Abstract
Anxiety disorders are the most prevalent psychiatric disorders, exhibiting strong female bias. Clinical studies implicate declining estradiol levels in the exacerbation of anxiety symptoms in the premenstrual phase of the menstrual cycle. This study aimed to simulate estradiol fluctuation-linked anxiety behavior in larval zebrafish, using an estradiol treatment withdrawal model. Contrary to model aims, estradiol treatment withdrawal decreased both basal activity and anxiety-like hyperlocomotion (ANOVA main effect of dose, P < 0.0001 and P < 0.01, respectively) in the light/dark transition test. The accuracy of the estradiol washout model was not improved by longer durations of treatment or withdrawal. Basal activity was slightly altered by supraphysiological concentrations of WAY-200070 in the absence of added estradiol. Estrogen receptor (ER) β expression was not upregulated in larvae exposed to physiologically relevant, low concentrations of estradiol. Longer exposure to low concentrations of estradiol increased antioxidant capacity (P < 0.01). In addition, acute exposure to low concentrations of estradiol increased basal activity. Data suggest that in the current models, estradiol-associated altered activity levels were linked to more favorable redox status, rather than reflecting altered anxiety levels. As such, it is recommended that zebrafish larval behavioral analysis be conducted in parallel with mechanistic studies such as redox indicators, for investigations focused on ER signaling.
Introduction
While anxiety disorders are the most common class of psychiatric disorders 1 —with a global annual prevalence of approximately 11.6% 2 —the lack of specific genetic, neuroimaging, neurochemical, physiological or psychological diagnostic, and treatment biomarkers3,4 results in a persistent treatment gap. 5 Furthermore, anxiety disorder prevalence in females is approximately twice that of males,6,7 implicating sex hormones as role players in this context. For example, psychological symptoms significantly increase at specific phases in the menstrual cycle.8,9 Moreover, anxiety symptoms in women—with and without anxiety disorders—increase in the late-luteal/premenstrual phases of the menstrual cycle, when estradiol and progesterone levels are low. 10
Preclinical investigations into the role of estrogenic signaling in anxiety-like behavior pose a novel avenue through which anxiety disorders can be studied. Studies in female rats are traditionally used to improve our understanding of sex hormones in anxiety. Similar to humans, rodents display decreased anxiety-like behavior in the proestrus phase of the estrus cycle when estradiol levels are increased in comparison to progesterone.11,12 This premenstrual exacerbation in anxiety symptoms is suggested to be due to fluctuations in ovarian hormones, and in particular the acute, transient, decreasing levels of estradiol, rather than absolute hormone concentration. This likely implicates variations estrogen receptor (ER) saturation and/or signaling activity. In support of this, preclinical studies on rodents suggest that ERβ may be largely responsible for the anxiolytic activity of estradiol. 13 However, interpretation of data on the potential role of ER signaling in anxiety-like behavior in rodents are complicated by the homeostatic mechanisms controlling ER signaling. In this regard, zebrafish larvae are positioned as a novel high-throughput behavioral model through which the role of estrogenic signaling may potentially be examined in the absence of endogenous cyclic estradiol fluctuations, bridging the gap between current in vitro and in vivo models.
Thus, the current study aimed to use an estradiol treatment washout method to establish a model simulating declining levels of estradiol and associated exacerbations in experimentally induced anxiety-like behavior, as seen in the premenstrual phase and proestrus phase of the menstrual and estrus cycles, respectively. In addition, given that zebrafish larvae ER signaling occurs in the absence of endogenous fluctuations in estradiol levels, the role of ER signaling was probed using human ER modulators (WAY 200070, PHTPP and tamoxifen).
Methods and Materials
Ethical Clearance and Zebrafish Husbandry
Experimental protocols were cleared by the Stellenbosch University Research Ethics Committee for Animal Care and Use (ref#: ACU-2021-21677). Zebrafish embryos spawned from (pet store-purchased) wild-type Danio rerio zebrafish were obtained from the Zebrafish Husbandry Unit (Stellenbosch University). Zebrafish embryos and larvae were maintained in embryo medium (E3; 5 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl2 •2 H2O, 0.33 mM MgSO4 •7 H2O, 4.00 × 10−5% m/v methylene blue in reverse osmosis-filtered water) pH 7.4 at 28.5 °C, with a 14-h:10-h light:dark cycle. E3 was refreshed daily for the duration of the experimental protocol. All larvae were euthanized with 4 mg/ml tricaine methane sulfonate (Sigma, A5040) at 7 days post-fertilization (dpf).
