Abstract
Objective
Post-traumatic stress disorder (PTSD) is a mental disorder that manifests after exposure to a stressful traumatic event, such as combat experience. Accumulated evidence indicates an important genetic influence in the development of PTSD. The serotonin transporter (5-HTT) gene has been identified as a candidate for PTSD and a polymorphism of the serotonin transporter-linked promoter region (5-HTTLPR) is associated with the disorder in the general population. However, whether it is associated with PTSD in active military service members has not been investigated. This study aimed to investigate the relationship between 5-HTTLPR and PTSD in service members.
Methods
Leucocyte genomic DNA was extracted from service members, including those with PTSD (n = 134) or without PTSD (n = 639). The 5-HTTLPR polymorphism was detected by means of 2 stages of TaqMan fluorescent PCR assay. PTSD symptoms and symptom severity were assessed using the PTSD Checklist (PCL), a 17-item, DSM-based, self-report questionnaire with well-established validity and reliability. PTSD was determined based on endorsement of DSM-IV criteria and a PCL total score ≥ 44.
Results
Significant differences in biallele distribution were observed between PTSD and controls (χ2 = 7.497, P = .024). The frequency of SS, SL, and LL genotypes in the PTSD group was 0.17, 0.56, and 0.27 respectively, compared to the frequencies of 0.27, 0.43, and 0.29 in non-PTSD controls. Carriers of the L allele had higher scores for reexperiencing and arousal symptoms on the PCL, compared to SS homozygote carriers (P < .05). The triallele genotypes showed no significant differences in distribution between the PTSD and control groups (P > .05) and no relationship with PTSD symptom severity. The interaction of triallelic genotypes of 5-HTTLPR and traumatic life events was associated with re-experiencing, avoidance, and arousal (P < .05 for all). Multiple regression analysis revealed significant correlations between both biallelic and triallelic genotypes of 5-HTTLPR, the interaction of the number of stressful lifetime events, and 5-HTTLPR genotypes with PCL total score (P < .001).
Conclusion
Our findings suggested that 5-HTT might play a minor role in PTSD, and the interaction between 5-HTTLPR and the environment had effects on PCL score, complementing and emphasizing 5-HTT for PTSD, especially in the military population.
Introduction
Post-traumatic stress disorder (PTSD) is a stress-related condition, occurring in only a fraction of individuals exposed to traumatic events.1,2 Despite a lifetime traumatic incidence of 40% to 90% in the general population, the overall lifetime prevalence for PTSD ranges from 7% to 12%, 1 highlighting the complex interplay of multiple factors involved in its development.2,3
PTSD risk is influenced by environmental and genetic factors, as well as gene-environment interaction.4,5 The risk of PTSD varies with the type of trauma, including war-related experiences, physical violence, partner or sexual violence, accident, 6 and natural disasters. 7 Traumatic event experiences, pre-trauma personality, and their interplay further influence PTSD risk. 6 Genetic factors, particularly the serotonin transporter (5-HTT) gene (SLC6A4) 8 or mitochondria DNA copy number, 9 have been a focus of research in PTSD.
Serotonin transporter-linked promoter region (5-HTTLPR) is an incomplete 43 bp insertion/deletion polymorphism in the 5′ flanking promoter region regulating serotonin transport gene (5-HTT) expression. The deletion (short, S) allele is associated with lower 5-HTT protein expression, potentially leading to serotonin dysfunction as seen in anxiety disorders and depression.10,11 This polymorphism is also tri-allelic due to the presence of an A > G single-nucleotide polymorphism within the L allele, resulting in Lg and La alleles. These alleles impact 5-HTT expression and function 12 and have been extensively studied in neuropsychiatric disorders, including PTSD.
