Abstract
Introduction:
Posttraumatic arthritis (PTA) is a common sequela of proximal humerus fractures that is commonly managed with anatomic or reverse total shoulder arthroplasty (TSA). TSA for PTA is more challenging than that performed for primary osteoarthritis and frequently leads to worse patient outcomes. CPT uniformly classifies all cases of primary TSA, irrespective of procedural complexity and resource utilization. This study analyzes intraoperative differences and 30-day outcomes for anatomic and reverse TSA performed in the posttraumatic shoulder.
Methods:
Patients undergoing TSA from 2008 to 2015 were selected from the National Surgical Quality Improvement Program database and stratified according to concurrent procedures and administrative codes indicating posttraumatic diagnoses. Perioperative parameters and 30-day complications were recorded; multivariate analyses were performed to determine whether PTA was a risk factor for poor outcomes.
Results:
A total of 8508 primary and 243 posttraumatic TSAs were identified. Posttraumatic TSA patients were slightly younger (P = .003), more likely to be female (P < .001), smokers (P = .029), and diabetic (P = .003). Diagnosis of PTA was an independent risk factor for prolonged operative times ≥160 minutes (≥1 standard deviation above the mean, P = .003; odds ratio [OR]: 1.718; 95% confidence interval [CI]: 1.204–2.449) and increased bleeding requiring transfusion (P < .001; OR: 2.719; 95% CI: 1.607–4.600). Although posttraumatic TSA had a tendency for longer hospital admissions, 30-day readmissions were not significantly different between cohorts.
Conclusions:
Compared with primary osteoarthritis, a preoperative diagnosis of PTA is an independent risk factor for prolonged operative times and postoperative transfusion in anatomic or reverse TSA patients; such patients may be less than optimal candidates for same-day discharges or outpatient shoulder arthroplasty.
Keywords
Introduction
Proximal humerus fractures account for 6% of all fractures and are the third most common fracture in patients over the age of 65.1,2 The incidence of these fractures is rising secondary to increased life expectancy among an osteoporotic population.1–4 Epidemiological studies have demonstrated a parallel increase in the severity and displacement of proximal humerus fractures, placing those affected at risk for poorer functional outcomes. 2 Zyto et al. reported a 64% rate of posttraumatic arthritis (PTA) within 3 years among patients with displaced 3- and 4-part fractures treated nonoperatively or with internal fixation. 5 Possible causes of PTA include malunion and nonunion with joint surface incongruity, humeral head splitting injuries, tuberosity malunion, neglected or recurrent instability, and late-stage osteonecrosis. Ensuing pain and functional impairment from accelerated arthritic change frequently necessitates further intervention. Although commonly thought of as a treatment for natural degenerative shoulder arthritis, total shoulder arthroplasty (TSA) has become an increasingly common modality to treat sequelae of glenohumeral joint trauma.6–8
Surgical management of PTA presents a number of unique challenges. The navigation of boney and soft-tissue abnormalities from initial injury and/or prior surgery, removal of broken or failed hardware, and a higher risk of complications complicate TSA in the posttraumatic patient.8–15 The current study uses a large, multi-institutional, patient-specific data set to analyze intraoperative variables, risk-adjusted outcomes, and 30-day hospital readmissions for anatomic and reverse TSA performed in the posttraumatic shoulder. Specifically, we aim to (1) compare preoperative patient demographics and intraoperative parameters between nontraumatic TSA and posttraumatic TSA; (2) analyze the 30-day postoperative outcomes of TSA on posttraumatic shoulders; and (3) determine whether the posttraumatic shoulder is an independent risk factor for medical and surgical complications in this patient group.
Materials and Methods
Patients participating in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP) were examined. The structure of the ACS NSQIP has been described previously.16,17 In short, the program prospectively collects detailed data regarding patient demographics, preoperative comorbidities, laboratory values, and specific operative variables. Patients are then followed for 30 days after the index operation, and postoperative complications are collected regardless of whether the patient is an inpatient, has been discharged to their home or other facility, or has been readmitted to another hospital. Data are abstracted at each site by surgical certified reviewers using clinical records, physician charts, and by contacting patients directly. Surgical certified reviewers are intensively trained with continuing education courses to standardize data collection. Data definitions are rigorous and standardized across all participating institutions. Data consistency and reliability are assessed at each hospital through an on-site interrater reliability audit program. 18
Patients who underwent primary anatomic or reverse TSA were identified using current procedural terminology (CPT) codes from January 1, 2008, to December 31, 2015 (CPT 23472, Arthroplasty, glenohumeral joint; total shoulder [glenoid and proximal humeral replacement], which does not distinguish between anatomic and reverse implants). All patients with a history of posttraumatic osteoarthritis were further identified according to International Classification of Disease (ICD)-9 coding (diagnoses including traumatic arthropathy, nonunion of fracture, malunion of fracture, late effect of fracture of upper extremity) and CPT coding (removal of hardware deep, 20680, or superficial, 20670) not otherwise associated with prosthetic joint implants or rotator cuff pathology as evidenced by ICD-9 coding. All cases with incomplete diagnoses were excluded from analysis. In addition, all cases involving prior TSA, infections, periprosthetic infections, pathologic fractures, and broken or loose prosthetic joints or implants were excluded.