Estradiol Administration and Withdrawal
The principle of the method was to first supply (load) estradiol at physiologically relevant doses, followed by acute withdrawal (wash out). Since dose optimization using sensitive detection of actual estradiol concentrations to confirm absorbed doses was not available, doses were estimated from existing literature. Six concentrations of estradiol (17β-estradiol; Sigma, E2758) were prepared based on an estimated percentage uptake of 5.5% at 4 dpf14,15 and included 2.5 nM, 5 nM, 10 nM, 20 nM, 50 nM, and 100 nM to achieve absorbed estradiol concentrations of approximately 137.5 pM, 275 pM, 550 pM, 1100 pM, 2750 pM, and 5500 pM, respectively. The lowest estradiol concentration administered was chosen to be greater than the EC50 values for ERα, ERβ1, and ERβ2 (77 nM, 39 nM, and 118 nM, respectively). 16 Estradiol stock was prepared in ethanol and diluted in E3. Final ethanol concentration in treatment media was <0.01%. Estradiol treatments were administered for 48 h (4-6 dpf) via immersion (refreshed daily), followed by treatment withdrawal of different durations (48, 24, 20, and 16 h) prior to endpoint behavioral analysis. During treatment administration and withdrawal, larvae were maintained in 6-well plates at a density of 20 larvae per well. At 7 dpf, E3 medium was refreshed, and larvae were placed in individual wells of a 96-well plate (each containing 250 µl E3) for light/dark transition test (LDTT).
The Light/Dark Transition Test
At experimental endpoint (7 dpf) activity of zebrafish larvae were assessed using the following LDTT protocol: a 20-min dark (infrared illuminated) acclimation period (larval locomotor activity typically settles to basal levels after 10 min), 10-min of bright white light exposure, and finally another 10-min dark phase. The dark-light-dark transition elicits a robust anxiety-like response in larvae, characterized by relative hyperlocomotion during the final dark phase. Larval movement was captured at a 25 Hz frame rate using a DanioVision activity tracker and quantified with onboard EthoVision XT15 analytical software (Noldus, Netherlands), using a smoothing profile of 0.2 mm (minimum distance moved).
Administration of Estrogen Receptor Modulators
Three ER modulators were administered—WAY-200070 (Sigma, W1520), PHTPP (Sigma, SML1355), and tamoxifen (Sigma, T5648)in the presence and absence of 50 nM estradiol (administered form 4-7 dpf). Based on work by Chen et al (2015) on larvae at 7 dpf, each ER modulator was administered for 45 min prior to the LDTT. Tamoxifen was administered at 312.5 nM, 625 nM, and 1250 nM, based on a previous dose-response in zebrafish larvae, while concentrations of 500 nM, 2000 nM, and 5000 nM were administered for both WAY-200070 and PHTPP. These doses were based on work by Chen et al (2015) and prepared by dissolving in ethanol/DMSO before dilution in E3. Final ethanol/DMSO concentration of treatments were <0.01%.
Estrogen Receptor β Expression
Quantification of ER levels in estradiol-treated larval zebrafish was performed using immunofluorescent labeling of ERβ (see Supplemental material for full protocol). Since ERs are known to regulate embryonic heart rate in zebrafish 17 and since the heart is easily identified, this region was chosen as site for standardized quantification. Expression levels (mean fluorescent unit) were normalized to heart total area.
Redox Assays
At experimental endpoint, larvae (n = 50 pooled per sample) in 450 µl of 1x PBS (Sigma, P4417) and homogenized using a Bead Ruptor Elite (OMNI International)(6.95 m/s, for four cycles of 15 s at 1-min intervals). Homogenized samples were centrifuged at 4000 x g for 10 min at 4 °C. The supernatants were stored at −80 °C until assayed. Three redox indicators were determined: hydrogen peroxide concentration (proxy for free radical abundance), malondialdehyde (MDA) concentration (lipid peroxidation), and trolox equivalent antioxidant capacity (TEAC; total antioxidant activity). Values were expressed relative to sample protein content, as quantified using a Jenway 7415 NANO spectrophotometer (Cole-Parmer, United States).
Hydrogen Peroxide: An colorimetric assay kit (E-BC-K102-S Biochemical assay kit; Elabscience Biotechnology Inc., United States) was used according to manufacturer's instructions with minor modifications for a 96-well plate and absorbance read at 415 nm (Victor Novo Multimode plate reader; PerkinElmer, South Africa).
Lipid Peroxidation: The extent of lipid peroxidation in samples was measured based on the reaction of the lipid peroxidation product, MDA, with thiobarbituric acid (TBA) to form a pink MDA-2 TBA adduct. The absorbance of samples was read at 532 nM.
