Abstract
Introduction
Social isolation (SI) and loneliness are becoming increasingly significant health issues. Although adults and the elderly are at higher risk for SI and loneliness, people of all ages have experienced increased SI and feelings of loneliness, especially in the coronavirus disease 2019 (COVID-19) era. SI and loneliness were associated with a 29% increase in the risk of heart disease and a 32% increase in the risk of stroke. 1 In addition, higher rates of anxiety, depression, and suicide are associated with SI and loneliness.
According to the Centers for Disease Control and Prevention, heart disease is the leading cause of death in women in the United States, accounting for one in every five female deaths. Several cardiovascular disease (CVD) risk factors, including high blood pressure (BP) and cholesterol levels, smoking, obesity, physical inactivity, and poor diet, have been associated with SI and loneliness.2-7 Recent research indicates that either SI or loneliness can independently increase the risk of CVD in older women and that women who experience both SI and loneliness have a significantly higher risk of CVD. 8 Hypertension, a well-established risk factor for CVD and stroke, disproportionately affects elderly women, and socially isolated women are more likely to develop hypertension than are men.9,10 In adolescents and young adults, the burden of CVD can result in a loss of lifetime productivity and an increase in lifetime healthcare costs.
Adolescence is a critical developmental stage and a vulnerable period to a variety of stressors, including SI, a major source of psychosocial stress in adolescents.11-15 Several behavioral abnormalities, including aggression, anxiety, social withdrawal, and learning deficits, are observed in animals subjected to SI early in life, with some of these abnormalities being sex specific.16-22 Of particular interest, SI of mice after weaning has been proposed as an animal model for developmental disorders, including neuropsychiatric conditions such as depression, and their treatment. 23
Although the impact of SI on depression-like behavior has been reported,20,24 it is unknown whether SI affects BP in adolescent animals, and if so, whether these changes are dependent on the animal's sex. We hypothesized that 8 weeks of SI would increase BP and induce depression-like behavior in young mice, with females being more susceptible than males. Because environmental deprivation is a significant factor in human SI, these animals were not provided with cage enrichment.
Methods
Animals and Social Isolation
C57BL/6 mice (Jackson Laboratory) were maintained in an animal facility with a constant temperature (21 °C), 30% to 70% relative humidity, a 12-hour light/dark cycle, and free access to food and water, treated with irradiation. The animals were allowed to breed, and their offspring were used in these experiments. At 21 days of age, mice were weaned and randomly assigned (n = 6/group/sex) to one of two cohorts: group housed (GH) or socially isolated (SI). Animals in the SI group were housed individually for 8 weeks, while animals in the GH group were housed two to three per cage. SI and GH group animals were housed in separate adjoining rooms. The floor area of each cage was measured 5.75 by 12 inches, or 69 square inches. Each group of animals was maintained in an IVC (individually ventilated cage) system, equipped with HEPA filters to prevent pheromone exposure, specifically in the SI animals. An opaque partition was installed between the SI group cages to prevent animals from seeing each other. In addition, SI animals were not provided with cage enrichment (bedding nestlets and cardboard tunnels), as were group-housed animals. SI animals were physically, visually, and olfactorily isolated from other housed animals, had no contact or social interactions with conspecifics. Cage housing and experimental protocol were maintained for both the SI and GH groups until they were euthanized. All animals were handled during cage changes at regular intervals in accordance with our standard operating procedures. The body weight of the animals was measured at the baseline, 4 weeks, and 8 weeks after SI. The experimental timeline outlining the activities/measurements/tests conducted in chronological order was presented in Figure 1. All the studies conducted and reported conformed with the “Animal Research: Reporting of In Vivo Experiments” guidelines. 25 All animal procedures and experiments were performed in accordance with the scientific, humane, and ethical principles outlined in the Guide for the Care and Use of Laboratory Animals (Publication no. 86-23 revised, National Institutes of Health, U.S. Department of Health and Human Services) and a protocol approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Illinois College of Medicine Peoria.
