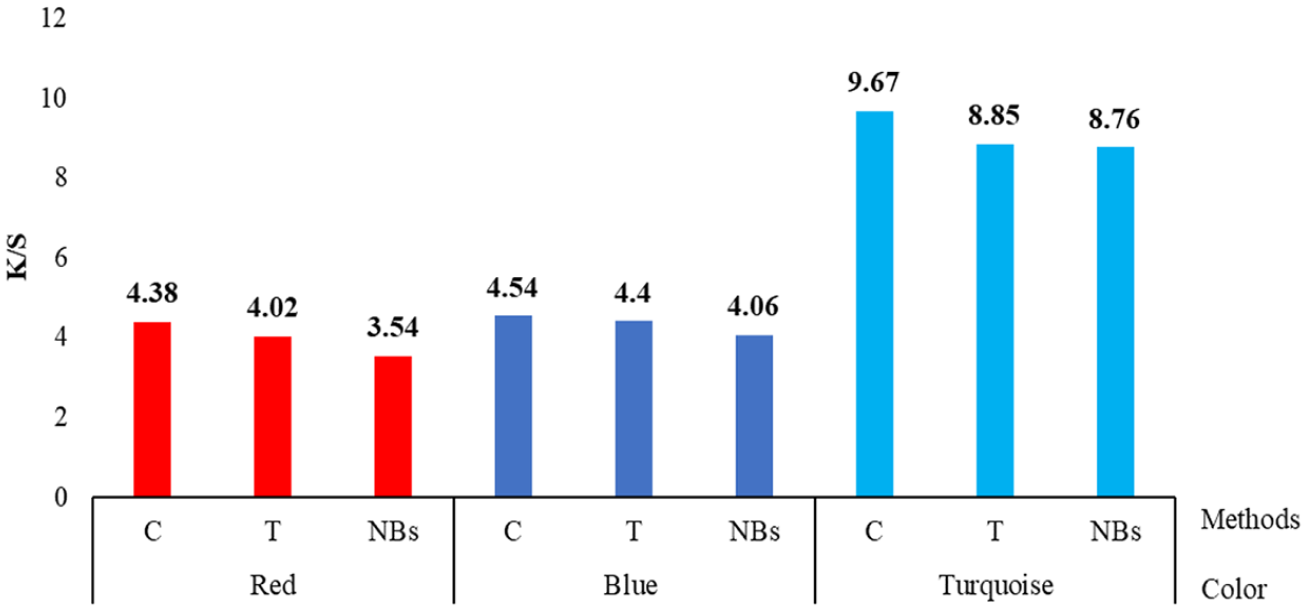
Color strengths of dyed and washed cotton towels.
Color difference values for different (C, T, NBs) washing methods.

Photographs of dyed and washed swatches.
Considering the color strengths on the basis of the color of the dye, it was observed that a higher color depth was obtained from turquoise compared to the other colors, even though an equal amount of dyestuff concentration was used in dyeings. Although the turquoise dye was monofunctional, it had higher tinctorial strength than the others, which was thought to be the reason for this situation.
The color strengths of cotton towels subjected to different washing processes after dyeing showed that the highest color depths were obtained after conventional washing. This situation was similar for dyeings conducted with three different colors, and the lowest depths were obtained after NBs washing. While conventional washing provided higher color depth compared to other methods, it resulted in lower washing fastness of approximately a 1/2 point compared to the others (Table 3). Despite the decreasing color depth with the NBs method, the increased fastnesses showed that water with nano-sized bubbles was more effective in removing the unfixed/hydrolyzed dyes on the fabric compared to the C and T methods. Especially in turquoise-dyed towels, a color difference value greater than 1 (∆E = 1.32) supported the idea that more dye was removed from the fabric surface with NBs (Table 2). On the other hand, NBs and T methods did not cause significant color difference (∆E < 1) in red- and blue-dyed fabrics compared to C. These cases about color differences were also supported by the photographs of swatches (Figure 4). It was seen that the color change was the greatest in turquoise and the least in blue after washing according to the NBs and T methods. Although these results show that NBs could be used instead of the conventional process, it was interpreted that it would require additional treatment and cost in terms of color loss and change.
