Abstract
Joint dysplasias always represent a great challenge for prosthetic surgeons. The common altered anatomical landmarks and the subversion of the anatomy of soft tissues surrounding the dysplastic joint are problems that can cause difficulties if approached with standard methods. Together with the resolution of functional issues related to dysplasia, the understanding of the underlying cause is fundamental. DNA analysis is generally performed via blood sampling; however, this might lead to misdiagnosis in case mosaicism is not detected in blood components. The etiology of genetic diseases can be further examined by means of whole exome sequencing and the detection of somatic mosaicism, recognized as a fundamental contributor to genetic diseases themselves. In this study, the clinical case of a patient suffering from a rare unilateral dysplasia localized to the left coxo-femoral and glenohumeral joint and treated at our center for reverse shoulder arthroplasty is reported. By virtue of the glenohumeral anatomical peculiarities, we had to devise a hybrid custom-made and navigated approach by means of a custom-made prosthetic stem and dedicated patient-specific instrumentation, using intraoperative GPS navigation for glenoid prosthesis. In addition, a genetic study was conducted on intraoperatively harvested bone marrow, which proved to be crucial in understanding the epigenetic basis of dysplasia. In fact, the patient resulted negative in blood but positive for a truncating variant of PTEN c.781C > T (p.(Gln261 *)) in 12% of the sequence analyzed in the bone marrow.
Case Report
A case of a 35-year-old man with congenital dysplasia of the left hip and shoulder is presented. The patient was affected by a gait deficit in the left limb since he was 10 years old. At 29 a left total hip replacement surgery was performed (Figure 1). He presented a functional range of motion (ROM) limitation to both active and passive movements and pain at the scapulohumeral joint level (Figure 2). At physical examination height was 173 cm (25°-50° percentile), weight was 81 kg (75°-90° percentile), occipital frontal circumference (OFC) was 55 cm (25°-50° percentile); discrepancy between the 2 sides of the body (right > left), doubtful left popliteal vascular malformation, enlarged right and left halluces with nail splitting, skin depression in the proximal right thigh unrelated to previous surgeries or trauma were observed. The preoperative radiological study (standard radiograms and 3-dimensional CT study) showed a peculiar dysplastic picture of the scapulo-humeral joint, with convex glenoid and saber aspect of the humeral diaphysis, flattening and retroversion of the proximal epiphyseal portion and a remarkable varus metaphyseal aspect (Figure 3(a) and (b)). Subversion of anatomy and poor joint function associated with young age inevitably required total shoulder prosthetic replacement.