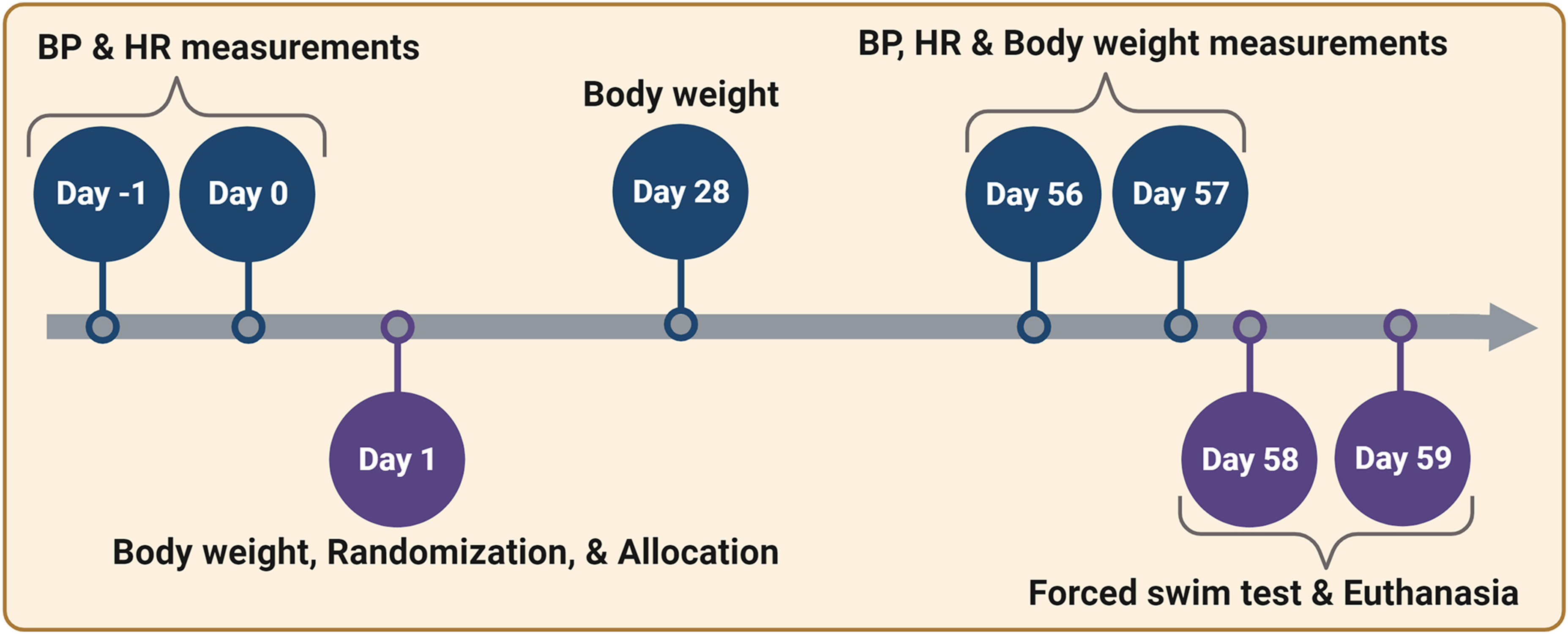
Schematic representation of the experimental timeline outlining the activities/measurements/tests conducted in a chronological order. BP, blood pressure; HR, heart rate; FST, forced swim test. This figure was created with biorender.com under a paid subscription.
Blood Pressure and Heart Rate Measurements
BP and heart rate (HR) were measured in mice under isoflurane (1%-2%) using a non-invasive, clinically validated BP monitoring system (CODA® Monitor, Kent Scientific Corporation, USA) with a tail occlusion cuff and a volume pressure recording (VPR) sensor. 26 In comparison to simultaneous radiotelemetry BP measurements, the VPR tail-cuff method accurately measures systolic BP but slightly underestimates diastolic BP in mice. 27 The anesthetized mouse was positioned prone on an infrared surgical warming pad, and a small occlusion cuff was placed around the base of the tail before the VPR tail cuff sensor was attached. During BP measurements, the tail occlusion cuff automatically inflates to 250 mm Hg within 1 second and deflates over 20 seconds, producing simultaneous values for systolic, diastolic, and mean BP, as well as pulse heart rate. The interval between cycles of inflation and deflation was 10 seconds. The BP and HR measurements were taken between 10 a.m. and 6 p.m. The average of five to ten BP and HR readings for each mouse was used for data analysis.
Forced Swim Test
The forced swim test (FST) was performed as described earlier during the light (inactive) phase of the animal's light/dark cycle.28,29 To prevent the potential impact of anesthesia on depression-like behavior, FST was performed after BP/HR measurements, with at least 1 day passing between the two (Figure 1). The mouse from each experimental group was placed in an inescapable, transparent glass cylinder (25 cm in height and 15 cm in diameter) filled with 15 cm of tap water at a temperature of 24 ± 1 °C. The behavior of the animal was video recorded during the 6-minute test for offline analysis. The amount of time each mouse spent “immobile” was measured. The mouse was judged immobile when it stopped struggling/swimming and assumed a motionless posture in the water, making only the necessary movements to maintain balance and keep its head above water. The duration of immobility indicates a depression-like state, although other interpretations exist.28-30 We also measured the latency to the first episode of immobility lasting more than one second. Two independent scorers, who were blind to treatment, evaluated the immobility duration and latency for each animal, with the average of their scores being used for data analysis. There was excellent agreement between the scorers for immobility duration (r(142) = 0.9797, p < .0001) and immobility latency (r(22) = 0.9941, p < .0001), as demonstrated by Pearson correlation analysis.
