Abstract
Introduction
Enterococci have emerged as the common cause of serious nosocomial urinary tract infections.
Aim and Objective
To isolate and identify uropathogenic Enterococci and to study the antibiogram, vancomycin-resistant Enterococci (VRE), and high-level aminoglycoside resistance (HLAR).
Materials and Methods
The urine specimens (3448) were cultured on CLED and MacConkey’s agar. Enterococcal isolates were identified and subjected to antibiotic sensitivity testing (minimum inhibitory concentration) by the Vitek 2 automated method. Statistical analysis was done using the Graph Pad InStat Software.
Result
Among the 3448 urine samples, Enterococci were responsible for 148 cases of urinary tract infections (UTIs) (4.29%). Enterococcus faecalis (69.6%) was the most common followed by E. faecium (19.6%). Enterococci were isolated from urine samples with various predisposing (45.95%) factors. Vancomycin resistance (28.38%) and HLAR (52.03%) were the common phenomena. The Enterococci have acquired resistance to erythromycin, tetracycline, and vancomycin. E. faecium was more resistant to most of the antibiotics, including vancomycin and HLAR, than E. faecalis. The emergence of linezolid resistance (27.7%) was noted in our study.
Conclusion
VRE, HLAR, and multidrug resistance were significantly observed more among patients with prolonged hospitalization, catheterization, and hypertension. Enterococcal UTI is an emerging infection in India. Wiser use of antimicrobial drugs is possibly guided by novel techniques for rapid microbiological diagnosis.
Introduction
Urinary tract infections (UTIs) stand among the most prevalent bacterial infections worldwide, with over 150 million cases reported annually. These infections are caused by a range of Gram-negative and Gram-positive bacteria, including E. coli, Klebsiella, Proteus, Staphylococci, and Enterococci. 1 Although Enterococci naturally reside in the gastrointestinal tract, they have recently become prominent as leading agents of serious hospital-acquired UTIs.2, 3 Over the recent decades, a worrying increase in resistance to commonly prescribed antibiotics among Enterococci has been observed, particularly with the emergence of vancomycin-resistant Enterococci (VRE), posing significant challenges in healthcare settings.
Among the Enterococcus species, Enterococcus faecalis is the most frequently identified, yet E. faecium presents greater resistance to a range of antibiotics, including vancomycin, 4 with a notable prevalence of high-level aminoglycoside resistance (HLAR). 5 This resistance complicates the treatment landscape, as the options for effectively managing enterococcal infections, especially serious UTIs caused by VRE, are severely limited. Furthermore, there is a noticeable lack of comprehensive data on the nature and treatment of enterococcal UTIs, highlighting a critical need for research in this area.
Given these challenges, our study aims to isolate and identify the species of Enterococci involved in UTIs and to evaluate their antibiotic-resistance profiles, with a specific focus on VRE and HLAR resistance patterns. Through this research, we seek to contribute valuable insights into the epidemiology and resistance mechanisms of Enterococci in UTIs, addressing a significant gap in current medical knowledge and potentially guiding the development of more effective treatment protocols for these increasingly resistant infections.
Materials and Methods
This study was designed as a prospective, cross-sectional investigation conducted from July 2020 to June 2023. It encompassed patients diagnosed with UTIs who provided informed consent to participate. The inclusion criterion was the isolation of Enterococci from the urine samples of these patients. The Institutional Ethical Committee granted the approval for the study, ensuring adherence to ethical standards.
We collected urine samples from 3,448 individuals suspected of having UTIs. These samples underwent a semi-quantitative culture using MacConkey agar and cystine lactose electrolyte-deficient (CLED) agar, sourced from Hi-Media Laboratory in Mumbai, India. 6
Identification of Enterococci species from the cultured urine samples was based on cultural characteristics and a series of conventional biochemical tests. For isolates that were difficult to identify or remained unidentified through conventional methods, the Vitek 2 automated system was employed for definitive species identification.
Subsequent to species identification, all enterococcal isolates were assessed for their antibiotic susceptibility. This involved determining the minimum inhibitory concentration (MIC) for a panel of commonly prescribed antibiotics. HLAR was specifically evaluated using the Vitek 2 system with concentrations of gentamicin (500 µg/ml) and streptomycin (1000 µg/ml). For quality control in antibiotic susceptibility testing, reference strains including Staphylococcus aureus (ATCC 25923), vancomycin-susceptible E. faecalis (ATCC 29212), and vancomycin-resistant E. faecalis (ATCC 51299) were utilized.
