Abstract
Addiction has been conceptualized as a three-stage cycle—
Conceptual Framework
What Is Stress?
Selye 1 defined stress as responses to demands (usually noxious) upon the body that historically have been defined by various physiological changes that include activation of the hypothalamic–pituitary–adrenal (HPA) axis. However, a definition of stress that is more compatible with its many manifestations in the organism is “anything which causes an alteration of psychological homeostatic processes.” 2 In fact, in a seminal paper, Mason 3 argued the importance of psychological stress for eliciting a stress response, even among physical stressors and that many physical challenges absent psychological stress are not stressful.
The physiological response that is most associated with a state of stress is an elevation of glucocorticoids that derive from the adrenal cortex. This response is controlled by the HPA axis. Vale et al. 4 first demonstrated that corticotropin-releasing factor (CRF) initiates the HPA axis neuroendocrine stress response (adrenocorticotropic hormone and ultimately glucocorticoids) by binding CRF1 receptors in the anterior pituitary after release into portal blood. CRF from the paraventricular nucleus of the hypothalamus was then identified as the primary controller in the HPA axis. Glucocorticoids function to increase and maintain blood sugar by elevating gluconeogenesis, and they decrease immune function by blocking proinflammatory proteins. These responses facilitate mobilization of the body in response to acute stressors. However, we now know that neurocircuits in the brain mediate behavioral responses to stressors and play a major role in “psychological homeostasis.”
Of relevance for this review, comorbidity between addictive and stress-related disorders is high. In the third wave of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), the 12-month odds ratio for posttraumatic stress disorder (PTSD; i.e., the psychiatric disease most directly linked to stress exposure) and any substance use disorder was 1.3; the lifetime odds ratio was 1.5. 5 Furthermore, in the National Comorbidity Survey-Replication, a diagnosis of PTSD at Time 1 was associated with odds ratios of 3.2 and 5.4 for alcohol and illicit drug dependence, respectively, at Time 2, 10 years later, among those individuals not substance dependent at Time 1. 6
What Is Addiction?
Addiction can be defined in many different ways, but one definition that has been generally adopted in the field is that addiction is a chronic, relapsing disorder that is characterized by a compulsion to seek and take drugs and the loss of control over drug intake. Others have emphasized a further characteristic, notably “the emergence of a negative emotional state (e.g., dysphoria, anxiety, and irritability) that defines a motivational withdrawal syndrome when access to the drug is prevented.”
7
Indeed, some theorists have argued that such a negative emotional state is the defining feature of dependence on a drug: The notion of dependence on a drug, object, role, activity or any other stimulus-source requires the crucial feature of negative affect experienced in its absence. The degree of dependence can be equated with the amount of this negative affect, which may range from mild discomfort to extreme distress, or it may be equated with the amount of difficulty or effort required to do without the drug, object, etc.
8
Another framework with which to conceptualize drug addiction is the impulsivity–compulsivity continuum, in which
In contrast, “compulsivity can be characterized by perseverative, repetitive actions that are excessive and inappropriate to a situation.” 15 Individuals who suffer from compulsions often recognize that the behaviors are harmful, but they nonetheless feel emotionally compelled to perform them. Performance of these behaviors reduces tension, stress, or anxiety.15,16 Operationally, in animal models, responding for a drug or alcohol in the face of adverse consequences 17 or responding for a drug or alcohol on a progressive-ratio schedule of reinforcement 18 has been argued to reflect compulsivity. Thus, in addition to the positive reinforcement associated with high impulsivity linked to the early stages of the addiction process, an additional source of motivation is recruited, namely negative reinforcement.
This impulsivity–compulsivity continuum has a nosological history. Subjects with classic atypical impulse control disorders, such as kleptomania, experience an increasing sense of tension or arousal before committing an impulsive act; pleasure, gratification, or relief at the time of committing the act; and regret, self-reproach, or guilt following the act.
19
In contrast, subjects with classic compulsive-like disorders, such as obsessive–compulsive disorder, experience anxiety and stress before committing a compulsive repetitive behavior and relief from the stress by performing the compulsive behavior.
19
We have argued that drug addiction progresses from a source of positive reinforcement that may indeed involve more elements of impulsivity to a source of negative reinforcement that may involve more elements of compulsivity (Figure 1).