Enzyme-Linked Immunosorbent Assay
The serum concentrations of corticosterone and metanephrine were determined using commercially available enzyme-linked immunosorbent assay (ELISA) kits for corticosterone (Abcam; Catalog # ab108821) and metanephrine (LS Bio; Catalog # LS-F56023), according to the manufacturer’s instructions. For both the corticosterone and metanephrine assays, absorbance was measured at 450 nm using Microplate Manager Software and a Bio-Rad xMark Microplate Absorbance Spectrophotometer. All samples were run in either duplicates (metanephrine) or triplicates (corticosterone).
Statistical Analysis
Statistical analysis of the data was performed using GraphPad Prism 8.4.3 for Windows. The quantitative data from each experiment was first tested for normality and equality of variances. Based on the number of experimental groups, type of data (between or within subjects), and results of the normality and variance tests, the most appropriate statistical test was selected to analyze the data, as noted in the text and figure legends. Differences between groups were considered statistically significant at p < .05. All data are expressed as mean ± SEM.
Results
Blood Pressure is Significantly Higher in Socially Isolated Female Mice
Young mice subjected to 8 weeks of SI had increased systolic (12% in males and 22% in females), diastolic (11% in males and 28% in females) and mean (11% in males and 26% in females) BP and HR (18% in males and 16% in females), in comparison to age- and sex-matched mice housed in groups (Figures 2 and 3). SI females exhibited a significant increase in systolic (unpaired t test, two-tailed, t(10) = 2.384, p = .0384), diastolic (unpaired t test, two-tailed, t(10) = 2.958, p = .0143), and mean (unpaired t test, two-tailed, t(10) = 2.784, p = .0193) BP. In contrast, SI males exhibited no significant changes in BP compared to GH males. The increased HR of SI animals compared to GH animals was significant in both males (unpaired t test with Welch's correction, two-tailed, t(6.06) = 2.958, p = .025) and females (unpaired t test, two-tailed, t(10) = 2.805, p = .0186) (Figure 3A). However, at the start of the SI experiment, there were no differences in baseline HR between SI and GH male or female mice. We were unable to measure the baseline BP of very young mice due to their small tail size and limited blood flow.

Social isolation significantly increases the systolic, diastolic, and mean arterial blood pressure of young female mice, but not male mice. The column scatter plots depict the systolic, diastolic, and mean arterial blood pressure (BP) of young male and female mice that were socially isolated (SI) or group-housed (GH). The columns and error bars represent the mean and SEM, respectively. The data were analyzed using a two-tailed unpaired t-test. *p < .05.