Statistical analyses were conducted using GraphPad InStat software. The χ2 test was applied to assess the significance of our findings, with a P value of less than .05 deemed indicative of statistical significance. The analysis also included calculations of odds ratios to further interpret the data.
Results
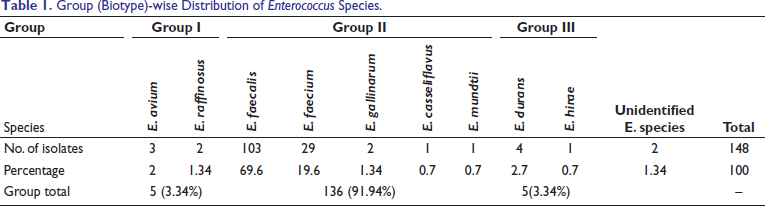
Among the 3,448 urine samples examined, 1,037 showed evidence of growth, with 148 (4.29%) identified as having Enterococci. Of these 148 isolates, 104 were successfully speciated using conventional biochemical methods, while 44 remained unclassified. However, utilizing the Vitek 2 automated method, 42 of these previously unclassified strains were identified at the species level, leaving two isolates unidentified.
E. faecalis was the predominant species, accounting for 69.6% of the isolates, followed by E. faecium at 19.6%. Among the total isolates, 54.05% were obtained from UTI cases lacking any predisposing factors, while 45.95% were associated with predisposing factors such as catheterization, diabetes, and pregnancy.
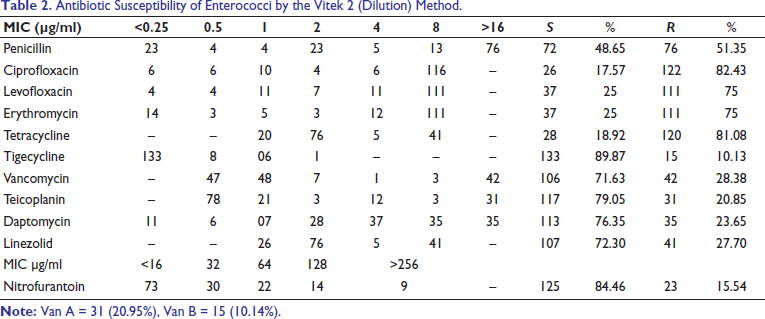
Regarding antibiotic resistance, 28.38% of the enterococcal isolates exhibited resistance to vancomycin, while 52.03% were resistant to high-level aminoglycosides (HLAR). Detailed species-specific resistance patterns are presented in Tables 1 and 2.
Group (Biotype)-wise Distribution of Enterococcus Species.
Antibiotic Susceptibility of Enterococci by the Vitek 2 (Dilution) Method.
Discussion
The rise of enterococcal UTIs presents a significant challenge in healthcare settings, particularly among hospitalized patients with disrupted mucosal or epithelial barriers or altered normal flora due to antibiotic therapy. 7 UTIs due to Enterococci are increasing among hospitalized patients, with risk factors such as antibiotic therapy (particularly cephalosporin), urinary catheterization, 3 and recurrent UTIs. 8 A 20-fold increase in the incidence of Enterococci causing nosocomial UTI has been documented by Sexena et al. 9 Our study sheds light on the epidemiology, species distribution, antibiotic-resistance patterns, and predisposing factors associated with enterococcal UTIs.
