Abstract
Objective
To investigate the optimal dose of dexmedetomidine for attenuating the haemodynamic response during emergence from anaesthesia.
Methods
Patients undergoing laparoscopic total hysterectomy were randomly allocated to receive 0.9% normal saline (control group) or dexmedetomidine (0.5 µg/kg, 0.75 µg/kg or 1.0 µg/kg 30 min) before extubation. Heart rate, systolic and diastolic arterial pressure and extubation time were measured before drug administration, immediately after the end of drug administration, 10 min after the end of drug administration, immediately after extubation and 5 min after extubation.
Results
A total of 120 patients were included in the study (30 per group). Systolic and diastolic arterial pressure and heart rate after drug administration were significantly lower in all three dexmedetomidine groups compared with controls. Extubation times in the 0.75 and 1.0 µg/kg dexmedetomidine groups were significantly longer than in the control group.
Conclusion
Intravenous infusion of 0.5 µg/kg dexmedetomidine 30 min before the end of surgery attenuated the haemodynamic responses during emergence without prolonging the extubation time. Dexmedetomidine doses higher than 0.5 µg/kg did not exert additional positive effects on cardiovascular responses, but did significantly prolong the extubation time.
Introduction
Emergence from anaesthesia and tracheal extubation are commonly associated with haemodynamic and plasma catecholamine responses due to postoperative pain, tracheal irritation and the use of anticholinergic drugs.1,2 When these responses are excessive, they may result in adverse effects such as increased intracranial and intraocular pressure, myocardial ischaemia and arrhythmias.3,4 Many studies have investigated strategies to attenuate hyperdynamic responses, including extubation under deep anaesthesia and the use of calcium-channel blockers, β-blockers or short-acting opioids.5–7 However, although extubation during the deep planes of anaesthesia avoids cardiovascular stimulation, it can also depress the respiratory and circulatory systems and occasionally results in difficulty managing the upper airway; 5 drug treatments have not been fully successful.6,7
Dexmedetomidine is a highly specific α2-agonist with sedative, analgesic and sympatholytic effects without significant respiratory depression.8–10 When dexmedetomidine was administered before and during general anaesthesia, its sympatholytic effect was manifested by decreases in arterial blood pressure, heart rate and noradrenaline release during emergence from anaesthesia and extubation.11,12 Research has focused on the haemodynamic and sedative effects of dexmedetomidine during emergence from anaesthesia,11–14 but to date the optimal dose of dexmedetomidine for stable haemodynamics during emergence has not been studied. The present study aimed to determine the optimal dose of dexmedetomidine for attenuating the haemodynamic response during emergence from anaesthesia.
Patients and methods
Patients
Female patients aged 18–60 years, with American Society of Anesthesiologists (ASA) status I or II, scheduled for total laparoscopic hysterectomy between August 2012 and January 2013 at St Vincent Hospital, Catholic University of Korea, Suwon, South Korea, were enrolled. Patients with asthma, hypertension, dysrhythmia, morbid obesity (body mass index > 25 kg/m2) and those who required transfusion during surgery were excluded from the study.
Patients were randomly allocated in equal numbers to one of four groups: control group S (saline), group D0.5 (0.5 µg/kg dexmedetomidine), group D0.75 (0.75 µg/kg dexmedetomidine) or group D1.0 (1.0 µg/kg dexmedetomidine). The group allocation and names were coded and remained concealed until completion of the statistical analyses. Randomization was performed using a block randomization technique with opaque sealed envelopes, performed by an anaesthesiologist who was not involved in the anaesthetic management of the patients or data collection. Random numbers were grouped in blocks in an allocation ratio of 1:1 for the four study groups. Study drugs were prepared by nurses who were not involved in the study; all of the drugs were diluted in a total volume of 50 ml normal saline.
All patients provided written informed consent. The study protocol was approved by the institutional review board of St Vincent Hospital (VC12MISION124) and listed on a World Health Organization-recognized registry (KCT0000822).