Anteroposterior radiographs before and after left hip total replacement.
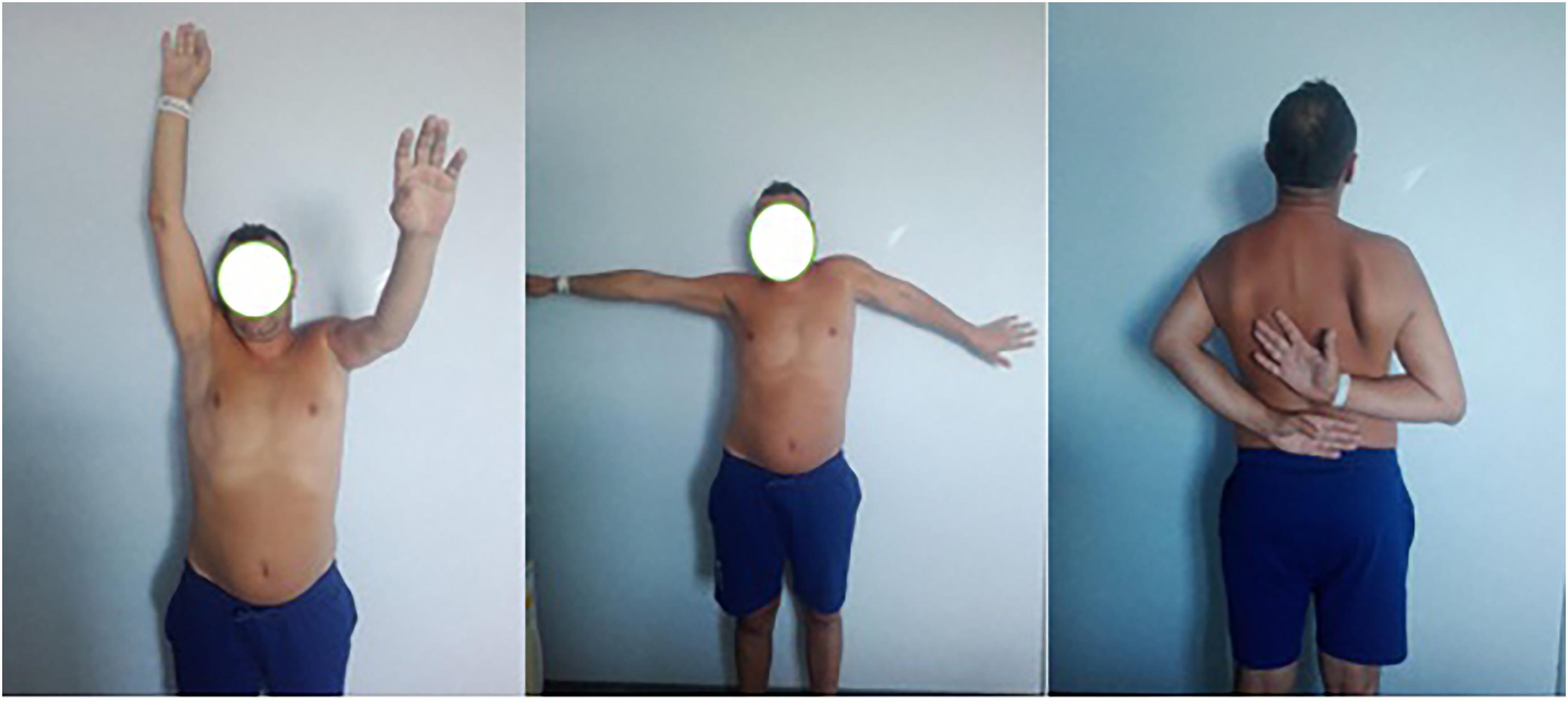
Preoperative functional assessment.

(a) Anteroposterior preoperative radiographs with arm in internal rotation, external rotation and abduction. (b) Preoperative CT scans. Morphological alterations of the glenoid and humerus are evident.
Materials and Methods
A CT scan was performed with 3D reconstructions. The obtained CT file was uploaded to the surgical planning software by the processor and turned into a 3-dimensional model; CT scans were then loaded to the reverse shoulder arthroplasty (RSA) planning software (Exactech, Gainesville, FL, USA) and interactively segmented by the manufacturer to reconstruct the shoulder in a 3-D model for preoperative planning.1,2 Glenoid features including version, inclination, and any other wear or deformity were accurately measured, while the best virtual base plate positioning was provided on the planning software by the surgeon (Figure 4). The elaborate planning involved the use of a baseplate augmented superiorly. Conversely, the 3-dimensional images obtained were sent to the Canary Islands Institute of Technology (ITC) for the production of a custom-made stem that could adapt to the saber morphology of the diaphyseal-metaphyseal area. The stem was manufactured from titanium alloy (T16Al4V ELI) with additive manufacturing (3D printing with electron beam technology—EBM) and numerically controlled (CNC) machining. A dedicated patient-specific instrumentation (PSI) 3 was consensually produced to guide the cutting of the humeral head and to broach the humeral canal with 3 progressive broaches conformed to the final design of the custom stem. An uncemented stem was implanted with a good fit. A 45° abduction brace was placed postoperatively for 4 weeks. We allowed passive rehabilitation of the shoulder in elevation only and elbow mobilization in all planes. At week 4, the brace was removed and we allowed active and passive rehabilitation of the shoulder along all planes. The patient was evaluated preoperatively with DASH and CONSTANT score clinical tests and subsequently monitored with clinical and radiological follow-up at 1-3-6 and 18 months.

Preoperative 3D CT-based planning.