In the present study, we observed a notable prevalence of enterococcal UTIs among patients with predisposing factors, such as urinary catheterization (16.9%), diabetes (12.16%), and pregnancy (11.49%). This aligns with previous findings highlighting the association between these risk factors and enterococcal UTIs.3, 8 Catheter-related UTIs, in particular, provide a conducive environment for enterococcal colonization and subsequent infection due to bacterial adhesion and mucosal irritation. The urinary stasis, vesico-urethral reflux, and enlarged belly increase the risk of UTI among pregnant women. 10 The reported frequency of enterococcal UTI is variable in different studies. Miskeen and Deodhar 11 have isolated Enterococci among 7.4% of the UTI patients, and Jaylaxmi and Jaram 12 have reported 2.12% enterococcal incidence in asymptomatic bacteriuria in pregnant women. The bladder, prostate, and kidneys are commonly infected by Enterococci. 13
In the present study, conventional tests and Vitek 2 together identified 146 (98.66%) of enterococcal isolates, but 2 (1.34%) enterococcal isolates remained unidentified by both methods. Rouff et al 14 and Bhat et al 15 were also unable to identify 0.3% and 0.7% isolates, respectively, up to the species level. Therefore, Vitek 2 gives a larger spectrum of tests in order to recognize species within the genus. Species identification is crucial for understanding antibiotic-resistance patterns, as different Enterococcus species exhibit varying susceptibilities to antibiotics. 3 Our study identified E. faecalis (69.6%) as the most common species, followed by E. faecium (19.6%), consistent with previous reports. we came across 10.8% non-E. faecalis and non-E. faecium species among the enterococcal isolates. E. durans (2.7%) was predominant among these species followed by E. avium (2%), E. gallinarum, and E. raffinosus (1.34%), while we identified the rare species E. casseliflavus, E. mundtii, and E. hirae (2.1% together) (Table 1). The non-E. faecalis and non-E. faecium species differ from 0% to 24% in various studies.3, 11, 14, 15 In Miskeen and Deodhar’s study, 11 E. durans was found to be the predominant species, at 2%. However, other studies have reported different prevalence rates for unusual species of Enterococci in India. Desai et al 3 reported a prevalence of 14.85%, while Vittal et al 16 reported 19%, and Ghosh et al 17 reported 24%. For these unusual species of Enterococci from India, which is more than the present study, these rates are higher than what was found in the study by Miskeen and Deodhar. 11 It is worth noting that Vitthal et al and Desai et al specifically focused on nosocomial infections. Additionally, a study from Chandigarh reported different predominant species in urinary isolates, with E. faecalis being the most prevalent at 55%, followed by E. casseliflavus at 24% and E. faecium at 12%. 18
The Enterococcus species are intrinsically resistant to cephalosporin; low level of clindamycin; and acquired resistance to erythromycin, tetracycline, fluoroquinolone, and high levels of penicillin, clindamycin, aminoglycosides, and vancomycin. 19 Resistance to commonly used antibiotics poses a significant therapeutic challenge in the management of enterococcal UTIs. Resistance to penicillin was found to be 51.35%. This resistance pattern correlated well with the studies conducted by Bhat et al 15 and Parvati et al 8 (43%). However, lower resistance has been reported by Miskeen and Deodhar (23.13%) 11 and Udo et al (12%). 20
Earlier, ciprofloxacin was the drug of choice for the empirical treatment of UTI and was effective in 55.78% of strains tested. 11 In the present study, a majority of the isolates were resistant to ciprofloxacin (82.43%) and levofloxacin (75%) (Table 2). Parvati et al 8 reported 33% of the strains resistant to ciprofloxacin. We also observed that Enterococci were more susceptible to penicillin (48.65%) than ciprofloxacin (14.87%). This observation corelates with the observation made by Miskeen and Deodhar 11 and Udo et al. 20 Therefore, ciprofloxacin may not be the best alternative drug for the treatment of serious enterococcal infections. Erythromycin was the non-effective (75%) drug against Enterococci in the present study. This resistance pattern is higher than that in the study by Udo et al, 20 which has reported 63% enterococcal isolates resistant to erythromycin.
The present study indicates that the majority of enterococcal strains exhibited higher susceptibility to tigecycline (90%) and nitrofurantoin (85%). Some strains also showed susceptibility to vancomycin (71.63%), linezolid (72.3%), daptomycin (76.35%), and teicoplanin (79.05%), although these drugs are considered reserve options. These resistance patterns are similar to those found in a study conducted by Mendiratta et al. 19 Nitrofurantoin, tigecycline, vancomycin, and linezolid were more effective than all other antibiotics tested, and this difference is statistically significant. E. faecalis was less resistant to these antibiotics than E. faecium. Desai et al. 3 and Baldir et al 21 also reported less than 2% enterococcal strains resistant to linezolid. Rahangdale et al 22 reported 100% sensitivity to linezolid. Even though linezolid is a more versatile and highly active drug against multi-drug-resistant E. faecium as well as E. faecalis, but it should not be used indiscriminately. 8 Enterococci have been demonstrated to spread linezolid resistance in hospital environments. This favors the emergence of linezolid resistance. The emergence of linezolid resistance occurred infrequently among VRE isolates recovered during compassionate-use program (1.8% isolates) in Chicago. 23 In the present study, 27.7% isolates are resistance to linezolid.
