Abstract
Background:
Renal cell carcinoma (RCC) accounts for approximately 90% of kidney cancers, with a significant percentage of patients presenting with metastatic disease. Recent evidence suggests a notable role of the human microbiome in the onset, progression, and therapeutic outcomes of RCC.
Objective:
This systematic review aims to synthesise current knowledge on the association between the microbiome and RCC, focusing on pathogenesis, progression, and response to therapy.
Methods:
Complying with PRISMA guidelines, databases including PubMed, Embase, and Web of Science were searched for relevant studies up to December 7, 2023. The inclusion criterion was English-language articles that discussed RCC in relation to the microbiome of any body region. Screening was performed in a two-phase manner by three authors.
Results:
From 570 articles, 65 met the inclusion criteria. The gut microbiome (GM) emerged as a potential RCC pathogenesis driver, with certain bacteria associated with increased or decreased risk. Studies have also demonstrated that antibiotics and other medications can influence RCC therapeutic outcomes, reducing the effectiveness of immune-modulating therapies.
Conclusions:
While multiple bacterial species and antibiotics have been implicated in influencing RCC, further research is necessary to elucidate these relationships and investigate the efficacy of microbiome modulation on therapy effectiveness. Findings underscore the significant impact of the microbiome on RCC, suggesting the potential for microbiota-targeted therapeutics.
Introduction
Globally, there are over 400,000 newly diagnosed cases of kidney cancer per year, approximately 90% of which is attributed to renal cell carcinoma (RCC). 1 An estimated 17% of patients are diagnosed with metastatic disease at presentation. 2 Historically, individuals with RCC – particularly locally advanced and metastatic disease – face a poor prognosis. However, treatment strategies have significantly evolved in recent years, incorporating systematic therapies that often use immune modulation to drastically improve outcomes. 3 For decades, the immune system has been coopted in the fight against a variety of cancers including RCC. 4 More recently, evidence has begun to emerge that highlights the significance of the immune system and its interplay with the microbiome in the onset and progression of RCC. 5
The microbiome refers to collective genome of the microbes (composed of bacteria, fungi, protozoa, virus and other microorganisms), some symbiotic and some pathogenic, that subsist within the human body, including respiratory, gastrointestinal, and genitourinary tracts. 6 Some of these pathogens have been directly implicated in various disease processes, such as H. pylori and gastric cancer or human papilloma virus (HPV) and penile cancer.7,8 Study of these interactions has been formalised in an international effort termed the human microbiome project (HMP). 9 The genitourinary tract, thought to be sterile, was not included in early studies of the human microbiome. Yet, advances in methods such as 16S rRNA sequencing have increasingly supported the existence of a urinary microbiome.10,11 This urinary microbiome has been potentially linked to numerous urologic pathology such as overactive bladder, interstitial cystitis, chronic pelvic pain syndrome, in addition to bladder, prostate, penile and kidney cancer.12–16
In this systematic review, we explore the connection between the human microbiome and RCC – namely the role it may play in the pathogenesis, progression, and therapeutic outcomes.
Search terms and strategy
Search terms and strategy
This systematic review was conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The available scientific literature within PubMed, Embase, and Web of Science was reviewed for all studies up until December 7, 2023. The search terms used within PubMed included “(Microbiology [MeSH] OR Microbiota [MeSH] OR Bacteria [MeSH] OR RNA, Ribosomal, 16S [MeSH]) AND (Carcinoma, Renal Cell [MeSH] OR Kidney Neoplasms [MeSH])”. Search terms used within Embase were “microbiome AND renal cell carcinoma”. Search terms used within Web of Science included “(renal cell carcinoma OR kidney neoplasms) AND (microbiome OR microbiota OR bacteria OR 16S)”.