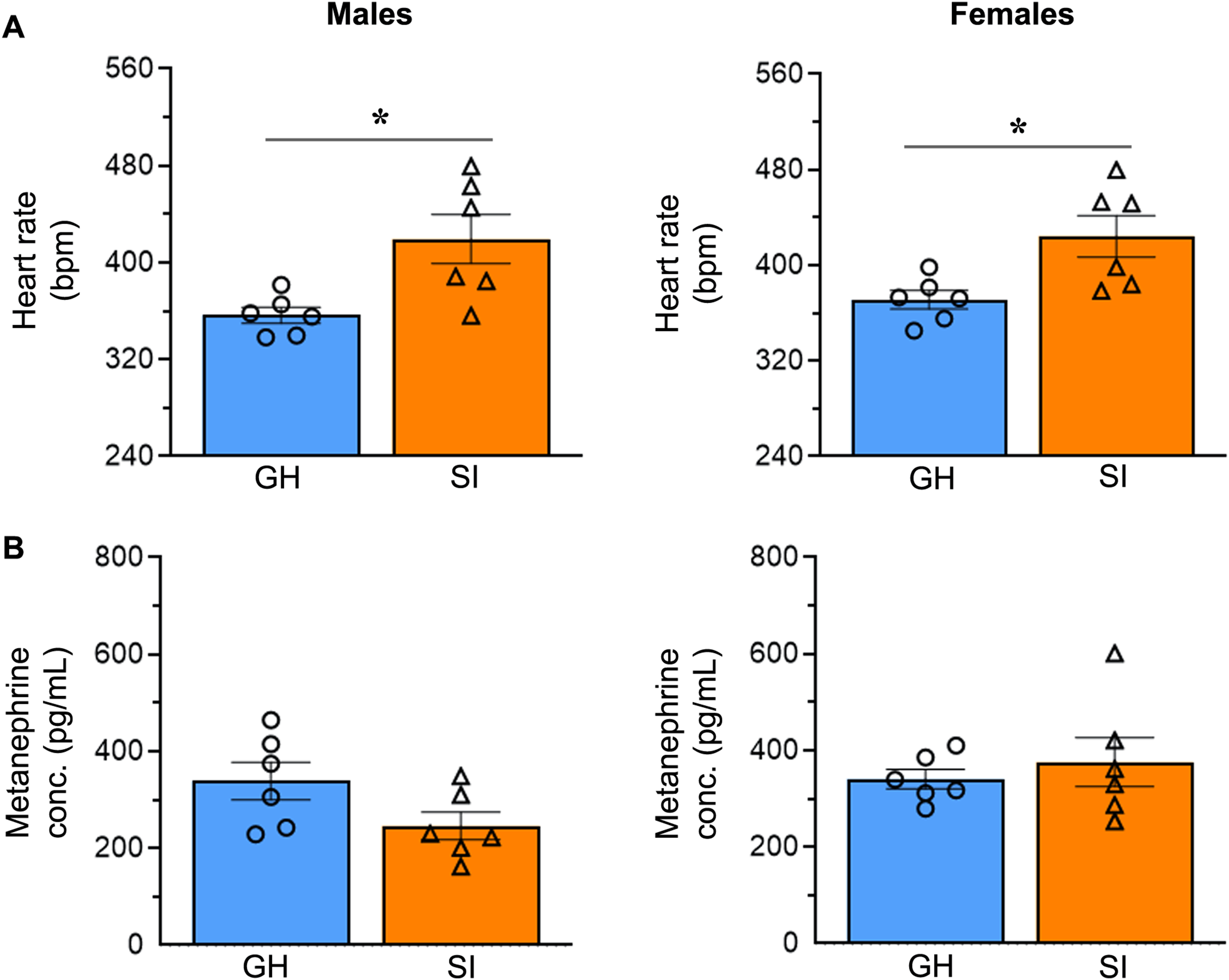
Social isolation increases the heart rate of young mice of both sexes. (A) The column scatter plots depict the heart rate in beats per minute (bpm) in socially isolated (SI) and group-housed (GH) male and female mice after social isolation. The columns and error bars represent the mean and SEM, respectively. The data were analyzed using a two-tailed unpaired t-test. *p < .05. (B) The column scatter plots depict the concentrations of metanephrine in the serum of male and female mice. The columns and error bars represent the mean and SEM, respectively. The data were analyzed using a two-tailed unpaired t-test.
In addition to BP and HR readings, the levels of the epinephrine metabolite metanephrine were determined. Serum metanephrine levels, which reflect sympathoadrenal activity, did not differ significantly between SI and GH male or female animals (Figure 3B).
Socially Isolated Female Mice Gain Weight Significantly
SI males exhibited a lower body weight gain (23% less after 4 weeks and 23% less after 8 weeks) compared to GH males (Figure 4A). However, SI females exhibited a dramatic increase in body weight gain (90% more after 4 weeks and 65% more after 8 weeks) compared to GH females. While the decrease in weight gain in SI males was not significant, the increase in weight gain in SI females was significant (two-way repeated measures ANOVA, F(1, 10) = 19.17, p = .0014; Sidak's multiple comparisons test, p = .0006 after 4 weeks and p = .0007 after 8 weeks of SI).

The effect of social isolation on body weight gain and the relationship between weight gain and diastolic blood pressure. (A) The weight gain was calculated as a percentage of the initial body weight. The column scatter plots depict the weight gain of young male and female mice that were socially isolated (SI) or group-housed (GH) over the 8-week experiment. The columns and error bars represent the mean and SEM, respectively. The data were analyzed using two-way repeated measures ANOVA and Sidak's multiple comparisons test. *p < .05 versus respective GH group. (B) There was a significant positive correlation (Spearman correlation test: r(10) = 0.6364; p = .0299) between body weight gain and diastolic blood pressure (BP) in young female mice (orange triangles represent socially isolated female; blue dots represent group-housed females).
There was a significant positive correlation between body weight gain and diastolic BP (Spearman correlation test: r(10) = .6364; p = .0299) in young female mice (both SI and GH animals combined) (Figure 4B).