20
The three-stage cycle of addiction, with the embedded conceptual sources of motivation of positive and negative reinforcement that parallel impulsivity and compulsivity (Figure 1), are not unique to drug addiction and generalize to non-drug or “process” addictions. In a recent review,
21
the authors identified three major domains of neurofunctional impairment related to gambling disorder, namely the loss of control, craving/withdrawal, and the neglect of other areas of life. These domains closely parallel the domains outlined in the three stages of the addiction cycle and the Addictions Neuroclinical Assessment (ANA) framework (see below).
(Top left) Diagram showing the stages of impulse control disorder and compulsive disorder cycles related to the sources of reinforcement. In impulse control disorders, an increasing tension and arousal occurs before the impulsive act, with pleasure, gratification, or relief during the act. Following the act, there may or may not be regret or guilt. In compulsive disorders, there are recurrent and persistent thoughts (obsessions) that cause marked anxiety and stress followed by repetitive behaviors (compulsions) that are aimed at preventing or reducing distress.
19
Positive reinforcement (pleasure/gratification) is more closely associated with impulse control disorders. Negative reinforcement (relief of anxiety or relief of stress) is more closely associated with compulsive disorders (taken with permission from Koob
20
). (Top right) Collapsing the cycles of impulsivity and compulsivity results in the addiction cycle, conceptualized as three major components: 
Neurobiology of Stress
Key highly conserved responses to stressors in the environment comprise fight or flight. A superstructure in the basal forebrain, the extended amygdala, processes fear, threats, and anxiety in humans (i.e., fight or flight responses)7,23 and engages the neurocircuitry of negative emotional states. The extended amygdala shares similarities in morphology, neurochemistry, and connectivity and is composed of the central nucleus of the amygdala (CeA), bed nucleus of the stria terminalis (BNST), and a transition zone in the posterior medial part (shell) of the nucleus accumbens (NAcSh). 24 The extended amygdala receives inputs from various regions of the brain that are involved in emotion, but most importantly the prefrontal cortex. The extended amygdala projects heavily to the hypothalamus and other midbrain structures that are involved in the expression of emotional responses.24,25 When animals are exposed to a stressor, they exhibit an enhanced freezing response to a conditioned fear stimulus, an enhanced startle response to a startle stimulus, the avoidance of open areas, open arms, and heights, and enhanced species-typical responses to an aversive stimulus. All of these responses are at least partially mediated by the extended amygdala. In psychopathology, dysregulation of the extended amygdala has been hypothesized to play a key role in disorders that are related to stress and negative emotional states, such as PTSD, general anxiety disorder, phobias, affective disorders, and addiction.26,27
Two neurochemical systems, CRF and dynorphin, play a key role in the extended amygdala to effect such behavioral changes. Both are also implicated in the psychopathology associated with the extended amygdala, and both are the focus of individual differences in stress pathology. The glucocorticoid response mobilizes the body for physiological responses to stressors; CRF plays another role by mobilizing the body’s behavioral response to stressors via brain circuits outside the hypothalamus. In an early study, CRF was intracerebroventricularly injected into the brain in naive rats, which produced hyperactivity and hyperarousal in a familiar environment but a very pronounced freezing-like response in a novel stressful environment. 28 Subsequent work showed that a prominent system that mediates such responses to CRF and fear and anxiety in general is the extended amygdala. The administration of competitive CRF receptor antagonists was shown to have opposite anti-stress effects. This observation was critical because it confirmed a role for endogenous CRF in behavioral responses to stressors (for review, see Koob and Zorrilla 29 ).
The dynorphin-κ opioid system also plays a key role in affecting behavioral responses to stressors. Dynorphins contain the leucine (leu)-enkephalin sequence at the
Other key neurotransmitter systems, all of which interact with the extended amygdala, that mediate behavioral responses to stressors include norepinephrine, vasopressin, hypocretin (orexin), substance P, proinflammatory cytokines, and key neurotransmitter systems that act in opposition to the brain stress systems, such as neuropeptide Y (NPY), nociceptin, and endocannabinoids. Altogether, these neurochemical systems set the tone and modulate emotional expression, particularly negative emotional states, via the extended amygdala (Figure 2).
33
These stress systems and their relevance for addiction are comprehensively reviewed in Koob.