Inclusion and exclusion criteria
Only articles in English with full text availability were included in the systematic review. Articles were considered relevant to the review topic if they included discussion of RCC and the microbiome of any region of the body as it relates to RCC. Articles were initially screened based on abstract composition, and then second pass screening was completed using full article text. Articles were excluded from the review if they were published as abstract only, case report, or an editorial or background article. Screening for eligibility was completed by three authors (SL, FM, and LB). Evidence synthesis was performed in a descriptive and narrative fashion.
Evidence synthesis
570 articles were retrieved from PubMed, Embase, and Web of Science. 76 of these articles were duplicates and were removed from the article pool. 494 articles underwent title and abstract screening, and a further 318 were deemed unacceptable for further review based on abstract content (wrong outcome, population, publication type, study design, or background article). The remaining 176 articles were sought for full text review, of which 7 were inaccessible. 169 full text articles were retrieved and underwent further screening; 89 were excluded based on inappropriate full text content and 15 additional articles were deemed duplicates. 65 articles remained for inclusion in the systematic review (Figure 1).
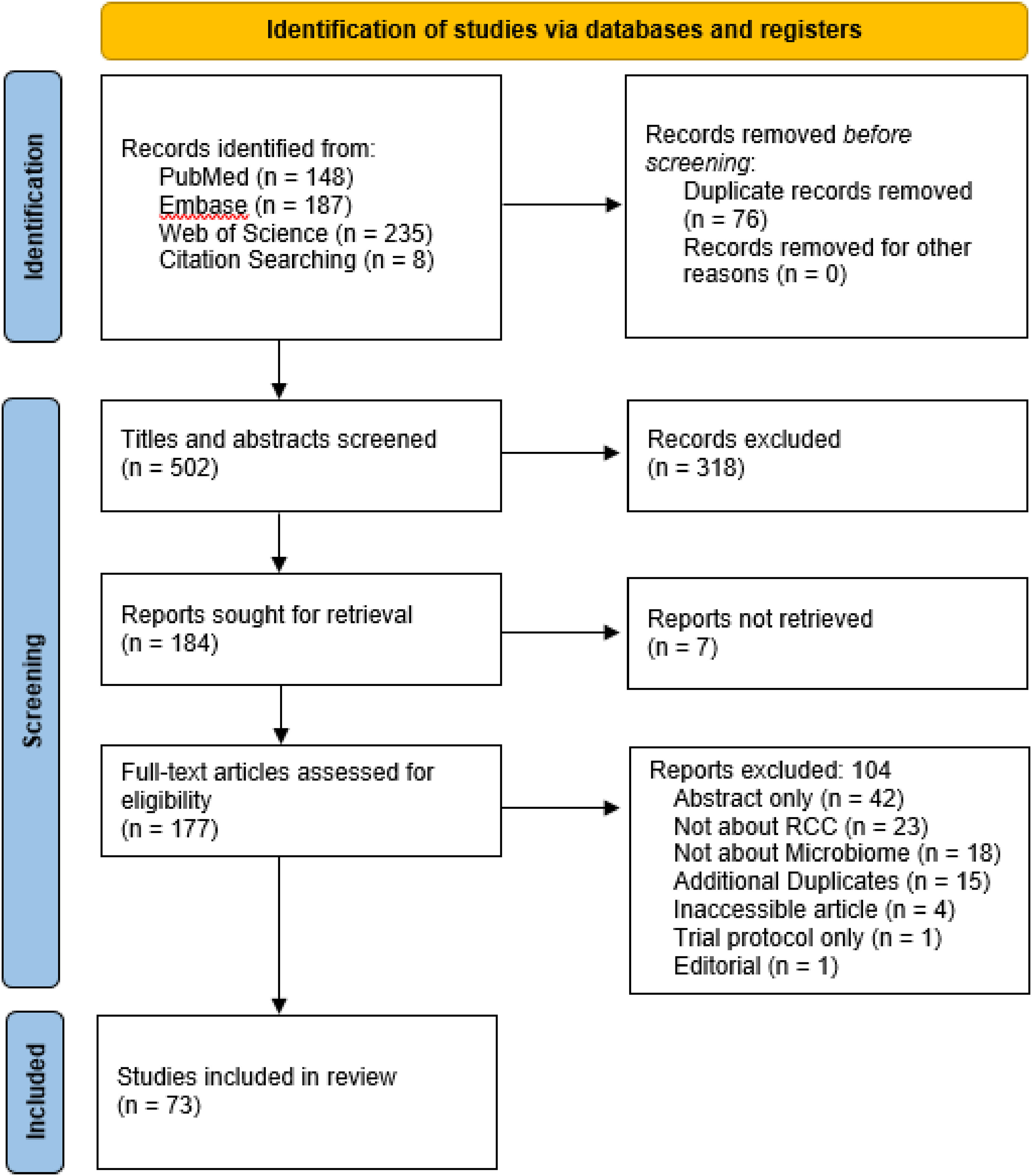
PRISMA flow diagram.