TEAC assay: The antioxidant capacity of samples was measured based on the quenching of 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) radicals after 24 h. Absorbance of the samples was measured at 734 nM.
Statistical Analyses
Statistical analyses were performed in GraphPad Prism Version 10.0.1 (http://www.graphpad.com, San Diego, CA). Prior to statistical analyses, outliers were removed using the ROUT method (Q = 1%) and the Shapiro-Wilk test was used to assess normality of data distribution. All LDTT locomotion data are displayed qualitatively as activity over time, binned into 1-min bins. Data are represented as mean distance moved (mm) ± standard error of mean. Quantitative analyses of the LDTT assay are displayed as total distance moved during either basal activity (LDTT min 10-20) or the hyperlocomotion response (LDTT min 30-40). All grouped data are represented as mean ± standard deviation (SD) and were analyzed with two-way ANOVAs and Tukey's multiple comparison test. A P-value of <0.05 was considered statistically significant. Prospective and retrospective power analyses were performed using G*Power 3.1 software, to calculate and confirm the use of sufficient sample size to achieve at least 80% power for all data sets.
Results
An Estradiol Treatment Withdrawal Protocol did not Simulate Estrogen-Linked Exacerbated Anxiety-Like Behavior in Zebrafish Larvae
Activity profiles in the LDTT (Figure 1) appear to be similar between control and treatment groups, with the exception that all treated groups—irrespective of withdrawal duration between 16 and 24 h—demonstrated lower basal activity levels, and while the dark-phase hyperlocomotion response magnitude seemed similar, treated groups seemed to resolve more quickly than control. Indeed, quantitative data confirmed decreased basal activity and hyperlocomotion responses (ANOVA main effect of dose, P < 0.0001 and P < 0.01, respectively), predominantly in larvae exposed to lower estradiol concentrations, compared to control (Supplementary Figure 1). However, withdrawal duration did not significantly impact activity.
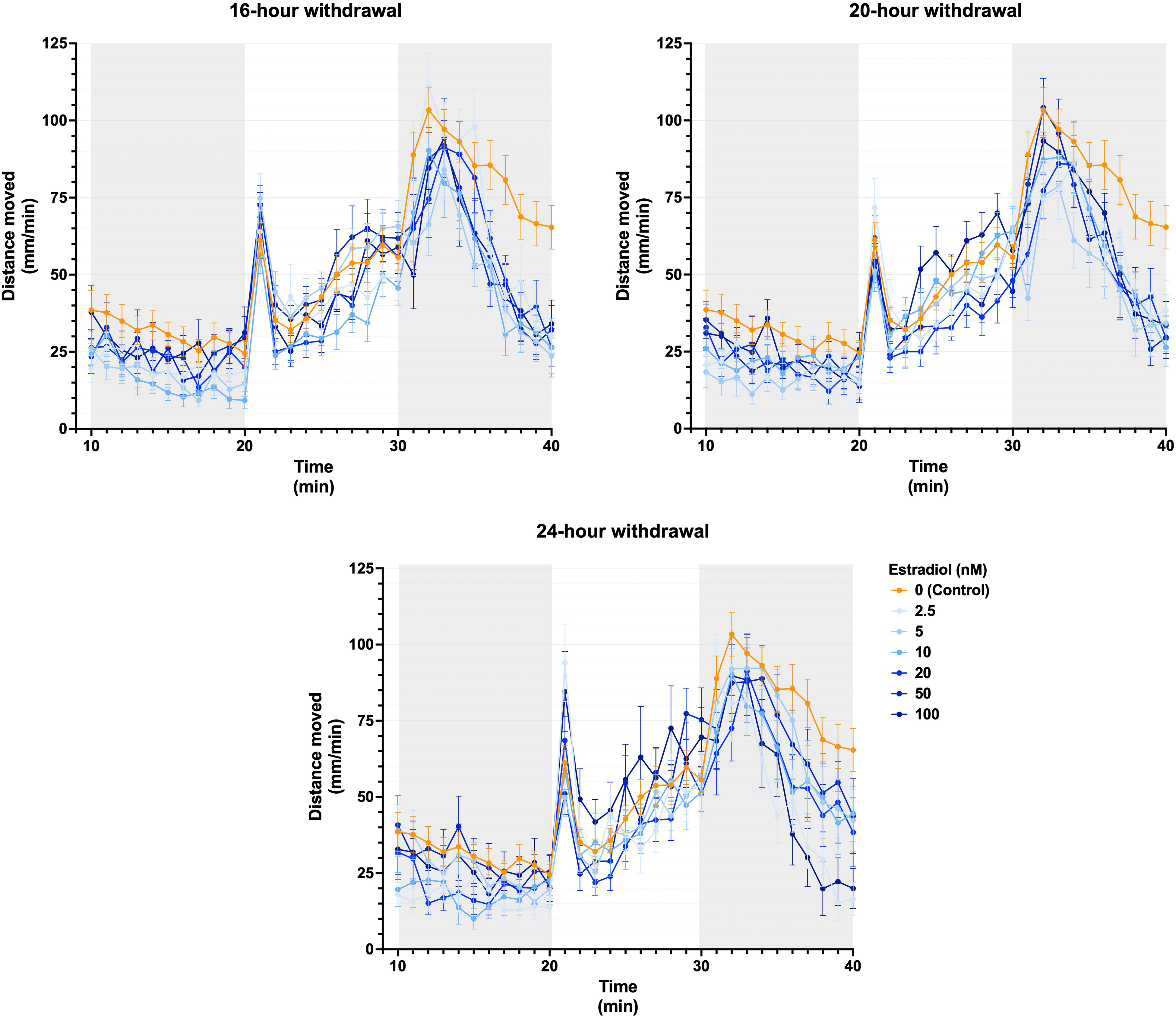
Larval activity over time following estradiol administration followed by acute estradiol treatment withdrawal. Estradiol withdrawal at 16, 20, and 24 h prior to behavioral analysis in zebrafish larvae is depicted. Data are displayed as means ± SEM (n ≥ 10 per group) from experiments repeated three times. SEM: standard error of mean.
Similar results were also obtained in a follow-up experiment, with estradiol washout periods of 24 and 48 h (Figure 2; Supplementary Figure 2).
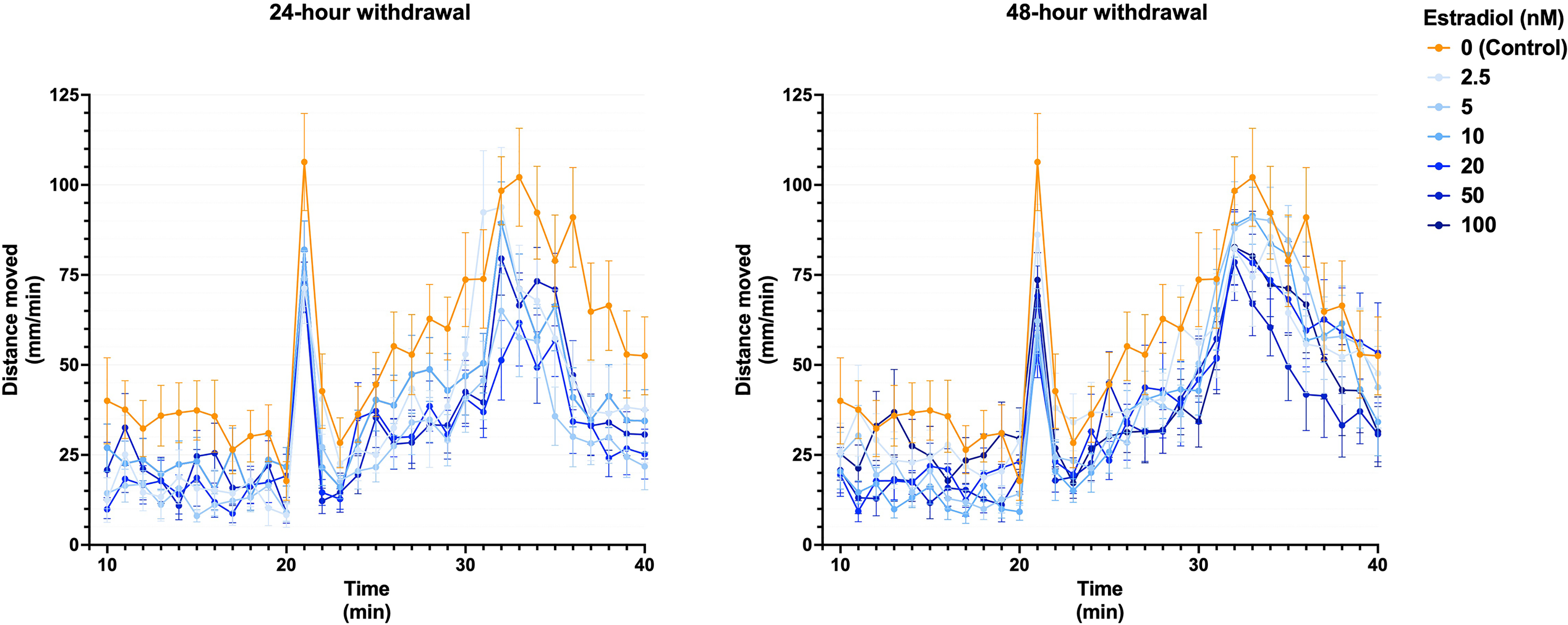
Larval activity over time following estradiol administration and subsequent withdrawal for 24 h and 48 h prior to the LDTT. Data are displayed as means ± SEM (n ≥ 13 per group) from experiments repeated three times. LDTT: light/dark transition test. SEM: standard error of mean.