Previous studies examining the association between 5-HTTLPR and PTSD have produced conflicting results. Some studies suggest that the S allele is associated with increased PTSD risk, 13 while others indicate the opposite. 14 For example, recent meta-analyses has not found consistent evidence linking 5-HTTLPR to PTSD in specific populations,5,15 while the studies of stratified by locus classification demonstrated stronger for the triallelic than for the biallelic locus subgroup. In addition, it is revealed that 5-HTTLPR influences the relationship between stress and PTSD. 16
There are needs to determine the reported results. In this study, we hypothesized the association between 5-HTTLPR polymorphisms, including biallelic and triallelic genotypes, and probable PTSD in active service members deployed to the Iraq and Afghanistan. We assessed PTSD symptoms and severity using the PTSD Checklist (PCL) and determined probable PTSD based on DSM-IV criteria and a PCL total score ≥ 44. We genotyped the 5-HTTLPR/rs25531 in PTSD and non-PTSD controls using polymerase chain reaction (PCR) amplification and amplified fragment length polymorphism. Our analysis included comparisons of genotype and allele frequency distributions and logistic regression to test the main effects of 5-HTTLPR on PTSD diagnosis. Our findings will contribute to the understanding of the complex relationship between 5-HTTLPR polymorphisms and PTSD, particularly in the military population.
Materials and Methods
Subjects
Eight hundred and ten service members with (n = 137; male: 123, female: 14) or without (n = 673; male: 606, female: 67) PTSD were collected during the Afghanistan or Iraq war between 2008 and 2016. Among them, 134 with probable PTSD and 639 without probable PTSD contributed blood samples. PTSD symptoms were assessed using the PCL. PTSD diagnosis was determined based on the endorsement of DSM-IV criteria and a PCL total score ≥ 44. The severity of PTSD symptoms was determined using the PCL total score. Subjects with depression, substance abuse, or any medication were excluded. The Institutional Review Board at the Uniformed Service University of Human Sciences approved all study procedures, and all participants were given written informed consent. Subjects with depression, substance abuse, or any medication use were excluded. In addition, demographic data, such as age, gender, and race, were collected.
Blood Sample Collection, DNA Extraction
2.5 mL forearm venous whole blood was collected using Qiagen DNA tubes. The blood samples were incubated at room temperature for 1 hr and then stored and transferred at −20 °C according to the manufacture's manual. DNA was extracted using the DNA extraction kits (Qiagen). Genomic DNA was extracted from 200 µL blood samples using the QIAmp (Qiagen, Chatsworth, CA) protocol. The quantity and quality of the genomic DNA isolate was determined by 260/280 ultraviolet spectrophotometry (Nanodrop SD-1000 spectrophotometer). Relative mtDNAcn was assessed by the real-time PCR-based assay. 17
5-HTTLPR Genotyping
Both biallelic and triallelic 5-HTTLPR genotyping were performed using published 2-stage methods.12,18 Briefly, for stage 1, the allele-discriminating probe (ADP) was capable of hybridizing once to the 43-bp L insertion, and an internal control probe (ICP) hybridized to a sequence located within the same amplicon but specific to a divergent repeat found only once in the amplicon and serving as positive DNA control. For stage 2, probes were explicitly designed to discriminate the La and Lg alleles. PCR was carried out in a 20 μL volume: 25 to 50 ng DNA in 2 μl, 10 μl PCR 2× Mastermix including probes (120 nmol ADP, 60 nmol ICP), PCR primers (200 nmol of each) and DMSO 4% by volume, 5 mmol/liter MgCl, 1× ABI Corebuffer, 0.2 mmol/liter dATP, 0.2 mmol/liter dGTP, 0.2 mmol/liter dCTP and 0.4 mmol/liter dUTP, 0.25 U/ml Taq Gold, and 0.01 U/ml AmpErase UNG. Each well of the 96-well optical plates (4306737, Alta.I) was sealed with optical caps (4323032, Alta.I). Amplification conditions were 2 min at 50 °C, 10 min at 95 °C, then 40 cycles at 96 °C for 15 s and 62.5 °C for 90 s. Genotypes were generated using ABIPRISM 7700 Sequence Detection system software. Genotyping standards were genomic DNA of SS, SL, and LL for insertion/deletion and LaLa, LaLg, and LgLg for A/G whose genotypes were known by sequencing. Genotypes acquired from these 2 stages were combined to classify samples as 1 of 6 genotypes: SS, SLa, SLg, LaLa, LaLg, and LgLg . The S and Lg alleles were associated with low HTT expressing and the L A allele was associated with higher expressing. To evaluate genotyping accuracy, one-fourth of the samples selected randomly were genotyped in duplicate. Error rate was <0.005.
Statistical Analyses
All analyses were performed using SPSS version 24.