Bacteria
As the largest microbial reservoir in the body, the gut microbiome (GM) has been repeatedly evaluated as a potential driver of RCC pathogenesis. Various genome-wide association studies have uncovered associations between gut bacteria and RCC. While results vary between studies, Yin et al. completed a Mendelian randomisation analysis and found the genera Streptococcus [Ruminococcus torques group] (OR 4.11, 95% CI 1.34–12.67, p = 0.014) and Eubacterium brachy (OR 2.14, 95% CI 1.05–1.90, p = 0.007) as well as the classes Alphaproteobacteria (OR 1.58, 95% CI 1.08–2.30, p = 0.018) and Bacilli (OR 1.41, 95% CI 1.05–1.90, p = 0.024) were associated with an increased risk of RCC. They also found some bacteria that were associated with a decreased risk of RCC, including the Family XI (OR 0.76, 95% CI 0.63–0.92, p = 0.006), the genera Coprococcus 2 (OR 0.63, 95% CI 0.44–0.89, p = 0.009), Intestinomonas (OR 0.69, 95% CI 0.53–0.89, p = 0.005), Lachnoclostridium (OR 0.53, 95% CI 0.37–0.77, p = 0.0008), and Lactococcus (OR 0.55, 95% CI 0.34–0.88, p = 0.014). 17 Mingdong et al. completed a similar study and similarly found Streptococcus [Ruminococcus torques group] (OR 3.80, 95% CI 1.15–12.50, p = 0.028) and Erysipelatoclostridium (OR 2.31, 95% CI 1.01–5.30, p = 0.048) were associated with RCC. 18 These findings provide some supporting evidence for an observational study that associated Streptococcus [Ruminococcus torques group] with RCC. 19
Additional studies have characterised the GM among subsets of patients treated with immune checkpoint inhibitor (ICI) or vascular endothelial growth factor-tyrosine kinase inhibitor (VEGF-TKI) therapy. Salgia et al. found the most significant species among patients who derived clinical benefit from ICI therapy were Bifidobacterium adolescentis (p = 0.002), Barnesiella intestinihominis (p = 0.006), and Bacteroides eggerthii (p = 0.009), although Prevotella_copri, Faecalibacterium_sp_CAG_74, Fermicutes_bacterium_CAG_194, Prevotella_sp_CAG_520, Ruminococcus_torques, and Odoribacter_splanchnicus were also associated. They also found that patients with clinical benefit from ICI therapy had greater alpha diversity of bacterial species (p = 0.001). Patients with no clinical benefit from ICI therapy were found with significant levels of Bacteroides_ovatus, Eggerthalia_lenta, Fusicatenibacter_saccharivorans, and Flavonifractor_plautii. Some patients in this study were also found to have generally increasing levels of Akkermansia species across time points in the study, while a few additional patients were found to have increasing levels of Prevotella copri. 20
In a similar fashion, Routy et al. found that A. muciniphila was most significantly enriched in a cohort of non-small cell lung cancer (NSCLC) and RCC patients treated with anti PD-1 therapy who exhibited favourable clinical outcomes (p = 0.004). A. muciniphila was also enriched in patients with a PFS >3 months, when compared to those with PFS <3 months. Other bacterial species including unclassified and classified Fermicutes, Akkermansia, and Alistipes were also found in patients who responded to therapy when compared to those who did not. 21 Similar to Salgia et al., Routy et al. found increased stool richness over the course of ICI therapy in RCC patients as well.20,21