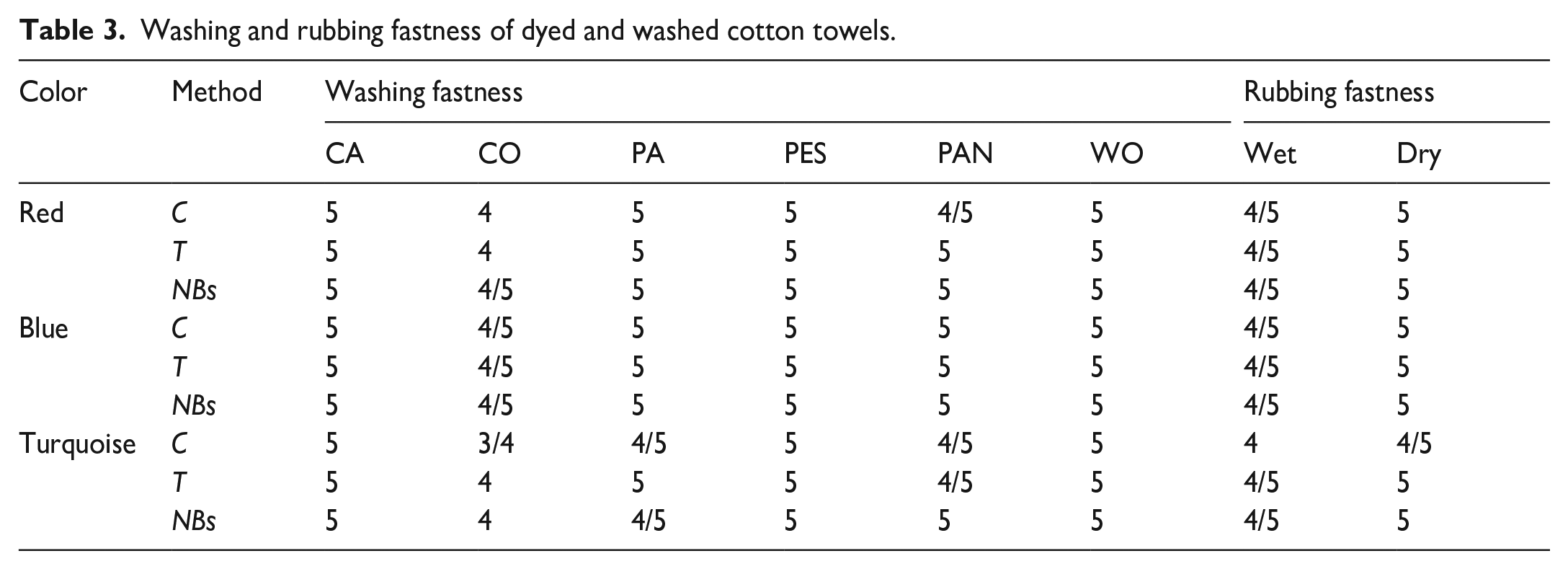
Washing and rubbing fastness of dyed and washed cotton towels.
Washing and Rubbing Fastness
Washing and rubbing fastness values of dyed cotton towels are given in Table 3.
Since reactive dyes are used in dyeing, the cotton fabrics in the multifiber stained ½ and 1 point more than the other fibers. The turquoise dye, on the other hand, stained the cotton more than the others. This was explained by the different functionalities of the dyes, that is, the blue and red dyes were bi-functional, and the turquoise was monofunctional. While the NBs washing process increased the washing fastness of turquoise- and red-dyed fabrics by approximately half a point, this difference was not observed in blue dye. Moreover, this could be supported by the lowest color difference being in blue and the highest in turquoise, as given by Table 2. Increased fastness with NBs revealed that it was more effective than the others in removing hydrolyzed/unfixed dyes. When the rubbing fastnesses were examined, it was observed that the wet rubbing values were half a point lower than the dry ones, but all of them were at good and excellent levels. Considering the color strengths of fabric (Figure 3) and functionality of dyes, it was not a coincidence that the lowest fastness values were obtained from turquoise-dyed fabrics.