DNA Extraction
DNA samples were extracted from peripheral blood and from mesenchymal stem cells isolated from bone marrow aspirate. DNA extraction was performed according to the manufacturer's instructions, using MagCore HF16 (Diatech Lab Line, Jesi, Ancona, Italy) and DNeasy Blood & Tissue Kit (Qiagen, Hilden, Germany), respectively. DNA was quantified using Qubit 2.0 Fluorometer (Life Technologies, Carlsbad, CA, USA). The Illumina DNA prep with enrichment manufacturer protocol was followed to construct the library preparation, while library fragments were hybridized to Illumina Exome Panel v1.2 (CEX) probes. Eventually, whole exome sequencing (WES) was performed on the Illumina NovaSeq6000 System (Illumina San Diego, CA, USA) according to the NovaSeq6000 System Guide. The Burrow-Wheeler aligner was used to map the reads against the hg19 reference genome. 4 An in-house pipeline which exploits the GATK Best Practices workflow was used to perform variant calling. 5 All variants were listed according to frequency, location, mutation category, literature, and mutation database data by means of ClinVar database, LOVD database, HGMD database. Polymorphisms (minor allele frequency <0.01) were excluded; intronic and synonymous variants were divided into benign and likely benign. CADD-Phred prediction tools for functional effect prediction foresaw that missense type variants are damaging, while frameshift, stopgain, and splice site variants were labeled as likely pathogenic. The following public databases were used for the interpretation of the variants: ClinVar (https://www.ncbi.nlm.nih.gov/clinvar/), LOVD (https://databases.lovd.nl/shared/genes), the Human Genome Mutation Database (HGMD, http://www.hgmd.cfac.uk/ac/index.php3).
Results
No complications occurred during surgery and the positioning obtained for the glenosphere was optimal in terms of version, inclination, and medialization, and the custom-made humeral component was adapted to the humeral dysplastic morphology (Figure 5). In the follow up at 1-3-6 and 18 months, a progressive improvement of the articular ROM, with a good functional recovery and quality of life of the patient (Constant Score 79, DASH Score 11) was obtained. No signs of early mobilization nor periprosthetic radiolucency are present (Figure 6). During the visit, the patient claimed to be satisfied with the surgery. He is able to perform heavy works (he is a bricklayer). No pathogenic variants that could explain the phenotype were detected by WES analysis on peripheral blood leukocytes DNA. The same analysis performed on bone marrow DNA was instead able to identify a PTEN mutation in 12% of the sequencing reads (43 out of 342 total reads), outlining the c.781C > T (p.(Gln261*)) variant in exon 7 of the PTEN gene. Such mutation, reported in the Clinvar and LOVD databases as a pathogenic variant, is thought to be responsible for premature protein truncation.
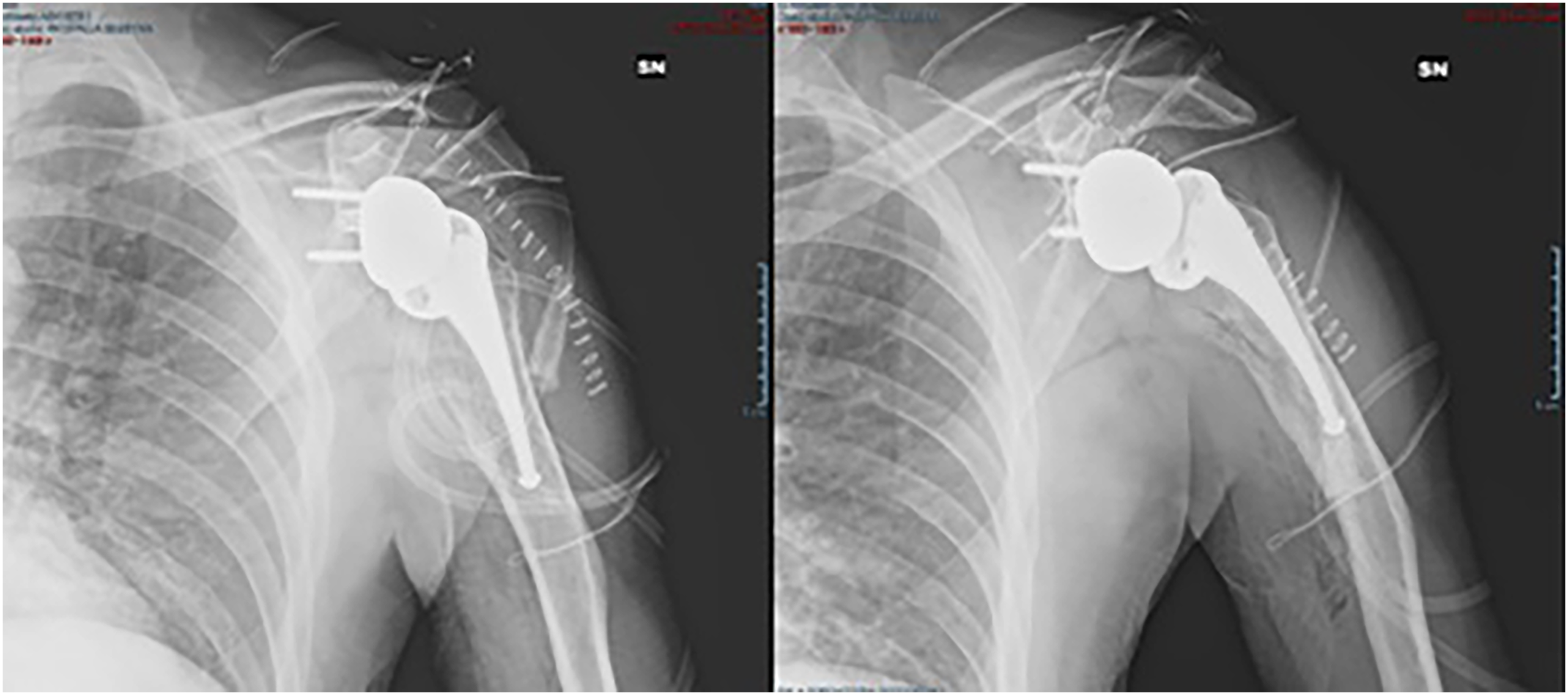
Anteroposterior postoperative radiographs.