Preoperative demographics, comorbidities, and 30-day complications were compared between the 2 data sets. Demographics included age, gender, height, weight, and body mass index. A comparison of the medical comorbidities between cohorts is demonstrated in Table 1.
Characteristics of 8751 Patients Undergoing Total Shoulder Arthroplasty.

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; CAD, coronary artery disease; CHF, congestive heart failure; pRBC, packed red blood cells.
aCalculated using Fisher’s exact test for categorical variables and independent samples t test for continuous variables.
bIndependent samples t test.
Postoperative variables included operative time, hospital length of stay (LOS), and 30-day reoperation and readmission rates. Postoperative complications encompassed death, deep venous thrombosis, pulmonary embolism, cerebrovascular accident, myocardial infarction, surgical site infections (SSI), and postoperative transfusion involving bleeding that required at least 1 unit of transfusion. Data regarding intraoperative use of tranexamic acid, intraoperative blood loss, and transfusion criteria at each of the participating hospitals was not available in the NSQIP data set. All variables were used as defined in the ACS NSQIP user guide. 19
SPSS Statistics version 22 (IBM, Armonk, NY) was used to perform all analyses in this study. In all cases, a P value of .05 was deemed statistically significant. Univariate analysis with Fisher’s exact test was used to compare categorical preoperative variables. The independent-sample t test assuming equal variances was used to compare continuous operative details between the 2 cohorts. To demonstrate whether a diagnosis of posttraumatic shoulder arthritis was independently associated with higher risk of complications, multivariate regression analyses were performed. Candidate preoperative variables for each regression were screened from those with P < .2 and at least 5 incidences in each of the cohorts from our previous univariate analysis. 20
Results
Out of 8751 anatomic or reverse TSAs performed during the study period, 243 (2.8%) were posttraumatic cases. There were significant differences in the demographics and preoperative characteristics between the posttraumatic and control groups (Table 1). The posttraumatic group included a higher percentage of female patients (P < .001), was younger (P = .003), and had a higher percentage of current smokers (P = .029) than the baseline TSA group. In addition, the posttraumatic group had a significantly higher percentage of diabetic patients (P = .003). Although slightly higher rates of baseline liver disease (ascites), bleeding disorders, and history of preoperative transfusion were observed in the posttraumatic group, overall incidence was extremely low and not statistically significant between cohorts. No other statistically significant differences in comorbidities between groups were identified.
Perioperative metrics and postoperative complications were different between study groups (Tables 2 and 3). Differences in perioperative variables between posttraumatic TSA and standard primary TSA were seen both in terms of time spent in the operating room and length of hospitalization. Average operative times were longer by 12.6 minutes (P < .001) and hospital lengths of stay were longer by 0.3 days (P = .006). Unplanned readmissions were equivalent between groups. Although percentage of unplanned reoperation was greater among posttraumatic TSAs, statistical significance was not reached. There were no statistically significant differences in major medical complications including death, deep venous thrombosis, pulmonary embolism, cerebrovascular accident, cardiac arrest, or myocardial infarction; however, posttraumatic patients were significantly more likely to incur bleeding requiring transfusion (P < .001). Surgical site complications were equivalent among groups.
Postoperative Complication Rates by Diagnosis.