Absorbance
In Figure 5, the absorbance graphs of the first and third baths of the washings are given. Pictures of washing bath swatches are shown in Figure 6. The maximum absorbance values of the first and third washings are presented in Table 4. The graphs of the first and third baths are given because they have the highest absorbance values. In the graph, numbers of baths are shown with 1 and 3. The maximum absorbance for red and blue dye solutions was recorded at 510–520 and 605 nm, respectively. For turquoise, the average absorbance values corresponding to 615 and 660 nm wavelengths were used.
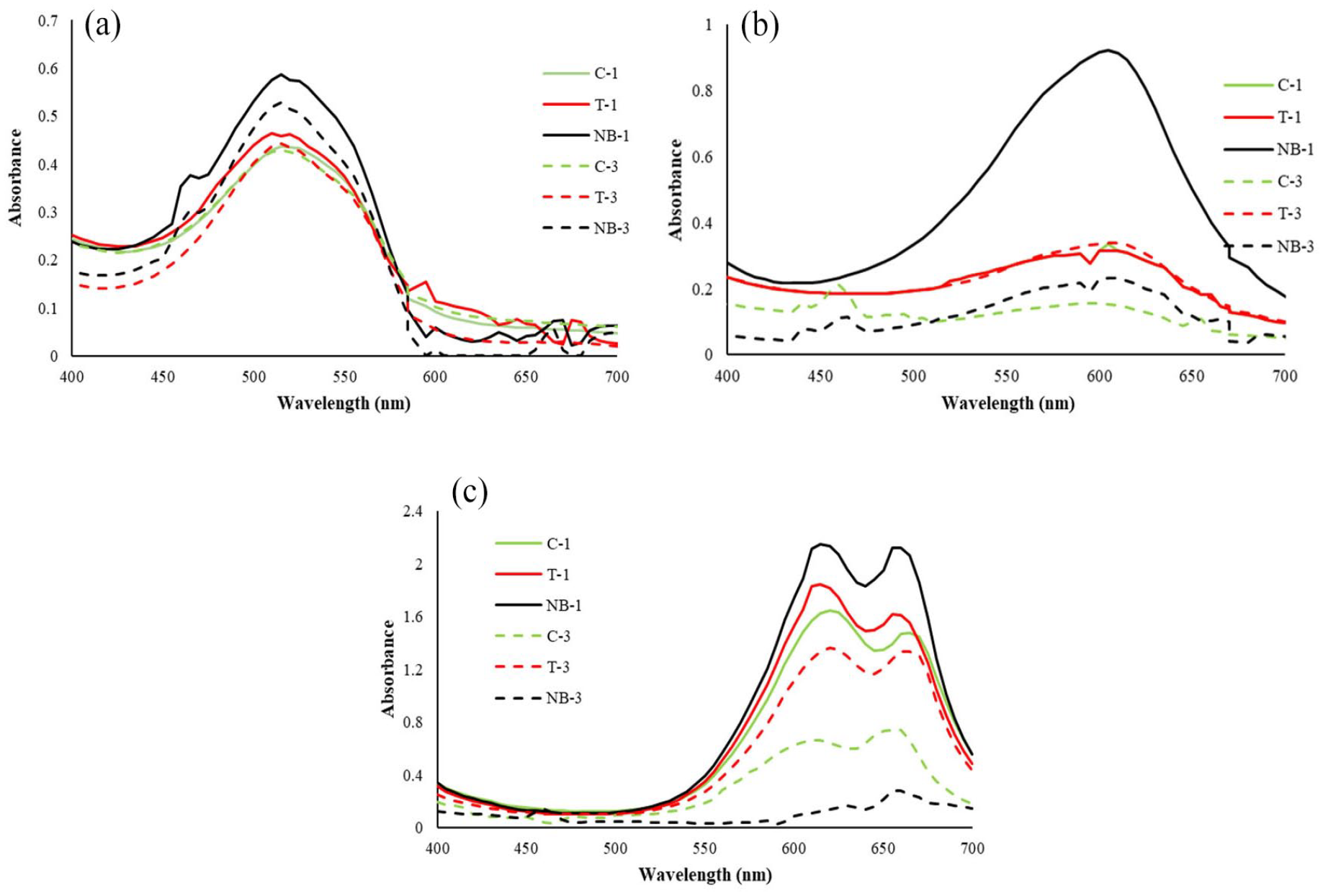
Absorbance graphs of first and third baths of washing sequences: (a) red, (b) blue, (c) turquoise.