Eight Weeks of Social Isolation Has a Mild Effect on Depression-Like Behavior
During a 6-minute FST, the cumulative immobility time of SI and GH mice was assessed at 1-minute intervals. In comparison to their respective GH counterparts, SI females exhibited a 12% increase in total immobility time, whereas SI males exhibited a 7% decrease in total immobility time (Figure 5A). Throughout the experiment, there were no statistically significant differences between SI and GH animals in the amount of time they ceased struggling or swimming and became immobile in the water. In addition, FST data were analyzed considering only the last four minutes of the test (i.e., 2 to 6 minutes), as is commonly done in mice.28,29 Contrary to our expectations, immobility duration during the last four minutes of FST was significantly decreased (instead of an increase) in SI males (unpaired t test, two-tailed, t(10) = 2.507, p = .0311) whereas the changes are not statistically significant in SI females (Figure 5B). When the animal's immobility time relative to body weight was analyzed, the differences between the GH and SI groups were not statistically significant in either males or females. The latency to the first episode of immobility lasting more than one second during FST was reduced by 18% in SI males and 20% in SI females in comparison to their respective GH counterparts (Figure 5C). As expected, the decreased latency to immobility was significant in SI females (unpaired t test, two-tailed, t(8) = 3.146, p = .0137) but not in SI males. Overall, there was no evidence of elevated immobility in FST although the latency to immobility was decreased in SI females. Serum levels of the stress hormone corticosterone were reduced by 55% in SI males and 17% in SI females compared to the levels in GH male and female mice, respectively (Figure 5D). The decrease in corticosterone levels was significant in SI males (unpaired t test, two-tailed, t(10) = 3.562, p = .0052), but not in females.
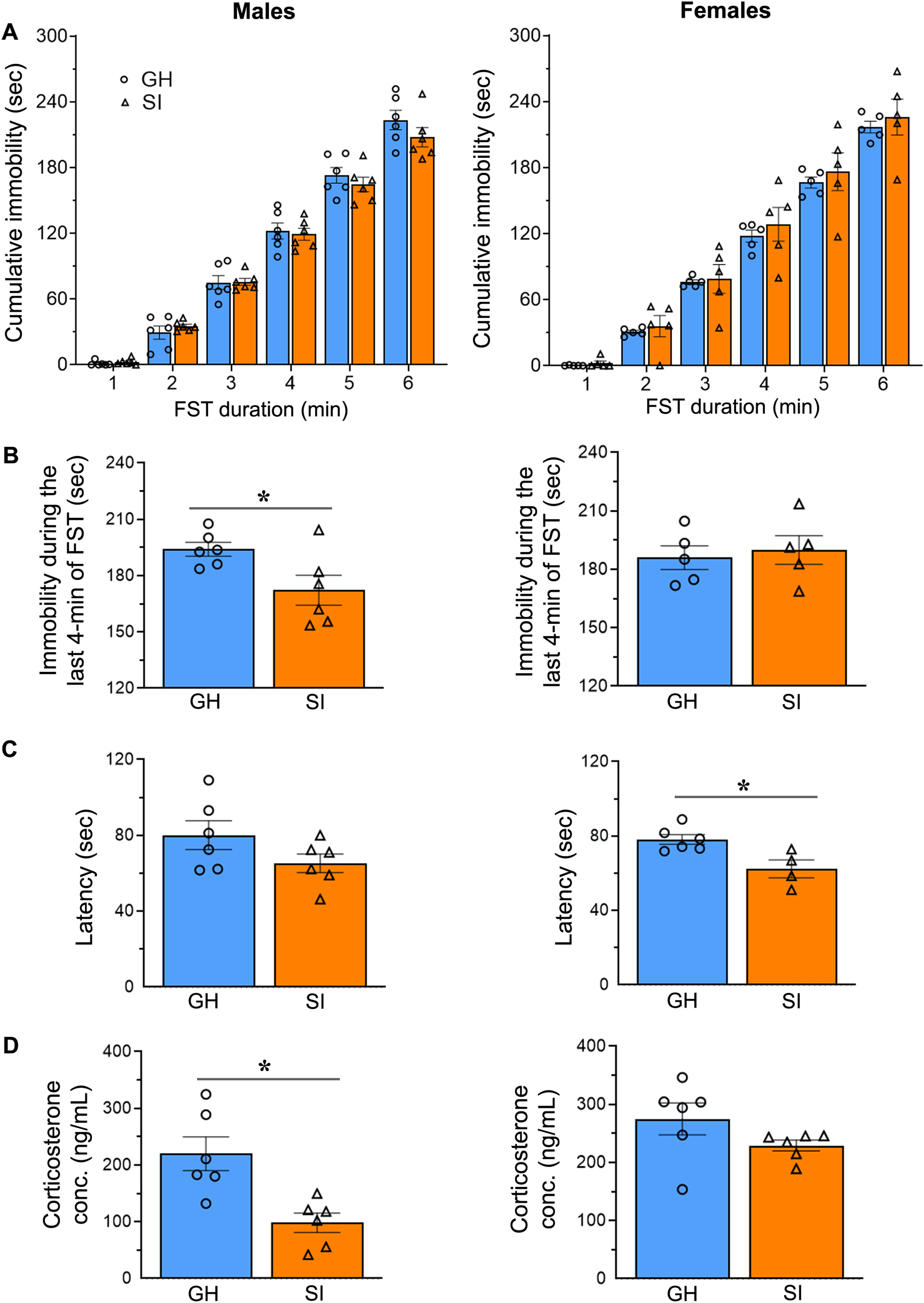
Evaluation of depression-like behavior in young mice following 8 weeks of social isolation. (A) The column scatter plots depict the cumulative immobility time of socially isolated (SI) and group-housed (GH) male and female mice that were subjected to a 6-minute forced swim test (FST). The columns and error bars represent the mean and SEM, respectively. The data were analyzed using two-way repeated measures ANOVA. (B) The column scatter plots depict the immobility duration during the last 4 minutes of the FST. The columns and error bars represent the mean and SEM, respectively. The data were analyzed using a two-tailed unpaired t-test. *p < .05. (C) The column scatter plots depict the latency to the first immobility episode lasting more than one second during the forced swim test in mice. The columns and error bars represent the mean and SEM, respectively. The data were analyzed using a two-tailed unpaired t-test. *p < .05. (D) The column scatter plots depict the concentrations of corticosterone in the serum of male and female mice. The columns and error bars represent the mean and SEM, respectively. The data were analyzed using a two-tailed unpaired t-test. *p < .05.
