Abstract
The subscapularis is the largest and most powerful muscle of the rotator cuff. Occupying the vast majority of the subscapular fossa, it is the only internal rotator of the rotator cuff. The subscapularis innervation is classically taught as a dual innervation of 1 upper subscapular and 1 lower subscapular nerve arising from the posterior cord of the brachial plexus. However, there is a large amount of research that suggests there is significant variance in the innervation of the muscle from multiple upper subscapular nerves to multiple lower subscapular nerves arising from various portions of the plexus. Although one of the main functions of the subscapularis is to internally rotate the humerus, there is substantial evidence that displays its importance in glenohumeral stability as well. The insertion of the subscapularis is both tendinous as well as muscular. The more superior tendinous portion inserts on the lesser tuberosity while the more muscular portion inserts inferior to the less tuberosity. The medial to lateral spread of the insertion is quite variable ranging from only on the lesser tuberosity to merging with fibers from the supraspinatus. Understanding the anatomy of the subscapularis improves subscapularis management during shoulder arthroplasty including techniques for takedown, release, and repair.
Keywords
Introduction
The initial descriptions of shoulder anatomy were recorded in the 500s BC. 1 From these writings, the design and function of the subscapularis has been partially understood. The subscapularis is the largest and most powerful muscle that exists in the rotator cuff. 2 Furthermore, it is the only internal rotator of the 4 rotator cuff muscles.1,3 Although other internal rotators of the shoulder exist (pectoralis major, teres major, and latissimus dorsi), the subscapularis remains one of the most important. 4 The subscapularis occupies the vast majority of the subscapular fossa on the anterior portion of the scapula with a complex insertion onto the humerus. 5 The following review details the known anatomical elements of the subscapularis muscle and tendon insertion and how the anatomy relates to performing a shoulder arthroplasty.
Innervation
The subscapularis is innervated by both the upper subscapular nerve (USN) and lower subscapular nerve (LSN).1,6 Both the USN and LSN arise from the posterior cord of the brachial plexus. 5 The USN and LSN receive contributions from both C5 and C6. 5 However, there is a significant amount of anatomical variation with regard to these nerves (Figure 1).
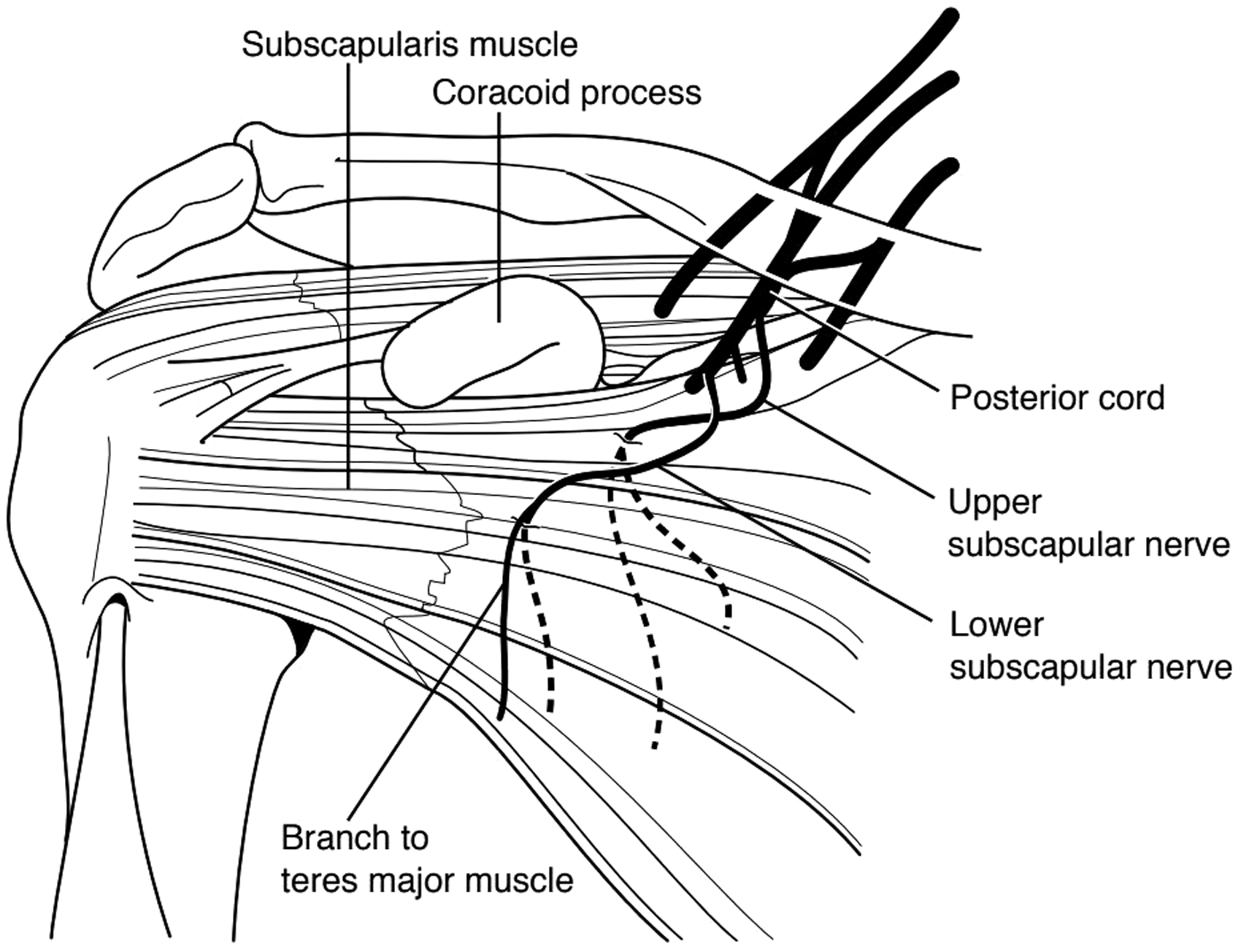
Innervation of the subscapularis muscle including the upper and lower subscapular nerves.