Anaesthesia induction and maintenance
In the operating room, routine monitoring was performed, including blood pressure measurement, electrocardiography, pulse oximetry and capnography. Anaesthesia was induced with 2–2.5 mg/kg propofol and 0.6 mg/kg rocuronium. After endotracheal intubation, anaesthesia was maintained with 6–8 volume% desflurane and 50% nitrous oxide in oxygen. Mechanical ventilation was adjusted to maintain the end-tidal carbon dioxide partial pressure at 30–35 mmHg. During surgery, 6 ml/kg per h Ringer’s lactate was infused; if blood loss occurred, the equivalent volume of 6% hydroxyethyl starch was also infused.
In all patients, intraoperative opioids were not administered and local anaesthetic was not infiltrated into the laparoscopic incision site.
Arterial blood pressure and heart rate were maintained between 80% and 120% of the preoperative values by increasing or decreasing the concentration of desflurane during surgery. Severe hypertension (defined as systolic arterial pressure [SAP] > 180 mmHg) or tachycardia (defined as heart rate > 100 beats/min lasting > 3 min) during the extubation period that could not be controlled by increasing the concentration of desflurane were treated with intravenous 20 µg/kg nicardipine or 0.5 mg/kg esmolol, respectively. After administration of the study drug, hypotension (defined as SAP < 80 mmHg) was treated with a 10-mg intravenous bolus of ephedrine; bradycardia (defined as heart rate < 50 beats/min) was treated with a 0.5-mg intravenous bolus of atropine.
Emergence from anaesthesia
Approximately 30 min before the anticipated completion of surgery, an anaesthesiologist blind to the drug and group allocation administered the study drug intravenously over 10 min. In addition, 0.5 mg/kg ketorolac tromethamine was given intravenously 10 min before the anticipated end of surgery; 5 min before the anticipated end of surgery the desflurane concentration was decreased to 5 volume%. Desflurane was discontinued at the end of surgery and the lungs were ventilated with 100% oxygen. Residual neuromuscular blockade was reversed with 6 µg/kg glycopyrrolate and 0.15 mg/kg pyridostigmine in all patients. Extubation was performed once the patient was breathing spontaneously.
Heart rate (HR), SAP and diastolic arterial pressure (DAP) values were recorded before study drug administration, immediately after the end of drug administration, 10 min after the end of drug administration, immediately after extubation and 5 min after extubation. The extubation time was defined as the time between discontinuation of inhalation agents and extubation.
Statistical analyses