Derosa et al. looked to correlate the GM with ICI therapeutic efficacy in terms of PFS and best overall response (BOR). They found that higher alpha diversity of bacterial species was associated with clinical response to therapy, which they defined as the absence of progressive disease at 12 months after initiation of ICI therapy. Further analysis found that A. muciniphila (p < 0.02), Bacteroidetes salyersia (p = 0.04), and Eubacterium siraeum (p = 0.01) were associated with response to therapy. E. bacterium (p < 0.01) C. hathewayi (p < 0.01), and Clostridium clostridioforme (p < 0.0009) were associated with non-responders. 22
A similar study described the bacterial profile among patients treated with VEGF-TKI therapy. The most common bacterial phyla were Bacteroidetes (67.91%) and Firmicutes (26.20%). Prominent bacterial genera were Bacteroides (47.37%) and Tannerella (0.84%) from the Bacteroidetes phylum and Faecalibacterium (3.75%), Ruminococcus (1.88%), and Blautia (1.09%) from the Firmicutes phylum. 23
Although there is some consensus regarding contributions to RCC from above mentioned bacterial taxa, the mechanistic nature of these associations need additional investigation. Chen et al. have suggested the TGF-β signalling pathway as a possible underlying mechanism based on in vitro studies that identified Streptococcus pneumoniae as a driver of TGF-β signalling in tympanosclerosis, asthma, and pulmonary infection. 19 In cancer, TGF-β typically regulates homeostasis and prevents tumor progression through apoptosis, cytostasis (inhibition of cell division), and differentiation. However, when premalignant cells lose responsiveness to this cytokine, tumor colonisation, invasion, and progression become possible. 24 Chen's study investigated this pathway by culturing RCC cell lines with Streptococcus lutentiensis or brain-heart infusion medium. They found that expression levels of TGF-β-related proteins were significantly elevated by RCC cells lines cultured with S. lutentiensis, thereby concluding that TGF-β promotes RCC development. 19
Streptococcus was observed to be more prevalent in clear cell Renal Cell Carcinoma (ccRCC) tissues compared to normal kidney tissues. The authors theorised that an infection with Streptococcus could lead to the activation of COX-2, triggering inflammation and carcinogenesis through the subsequent activation of interleukin-8 (IL-8), a strong factor promoting angiogenesis. Moreover, additional virulence factors such as the exotoxins SpeG (which encodes pyrogenic exotoxin G) and smeZ (which encodes mitogenic exotoxin Z) were found to elevate levels of C-C motif chemokine ligand 2. This increase aids in the recruitment of tumor-associated macrophages (TAMs), contributing to the progression of ccRCC.
Another study by Yang et al. associated alterations in pro-carcinogenic bacteria with development of RCC. In their study, they found that 11 species of bacteria were enriched, including Desulfovibrionaceae, and nine species were deficient, including four species of Lactobacillus. 25 Lactobacillus has been negatively correlated with inflammatory markers, including WBC counts, monocyte counts, and C-reactive protein (CRP). As a potential link between altered gut bacteria levels and inflammation, further analysis sought to identify metabolites in blood. In the RCC group, 358 different metabolites were identified, with notable changes in amino acid and lipid metabolism, especially involving taurine. Taurine itself has been linked to altered inflammatory responses by preventing the formation of reactive oxygen species (ROS), inducing apoptosis, and inhibiting the protein kinase B (Akt) signalling pathway. These actions have the potential to impede the development and advancement of ccRCC. The authors propose that when the Desulfovibrionaceae family metabolises taurine, it is converted into H2S, which may further contribute to carcinogenesis.