22
Neural circuitry associated with the three stages of the addiction cycle, with a focus on the 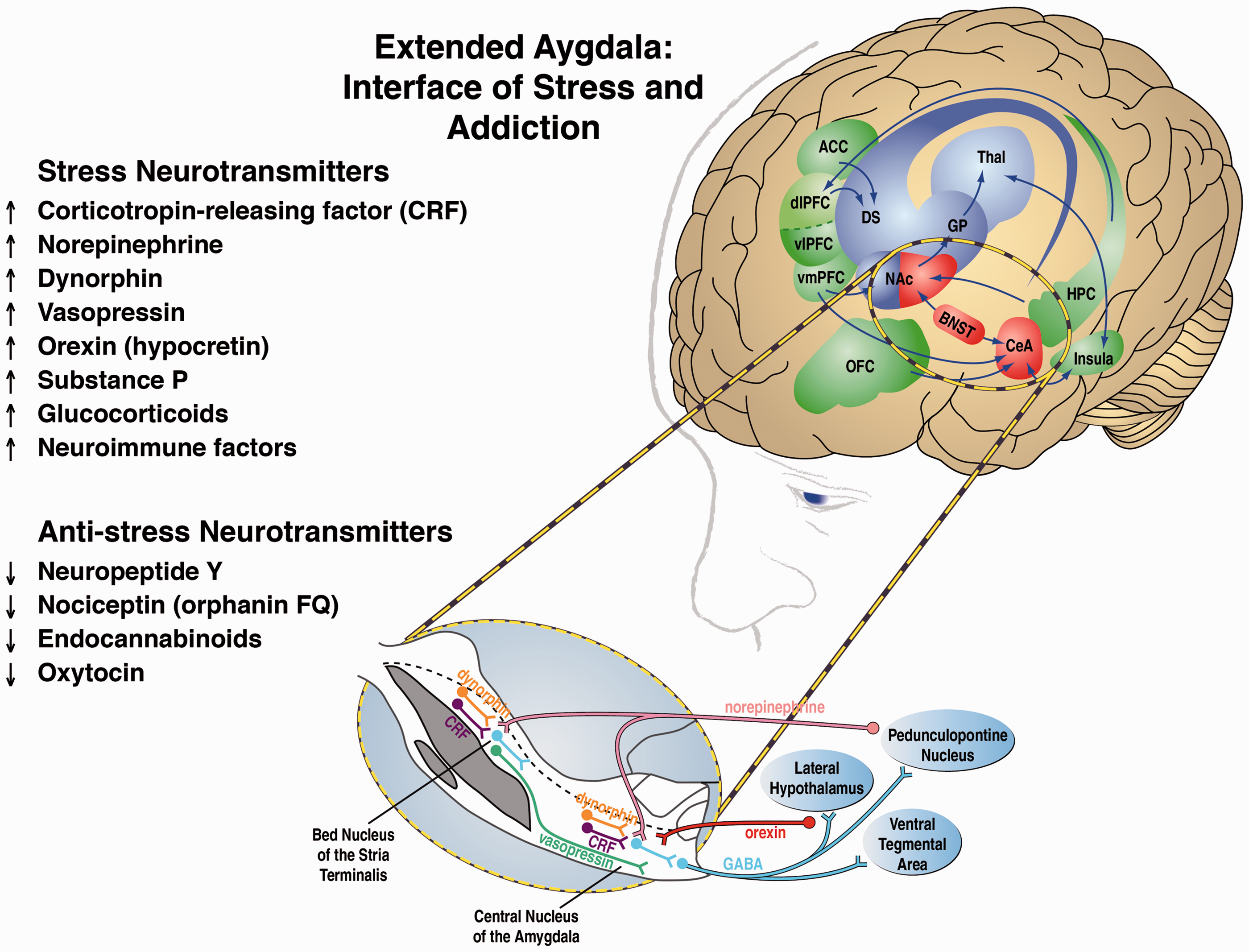
Neurobiology of Addiction
The neurobiological basis of the
Molecular neurocircuits of the
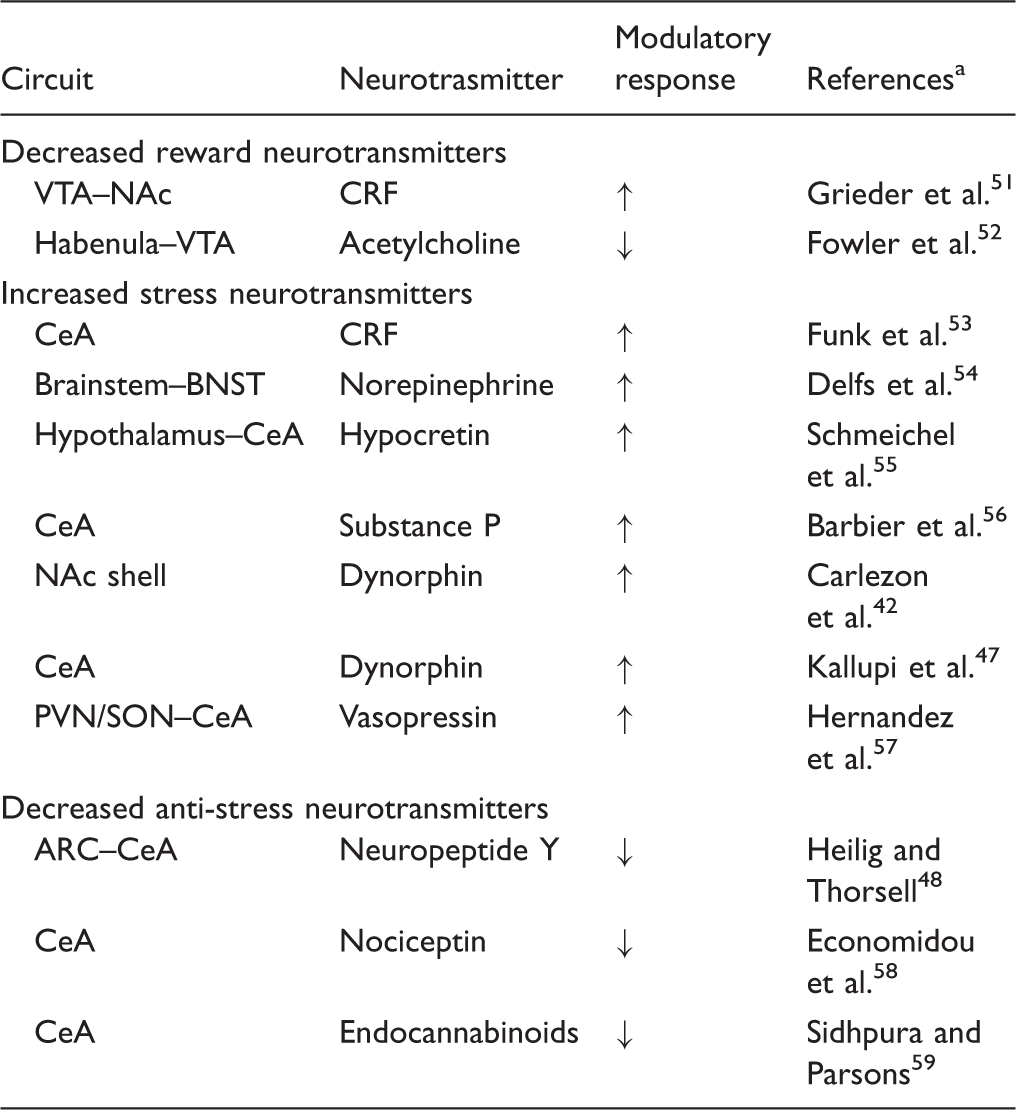
ARC: arcuate nucleus; CeA: central nucleus of the amygdala; NAc: nucleus accumbens; PVN: paraventricular nucleus; SON: supraoptic nucleus; VTA: ventral tegmental area.
References are key papers that show either direct evidence of the circuit outlined or hypothesize the existence of such modulation. The second column (Circuit) indicates either a neurotransmitter circuit or, where only one neuroanatomical site is listed, a local circuit. Arrows represent the direction of modulation.
Thus, multiple circuits that involve multiple modulatory neurotransmitter systems converge on the extended amygdala to mediate negative emotional states associated with the
The
Neuroclinical Assessment: From Reward to Stress and Back
The nosological research framework termed Research Domain Criteria (RDoC) originated as part of the National Institute of Mental Health (NIMH) 2008 strategic plan, with the goal of creating a research framework for studying psychiatric disorders. The NIMH framework was conceptually grounded in neuroscience research and spanned five domains: Negative Valence Systems, Positive Valence Systems, Cognitive Systems, Systems for Social Processes, and Arousal and Regulatory Systems. RDoC domains are organized by units of analysis, ranging from genes to paradigms (for an overview of the RDoC matrix, see http://www.nimh.nih.gov/research-priorities/rdoc/research-domain-criteria-matrix.shtml; accessed 20 January 2017), and this approach has generated much conceptual and methodological discussion.61–65 We have proposed a more parochial, within-disorder, research approach, the Addictions Neuroclinical Assessment (ANA) framework, 66 which captures information in three of the five original RDoC domains.