Chi-square tests were performed to test the differences of distributions of sex among the groups. All data are presented as a number or mean ± SD. The statistically significant differences of mtDNAcn among the male and female subjects were analyzed by Student's t-test. Moreover, under the multiplicative disease model, based on our case and control sample size and disease allele frequency of 0.45, the statistic power is greater than 0.92. We acknowledge that multiple comparisons can increase the risk of false positive findings and lead to overinterpretation of results. We performed a Bonferroni correction by adjusting the significance threshold (P-value) to account for the number of hypotheses being tested. Consequently, we adopted a more stringent threshold of significance (P < .05) to minimize the likelihood of false positive results.
Results
Demographic Characteristics
Table 1 shows the demographic characteristics of the participants with PTSD and non-PTSD controls. The subjects with PTSD were younger (mean: 27.4, SD: 6.0 years) than the non-PTSD controls (mean: 28.8, SD: 7.0 years) (P < .05). The subjects with educational experience, high school diplomas, and some college/technique schools had a higher risk of PTSD (P < .05). The distributions of gender, ethnicity, and marital status were not different between the participants with PTSD and non-PTSD controls. Multiple regression analysis did not show the effects of age×5-HTTLPR genotypes on PCL score (Beta = -0.078, P = .848 for biallelic genotypes; Beta = 0.081, P = .821 for triallelic genotypes).
Demographic Characteristics.

Distributions of 5-HTTLPR Genotype and Allele Frequencies Between PTSD and Non-PTSD Controls
Table 2 showed significant differences in biallele (insertion/deletion) genotype distribution between PTSD and controls (χ2 = 7.497, P = .024.). The genotyped variants of 5-HTTLPR were not consistent with Hardy-Weinburg Equilibrium (HWE) in non-PTSD controls (P < .05). The genotype distributions in PTSD did not show significant evidence for deviation from HWE (>0.05). The frequencies of SS, SL, and LL genotypes in PTSD were 21 (0.17), 68 (0.56), and 33 (0.27), respectively, in comparison to 160 (0.27), 256 (0.43), and 174 (0.29) non-PTSD controls. However, there were no significant differences in allelic distribution between PTSD and control (χ2 = 1.128, P = .288). S and L of