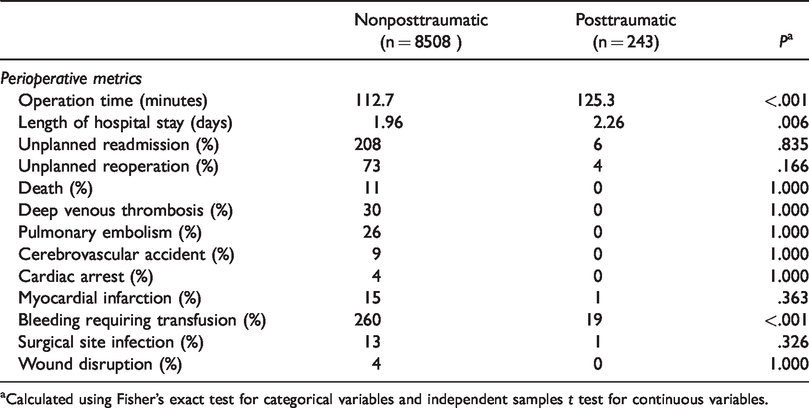
aCalculated using Fisher’s exact test for categorical variables and independent samples t test for continuous variables.
Risk-Adjusted ORs of 30-Day Adverse Events Among Patients Undergoing Total Shoulder Arthroplasty.
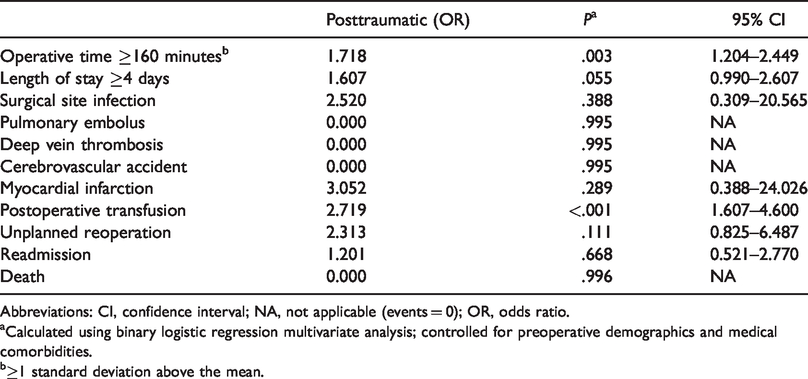
Abbreviations: CI, confidence interval; NA, not applicable (events = 0); OR, odds ratio.
aCalculated using binary logistic regression multivariate analysis; controlled for preoperative demographics and medical comorbidities.
b≥1 standard deviation above the mean.
After controlling for potentially confounding variables, the posttraumatic shoulder was shown to be an independent risk factor for prolonged operative time and postoperative transfusion (Table 3). Specifically, compared to standard primary TSA, the posttraumatic shoulder was an independent risk factor for increased operative time greater than or equal to 160 minutes, >1 standard deviation above the mean (odds ratio [OR]: 1.718; 95% confidence interval [CI]: 1.204–2.449). The posttraumatic shoulder was also a strong independent risk factor for transfusion after TSA (OR: 2.719; 95% CI: 1.607–4.600) compared to primary glenohumeral arthritis. The risk-adjusted OR for hospital stay greater than or equal to 4 days after posttraumatic TSA did not reach statistical significance (OR: 1.607; 95% CI: 0.990–2.607).
Discussion
We have demonstrated increased operative times and heightened transfusion risk after conversion TSA in the posttraumatic population. TSA in the posttraumatic glenohumeral joint is a procedure with considerable risk and difficulty compared to TSA for primary arthritis. Such procedures often require concomitant management of soft tissue and bony pathology and yield worse clinical outcomes compared to primary TSA.11–15,21 Clinical research efforts have come to outline the various considerations surgeons must make when managing the posttraumatic shoulder with arthroplasty.