Discussion
In this study, young mice that were socially isolated exhibited increased BP (systolic, diastolic, and mean) and body weight gain in females, decreased serum corticosterone levels in males, and increased HR in both males and females. In addition, serum metanephrine levels were not significantly different between SI and GH male or female animals. There was no evidence of elevated immobility in the FST in either SI males or SI females; however, the decreased latency to immobility in SI females suggests that 8 weeks of SI may have some influence on depression-like behavior. In our study, mice were both socially isolated and deprived of environmental enrichment because we believe that environmental deprivation is a significant factor in human SI (which we sought to replicate). Changes to the environment and/or enrichment promote social interactions and increase sensory, motor, and cognitive stimulation, which are often absent or severely limited in housebound or solitary individuals. Thus, all the observed effects in this study, including the depression-like behavior, BP, and HR, in the SI group represent the “sum” effect of social and environmental deprivation.
Multiple human studies involving middle-aged or older adults have found that a lack of social relationships or limited social participation is strongly associated with hypertension, whereas stronger social connections are associated with lower hypertension.10,31-35 In addition, SI in childhood and adolescence increases the risk of CVD risk and leads to hypertension in young adulthood. 36 In the present study, SI mice displayed significant changes in BP and HR. HR was significantly increased in both SI males and SI females; however, only SI females exhibited increased systolic, diastolic, and mean BP. Sex-specific changes in BP were recently reported in middle-aged and elderly adults with little to no social participation, compared to those who had social participation. 10 The risk of hypertension is higher among women who lack a partner, have limited social relationships, or have a small social network. In contrast, the risk of hypertension is lower among men who live alone compared to those who live with others. Our finding that SI female mice exhibited higher BP than GH female mice is consistent with the notion that women who lack social relationships are at higher risk for hypertension. Furthermore, similar to the findings in humans, young female mice may also be more susceptible to SI-related hypertension compared to male mice.
In addition to the changes in BP and HR observed in this study in SI animals, a recent study found that chronic social stress (8 weeks of SI with intermittent, twice-daily forced social interaction for 20 consecutive days) in C57BL/6 mice significantly altered cardiovascular parameters and responses to global ischemia and reperfusion in the isolated heart preparation. 37 The hearts of females subjected to social stress demonstrated an increase in coronary resistance, but no change in ischemic tolerance. In contrast, male mice subjected to social stress exhibited both coronary resistance and ischemic tolerance, in comparison to animals housed in same-sex social groups. In addition, male socially stressed mice demonstrated enhanced myocardial contractility and lusitropy (cardiac relaxation), but female socially stressed animals showed only increased lusitropy. These findings suggest that, as an outcome of SI stress, males may become more susceptible to myocardial ischemia/infarction (due to increased force of contraction and reduced ischemic tolerance), while females may become more susceptible to coronary ischemic events (due to coronary dysfunction). 37 Similar changes in cardiac function may occur in humans in response to prolonged SI stress and may contribute to or play a role in the development of hypertension or other cardiovascular conditions in men and women who experience SI stress.