Despite all the permutations in terms of treatment washout duration and concentration of added estradiol, the proposed model consistently reflected a seemingly anxiolytic effect. In order to understand the mechanisms responsible for this unexpected result, the role of ER signaling on behavior was considered.
Role of ER in Modified Activity Levels
The role of ER modulators WAY-200070 (ERβ-selective agonist), PHTPP (ERβ-selective antagonist), and tamoxifen (ER pan-antagonist), administered in the presence and absence of estradiol treatments (without a treatment withdrawal), on behavior are illustrated in Figure 3 (basal activity in left column and induced hyperlocomotion on the right). The activity profiles of larvae exposed to WAY-200070 (Figure 3A, B) were erratic and variable in comparison to control larvae. In terms of treatment effects, the highest concentration of WAY-200070 increased basal activity significantly (P < 0.0001)—an effect that was mitigated by the presence of estradiol. During the dark phase, no clear treatment effect was discernible. Similarly, LDTT activity profile of larvae treated with PHTPP were highly variable, and no clear treatment effect was evident (Figure 3C, D). While the activity levels in tamoxifen-administered larvae were generally less variable than that seen for WAY-200070- and PHTPP-treated larvae, again, no clear pattern for the effect of tamoxifen on either basal activity or hyperlocomotion was observed in the absence of estradiol (Figure 3E, F). However, the presence of estradiol, the addition tamoxifen (312.5 nM and 625 nM) increased basal activity (P < 0.01 and P < 0.05 respectively).
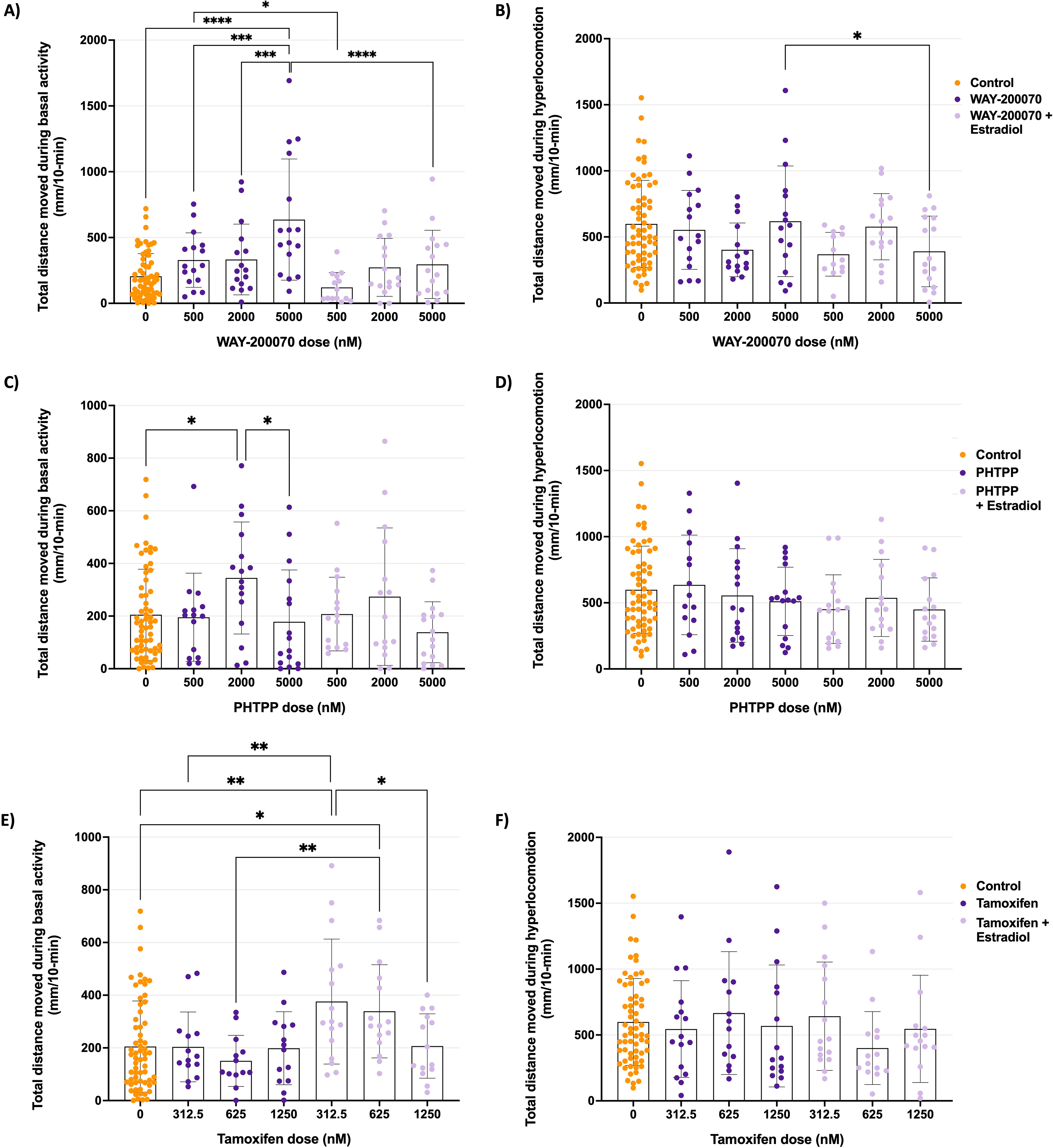
Total activity during basal and hyperlocomotion phases following acute treatment of the estrogen receptor modulators, WAY-200070 (A, B), PHTPP (C, D), and tamoxifen (E, F), in the presence and absence of prolonged exogenous estradiol treatment. Data is presented as the total distance moved (mean ± SD) for each treatment group (n ≥ 13). Data was calculated from experiments repeated twice. Statistical analyses: two-way ANOVA with Tukey's multiple comparisons; * = P < 0.05, ** = P < 0.01, *** = P < 0.001, **** = P < 0.0001. SD: standard deviation.