PTSD were 110 (0.54) and 134 (0.46), while controls were 576 (0.49) and 604 (0.51), respectively. There were also no significant differences in triallele genotype and allele frequencies between PTSD and control (P > .05; Table 3). The frequencies of SS/SLg/LgLg, Sla/LgLa, and LaLa in PTSD were 34 (0.28), 56 (0.46), and 32 (0.26), while the frequencies of those genotypes in non-PTSD controls were 198 (0.34), 236 (0.40), and 156 (0.26), respectively (Table 4).
Distributions of HTTLPR Genotypes and Alleles (Insertion/Deletion).

χ2 = 7.497, P = .024. χ2 = 1.128, P = .288.
Distributions of HTTLPR Genotypes and Alleles (Triallele).

P > .05.
Predictors for PCL Total Score.
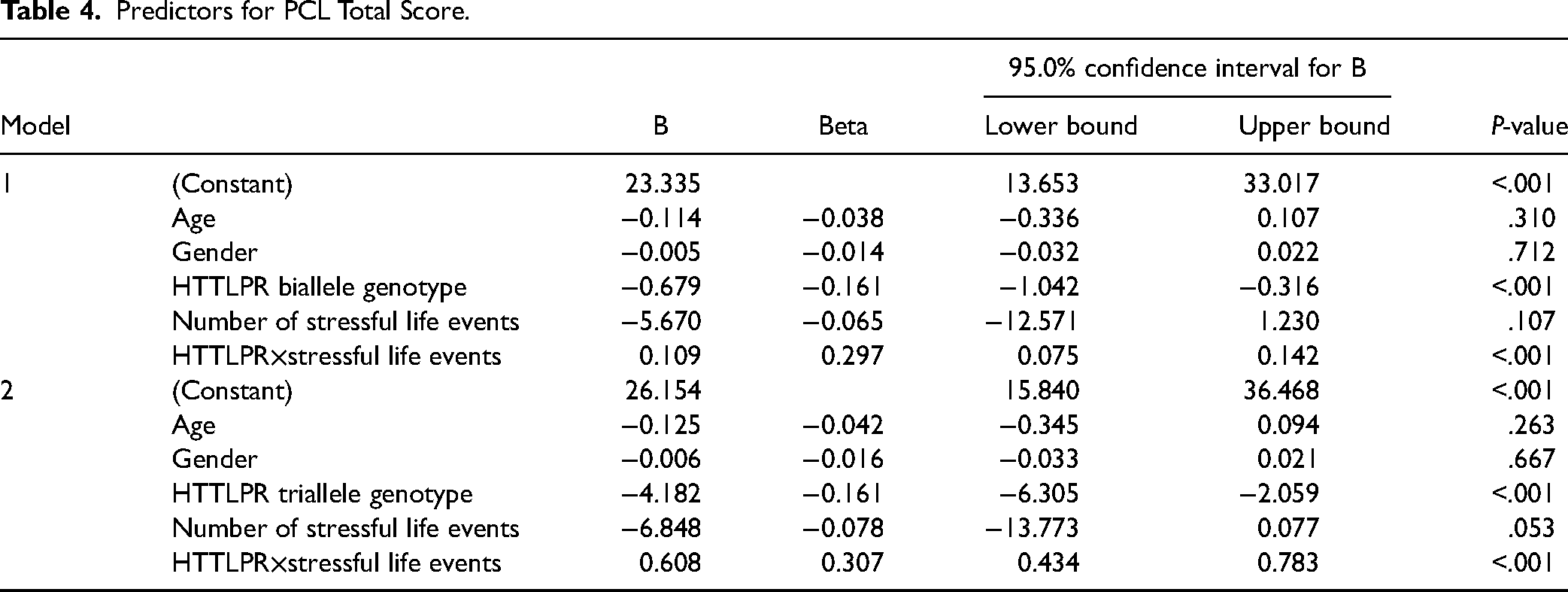
Although there was no correlation between triallele and the severity of PTSD symptoms, there was an association of SL/LL with total scores of PCL (P = .015), re-experience (P = .008), and arousal (P = .034) (Figure 1).

Comparison of PCL score between SS genotype and L allele carriers. (A) Participants with 5-HTTLPR SL or LL genotypes (high expression of HTT L allele carriers) tended to have higher PCL total score than those with homozygote of low HTT expression short allele (SS genotype carriers). Further analysis indicated that L allele carriers had significantly higher scores in re-experience and avoidance symptoms, compared to SS genotype carriers. (B) The triallele comparison and no significant difference among those groups.
The Effects of 5-HTTLPR Genotypes and Stressful Lifetime Events on PCL Score
The G×E relationship between 5-HTTLPR and stressful life events on total PCL score and PCL factor scores (re-experiencing, avoidance, and arousal) were analyzed. The effects of biallele- and triallele-based genotypes on total PCL scores are shown in Figures 2A and B, respectively. Those who experienced higher (4 or more) stressful life events tended to have higher total PCL scores than those who experienced lower (3 or fewer) stressful life events, no matter what their genotype (P < .05). Similar results were observed with the scores of re-experiencing, avoidance, and arousal (P < .05 for all). Multiple regression analysis showed that both biallelic and triallelic genotypes of 5-HTTLPR and the interaction of the number of stressful lifetime events and genotypes of 5-HTTLPR were significantly correlated with PCL total score (P < .001, Table 4).