USN is the most consistent, being found to have a limited amount of anatomic variation. The most common anatomic finding is 1 USN innervating the superior muscle belly. 7 Several studies have done anatomic dissection of the USN. In combination, 51% had 1 USN while 45% had 2 USNs. The remaining 4% was accounted by 2 shoulders that had 3 USNs.7,8 The overwhelming majority, greater than 90%, of these nerves originated from the posterior cord of the brachial plexus.7–9 One study, that only quantified the number of nerves to the subscapularis as opposed to naming them, had 56% of the most superior nerves to the subscapularis arising from the posterior division. The remaining superior nerve fibers arose from the posterior cord. 9
The LSN has a much larger variance. The most common variant, around 79%, is 1 LSN arising from the posterior cord of the plexus.7,8 However, numerous variants exist including 2 LSN from the posterior cord, 2 LSN from the thoracodorsal nerve, and 1 or 2 LSN from the axillary nerve. The LSN was always found to innervate the more inferior muscle belly of the subscapularis.7–9
In another descriptive study, 82% of shoulders contained 3 nerves that help innervate the subscapularis and 16% had 4 nerves. 9 In fact, the study found that it was the least common to only have 2 nerves innervating the subscapularis. This study further demonstrates the wide range of anatomic variation associated with both the USN and LSN. 9
The USN mostly receives fascicles from C5 and C6. There is evidence to suggest that a small minority of USN receives contributions from C7; however, when it does it is small and is most likely insignificant. However, the LSN contains significantly more contribution from the C7 nerve root. Chen et al. found that the C7 nerve root provided 40.5% of cross-sectional area of the LSN. The C5-C6 nerve roots provided the remaining 59.5%. 8
The site of innervation of both the USN and LSN occurs in a fairly predictable pattern. At the medial border of the coracoid, the most superior innervating nerve enters the muscle anywhere from 15 to 38 mm inferior to the upper border of the muscle on its superficial (anterior) surface. The most inferior innervating nerve enters the muscle anywhere from 35 to 56 mm inferior to the upper border of the muscle.9,10 In between these insertions can be anywhere from 0 to 3 nerves. 9 However, they all insert in a fairly linear pattern between the most superior and most inferior nerves. 9 Furthermore, the position of the shoulder significantly affects the location of these nerves in relation to anatomic landmarks. Denard et al. looked at the difference of insertion based off the medial border of the coracoid in neutral and external rotation. The average distance of the USN from the coracoid base was 31.6 ± 6.6 mm (range, 22–45 mm) in neutral rotation and 24.2 ± 7.4 mm (range, 11–35 mm) in external rotation. The LSNs insertion point averaged 42.6 ± 7.6 mm (range, 25–55 mm) from the coracoid base in neutral rotation and 33.9 ± 6.9 mm (range, 24–45 mm) in external rotation. They found that all the nerves innervating the subscapularis moved significantly closer to the coracoid with external rotation when compared to the shoulder in neutral rotation. 10
The subscapular nerves may be potentially injured during shoulder arthroplasty as a result of surgical release of the subscapularis tendon. Given the proximity of the innervation to the upper border, care needs to be taken during superior, medial, and anterior release especially of the coracohumeral, superior and middle glenohumeral ligaments. Similarly, blunt dissection on the undersurface of the conjoined tendon superficial to the subscapularis needs to be performed with care as these nerves may be subject to injury. During repair of the subscapularis, fixation should not be passed through the muscle or tendon medial to the conjoined tendons and coracoid as the fixation may place the nerves at risk for injury. Finally, muscle-splitting approaches in the setting of instability repairs may place the USN and LSN at risk. There is clearly a zone between these 2 nerves as they enter the muscle therefore staying lateral to the conjoint during initiation of the split, splitting in the middle of the muscle from the upper to lower borders and blunt dissection of the interval as it is developed medial to the conjoint will potentially limit iatrogenic injury.