Nitrofurantoin and fosfomycin are effective against some isolates, including VRE, especially from cystitis and other UTIs. Miskeen and Deodhar 11 also showed that nitrofurantoin resulted in complete inhibition of growth in 99.22% of the isolates tested. Suneeta et al. 2 shows 98%, 92.6%, and 81.4% enterococcal isolates, respectively, were susceptible to nitrofurantoin. Nitrofurantoin and tigecycline are outstanding drugs against both Gram-positive and Gram-negative bacteria. 23 Therefore, nitrofurantoin can be recommended for UTI, while tigecycline and linezolid can be used for other infections because the tissue penetration of tigecycline is excellent.
Of particular concern is the emergence of VRE, with a substantial proportion of isolates demonstrating resistance to vancomycin. This has been increasingly reported from all parts of the world. In the present study, among the 148 isolates, 42 (28.38%) were resistant to vancomycin. Among these 42 VRE strains, 31 (20.95%) isolates showed high-level resistance to vancomycin as well as teicoplanin and belonged to the Van A phenotype. Even though 15 (10.14%) isolates of Enterococci belonged to the Van B phenotype, of these, 11 isolates were high-level resistant while 4 isolates were low-level resistant to vancomycin but susceptible to teicoplanin (Table 2). E. faecalis (77.67%) was sensitive to vancomycin, while 51.72% isolates of E. faecium were sensitive to vancomycin. Resistance to vancomycin has also been reported among E. gallinarum and E. faecalis. 24 Miskeen and Deodhar 11 (100%) and Udo et al (99.6%) 20 reported that many isolates were susceptible to both vancomycin and teicoplanin, and all the organisms were inhibited at concentrations ranging from 0.5 to 4 µg/ml.
Udo et al 20 have reported that VRE isolates expressed low MIC values, but they were resistant to ampicillin, erythromycin, ciprofloxacin, and high-level streptomycin and gentamicin. So, the treatment of VRE infections, particularly E. faecium, is tremendously challenging. Penicillin or ampicillin with or without a synergizing aminoglycoside would be a rational option in a nonallergic patient infected with vancomycin-resistant E. faecalis. 23 The prevalence of VRE has increased from 0.3% to 47% in the United States 25 and European countries. 23 The use of avoparcin (glycopeptide) as a growth promoter in animal feed seems to be the major contributor to vancomycin resistance in European countries. In contrast, the United States has not permitted avoparcin in animal feed, but the use of vancomycin in treating human infections has increased more than 100-fold in the last 50 years. 23 In India, neither avoparcin is used as a feed supplement for animals nor vancomycin is used much in clinical practice. Therefore, few earlier studies from India report a meager percentage of vancomycin resistance,3, 8 11 15 24 while in the present study, we observed several-fold (2%–28.29%) 26 increase in vancomycin resistance among uropathogenic Enterococci during the last 5 years. The same observation (1.4%–7.9%) was reported by Tripathi et al. 27
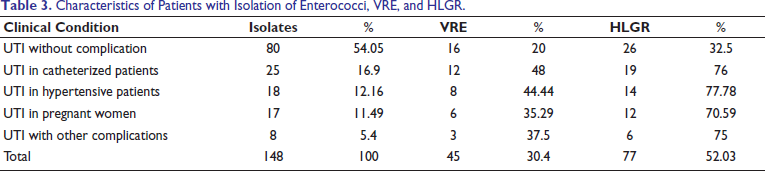
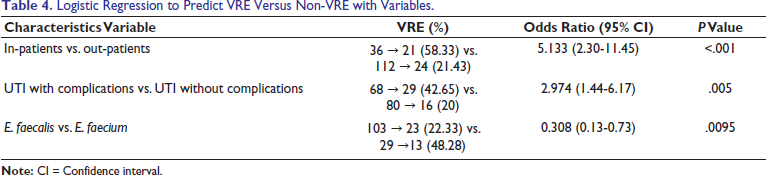
In the present study, among the UTIs without any predisposing factors, 20% enterococcal isolates were resistant to vancomycin, while among the UTIs with various predisposing factors, 48% VRE were reported from catheterized patients, 44.44% from diabetic patients, and 35.29% from pregnant women (Tables 3 and 4). The National Nosocomial Infection Surveillance System reported 28% nosocomial VRE from hospitalized patients in various US hospitals. Tripathi et al 27 also reported 57.6% nosocomial VRE in their study. In the present study, VRE are found to be significantly more among hospitalized (58.33%) than among non-hospitalized (21.48%) patients. VRE were found to be the most common among UTIs with predisposing factors (42.65%) than UTIs without predisposing factors (20%) and among E. faecium (48.28%) than among E. faecalis (22.33%) with P value < .05 (Table 4). Suneeta et al 2 reported VRE among E. faecium (41%) was significantly more than that among E. faecalis (11.11%). They also noted that the use of invasive devices, old age, and patients hospitalized for a longer time, with urinary catheter in the ICU, were found to be significantly associated with VRE infections; this is in concordance with the present study. Recent appearances of VRE have gravely affected the treatment of patients with this infection. This gives very limited choices to physicians.