The number of patients required in each group was determined using a power calculation based on data from our preliminary study of dexmedetomidine and its influence on the haemodynamic responses (unpublished). This calculation established that 30 patients were required in each group for a type I error of 0.05, a type II error of 0.2, Δ = 18 and SD = 20 for the SAP parameter. Recruitment was increased by 10% to compensate for unexpected loss.
The within-group data for SAP, DAP and HR were analysed using repeated-measures analysis of variance (ANOVA); between-group differences were analysed using the Student’s t-test. Comparisons among groups were made using covariance analysis followed by Bonferroni post-hoc testing. Extubation times were compared between groups using one-way ANOVA followed by Dunnett T3 post-hoc testing. A P-value < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS® software, version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
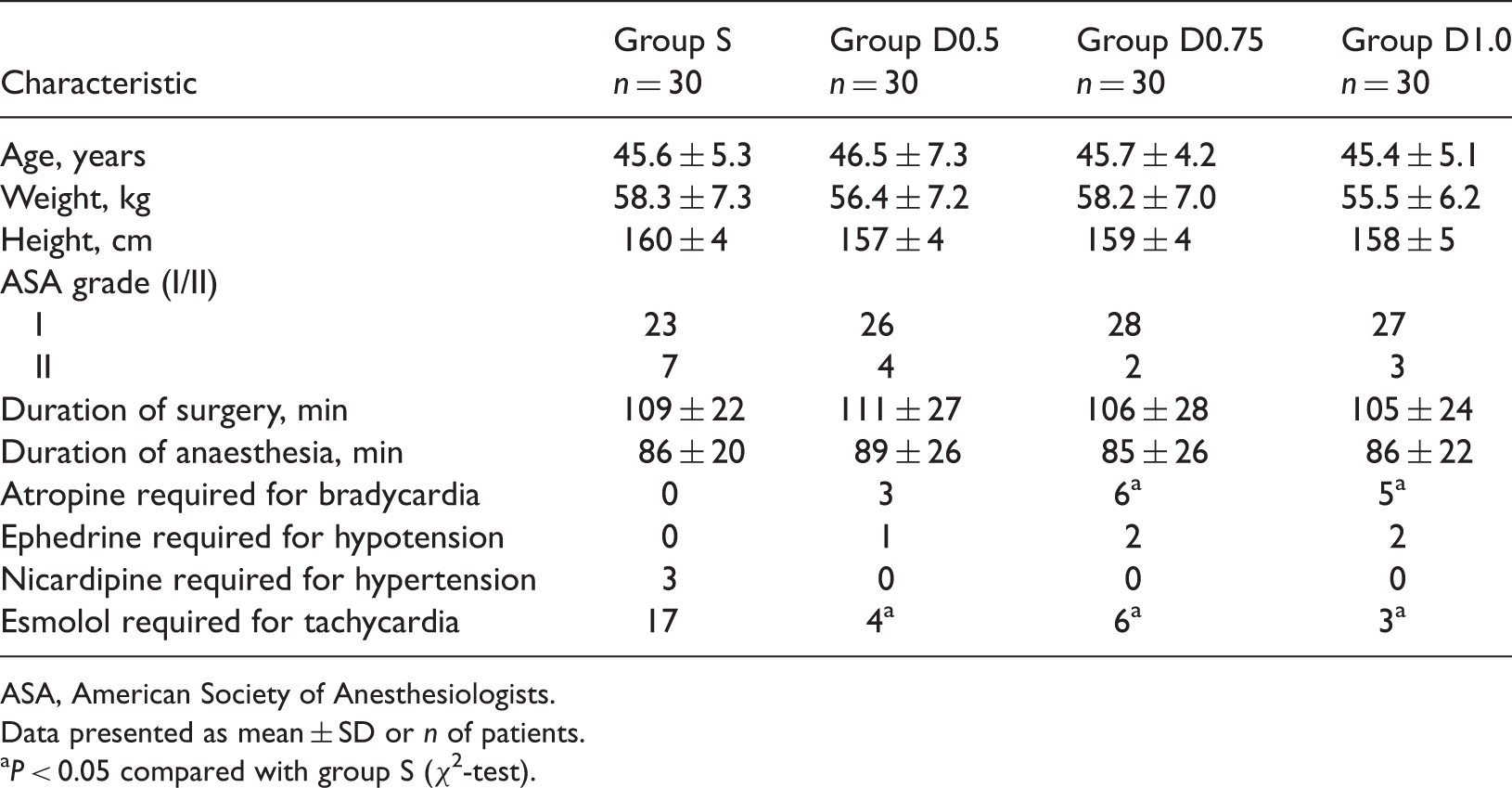
A total of 132 patients were randomized into the study. Of these, 12 were excluded due to transfusion or change to open abdominal hysterectomy. Of the remaining patients, 30 patients were allocated to each intervention group and received the allocated treatment (Figure 1). Baseline patient characteristics and the duration of surgery and anaesthesia were similar in the four groups (Table 1).
Flow diagram of progress through the study in an investigation of the optimal dose of dexmedetomidine for attenuating haemodynamic response during emergence from anaesthesia. Characteristics of, and perioperative data from, patients undergoing total laparoscopic hysterectomy who received saline (group S) or dexmedetomidine at doses of 0.5 µg/kg (group D0.5), 0.75 µg/kg (group D0.75) or 1.0 µg/kg (group D1.0) before extubation. ASA, American Society of Anesthesiologists. Data presented as mean ± SD or n of patients. P < 0.05 compared with group S (χ2-test).
Hypertension requiring treatment with nicardipine was observed in three patients in group S during extubation, but not in any of the dexmedetomidine groups. Tachycardia requiring treatment with esmolol occurred during extubation in significantly more patients in group S than in the dexmedetomidine groups (P < 0.05, Table 1). The number of patients who received ephedrine for hypotension after administration of the study drug did not differ significantly among the four groups. However, significantly more patients in the D0.75 and D1.0 groups required atropine to treat bradycardia after administration of the study drug compared with group S (P < 0.05, Table 1).