Consensus on the important bacterial players in RCC pathogenesis is limited, with various studies identifying a range of bacterial alterations in patients with RCC. Ruminococcus torques group frequently emerges as a significant entity in this context. However, beyond individual genera, there is evidence that the diversity of bacterial species differs significantly between healthy and cancerous tissue. Kovaleva et al. looked at the tumor microbiome composition for clear cell, papillary, and chromophobe RCC and compared the bacterial species to normal kidney tissue. Although they did not find differences in dominant bacteria subtypes, their analysis revealed that clear cell, papillary, and chromophobe subtypes had less taxonomic bacterial diversity than normal tissue when evaluated with Chao1 (clear cell p = 0.0019, papillary p = 0.0083, chromophobe p = 0.0009) and Shannon (clear cell p = 0.0049, papillary p = 0.0298, chromophobe p = 0.0049) indices, respectively (Table 1).
Summary of included studies about the bacterial microbiome in RCC.

Viruses and fungi
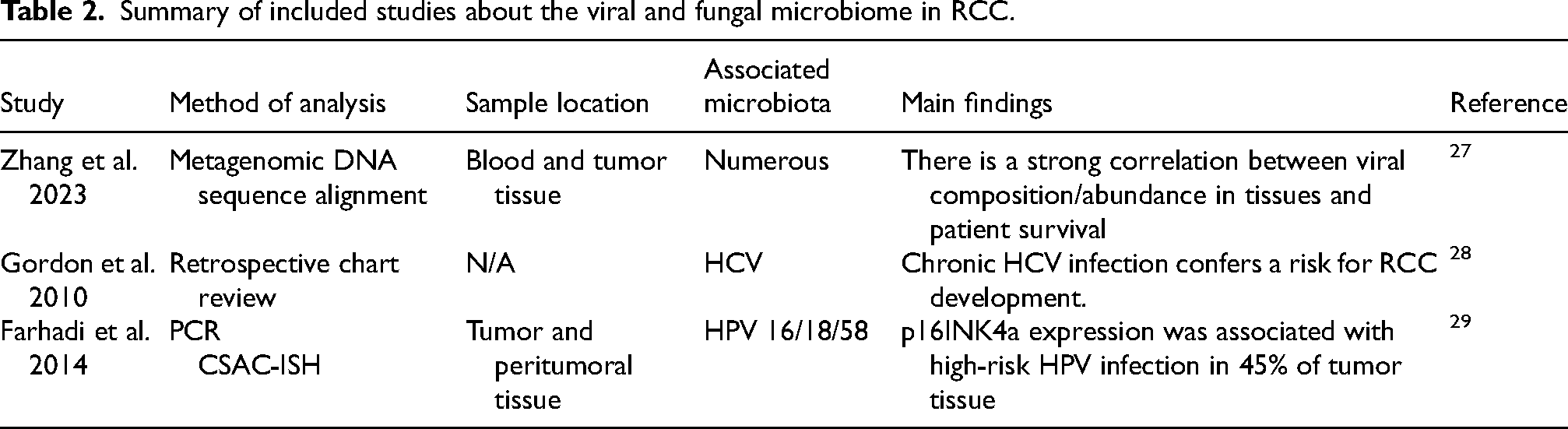
Strongest evidence of a viral component within RCC microbiome originates from foundational study by Zhang et al. Their research aimed to identify viral presence in cancer tissue and blood and to examine the potential of these viral markers as prognostication of cancer survival. They obtained genetic data from 9340 tissue samples spanning 31 different cancer types and matched sections of these genomes that were inconsistent with the human genome to known viral, bacterial, and fungal genomes. 27
Among the wealth of data (Table 2) put forth by their study, RCC was one of four cancers whose prognosis was correlated with viral components. RCC prognosis was associated with 25 different viruses, and regression analysis showed the survival of patients with RCC was associated with different relative abundance of Simplexvirus (p < 0.0001), Betapartitivirus (p = 0.00031), Alphapartitivirus (p < 0.0001), and Gammaretrovirus (p = 0.0011). Whereas less-than-median level presence of Simplexvirus and Gammaretrovirus was significantly associated with a lower probability of survival, less-than-median levels of Betapartitivirus and Alphapartitivirus was significantly associated with a higher probability of survival. Thus, these viruses were able to independently predict post-operative survival of patients. 27
Summary of included studies about the viral and fungal microbiome in RCC.