The failure of these data to illustrate a clear role for ER in zebrafish larval behavior raised the question of whether this model may be unsuitable for the study of ER signaling in the context of anxiety. Given the reported modulatory role of ERβ in anxiety already mentioned, we evaluated the effect of equivalent estradiol concentrations corresponding to follicular (20 nM) versus menstrual (2.5 nM) phase in humans, on the expression of ERβ protein in zebrafish larvae at 7dpf, by immunofluorescent staining. No significant differences in ERβ expression were observed between groups, regardless of estradiol concentration or treatment duration (1 h, 24 h or 48 h) (Figure 4; representative images in Supplementary Figure 3).

Erβ protein expression levels in the heart of estradiol-treated zebrafish larvae. Zebrafish larvae were treated with estradiol concentrations of 2.5 nM and 20 nM, for a duration of 1 h, 24 h, and 48 h each. Data is displayed is mean fluorescent intensity (mean ± SD) for each treatment group (n ≥ 6). Statistical analyses: two-way ANOVA with Tukey's multiple comparisons. SD: standard deviation.
As our current understanding of the role of estradiol and ER signaling does not adequately describe the role of estradiol on larval basal activity and anxiety-like behavior observed in larval zebrafish, the known direct antioxidant effect of free estradiol18,19 was investigated as potential confounder in this model.
Effects of Free Estradiol on Activity and Redox Profile
Firstly, the direct effect of exogenous estradiol (2.5 nM or 20 nM) treatment on activity was assessed. Activity profiles (Figure 5) indicated that basal activity did not appear to be dependent on estradiol concentration, but the stratification of activity levels by treatment duration was clear. Indeed, quantitative assessments confirmed an ANOVA main effect for treatment duration on basal activity (P < 0.0001), but not the hyperlocomotion response. Specifically, larvae treated with both 2.5 nM and 20 nM estradiol for 48-h displayed significantly higher basal activity levels compared to control (Supplementary Figure 4, P < 0.0001 and P < 0.05, respectively). These increases in activity in the presence of estradiol are in line with the decreases in basal activity seen previously in the estradiol washout model.

Locomotor activity of zebrafish larval after estradiol treatment. Larvae were treated for 1 h, 24 h, or 48 h with either 2.5 nM or 20 nM of estradiol. Data is displayed as the distance moved per minute (mean ± SEM) for each treatment group (n ≥ 28), calculated from experiments repeated twice. SEM: standard error of mean.