The proposed mechanisms underlying the increase in BP associated with SI and loneliness are impaired vasodilation by glucocorticoid and epinephrine-mediated vasoconstriction. 38 SI and loneliness activate the hypothalamic–pituitary–adrenal (HPA) axis and the sympathetic adrenomedullary axis.39,40 The activation of the HPA axis stimulates the adrenal cortex to release glucocorticoids, primarily cortisol in humans and corticosterone in rats and mice.41-46 Glucocorticoids inhibit the expression of the endothelial nitric oxide synthase gene, resulting in decreased nitric oxide production and impaired vasodilation. 38 When the sympathetic adrenomedullary axis is activated, epinephrine (and smaller amounts of norepinephrine and dopamine) is released from the adrenal medulla into the bloodstream. 40 Epinephrine, a powerful vasoconstrictor, elevates BP. Because serum levels of corticosterone (a rodent stress hormone) and metanephrine (a metabolite of epinephrine) were not significantly elevated in SI females, it appears that the increase in BP observed in this study is unrelated to increased adrenocortical or adrenomedullary activity. Reduced levels of corticosterone in SI males did not result in significant changes in BP. This may be because reduced corticosterone levels are still within the physiological range. 47 In support of our conclusion, chronic social stress in the aforementioned study had no effect on circulating levels of norepinephrine, epinephrine, or corticosterone in female mice; however, norepinephrine was elevated in male mice. 37
Based on our current understanding of the stress response, chronic stress may induce persistent hypercortisolemia, which promotes an increase in food consumption and body weight gain. However, while some studies in SI animals have found an increase in food intake, adiposity, and weight gain, others have found a decrease in body weight gain as compared to GH animals.48-56 Although differences in the tested animal strains, duration or intensity of SI stress, and diet may contribute to these discrepancies, studies in humans have also shown that chronic stress can lead to both an increase and a decrease in food consumption and body weight, similar to animal studies.57-60 Interestingly, our study revealed sex-specific differences (opposite effects in both males and females) in body weight gain in response to SI. In comparison to GH animals of the same sex, young female mice housed alone gained significant weight, whereas male mice housed alone tended to lose weight (p = .0618). In addition, the positive correlation between body weight gain and diastolic BP in female mice suggests that the elevated BP of SI females could be attributed to an increase in total peripheral vascular resistance due to excessive weight gain.
SI and/or loneliness can exacerbate or precipitate mood disorders such as depression in humans and depression-like behaviors in rodents. An increase in total immobility time during FST is indicative of depression-like behavior in the animal.28,29 Previous studies reviewed by Bogdanova et al. in 2013 indicate that the animal's age, strain, sex, and level of SI stress can affect the outcome of this test, resulting in different or contradictory results. 61 The decreased latency to immobility was statistically significant in SI females but not in SI males. The decreased latency to immobility in the FST in SI females suggests that there may be some influence of SI on depression-like behavior. The immobility time (during the 6-minute of FST) of either young male or female mice subjected to SI was not statistically different from GH animals of the same sex. However, significantly decreased immobility time in SI males during the last 4 minutes of FST reveals that males are less sensitive than females for the SI-induced depression-like behavior. Overall, this study demonstrates that 8 weeks of SI produces mild depression-like behavior in young female mice. In part, our results in young mice are consistent with those of Almeida et al., who revealed a significant association between SI and depression in children and adolescents. 62 Depression is associated with weight changes (gain or loss) in humans. In this study, SI females that exhibited mild depression-like behavior showed significant weight gain during the SI period.
Our results corroborate to some extent with those of a recent study that used the same strain and age of animals of both sexes and chronic SI as in our study and found no depression-like behavior in either males or females. 63 However, the FST by Wu et al., was conducted during the dark cycle, included the adaptation stage (pre-test one day before the actual test), and all mice were able to smell one another. Based on additional evidence from this study, neither male nor female mice developed depression-like symptoms after 8 weeks of SI. Conducting FST during the dark phase, as opposed to the inactive light phase, could have impacted the behavioral outcome in our study because the SI circumstances in our study were more rigorous (socially isolated animals could not smell one another). In the study by Wu et al., in response to the highly palatable/rewarding properties of sucrose, SI animals did not exhibit anhedonia (a core symptom of depression in humans) during the sucrose preference test. In addition, SI animals did not exhibit behavioral despair in the tail-suspension test. However, these behavioral tests were performed up to 12 days after SI, and it is uncertain whether the behavioral effects of SI last that long, despite the increased peripheral cytokines and neuroinflammation found in their study. In addition, 1 month of SI in 18-month-old female mice had no effect on the immobility duration in the FST, suggesting that SI also has no effect on depression-like behavior in older females. 64 On the contrary, 6 weeks of SI in young male rats 24 and mice 20 as well as 4 weeks of SI in adult mice 65 and prairie voles 66 induced depression-like behavior. However, these results were not replicated either in our study or the study by Wu et al. 63 Differences in species, strain, age, cage enrichment, housing conditions, and duration of SI may account for the lack of substantial depression-like behavior in socially isolated animals in this study.