Radiographical assessment at 18 months follow up.
Discussion
Evidence of scapular and iliac dysplasia association was described only in Kosenow’s syndrome (Plevis-Shoulder Dysplasia), although very few cases have been described.6-8 Such syndrome was first delineated by Kosenow, Niederle, and Sinios in 1970 9 as a rare dysplasia involving symmetrical hypoplasia of iliac wings and scapulae, often associated with unstable hips in infancy, a prominent lumbar lordosis, and waddling gait. No additional cases of association of ipsilateral hip and shoulder dysplasia are described in the literature. The patient analyzed in this research suffered from hip and shoulder congenital dysplasia and unilateral discrepancy, but no remarkable medical family history was recorded. However, this clinical case does not meet all the criteria associated with Kosenow syndrome, and genetic analysis on peripheral blood showed no mutations that could account for the presented phenotype. For this reason, the focus was shifted to genetic mosaicism, so that a wide variety of other morphological and clinical manifestations might be recognized. PTEN protein is responsible for a wide range of physiological and pathological processes, a wide spectrum of disorders can arise from its modifications. Germline PTEN mutations have been recognized in patients with heterogeneous tumor syndromic diseases, called PTEN hamartoma tumor syndrome, which include Cowden syndrome and Bannayan-Riley-Ruvalcaba syndrome (BRRS). 10 PTEN genes mosaic mutations are responsible for overgrowth disorders specifically limited to the tissues where the modifications occurred. Somatic mutations are at the base of the patchy distribution of lesions localized in various districts of the body. Moreover, as these variants may be undetectable in circulating blood, it is recommended to test more than 1 tissue type. In order to delineate a differential diagnosis and to deeply understand the pathways leading to the disease of our patient, WES from both blood and bone marrow was performed: a low-level truncating PTEN variant (c.781C > T; p.(Gln261*)) was found exclusively in bone marrow cells (12%). The Patient’s genome analysis proposed in this paper adds a further contribution in the understanding of the dysplastic aspect of hip and shoulder joints.
The obtained results reinforce the importance of genetic tests to be performed in different tissue samples, especially the main tissue involved. In fact, although the conventionally analyzed tissue is blood, the nonsense pathogenic variant (c.781C > T; p.(Gln261*)) was detected in bone marrow cells only. This was already demonstrated in a study performed in 2019, which recommended performing genetic analysis of additional tissues in order to unmask mosaic PTEN mutations. 11 A mosaic deletion involving PTEN in buccal mucosa (patient 1) or in a surgical specimen of the thyroid gland (patient 2) was identified, while the same alteration was absent in blood. For this reason, the diagnosis of a 10q23 deletion syndrome—clinically presented as BRRS—was established in both cases. Since pathogenic variants confined to bone marrow may lead to particular phenotypes, studies on animal models demonstrated that chondrocyte-specific PTEN deletion is responsible for skeletal abnormalities and lipoma formation. 12 As matrix synthesis and chondrocyte survival processes are modulated by PI-3/AKT pathway, in-vivo analysis showed skeletal elements overgrowth, disorganized growth plates, and accelerated hypertrophic differentiation of chondrocytes.