The ANA domains were derived from the conceptual framework outlined above, in which drug addiction derives from a three-stage cycle with conceptual roots in impulsivity and compulsivity, the recruitment of positive and negative reinforcement, and interactions between the neurobiological substrates of reward and stress. Three functional domains—executive function, incentive salience, and negative emotionality—were proposed as described above. The
Negative Emotionality
Although often not emphasized, the reports of individuals who suffer from drug addiction are replete with descriptions of overall self-reported dysphoria and various manifestations of negative emotional states.68,69 Such descriptions include depression, anxiety, anhedonia, dysphoria, malaise, alexithymia, hyperkatifeia, emotional pain, physical pain, irritability, and sleep disturbances. A self-medication hypothesis has long infiltrated theories of addiction but has been dismissed, usually based on the grounds that both humans and animals will self-administer drugs without undergoing physical withdrawal. However, a rather common misunderstanding of tolerance and withdrawal in addiction is that they represent purely “physical” phenomena,70–73 rather than motivational constructs. Indeed, both tolerance (defined as increased reward seeking and taking more drug to produce the same effect) 74 and withdrawal (defined as a motivational withdrawal syndrome characterized by dysphoria, anxiety, and irritability when the reward that is sought is unavailable)67,75 are present in all drug and behavioral addictions.76,77 For example, a complete assessment of reward constructs must include measurements of hypohedonia. 78 Hypohedonia is widely documented as a clinical feature of addiction79–83 and is highly associated with increased craving for drugs of abuse 84 and relapse. 85
Opponent Process as a Guiding Principle
The interaction between reward and stress is dynamic both phenotypically and neurobiologically. Low levels of acute stress have long been considered rewarding. Glucocorticoids have rewarding properties and can even be self-administered by animals.
86
However, chronic stress generally leads to malaise, irritability, and dysphoria, which drive mechanisms of negative reinforcement. Neurobiologically, accumulating evidence links excessive activation of the reward system as a causal mechanism for activation of the brain stress systems (see below). In the domain of motivation in addiction, the interaction between reward and stress was inextricably linked with hedonic, affective, or emotional states in the context of temporal dynamics by the opponent-process theory of motivation.
87
Here, hedonic, affective, or emotional states, once initiated, are automatically modulated by mechanisms that reduce the intensity of hedonic feelings, presumably mediated by the central nervous system. Solomon and Corbit argued that there are affective or hedonic habituation (or tolerance) systems and affective or hedonic withdrawal (abstinence) systems. They defined two processes: the (a) Schematic of the progression of drug and alcohol dependence over time, illustrating the shift in underlying motivational mechanisms. From initial, positive-reinforcing, pleasurable effects of drugs and alcohol, the addiction process progresses over time to being maintained by negative-reinforcing relief from a negative emotional state. Neuroadaptations that encompass the recruitment of extrahypothalamic CRF systems are key to this shift (taken with permission from Heilig and Koob
88
). (b) The 
Such an opponent process has been demonstrated in animals. 89 In an early study, chronic binge-like cocaine self-administration resulted in an opposite effect on brain stimulation reward thresholds (i.e., a measure of hedonic activity in the brain), namely an elevation of brain-stimulation reward thresholds. 89 Subsequent studies showed that the elevation of brain reward thresholds that was associated with withdrawal from chronic administration of drugs of abuse is a common element of all drugs of abuse, including cocaine, 89 amphetamine, 90 opioids, 91 cannabinoids, 92 nicotine, 93 and alcohol. 94 A series of studies revealed elevations of brain reward thresholds during withdrawal in animal models. Key neuropharmacological evidence has been generated that shows that both reversing reward deficit neurotransmission and reversing stress surfeit neurotransmission can block the elevation of reward thresholds produced by drug withdrawal. 95
A key component that drives negative emotional states in general and hypohedonia in particular and is associated with the
Neuroclinical Assessment: Anhedonia, Hypohedonia, and Dysphoria
Animal models for negative emotional states.

EEG: electroencephalogram.