Given that estradiol treatment concentration did not seem to be a major determining factor, the effect of 20 nM only estradiol exposure on redox status in larval zebrafish was assessed (Figure 6). TEAC appeared to be the most robust redox indicator of the panel employed, exhibiting a significant increase in TEAC with longer treatment duration. Of further interest, this increase was paralleled by a decrease in MDA levels (but not hydrogen peroxide), suggesting that longer exposure to free estradiol may benefit redox status in zebrafish larvae.

Redox status in estradiol-exposed zebrafish larvae. Data is displayed as (A) Hydrogen peroxide concentration, (B) Malondialdehyde (MDA) levels, and (C) Trolox equivalent concentration, per mg/ml protein (mean ± SD) with three biological repeats per treatment group. Statistical analyses: One-way ANOVA with Tukey's multiple comparisons; *P < 0.05, **P < 0.01. SD: standard deviation.
Discussion
The current study elucidated several main findings. Firstly, an estradiol treatment withdrawal protocol did not simulate premenstrual estradiol-linked anxiety-like behavior in larval zebrafish. In fact, instead of the increased hyperlocomotion (indicative of exacerbated anxiety) that was expected, decreased locomotor activity was observed following estradiol withdrawal. Secondly, the lack of changes in activity level following acute ER modulation and estradiol-induced ER expression suggests that the locomotion changes induced by the estradiol treatment withdrawal protocol may not be mediated via ER signaling alone. Finally, the role of free estradiol was probed in the context of redox status, and data seemed to link increased activity levels in the presence of estradiol to better antioxidant capacity.
In both humans and rodent models, higher levels of estrogen—estradiol in particular—are associated with reduced fear and relative anxiolytic outcome when compared to other—low estradiol—stages of the menstrual cycle.10,11 This suggests that a transient downward fluctuation of estradiol exacerbates the associated anxiety outcome. As such, an increase in anxiety-like behavior—reflected as elevated activity during the hyperlocomotion phase—was expected during estradiol washout in the current study. However, in larval zebrafish estradiol treatment withdrawal decreased locomotion activity compared the control. While this demonstrates that estradiol treatment indeed affects larval zebrafish behavior, the effect was opposite to what was expected when estradiol availability was diminished. This suggests that our model did not accurately simulate premenstrual exacerbation of anxiety symptoms.
While literature on the performance of larvae in the LDTT in the context of estradiol supplementation is sparse, a previous study reported similar results to ours after acute (45 min) exposure to estradiol treatments, 20 which they interpreted as an anxiolytic outcome. While these results are in line with the current data, our redox analysis suggests that activity may be significantly affected by antioxidant status in addition to anxiety state. While these results illustrate the robustness of data generated in zebrafish across research groups, the TEAC data highlights the importance of considering potential confounding effects when interpreting behavioral data.
To our knowledge, this is the first study to link increased activity levels in zebrafish larvae to better antioxidant capacity, in the context of exogenous estradiol availability. Indeed, increased basal activity seemed to reflect overall improvement in wellbeing, an interpretation in line with the decreased activity (both basally and in the LDTT) reported in larvae with increased inflammation and oxidative stress, 21 which reflects sickness behavior. This interpretation is supported by literature from other preclinical models, confirming the antioxidant effect of estrogen,19,22 as well as by previous work from our own group in larval zebrafish. 23 Although the lipid peroxidation data was slightly variable at the 1-h time point, it is perhaps relevant to note that increased TEAC was paralleled by decreased lipid peroxidation. This suggests that the magnitude of antioxidant outcome after estradiol treatment was indeed likely sufficient to achieve improved energy levels and thus activity in the LDTT.
It is important to point out that an improved redox status will also indirectly have benefit in the context of anxiety outcome.24,25 Thus, our observation does not invalidate all previous research using the LDTT to make conclusions pertaining to anxiety. Rather, our data serves to caution about the importance of considering exact mechanisms at play to derive at a behavioral outcome, which may be the net effect of various contributors. This is of course of particular importance in studies focused on treatments which may have multi-pronged mechanisms of action, such as estradiol. Although we did not achieve the anticipated simulation, the sensitivity of the larval model to reflect dose- and time effects in a repeatable manner in the current study, highlights its potential as high-throughput in vivo screening tool under standardized conditions.