The SAP was significantly lower in all three dexmedetomidine groups compared with group S at all times from 10 min after the end of drug administration (P < 0.001, Figure 2). In addition, the SAP in groups D0.5 and D0.75 was significantly lower than in group S immediately after the end of drug administration (P < 0.001, Figure 2).
Changes in systolic arterial pressure (SAP) during emergence from anaesthesia in patients undergoing total laparoscopic hysterectomy who received saline (group S) or dexmedetomidine at doses of 0.5 µg/kg (group D0.5), 0.75 µg/kg (group D0.75) or 1.0 µg/kg (group D1.0) before extubation. T1, before drug administration; T2, immediately after the end of drug administration; T3, 10 min after the end of drug administration; T4, immediately after extubation; T5, 5 min after extubation. Data presented as mean ± SD; *P < 0.001 compared with group S (repeated-measures analysis of variance).
The DAP was significantly lower in all three dexmedetomidine groups compared with group S 10 min after the end of drug administration (P < 0.05, Figure 3). In addition, the DAP was significantly lower in group D0.5 immediately after the end of drug administration and in group D1.0 immediately after extubation and 5 min after extubation, compared with group S (P < 0.05, Figure 3).
Changes in diastolic arterial pressure (DAP) during emergence from anaesthesia in patients undergoing total laparoscopic hysterectomy who received saline (group S) or dexmedetomidine at doses of 0.5 µg/kg (group D0.5), 0.75 µg/kg (group D0.75) or 1.0 µg/kg (group D1.0) before extubation. T1, before drug administration; T2, immediately after the end of drug administration; T3, 10 min after the end of drug administration; T4, immediately after extubation; T5, 5 min after extubation. Data presented as mean ± SD. *P < 0.05 compared with group S (repeated-measures analysis of variance).