Insertion/Origin
The origin of the subscapularis is predictable and arises from the anterior portion of the scapula called the subscapularis fossa.1,5,11,12 The muscle occupies almost the entire fossa making it the largest muscle attached to the scapula.2,5
The insertion of the subscapularis muscle has been well described. The majority of the subscapularis inserts into the lesser tuberosity.1,5,13 The shape and width of that insertion have been evaluated by several authors with some differences in the findings. There are a several consistent findings among the studies. First, the tendon has a wider insertion more superiorly and tapers inferiorly and is shaped liked a trapezoid.14–17 Richards et al. likened the shape of the insertion to the outline of the State of Nevada. 14 The typical dimensions are approximately 2 cm in a mediolateral dimension and 2.5 cm in a superioinferior dimension. Second, there is both a tendinous and muscular insertion of the subscapularis. The tendinous insertion is the majority of the insertion and is roughly the superior 60% of the footprint. The muscular portion is the inferior 40% and consists of direct attachment of muscle fibers to bone.1,11–13,15,18 The muscular attachment is distal to the lesser tuberosity and inserts onto the anterior humeral metaphysis.11,16,17 The tendinous portion of the insertion is created from 4 to 6 collagenous bundles that run superiorly as the tendon travels laterally toward the lesser tuberosity.12,19 The overall tendon insertion length from superior to inferior has been described to range from 2.5 to 6 cm.13,14,16
Some controversies exist with regarding the insertion of subscapularis. Many authors have researched whether the tendon exclusively inserts onto the lesser tuberosity or extends beyond in the medial to lateral direction. Although the textbooks and some studies suggest that the tendon inserts exclusively on the lesser tuberosity,13,16 the majority of the studies would argue that the tendon reaches lateral to the tuberosity.11,14,20,21 Cadaveric dissection with microscopic assessment along with magnetic resonance imaging studies have shown that the tendon extends at least as lateral as the bicipital groove, and the majority of specimens have attachments on the greater tuberosity as well.11,18,19 These lateral attachments mostly travel anterior to the long head of the biceps (LHB); however, some travel both anterior and posterior enveloping the LHB. Some have described this extension of the subscapularis tendon lateral to the bicipital groove as the transverse humeral ligament. Most recent histological and cadaveric studies refute the existence of a discrete transverse humeral ligament but rather support a lateral extension of the subscapularis tendon. 18 Care must be taken when releasing this in the setting suprapectoral biceps tenodesis as preparation of the tendon with transverse humeral ligament release may weaken this lateral extension.
Knowledge of the insertion of the subscapularis aids in takedown and repair techniques. If a tenotomy is performed, understanding the dimensions of the footprint can guide location of tenotomy such that a sufficient stump is left attached to the lesser tuberosity for repair. In general, the tenotomy should be performed about 2 to 2.5 cm medial to the medial edge of the bicep groove to allow adequate stump for repair. If an osteotomy is performed, care should be taken to remove the entire tendon with bone; therefore, a 2 cm wide and 2.5 cm superior inferior length osteotomy piece should be planned during removal and avoid a significantly smaller or larger piece.
Mechanism of Action
The primary mechanism of action of the subscapularis has traditionally been understood to be internal rotation of the glenohumeral joint, though as further investigation has shown, the subscapularis likely plays a more multifaceted role in facilitating glenohumeral motion. Furthermore, it has been hypothesized that the subscapularis plays a dynamic and passive role in providing anterior glenohumeral joint stability, though the specifics of this role remain debated.