They further found that viral composition of the tumor was predictive of clinical phenotypes. By dividing tumor samples into two groups based on the presence of prognostic viral signatures from various viruses including Simplexvirus, Betapartitivirus, Flavivirus, Tymovirus, Molluscipoxvirus, Bracovirus, Carlavirus, Polyomavirus, Gammaretrovirus, Taupapillomavirus, Prymnesiovirus, Alphavirus, and Potyvirus. Comparative analysis of clinical parameters between the two subgroups revealed notable disparities in disease stage (p = 0.003) and tumor size (p = 0.0004). Consequently, Zhang et al. suggest that the viral makeup of RCC tissues could serve as an effective predictor of disease prognosis, though further confirmatory research is required. 27
Zhang et al. also explored the relationship between viral and bacterial communities in tumor tissue. 27 Based on prior studies, they hypothesised that viral and bacterial communities have synergistic effects and influence prognosis (Table 2).28,30 Their Procrustes analysis found significant overlap between these two communities with a correlation coefficient of 0.133 (p = 0.001) as well as a correlation between viral and bacterial community richness (r = 0.178, p < 0.001). They ultimately concluded that viral and bacterial community have a possible non-competitive relationship within RCC tumor tissue. 27
Subsequent research has linked hepatitis C virus (HCV) with the development of RCC. Gordon et al. leveraged a comprehensive cancer registry to compare the rate of HCV infection in individuals with RCC against those without cancer. They found that 0.6% (17 of 3057) RCC patients had chronic HCV infection compared to 0.3% (177 of 64,006) non-cancer controls. A multivariate analysis found the hazard ratio (HR) for RCC in HCV patients was 1.77 (95% CI 1.32–3.67, p = 0.0025). 31 The mechanism for this association is unclear, although studies of HCV in hepatocellular carcinoma patients leads to activation of peroxisome proliferator-activated receptor α, a regulator of transcription genes for fatty-acid metabolism and an essential component of hepatocellular carcinoma development by HCV in mice. 32
The presence of high-risk human papillomavirus (HPV) has also been found in RCC tissue. One study found that 30.3% (37 of 122) of RCC tissue samples were positive for human papilloma virus (HPV) strain 16 or 18 and 29.7% (11 of 37) of these cases exhibited double infection with more than one HPV strain. Additionally, a significant association was found between the presence of HPV DNA and the expression of p16INK4a (p < 0.001). Among the HPV-positive RCC samples, 79.2% showed p16INK4a expression, compared to only 20.8% of HPV-negative tumors. P16INK4a is a protein involved in the regulation of the cell cycle, and its activity has been linked to both tumor suppression and high-grade tumors alike. While up to 70% of tumors exhibit p16INK4a inactivation—often associated with tumor progression—overactivity of p16INK4a is also a common finding in HPV-related cancers. 29 It's suggested that the function of p16INK4a varies based on its location within the cell; while typically located in the nucleus of the cell, cytoplasmic sequestration of p16INK4a has been observed in malignant cells, a phenomenon that could contribute to the malignancy of HPV-associated tumors. 33
Antibiotic effects on the microbiome
The microbial mechanisms that modulate anti-tumor processes are numerous and complex. These natural microbial processes are beneficial as evidenced by repeated studies that document the detrimental effects of antibiotics on certain anti-cancer therapies, particularly immune-modulating therapies such as immune-checkpoint blockade (ICB). Without the influence of antibiotics, the microbiome has the capability to activate and/or recruit various innate and adaptive immune cells, including dendritic cells (DC), natural killer cells (NK), macrophages, CD8+, and CD4 + cells. For example, DC are mobilised by microbiome antigens and metabolites with immunomodulators and can reverse induced immune tolerance. Furthermore, the microbiome can initiate Interferon-I signalling in tumor-associated monocytes, which in turn stimulates the production of anti-tumor macrophages, promotes immune communication, and aids in the shift from innate to adaptive immunity. Microbiome metabolites, notably inosine, also play a role in anti-tumor activity. It not only boosts the activity of the immune cells but also supports their activation, and meets their energy needs in the tumor's nutrient poor environment. 34 The effectiveness of these microbiome mediated processes is compromised by antibiotics, which reduces microbial count and diversity and undermines the beneficial effects the microbiome has in cancer therapy. 35