Young adult male mice that were exposed to 12 weeks of SI and 6 weeks of unpredictable chronic mild stress during the first 6 weeks of SI demonstrated robust depression-like behavior. 67 These mice exhibited a 25% increase in immobility and a 15% decrease in sucrose consumption during the sucrose preference test. Furthermore, C57BL/6 mice that were subjected to chronic SI for 8 weeks, along with brief periods of forced social interaction twice a day for the last 20 days, exhibited more evident physiological and behavioral changes, compared to the preceding SI period alone. 37 Together, these findings suggest that the likelihood of developing depression-like behavior increases when SI is combined with other stressors. In the present study, the induction of 8 weeks of SI and the lack of cage enrichment induced only mild depression-like behavior in the FST in female mice.
Interestingly, in our study, blood corticosterone levels were reduced in male SI mice compared to GH mice, consistent with previous studies in rats and mice.65,68,69 Contrary to the observed decrease in corticosterone, elevated glucocorticoid levels are often associated with depression in humans and animal models of depression. 70 Because the significantly lower corticosterone levels in SI males were still within physiological range, we did not expect or find evidence of depression-like behavior in FST.
Recent research has highlighted the importance of social touch for maintaining physical and mental health.71,72 The skin is richly innervated with sensory fibers (C-tactile afferents), whose primary function is to detect and convey to the brain the emotional and hedonic components of tactile stimuli. In a recent study, paired-housed male mice were found to spend approximately 54% of their 24-hour day in direct physical contact. 72 Despite male mice being extremely territorial and aggressive toward one another, they prefer to live in close proximity to each other, demonstrating the significance of social touch in their daily activities. Positive physical contact promotes relaxation and reduces stress, anxiety, and pain. These beneficial/therapeutic effects are mediated in part by increased vagal activity (which reduces HR), and the release of endogenous opioids and the neuropeptide oxytocin, which inhibits the HPA axis, pain pathways, and the sympathetic nervous system. The increased HR observed in our SI male and SI female mice may therefore be attributed to a decrease in vagal tone caused by prolonged lack of social contact. In addition, because females are typically more gregarious and sociable than males and engage in more positive social touch, they may be more affected by SI due to a lack of physical contact, which may contribute to their higher BP and HR. It has been shown that tactile stimulation decreases BP and HR in conscious rats and inhibits adrenal sympathetic efferent nerve activity and catecholamine secretion in anesthetized animals, although neither male nor female SI mice in the present study exhibited elevated levels of metanephrine.73-75 Interestingly, split-housed male mice that were deprived of social touch for 1 month, but allowed olfactory, auditory, and visual communication, exhibited more anxiety-like behavior in the open field test and light box test than single- or pair-housed mice. However, neither the duration of immobility in FST nor in the tail suspension test was significantly altered in either the single- or split-housed animals, suggesting that a lack of physical touch, in and of itself, does not elicit a notable depression-like behavior, which is consistent with our results. 72
This study is limited by the measurement of BP and HR in mice under isoflurane anesthesia. Although isoflurane is the most commonly used inhalational anesthetic in animal research and clinical practice, it can affect cardiovascular function. When used at a concentration of 3%, isoflurane significantly reduces the BP and HR in rodents. 76 To minimize the impact on cardiovascular parameters, all experimental animals in this study were administered isoflurane at or below 2%. This anesthetic treatment was sufficient to eliminate the righting reflex and all bodily movements. This study also lacks monitoring of the female estrous cycle, which typically lasts 4 to 5 days. Some evidence suggests that the phase of the estrous cycle can influence tonic immobility, as measured by the tail suspension test. 77
In summary, this study found that SI in young mice increased the heart rate of both males and females but only the BP in females. Increased BP is associated with excessive weight gain in SI females. SI for 8 weeks had mild effect on depression-like behavior. Our findings may shed light on the relationship between SI or loneliness and CVD risk factors in adolescents.
Footnotes
Acknowledgments
The authors thank Christina Constantinidou for her assistance with the formatting of the manuscript.
Author Contributions
KKV, PG, SM, and MBS contributed to conceptualization and supervision. JB, SRC, and CAF were involved in animal care and social isolation procedures. CAF, SRC, BCW, JB, RED, and PU were involved in the methodology, investigation, and acquisition of data. KKV and CAF performed data analysis and interpretation. KKV prepared, and CAF edited the manuscript. All the authors reviewed and approved the final manuscript.
Data Availability Statement
All relevant data supporting the key findings of this study are available within the article. Any relevant raw data will be shared upon a reasonable request to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Illinois College of Medicine Peoria. We declare that all animal procedures were conducted in accordance with the IACUC-approved animal protocol.
Funding
The authors disclosed receipt of the following financial support: KKV and MBS are the recipients of National Institutes of Health research grants R01NS102573 and OT2OD026557, respectively. The funders had no role in study design, data collection, analysis, interpretation, manuscript preparation, or publication decision. The content of this study is the sole responsibility of the authors and does not necessarily reflect the official position of the funders.