Given the increased technical demands and pathology unique to TSA performed for sequelae of trauma, it is no surprise that our data demonstrated PTA to be an independent risk factor for prolonged operative times. Associated rotator cuff tears, soft tissue contractures, deltoid scarring, residual malalignment, malunion, nonunion, tuberosity displacement, bone loss, and retained hardware are potential difficulties encountered while performing TSA on a posttraumatic joint. Prior studies have addressed optimal operative strategies for the spectrum of proximal humerus fracture sequelae. Boileau et al. anatomically classified common late physical manifestations of these injuries. 9 These authors further categorized sequelae types regarding typical surgical treatment and postoperative prognosis, 9 since single- and multicenter studies have contributed to the understanding of complex TSA as conversion procedures for failed internal fixation of such fractures.10,12–14,21–27 These studies demonstrate the need for alternative surgical techniques, use of reverse or custom designed implants, and the increased rate of postoperative complications for TSA performed in the posttraumatic shoulder. In the presented study, average operative time was 13 minutes longer in the posttraumatic group, with an OR of 1.718 for operative time >160 minutes compared to the control. Prolonged surgical time not only increases patient exposure to anesthesia and potential for blood loss but may also increase the risk of SSI. Although not previously studied in the shoulder, 20 minutes of additional operative time has been associated with nearly a 25% increased risk of subsequent periprosthetic joint infection after primary total hip and knee arthroplasty. 28
Increased risk of blood transfusion after TSA for PTA has been identified. On multivariable analysis, Padegimas et al. demonstrated a diagnosis of PTA to be an independent predictor of transfusion after shoulder arthroplasty with an OR of 11.0 (95% CI: 2.9–42.2, P < .001). 29 This is much greater than the risk-adjusted OR of 2.72 in the presented study. In another single-center analysis of 407 consecutive patients, transfusion among patients undergoing TSA for sequelae of trauma was significantly greater compared to osteoarthritis: 15.8% versus 4.6%, respectively. 30 Conclusions from the literature on transfusion after shoulder arthroplasty can partially explain the higher rate of transfusion following conversion TSA in the posttraumatic shoulder. Hardy et al. showed higher intraoperative blood loss to be a significant risk factor for transfusion status-post any form of shoulder arthroplasty, primary or revision. 31 Ahmadi et al. demonstrated that increased operative time independently predicts the need for transfusion following revision TSA. 32 Increased age,32–34 female gender,30,33 history of diabetes, 32 and use of reverse-style implants 33 have each been shown to independently predict risk of postoperative transfusion. These findings reflect the typical patient population and favored implant selection for many posttraumatic TSA cases. Although there was a significantly higher percentage of both diabetics and females among posttraumatic patients in our study, we found the etiology of PTA to independently predict both prolonged operative times and transfusion risk, irrespective of diabetic and gender status. In addition to the implications for the patient, postoperative blood transfusion has the potential to affect both hospital stay and system costs.
Sperling et al. found the mean LOS following TSA for fracture sequelae to be 1.5 times the standard deviation of patients with osteoarthritis. 30 Among patients undergoing elective primary TSA, a postoperative LOS of at least 2 days has been associated with significantly increased rates of urinary tract infection, blood transfusion, and 30-day complications. 35 Although PTA appeared to be associated with prolonged admission in the presented study, this difference did not reach statistical significance when controlling for preoperative demographics and medical comorbidities. This is consistent with previous literature that correlates increased age, female gender, and medical comorbidities with longer LOS after primary TSA.36–38 The independent risk of prolonged operative time and transfusion among our posttraumatic cohort do suggest a tendency toward lengthier subsequent admissions in parallel with other literature findings. Using the NSQIP data, Dunn at al. demonstrated operative time >174 minutes (OR: 1.94, P = .0041) predict increased LOS after primary TSA. 36 Among patients who underwent shoulder arthroplasty for PTA, Sperling et al. found the mean length of hospital stay to be 5.2 versus 3.9 days for those requiring transfusions compared to those who did not. 30 Preoperative diagnoses other than osteoarthritis, rheumatoid arthritis, and cuff arthropathy, as well as osteonecrosis have also shown to be predictive of increased LOS following anatomic and reverse TSA, respectively. 37 Although Menendez et al. do not cite PTA as a potential diagnosis in their study, 37 many subjects in the osteonecrosis (a well-known complication of proximal humerus trauma) or other cohorts may have fit this presentation.
Aside from hospital LOS, inpatient versus outpatient status will become an increasingly important consideration when comparing costs between primary and posttraumatic TSA. Recent data by Leroux et el. showed no difference in 30-day readmission or adverse events between outpatient and inpatient primary TSA; however, preoperative diagnosis was not included as a variable in this analysis. 39 Increased age, female gender, and prolonged operative times were significant differences among their inpatient TSA cohort, consistent with typical patient demographics and our data on operative times for posttraumatic TSA. In conjunction with the increased risk for postoperative transfusion, the current literature does not support the practice of outpatient TSA for the strong majority of patients with posttraumatic sequelae.
Recent decades have shown a dramatic increase in the utilization of and indications for TSA.40–43 Given the ever-mounting concern for health-care cost containment, orthopedists must identify specific factors affecting the cost of shoulder arthroplasty. From 1993 to 2010, average hospital charges for TSA increased at a rate greater than that of both coronary bypass and total hip arthroplasty (THA), while average hospital charges in 2010 for TSA were more than double that of THA. 44 Research has demonstrated the increased technical demand, need for advanced implants, and risk of poorer long-term outcomes in cases of posttraumatic TSA. NSQIP data demonstrated that hospital admissions for posttraumatic TSA are associated with a 12.38% significant increase in cost compared to that of TSA for primary osteoarthritis, irrespective of rehabilitation, readmissions, and complications treated outside the initial hospital stay. 44 Our presented data further highlight PTA as an independent risk factor for greater hospital resource consumption through increased operative times, need for blood transfusion, and a tendency for prolonged LOS among the affected population.