Pictures of washing baths swatches.
Maximum absorbance values of first and third washing baths.
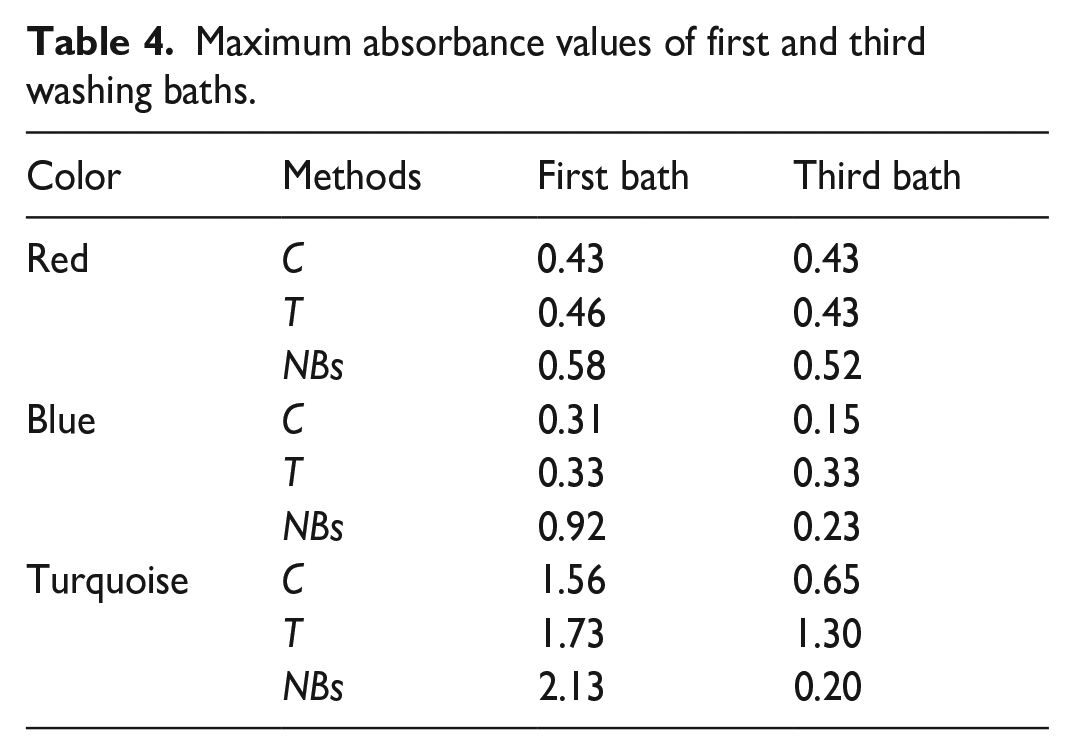
Among the first washing baths, the highest absorbance values were obtained with NBs. This means that more hydrolyzed/unfixed dyes with NBs passed into the bath. The absorbance values of the T process were also higher than that of the C process. In other words, the increased temperature increased the amount of hydrolyzed/unfixed dyes passing into the bath. This was particularly evident in turquoise washing baths (Table 4). Since a large amount of hydrolyzate was removed in the first bath, lower absorbance values were obtained from the diffusion phase (third) compared to the first bath. This was more pronounced in blue and turquoise and it is clearly seen in the bath images shown in Figure 6. Another remarkable point is that the absorbance values of the turquoise washing baths were higher than those for the others, which is supported by Figures 3 and 4. This case, which means the removal of a higher amount of hydrolyzed/unfixed turquoise dye, is also supported by the higher color difference of turquoise-dyed fabrics compared to others (Table 2). NBs and T, which had higher absorbance values than C, showed that the soaping-off step could be eliminated in washings after reactive dyeing.
COD Measurements
The COD values of the soaping-off baths are given in Figure 7.