Turning attention to the assessment of ER signaling, in the absence of added estradiol, ER modulators had little effect on basal activity, and none on the hyperactivity response. The suitability of zebrafish as a model, in terms of homology to humans in the context of ER, is supported by the fact that estrogenic signaling is highly conserved between vertebrates and the fact that zebrafish larvae have been demonstrated to have functioning ERs (also in the brain) from 24 h post fertilization (hpf).26,27 Specifically, estrogenic signaling in zebrafish is homologous to humans in that zebrafish also possess membrane-associated ER GPER1. 28 Thus, zebrafish may be good models for elucidating the role of this membrane ER, which may provide valuable information on the link between estrogen signaling and hippocampal redox status in zebrafish larvae, which was recently linked to an antidepressant outcome in aged, female rats. 29
However, other factors may pose limitations to the immediate use of the larval model in the current context. Firstly, decreases in estrogenic signaling were recently shown to adversely affect the larval zebrafish visual system by altering retinal function to result in a relative photosensitivity, 30 which may alter activity patterns in the LDTT. Secondly, the ER modulators employed (WAY-200070, PHTPP and tamoxifen) may not have had sufficient specificity for zebrafish ER, or may have different affinity for different ERs when compared to humans. For example, WAY-200070, a mammalian ERβ agonist, demonstrated greater selectively for zebrafish ERα than either of the ERβ receptors.16,26 Furthermore, PHTPP appeared to act as an ER pan-antagonist.16,31 Thus, ER agonists traditionally used in mammalian models seem to have altered specificity in zebrafish larvae, while selective ER antagonists are not equally effective at inhibiting zfER signaling, as suggested earlier. 32 Furthermore, the stimulatory or inhibitory effect of at least some of the ER modulators are dependent on the coactivators or corepressors present in tissue, 33 which of course may differ slightly between species. Thus, before zebrafish can reach their full potential as model for sex bias in anxiety—in particular where ER signaling is a focus—more suitable reagents for probing of ER signaling need to be developed.
Finally, on a technical note, despite strong reproducibility and robustness of behavioral tests in general, some distinct, different behaviors have been reported for different strains of (adult) zebrafish. 34 Thus, standardization of strain should be a consideration in future studies. Furthermore, although the LDTT is widely used to assess modulation of anxiety-like behavior, it is important to consider that measured behavior may be influenced by factors other than anxiety state. It is therefore recommended that other anxiety indicators—such as levels of serotonin, norepinephrine, and GABA—are measured in conjunction with behavioral tests such as the LDTT to validate whether effects seen are indicative of anxiety.
Conclusion
In conclusion, while it seems that insufficient reagent availability is currently hampering the use of zebrafish as model for sex bias in anxiety, current data demonstrates a link between increased larval activity and relatively improved antioxidant capacity. Thus, increased basal activity may be a marker of improved wellbeing in zebrafish larvae. Under tightly controlled experimental conditions and the absence of alternative potentially confounding stimuli, basal activity in particular may act as proxy for redox status in zebrafish larvae, although further experimental confirmation of this effect is required.
Supplemental Material
sj-docx-1-css-10.1177_24705470241261781 - Supplemental material for Modeling Sex-Bias in Anxiety: Pros and Cons of a Larval Zebrafish Model
Supplemental material, sj-docx-1-css-10.1177_24705470241261781 for Modeling Sex-Bias in Anxiety: Pros and Cons of a Larval Zebrafish Model by Lesha Pretorius, Aidan G. Balshaw, Kelly S. Ross and Carine Smith in Chronic Stress
Supplemental Material
sj-xlsx-2-css-10.1177_24705470241261781 - Supplemental material for Modeling Sex-Bias in Anxiety: Pros and Cons of a Larval Zebrafish Model
Supplemental material, sj-xlsx-2-css-10.1177_24705470241261781 for Modeling Sex-Bias in Anxiety: Pros and Cons of a Larval Zebrafish Model by Lesha Pretorius, Aidan G. Balshaw, Kelly S. Ross and Carine Smith in Chronic Stress
Footnotes
Author Contributions
LP and CS contributed to the study conceptualization and design. Material preparation, data collection, and analysis were performed by AGB, under the supervision of LP and CS. KR supervised the redox data collection. The first draft of the manuscript was written by LP and all authors edited the manuscript. All authors read and approved the final manuscript.
Data Availability
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors would like to acknowledge Prof Jean Millar and Prof Robert Fenn for their financial contribution toward researcher salary and zebrafish unit maintenance. The Harry Crossley Foundation is acknowledged for financial support for running expenses. The Office of the Vice-rector: Research, Innovation and Postgraduate studies, Stellenbosch University, is acknowledged for financial support enabling the preparation of this manuscript.
Supplemental Material
The corresponding quantitative data to Figure 1, Figure 2, and Figure 5 are represented in Supplementary Figure 1, Supplementary Figure 2, and Supplementary Figure 4 respectively. The full protocol for whole mount immunofluorescent staining of ERβ is presented in the Supplemental material and representative fluorescent micrographs that correspond to quantitative data present in Figure 4 are depicted in ![]() .
.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