When considering the function of the subscapularis, it is important to account for the multipennate nature of the muscle as well as its dual innervation. As the subscapularis has a broad origin involving 90% of the anterior surface of the scapula, the mechanical advantage of the superior portion of the muscle is substantially different than the inferior portion of the muscle; furthermore, the moment arms of the subscapularis are affected by the position of the glenohumeral joint.22,23 In a cadaveric study by Halder et al., the stiffness of the superior portion of the subscapularis was significantly higher in 60° of glenohumeral abduction (correlating to 90° of shoulder abduction) compared to the hanging arm position. 22
The subscapularis is activated most consistently with an internal rotation force at the glenohumeral joint.24–26 From a clinical perspective, this concept can be best demonstrated by the Gerber lift off test, which has been shown to be the optimal clinical test for reliably activating the subscapularis.27,28 The Gerber lift-off test is performed by pushing the hand posterior and lifting off posteriorly from the mid-lumbar spine, which both extends and internally rotates the shoulder. 29 The other clinically useful tests of subscapularis function include the belly-press test and the bear-hug test.30,31 The belly-press test is performed with the arm at the side and the elbow flexed with the patient pressing palm into his or her abdomen by internally rotating the shoulder.30,31 A positive test for subscapularis dysfunction is considered when the patient pushes the hand against the belly with a flexed wrist. The bear-hug test is performed with the palm of the involved side placed on the opposite shoulder with fingers extended and the elbow anterior to the body.30,31 The patient is then asked to hold the position as the examiner tries to pull the patient’s hand from the shoulder with an external rotation force to the forearm. The test is positive when the patient cannot resist the examiner and when weakness is exhibited when compared to the contralateral side. Yoon et al. determined that for differentiating any tears from an intact subscapularis, the most sensitive test is the belly press (27.8%) and the most specific is the lift-off test (100%). For differentiating a full-thickness tear from a partial tear, the most sensitive test is the belly-press test (56.8%) and the most specific is the lift-off test (96.9%). 31 The authors recommend performing all 3 tests to accurately assess the function of the subscapularis.
When considering the overall function of the rotator cuff with regard to shoulder mechanics and motion, one aspect that has been investigated is the role of the inferior cuff (subscapularis, infraspinatus, and teres minor) in maintaining glenohumeral positioning, specifically in resisting the superior shearing force of the deltoid during abduction.24,32–34 It is this function that allows the deltoid to retain its mechanical advantage, rather than simply driving the humeral head into the acromion during forceful abduction. In an early set of observations made by Inman et al. in 1944, it was noted that the subscapularis activity peaked around 60° to 80° of abduction as it worked to resist humeral head migration. This activity could be further isolated by creating a rotatory resistance and weaken the subscapularis and hinder the abduction force coupling. 34
However, it is worth noting that the subscapularis likely plays a role in not just limiting superior migration of the humeral head but also in maintaining the anteroposterior position of the humeral head. This was demonstrated by both Rathi et al. and in a systematic review by Sangwan et al., where the rotator cuff musculature was found to limit anteroposterior translation of the humeral head.24,26,35
The subscapularis also likely contributes to glenohumeral stability and prevention of anterior dislocation. In 1967, DePalma et al. postulated, based on cadaveric dissections, that the subscapularis must play a significant role in anterior stabilization given the thin and redundant nature of the anterior capsule. 36 In another cadaveric study, Turkel et al. performed sequential release of anterior structures with external rotation stress applied to determine the relative contribution of these structures to glenohumeral stability. The subscapularis tendon contributed substantial anterior stability at 0° and 45° of shoulder abduction. 37 However, when considering its role as a dynamic stabilizer, several studies have suggested that the subscapularis may actually function to prevent posterior translation, while the infraspinatus and teres minor resist anterior translation.24,26,35 This concept was demonstrated in a recent study by Rathi et al., where electromyography and ultrasound were used to determine how activated cuff musculature affected humeral head translation; activated subscapularis prevented posterior translation, while activated posterior cuff muscles prevented anterior translation. 24
Blood Supply
The blood supply to the subscapularis is thought to arise from the subscapular artery. 1 The subscapular artery arises as the first branch from the third part of the axillary artery. 5 However, Bartlett et al. dissected 50 cadaveric shoulders and found that approximately 85% did not have any significant branches off of the subscapular artery that would supply the subscapularis muscle. 38 There is some evidence supporting the blood supply is actually derived from the anterior humeral circumflex (AHC) artery. 1 The AHC artery arises as the second branch from the third part of the axillary artery. 5 It courses along the inferior half of the anterior surface of the subscapularis. As the artery courses laterally over the tendon, the AHC artery divides the superior two-thirds of tendinous portion from the inferior muscular insertion.5,17 The venous drainage from the subscapularis muscle has been shown to occur through the circumflex scapular veins. In the same study, the investigators compared the “venosomes” with the corresponding angiosomes of certain muscles, including the subscapularis, which showed a similar distribution. This may suggest importance of the circumflex scapular artery in supplying blood to the subscapularis. 39
Conclusions
The subscapularis is the primary internal rotator of the rotator cuff. It has a broad scapular origin with a variable musclotendinous insertion onto the humerus. The innervation is primarily through the USN and LSN with a variable number of branches all primarily entering the muscle medial to the coracoid. It functions as an internal rotator as well as a dynamic stabilizer both in superoinferior and anteroposterior directions. During shoulder arthroplasty, care needs to be performed during subscapularis release to avoid deinnervation with aggressive medial superior or anterior release. Similarly, during takedown, the dimensions need to be taken into account to correctly place the tenotomy in the substance of the tendon as well as to correctly size the lesser fragment removed during osteotomy. Understanding the subscapularis during arthroplasty will allow surgeons to optimize repair techniques as well as maximize outcomes through restoration of anatomy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