Despite these findings, there is no categorical differentiation between primary TSA for osteoarthritis and TSA for the posttraumatic joint in the United States. CPT has a single code for primary TSA (CPT 23472, Arthroplasty, glenohumeral joint; total shoulder—glenoid and proximal humeral replacement) and 2 codes for revision TSA (revision of TSA, including allograft when performed; humeral or/and glenoid component, CPT 23473/CPT 23474). 45 Recent research has demonstrated differences in intraoperative parameters and 30-day outcomes between total knee arthroplasty performed as primary versus conversion procedures in patients with PTA. 46 Furthermore, current procedural classification already differentiates “conversion THA” in the posttraumatic, previously operated on hip (conversion of previous hip surgery to THA, with or without autograft or allograft, CPT 23132) from primary THA, as well as 3 separate revision THA codes. 45 For the shoulder, however, posttraumatic conversion cases are coded and bundled as primary TSA, irrespective of procedural complexity, resource utilization, and outcomes. Amidst the contemporary emphasis on health-care cost containment, arguments can be made that posttraumatic shoulder arthroplasty ought to be considered separately from primary TSA. Whether coded as a revision or reclassified as a conversion/complex procedure, such practice would better reflect the resources needed for the procedure, improve documentation, and allow for appropriate institutional reimbursement. Heightened costs associated with TSA in the posttraumatic shoulder may create a relative disincentive to undertake complicated conversion procedures in a population with already high rates of end-stage arthritis, which will only increase as more primary TSA are being performed on an outpatient basis. Classification reform could be a significant step forward in quality of care.
We acknowledge several limitations to this study. First, ACS NSQIP represents hospitals with a presumed interest in quality improvement and the financial means to participate. 17 These institutions are typically large, urban academic centers that may differ from other acute care hospitals in the United States. 17 Despite the 517 participating hospitals included, postoperative complications and readmission rates may not reflect nationwide trends. Second, we attempted to capture all patients undergoing TSA who had a prior history of shoulder trauma. Limitations in reporting prevented us from assessing severity of injury, degree of intraarticular involvement, or the time interval between injury and arthroplasty. We also cannot fully assess the degree of PTA, control for baseline shoulder function, or track orthopedic specific outcomes (range of motion, pain, functional outcomes). Third, as CPT coding does not differentiate reverse from anatomic TSA, we are unable to report potential differences between patients receiving one form of implant versus the other. The NSQIP data additionally does not capture incidence of intraoperative fracture or subscapularis failure as potential complications associated with posttraumatic TSA. This study only tracks 30-day outcomes and may underestimate longer term complications potentially associated with posttraumatic TSA—such as subscapularis failure and infection. Finally, outpatient complications and readmissions to non-ACS NSQIP hospitals are potentially missed by chart reviewers, which would also increase the proportion of postdischarge events. Despite these limitations, ACS NSQIP offers a highly standardized source of clinical data for the accurate assessment of surgical quality. 18 It should be noted that the posttraumatic subset represents only 2.8% of TSA patients, and thus, the absolute number of complications may not be clinically significant for individual hospitals. It is difficult to assess whether there is any clinical significance regarding the increase in operative time and hospital LOS for individual patients. However, we believe that operative times and LOS are significant from a hospital’s financial standpoint, and that inappropriate reimbursement for a several events per year can have a large impact over a longer time course. The epidemiologic trends of osteoporotic fractures, increasing numbers of TSA performed, and emphasis on outpatient surgery will continue to magnify this impact in the future.
Conclusion
The presented study is the first of its kind to evaluate independent risk factors associated with patient demographic factors, hospital resource utilization, complications, and readmissions associated with performing anatomic and reverse TSA for posttraumatic shoulder arthritis. We have demonstrated increased operative times and transfusion requirements after conversion TSA in this cohort. In contrast to THA, current diagnosis and reimbursement schemes do not differentiate posttraumatic patients from atraumatic groups undergoing TSA. Our data add to the current literature regarding the increased technical demands, advanced implant requirements, and long-term complications associated with TSA performed on the posttraumatic joint. Especially, as emphasis on outpatient TSA increases, we believe that classification reform would improve medical documentation and improve patient care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Each author certifies that he or she has no commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements) that might pose a conflict of interest in connection with the submitted article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