Human laboratory tests for negative emotional states.

Neuroclinical Assessment: Anxiety, Stress Reactivity, and Irritability
The neurocircuitry of anxiety, stress, and irritability are hypothesized to involve “between-system” changes that include activation of neurocircuits involved in stress (CRF, norepinephrine, vasopressin, and hypocretin in the CeA and BNST; Table 1). Animal models with construct validity for anxiety-like behavior, stress reactivity, and irritability-like behavior that have helped elucidate the neurocircuitry associated with anxiety, stress, and irritability include the elevated plus maze, defensive withdrawal test, defensive burying test, marble burying test, and social interaction test (Table 2). Human laboratory assessments of anxiety, stress reactivity, and irritability range from standard self-report measures, such as the Beck Anxiety Inventory and Hamilton Depression Rating Scale, to those that focus selectively on trauma constructs, such as the Childhood Trauma Questionnaire (Table 3). More operational measures of anxiety, stress reactivity, and irritability include the Cyberball Test, Trier Social Stress Test, and Buss-Durkee Hostility Inventory (Table 3).
Neuroclinical Assessment: Pain and Hyperkatifeia
The neurocircuitry of pain and analgesia are hypothesized to involve “between-system” changes that include the activation of pain circuits and also neurocircuits that are involved in stress (CRF, norepinephrine, vasopressin, and substance P in the CeA and BNST; Table 1). Animal models with construct validity for pain and hyperalgesia that have helped elucidate the neurocircuitry associated with pain and the interaction between pain and stress include the hot plate test, tail flick test, and von Frey test (Table 2).
Both hyperalgesia and hyperkatifeia have been observed in humans during withdrawal from opioids and alcohol.126,127 Hyperalgesia can be defined as an increased sensitivity to pain. Hyperkatifeia (derived from the Greek word
Neuroclinical Assessment: Malaise, Sleep Disturbances, and Arousal
The neurocircuitry of malaise, sleep disturbances, and arousal are hypothesized to involve both “within-system” changes in the mesocorticolimbic dopamine system for arousal and malaise, but also “between-system” changes in neurocircuits that are involved in malaise (CRF, norepinephrine, vasopressin, and hypocretin in the CeA and BNST) and sleep/arousal (hypocretin in the hypothalamus; Table 1). Indeed, hypocretin (orexin) has been shown to play a critical role not only in addiction, as described above, but also in regulating arousal and coordinating the alertness that is necessary to pursue goal-directed behaviors. 129 Animal models with construct validity for malaise, sleep disturbances, and arousal that have helped elucidate the neurocircuitry associated with these constructs in humans include activity measures, electroencephalography, and observations of peripheral physiological arousal. Patients who are addicted to various agents have present self-reported malaise, 130 sleep disturbances,131,132 and disruptions in arousal. 133 Malaise may be defined as an undefined sense of illness or unease without a specific cause. Within addictive disorders, sleep disturbances often take the form of insomnia and changes in sleep architecture.131,132 Dysregulated arousal may appear as hyperarousal in response to stressful stimuli or drug cues compared with individuals who are not addicted. 133 Relatedly, hyperarousal is a key diagnostic criterion for PTSD, which is highly comorbid with addiction to various substances. 134 Human laboratory assessments of these constructs include polysomnography for the evaluation of sleep and electroencephalography and peripheral signals (e.g., galvanic skin response, respiration, and heart rate) for the evaluation of arousal, in addition to self-report measures, such as the Malaise Inventory, Pittsburgh Sleep Quality Index, and Behavioral Activation System Scale (Table 3).
Implications for Nosology of Addiction
Over time, the nosology of addictions has remained relatively static. The most recent iteration of the
Even without a specific and definitive neurobiological marker, an emphasis on stress and negative affective states in addictive disorders, as discussed herein, could lead to the inclusion of these in future iterations of addiction diagnoses. For example, specifying whether an individual experiences significant dysphoria or relief craving during withdrawal, while still being symptom-based, would be one step closer toward a neurobiologically informed addiction diagnosis. It would also critically allow clinicians to identify treatments that would more closely align with a specific subtype of addiction. Overall, a strong emphasis on negative affective states that are associated with addiction could further the integration of neurobiology into the addiction nosology and improve treatment outcome. Given the significant public health costs associated with addictions, these improvements would be well worth the time and effort to further explore the role of stress and negative affect in addictions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