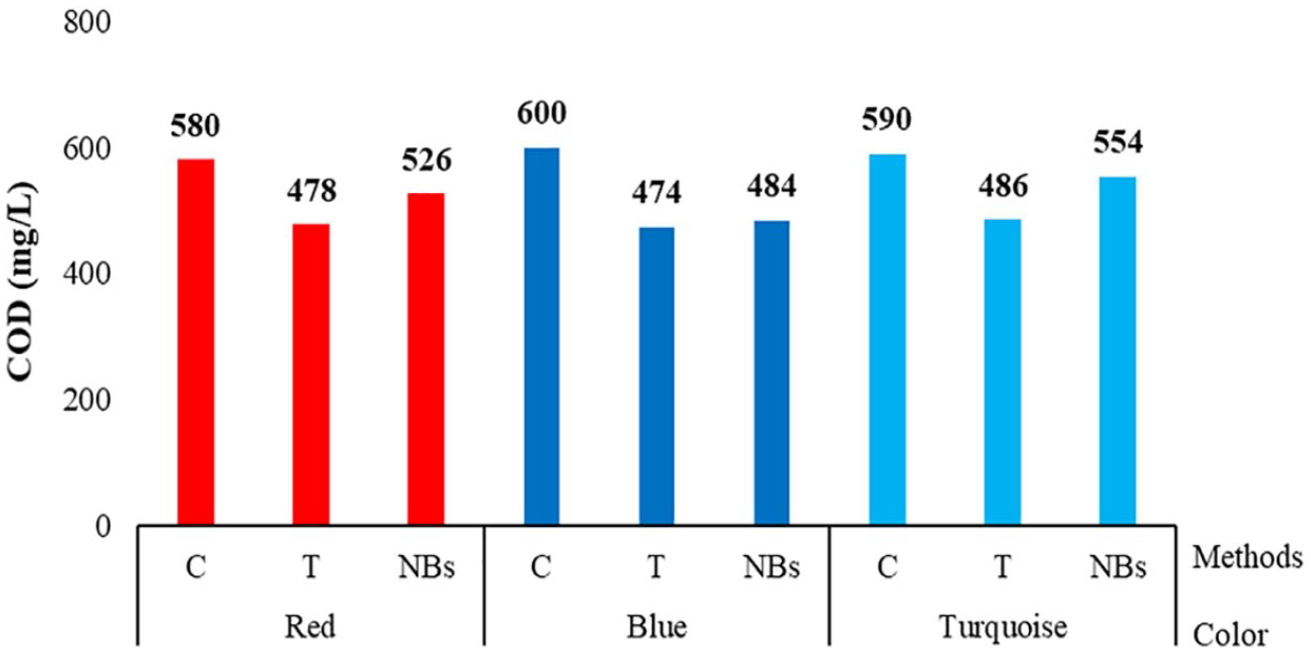
COD values of soaping-off washing baths.
It was observed that both alternative washing processes (T-NBs) provided a reduction in COD values, that is, higher COD values were obtained in the washing processes performed with the conventional method (C). The COD values of the T process were at the lowest level. The higher COD values of the NBs process compared to T were thought to be associated with more hydrolyzate removal from the fiber surface.
Conclusion
To develop an environmentally friendly washing process, the elimination of detergent use in soaping-off, which is one of the washing steps after reactive dyeing that causes intense hydrolyzate pollution, was examined with temperature increase (T) and nanobubbles (NBs). The results showed that with new technologies such as the nanobubble generator, the use of detergents in the soaping-off step could be abandoned and more ecological washings could be conducted without compromising the fastness parameters. Since these green washing processes eliminated the use of soap, they reduced the COD values of the washing baths. With the use of nanobubble technology in washing processes, the extra treatment cost of wastewater with higher color levels and the cost of the generator should also be considered. However, it was thought that these extra costs could be compensated for by the reduced washing costs by not using soap and the lower treatment costs of washing wastewater that did not contain soap. In addition, although it was thought that the color loss caused by alternative washings would increase the dyeing cost, it should be taken into account that these losses were in very low amounts. The effects of nanobubble technology in other areas of textile are also worth examining.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their sincere thanks for funding this study with the project “Yeni ve Çevreci Teknolojilerin Reaktif Boyama Sonrası Yıkamada ve Atık Suyun Dekolorizasyonunda Kullanımı (Project Number: TEYDEB 1505 - 5180061)” by the Scientific and Technological Research Council of Turkey (TÜBİTAK).