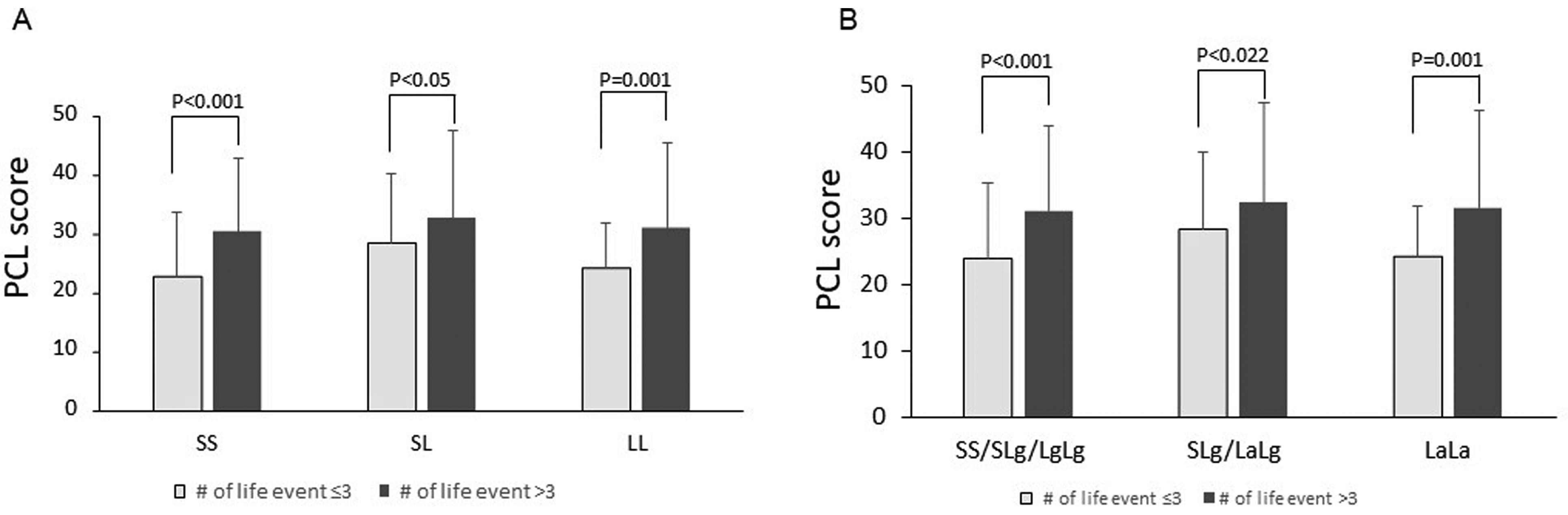
Effects of interaction of 5-HTTLPR genotype and life events on PCL score. No significant differences were shown in PCL total score and the 3 symptom scores between participants with 5-HTTLPR SLa, LaLg, or LaLa genotypes (high expression of HTT La allele carriers) and those with homozygote of low HTT expression short allele (SS, SLg or LgLg genotypes carriers).
Discussion
In our study, we observed that younger subjects were more likely to have probable PTSD, and those with lower education faced a higher risk of PTSD. We found a significantly lower frequency of the SS genotype in individuals with PTSD compared to non-PTSD controls. However, no significant differences were detected in the allelic distribution or triallelic genotype and allele frequencies between PTSD cases and non-PTSD controls. Carriers of the L allele (SL or LL) exhibited higher total scores for PCL re-experience and arousal than SS homozygote carriers. Notably, these differences disappeared when analyzing triallelic genotypes.
Our observation indicating a higher likelihood of probable PTSD among younger individuals underscores the importance of understanding the complex interplay between age and PTSD susceptibility. Younger individuals may encounter unique stressors and vulnerabilities, such as exposure to traumatic events during critical developmental stages, societal pressures, or limited coping mechanisms. Numerous epidemiological studies have consistently reported a higher prevalence of PTSD among younger age groups. For example, a meta-analysis by Koenen et al found that younger age was significantly associated with an increased risk of developing PTSD following exposure to trauma. 19 Moreover, younger individuals are more likely to experience certain types of traumatic events, such as physical or sexual abuse, neglect, or witnessing violence, which are known risk factors for PTSD. 20 The vulnerability of younger populations to such traumas may contribute to their heightened risk of developing PTSD symptoms. In addition, age-related factors like cognitive development and emotional regulation could influence the manifestation and recognition of PTSD symptoms. Research suggests that developmental differences in brain structure and function may influence the risk of developing PTSD. Younger individuals, whose brains are still undergoing significant maturation processes, may exhibit heightened emotional reactivity and impaired emotion regulation, predisposing them to a higher likelihood of PTSD following trauma exposure.21,22 This result also highlights the necessity for targeted interventions and support systems tailored to address the needs of younger populations at risk of PTSD. Early identification and intervention strategies, including accessible mental health resources and trauma-informed care, are crucial for mitigating the long-term impact of PTSD among younger individuals. Additionally, longitudinal studies examining the trajectory of PTSD symptoms across different age groups can provide deeper insights into the evolving nature of this disorder and inform preventive measures. Furthermore, our findings emphasize the significance of age-sensitive approaches in clinical assessments and treatment planning for PTSD. Healthcare professionals need to be attuned to age-related nuances in symptom presentation and treatment response to optimize therapeutic outcomes. Overall, our observation underscores the multifaceted relationship between age and PTSD, emphasizing the importance of holistic and age-appropriate interventions in addressing this significant public health concern.