Characteristics of Patients with Isolation of Enterococci, VRE, and HLGR.
Logistic Regression to Predict VRE Versus Non-VRE with Variables.
Aminoglycoside-modifying enzymes can inactivate all available aminoglycoside and are responsible for HLAR. 28 Only streptomycin, gentamicin, and, occasionally, kanamycin should be considered for synergy screening. HLAR Enterococci often have plasmid, which also carries determinants encoding resistance to other antibiotics.
In the present study, a total of 52.03% isolates showed high-level resistance to gentamicin by the Vitek 2 method (Table 3). This correlated well with a study conducted by Jadhav (50.8%) and Bose (58%). Mendiratta et al (46% HLGR), 19 Rouff et al, 14 and Udo et al 20 reported 14% HLGR. Combination therapy with cell wall active agents (penicillin, ampicillin, or vancomycin) and aminoglycoside (gentamicin or streptomycin) is often used empirically for serious infections, which improve the outcomes and are thus recommended for the treatment of serious enterococcal infections like meningitis or endocarditis, especially caused by VRE. Among the VRE, 30% strains can produce multiple enzyme types and are highly resistant to all known aminoglycosides. 28
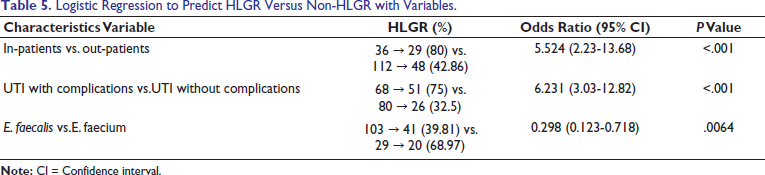
In the present study, among the UTIs without predisposing factors, 32.5% enterococcal isolates were resistant to high-level gentamicin resistance, while among the UTIs with predisposing factors, 76% HLGR was reported from catheterized patients, 77.78% from diabetic patients, 70.59% from pregnant women, and 75% from patients with other complications (Table 3). High-level gentamicin resistance among E. faecium (68.97%) was significantly more than among E. faecalis (39.81%). Mendiratta et al 19 and Rouff et al 14 also reported the same observation. In the present study, 80% HLGR were reported among admitted patients, while only 42.86% were found among out-patients, and this difference was statistically significant (P < .001) (Table 5). The increased prevalence of HLGR in metropolitan hospitals may be due to a longer therapy, increased stay of chronic cases, and wider usage of broad-spectrum antibiotics. Moreover, E. faecium has become difficult to treat by using glycopeptides and aminoglycosides in combination with penicillin.
Logistic Regression to Predict HLGR Versus Non-HLGR with Variables.
Conclusion
In conclusion, our study underscores the pressing challenge posed by multidrug-resistant enterococcal infections, particularly VRE and HLAR strains. These pathogens are increasingly encountered in hospitalized patients, especially those with prolonged stays and catheterization, highlighting the importance of infection control measures in healthcare settings. Our findings highlight the efficacy of nitrofurantoin, tigecycline, vancomycin, and linezolid as reliable treatment options for enterococcal UTIs, given their consistent sensitivity across all species tested. However, the emergence of resistance to these antibiotics underscores the need for prudent antimicrobial stewardship practices and the development of novel therapeutic approaches. Moving forward, the development of new antimicrobial agents tailored to combat multidrug-resistant Enterococci, including VRE, offers the promise for more effective mono-therapy. Additionally, the implementation of rapid microbiological diagnostic techniques and the rational use of antimicrobial drugs may help mitigate the selective pressures driving the emergence and spread of VRE.
Footnotes
Authors’ Contribution
Concept, design, and definition of intellectual content: Sanjay M. Wavare, Rashmi M. Karigoudar, Annapurna G Sajjan
Literature search, experimental studies, and data acquisition: Sanjay M. Wavare, Rashmi M. Karigoudar, Smitha Bagali
Data analysis and statistical analysis: Sanjay M. Wavare, Praveen R. Shahapur
Manuscript preparation, manuscript editing, and manuscript reviewing: Sanjay M. Wavare, Rashmi M. Karigoudar, Annapurna G. Sajjan, Praveen R. Shahapur
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