The HR results were similar to those for SAP. The HR was significantly lower in all dexmedetomidine groups compared with group S at all times after drug administration (P < 0.05), except for group D0.5 5 min after extubation (Figure 4). Among the dexmedetomidine groups, HR was significantly lower in group D1.0 than in group D0.5 5 min after extubation (P < 0.05, Figure 4).
Changes in heart rate during emergence from anaesthesia in patients undergoing total laparoscopic hysterectomy who received saline (group S) or dexmedetomidine at doses of 0.5 µg/kg (group D0.5), 0.75 µg/kg (group D0.75) or 1.0 µg/kg (group D1.0) before extubation. T1, before drug administration; T2, immediately after the end of drug administration; T3, 10 min after the end of drug administration; T4, immediately after extubation; T5, 5 min after extubation. Data presented as mean ± SD. *P < 0.05 compared with group S and †P < 0.05 compared with group D0.5 (repeated-measures analysis of variance).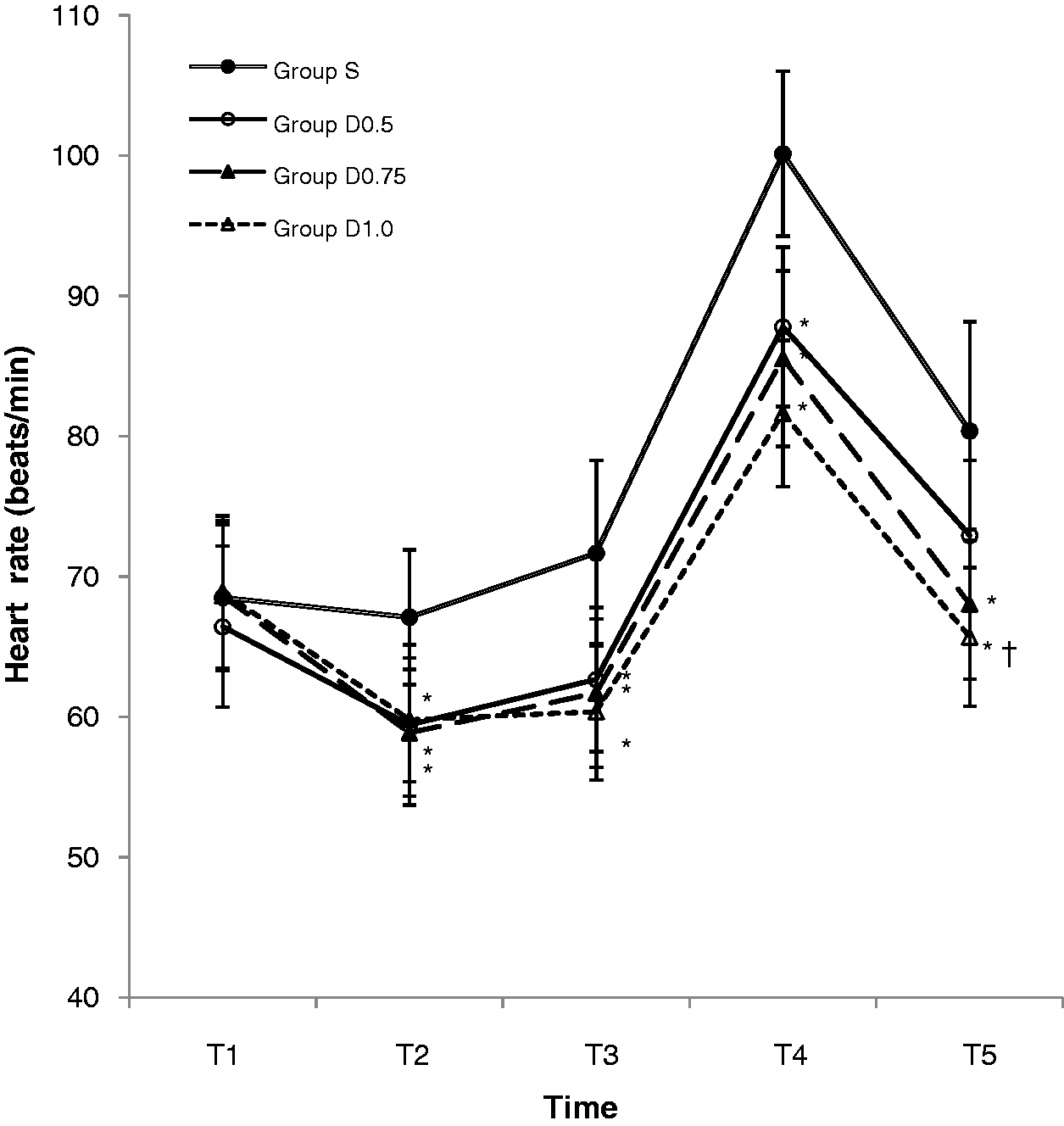
Extubation times in the four groups are shown in Figure 5. The time taken for extubation was significantly longer in groups D0.75 and D1.0 compared with group S (P < 0.05).
Extubation time in patients undergoing total laparoscopic hysterectomy who received saline (group S) or dexmedetomidine at doses of 0.5 µg/kg (group D0.5), 0.75 µg/kg (group D0.75) or 1.0 µg/kg (group D1.0) before extubation. Data presented as mean ± SD. *P < 0.05 compared with group S (one-way analysis of variance).
Discussion
The present study demonstrated that prophylactic use of dexmedetomidine 0.5 µg/kg was effective for attenuating cardiovascular responses during emergence without prolonging extubation time.
Substantial haemodynamic stimulation during tracheal extubation may adversely affect patients. 2 Changes in ejection fraction and cardiac work during recovery can induce undesirable complications, such as myocardial ischaemia, in vulnerable individuals.15,16 Stabilization of blood pressure and heart rate throughout emergence and the recovery period is especially important in patients with coronary artery disease. 17 Anaesthesiologists need to be able to perform a smooth extubation with stable haemodynamics.