In conclusion, the obtained results demonstrate that deep sequencing is a key tool to detect low-level mosaicism, which can be helpful for determination of the best testing strategy for the extended family. The suggestion is therefore to consider deep sequencing whenever a high clinical suspicion of a PTEN mutation is present, particularly when a negative family history is recorded. Mosaicism detection is not correlated to an increased risk for siblings but it suggests low but indefinite risk for the offspring, according to the involvement of germinal tissues. Since PTEN is a tumor suppressor, germlines mutations are responsible for the loss of functional alleles, thus resulting in an increased susceptibility to develop multiple tumors. In fact, tumorigenesis is characterized by inactivation of the second allele, generally by means of a second-hit somatic mutation in the wild-type allele. However, in this study it remains unclear if the recorded PTEN variant is confined to bone marrow and whether the patient is prone to an increased risk of tumors or not.
Another aspect addressed in our case report was how aforementioned deformities should be approached during surgery. Because of the anatomical and functional peculiarities of the affected patient, we could not approach prosthetics with standard techniques and instrumentation, thus we had to adapt the implant to the patient's unique morphologic variables and assist navigation for proper component placement due to the absence of common landmarks. The accurate preoperative 3-dimensional CT study associated with virtual planning of glenoid component positioning—which was then navigated intra-operatively using GPS technology—was crucial. At the same time, the conformation of the humerus required the fabrication of a custom prosthetic stem that could adapt to the peculiar humeral morphology presented, as well as of a dedicated PSI instrumentation for its preparation and processing (Figure 7).

Intraoperative steps. (a) 3-dimensional humeral models and the 3 patient specific broaches. (b) Placement of the custom-made cutting guide. (c) Final result of the humeral osteotomy.
Glenoid component positioning in reverse shoulder prosthesis represents a crucial point for the success of the surgical intervention and for a correct restoration of functional capacities. The complete coverage of the baseplate surface, the correct positioning in terms of version, inclination, and off-set, as well as the positioning of the screws are fundamental. The latter must be as long as possible and completely intraosseous to avoid lesions of the adjacent vasculo-neural structures and the risk of impingement with the humeral component. However, in addition to the arthritic glenoid abnormalities in terms of wear and bone-stock, glenoid anatomy presents a notable subjective morphologic variability. Moreover, the difficult surgical exposure of such anatomical target, its limited size and the difficulty in identifying common landmarks necessary for anatomical orientation may affect proper placement and valid fixation results. Fixation screws mispositioning might be responsible for soft tissues and surrounding vasculo-nervous structures injuries, as in case of axillary nerve13,14 or suprascapularis 15 damages due to superior and inferior screws mispositioning, respectively, as well as blood vessels or even the rotator cuff itself. A dysplastic condition that totally subverts the common anatomical parameters surely contributes to further increase the surgical difficulties. In our opinion, it is not possible to face a prosthetic intervention without a careful CT study with close cuts and 3-dimensional reconstructions. The possibility to delineate a propaedeutic study and a digital planning of the surgical result to be achieved is thus fundamental; however, the radiological study alone cannot be considered the winning weapon for the optimal result. Difficulties in orientation on the operating field and the impossibility to visualize the scapular anatomy beyond the glenoid surface can lead to defective positioning or invalid fixation. Thanks to the intra-operative finding of the key features of the glenoid surface and the coupling with the 3-dimensional reconstructions captured preoperatively, GPS navigation allows to guide the surgeon step by step, continuously monitoring the version, the inclination, and the off-set of the prosthesis, thus obtaining an accurate reproduction of the planning performed (Figure 8). In our clinical case, the difficulty was also increased by the dysmorphic humeral component that could not accommodate a standard prosthetic stem. The saber aspect of the humeral diaphyseal-metaphyseal portion did not allow to locate a conventional prosthetic design in its intramedullary canal. On the other hand, the extreme lateralization resulting from the curvature of the humerus must be taken into consideration. For this reason, it was necessary to develop an ad-hoc stem that would adapt to the intramedullary anatomy and that would not increase the already present lateralization. Considering the young age, an uncemented stem was implanted. There are no clinical cases of custom humeral stems in the literature, as opposed to what concerns hip replacements.

Intraoperative GPS-assisted navigation.
Footnotes
Author Contributions
GBC, ET, AGDS, LDS, AR, NM wrote and reviewed the paper, cured editing. ET and LDS cured data acquisition, collection and analysis, interpreted the data and drafted the work. GBC, AR, and SG conceptualized the study, wrote, and reviewed the paper. All authors approved the submitted version and agreed to be accountable for all aspects of the work.
Availability of Data and Material
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
The patient gave his informed consent to data collection and their anonymous use for scientific and teaching purposes.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
At our Institution, no Institutional Review Board nor Ethical Committee Approval is necessary for retrospective studies.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