Our results of the relationship between education and PTSD are consistent with previous reports. Cerda et al. and Goldmann et al reported that individuals with lower levels of education often reside in socioeconomically disadvantaged environments, which may increase their exposure to traumatic events such as violence, natural disasters, or interpersonal conflicts.23,24 This elevated exposure to trauma serves as a significant risk factor for developing PTSD symptoms.
In addition, McLaughlin et al showed that lower education levels are associated with reduced access to resources and support systems that are crucial for coping with traumatic experiences and seeking appropriate mental health care. 25 Limited access to healthcare services, including mental health treatment, may exacerbate PTSD symptoms and hinder recovery among individuals with lower educational attainment. Our examination of the E×G interaction revealed that individuals who experienced more than 3 stressful life events tended to have higher total PCL scores, regardless of their genotype. Previously the studies have shown inconsistent results. For example, recent meta-analyses showed no association between 5-HTTLPR and PTSD in specific populations,5,15 while the other studies demonstrate stronger for the triallelic than for the biallelic locus subgroup, and influence of 5-HTTLPR on the relationship between stress and PTSD. 16 In the present study, both biallelic and triallelic genotypes of 5-HTTLPR, along with the interaction of the number of stressful lifetime events and genotypes of 5-HTTLPR were significantly correlated with PCL total score. However, we did not find any significant effects resulting from the interaction between age and 5-HTTLPR on PCL total score. Our data suggested that ins/del genotypes of 5-HTTLPR were associated with probable PTSD, with a higher frequency of SS in non-PTSD control, indicating that the SS genotype might be a protective factor. However, the association between 5-HTTLPR and PTSD disappeared when the low expression-related allele Lg was removed from the L allele, which contrasts with a previous study that reported no association between 5-HTTLPR and the PTSD symptoms severity in ethnically homogenous groups of veterans with matched combat experience. 26
Regard to the G×E interaction, a meta-analysis suggests a modulation effect of the 5-HTTLPR on the association between stress and PTSD. 16 However, studies have produced varying results, such as no evidence for combat×5-HTTLPR effects on PTSD in certain groups of veterans. 27 Notably, there is a significant interaction of the S allele with the risk of PTSD in the individuals who experienced specific traumatic events, as seen in the 2008 Wenchuan earthquake in China. 28 In our study, we found that individuals who experienced more than 3 stressful life events tended to have higher PCL total score, regardless of their genotypes. Both biallelic and triallelic genotypes of 5-HTTLPR, in conjunction with the interaction of the number of stressful lifetime events and genotypes of 5-HTTLPR, were significantly correlated with the PCL total score. Nevertheless, we did not observe any significant effects resulting from the interaction between age and 5-HTTLPR on PCL total score.
The main limitation is that the present study focused on one polymorphism of the candidate gene for evaluating the association with PTSD. Multiple functional polymorphisms should be included in the future studies. Our study focused on a single candidate-gene polymorphism within a moderate sample size, which may potentially affect reliability of our genetic analyses. Delving deeper into this limitation, various aspects arise, such as the potential oversight of other relevant genetic variants and the limitation in generalizing findings to broader populations. Therefore, caution is warranted in drawing definitive conclusions solely based on the analysis of a single genetic marker. It is important to note that comprehensive studies with larger sample sizes and consideration of multiple variants would be necessary in the future.
Conclusion
In summary, our results suggest that while the ins/del polymorphism of 5-HTTLPR is associated with PTSD, the triallele-based polymorphism, where the low expression-related Lg allele, is not linked to PTSD. Further analysis revealed that the interaction between 5-HTTLPR and traumatic events was correlated to PCL score. Therefore, our findings underscore the importance of the 5-HTTLPR polymorphism in the context of PTSD, especially in the military population.
Footnotes
Abbreviations
Author Contributions
L.Z. and XZ.H. have the responsibility for the study design, conducted the analysis of the data, and wrote the original draft of the article. X.L. participated in the DNA extraction. XZ.H. performed the PCR for 5-HTTLPR genotype discrimination. Biomarker Study Group, D.B., and R.U. participated in the study design, the sample collection and edited the manuscript. All authors contributed substantially to this work and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Center for the Study of Traumatic Stress.