Dexmedetomidine, like other α2-adrenergic agonists, exerts sympatholytic effects by activating inhibitory α2 receptors in both the central nervous system and the peripheral sympathetic nerve endings, inhibiting noradrenaline release. 18 The inhibition of sympathetic transmitter release can be measured in humans as a decline in the plasma concentration of noradrenaline. 19 The prevention of a catecholamine reaction to stress by dexmedetomidine may be of clinical importance, since there is a clear relationship between surgical events known to produce intense sympathetic stimulation and perioperative myocardial infarction. 16
Dexmedetomidine causes brief biphasic, dose-dependent cardiovascular effects via the stimulation of α2 receptors. 20 At high doses or doses that are quickly administered, dexmedetomidine stimulates the α2 receptors in vascular smooth muscle, and causes a transient increase in blood pressure and a reflex drop in heart rate. 10 The initial transient increase in blood pressure, presumably mediated by peripheral vasoconstriction, is followed by a longer lasting reduction in blood pressure, presumably due to both centrally and peripherally mediated sympatholytic actions. 4 A reduction in circulating catecholamine is consistent with the long-lasting reduction in sympathetic tone caused by this compound.
Bloor et al. 21 reported that when dexmedetomidine was given as a 2-min intravenous infusion in healthy volunteers, lower doses (0.25 and 0.5 µg/kg) resulted in a monophasic reduction in mean arterial blood pressure, whereas larger doses (1.0 and 2.0 µg/kg) resulted in biphasic responses. With larger doses, the maximal early transient pressor effect was associated with a doubling of the systemic vascular resistance and a reduction in cardiac output. The second, biphasic pressure effect involved a longer lasting reduction in mean arterial blood pressure, with concomitant decreases in HR and cardiac output. 21 Dexmedetomidine also reduced plasma noradrenaline levels for >4 h at all four doses. 21 In the present study, dexmedetomidine was infused over 10 min, and no biphasic blood pressure responses were seen, even with a dose of 1.0 µg/kg. This suggests that the transient pressor phase can be avoided by using a slower infusion rate, which is important for patients with impaired cardiac function.
In the present study, the effects of 0.5 µg/kg dexmedetomidine on haemodynamic responses during extubation were not significantly different from those of 0.75 or 1.0 µg/kg dexmedetomidine. However, 0.5 µg/kg dexmedetomidine did not prolong extubation time compared with the control group, while doses of 0.75 or 1.0 µg/kg dexmedetomidine did delay extubation by increasing amounts, suggesting that prolongation of the extubation time was dose dependent. Although the extubation time in the 0.5 µg/kg dexmedetomidine group was longer than that in the control group, the difference was not statistically significant. These findings are comparable with those of previous studies. Guler et al. 14 reported that single-dose 0.5 µg/kg dexmedetomidine attenuated airway and circulatory reflexes during extubation, but there was no significant difference in extubation time between the dexmedetomidine and control groups. In addition, Bicer et al. 22 reported that patients who received 1.0 µg/kg dexmedetomidine demonstrated a prolonged extubation time compared with a control group after surgery. In the present study, although extubation times were prolonged in group D0.75 and D1.0 compared with group S, these doses may deserve consideration for providing haemodynamic stability throughout emergence and recovery in particular circumstances (such as in patients with myocardial ischaemia).
The present study has a number of limitations. First, because 0.5 µg/kg dexmedetomidine was the lowest dose tested, it is not possible to state that this is the minimum required dose. Bloor et al. 21 reported that infusions of 0.25 or 0.5 µg/kg dexmedetomidine decreased mean arterial blood pressure by 14% and 16%, respectively, in healthy volunteers. Therefore, a dexmedetomidine dose <0.5 µg/kg may be sufficient to attenuate cardiovascular responses during anaesthesia emergence. Secondly, the occurrence of coughing or other airway reflexes was not investigated. The presence of the endotracheal tube leads to reflex responses, the most common of which is coughing, which can cause hypertension and tachycardia. Dexmedetomidine has been reported to facilitate tolerance of the endotracheal tube and to significantly reduce coughing during extubation. 14 Thirdly, the sample population consisted of only young adult female patients who were relatively healthy. It needs to be determined whether the present findings can be generalized to male patients, and other age groups.
In conclusion, intravenous infusion of 0.5 µg/kg dexmedetomidine 30 min before the end of surgery attenuated haemodynamic responses during emergence, without prolonging the extubation time, in patients undergoing total laparoscopic hysterectomy. Dexmedetomidine doses higher than 0.5 µg/kg did not exert additional positive effects on the cardiovascular responses, but did significantly prolong the extubation time.
Footnotes
Declaration of conflicting interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