In treatments that leverage the diversity and richness of the bacterial community, the use of antibiotics poses a risk to the effectiveness of these therapies, as demonstrated by numerous studies examining their impact on clinical outcomes. It is common to see reduction in objective response rate (ORR), progression free survival (PFS), and overall survival (OS) in the setting of RCC. Lalani et al. document these changes in their retrospective study, which looked at both an institutional cohort who received PD-1/PD-L1 therapy and a trial database cohort which received interferon-α, mTOR, or VEGF-TKI therapy in phase II/III studies. The window within which antibiotics were used was 8 weeks prior and 4 weeks post-initiation of anticancer therapy. In the institutional cohort, antibiotic use was associated with a decline in ORR from 34.8% to 12.9% and a hazard ratio (HR) for PFS of 1.96 (95%CI 1.20–3.20, p = 0.007). (Table 3) Additionally, a decrease in OS (HR 1.62) in patients treated with interferon or VEGF-TKI with prior cytokine therapy, but not in those without prior cytokine therapy, was observed. 36
Summary of included studies about antibiotic effects on the microbiome in RCC.

These results have largely been replicated by many studies. Derosa et al. reported that patients who used antibiotics within 30 days before starting ICI therapy experienced a significantly shorter PFS of 1.9 months, as opposed to 7.4 months for patients who did not receive antibiotics. This group also experienced a reduced OS of 17.3 months compared to 30.6 months for non-antibiotic users, in addition to higher rate of disease recurrence (75% in antibiotic users vs. 22% in non-users) (Table 3). 37 Similarly, Guven et al. retrospectively looked at patients who were prescribed antibiotics within 90 days of ICI initiation. In their findings, the hazard ratios for PFS and OS in the antibiotic group were 2.2 and 2.3, respectively, indicating a significant association of antibiotic use with adverse outcomes in both PFS and OS upon multivariate analysis. 38
Spakowitz et al. further expanded on this and looked at specific classes of antibiotics and their effect on OS. Among individual antibiotics, vancomycin stood out as the only one linked to a HR greater than 1 for RCC patients. However, overall analysis that combined data from all antibiotics again showed a HR greater than 1. Furthermore, while the HR was comparatively lower than when antibiotics were started prior to ICI therapy, antibiotic initiation subsequent to ICI therapy was still statistically significant for up to 120 days later. 39 Various other studies have yielded similar results in terms of worse outcomes for patients who receive systemic antibiotics around the time of ICI initiation.22,40–45
Hahn et al. took unique perspective in studying the microbiome's influence on cancer treatment effectiveness. It has been reported that higher levels of Bacteroides in the stool are associated with diarrhoea for patients on VEGF-TKI therapy. 23 As diarrhoea is often a limiting side effect of these medications, and they hypothesised that antibiotics targeting Bacteroides, reducing their gut levels, might alleviate diarrhoea, thereby enhancing patient outcomes by allowing for longer and more tolerable treatment durations. Interestingly, in their retrospective study, they found that patients who received antibiotics with Bacteroides coverage had longer PFS (18 months) than those treated with antibiotics that did not cover Bacteroides (9 months) or patients who did not receive antibiotics at all (8 months). 46 This suggests that while antibiotics may reduce the effectiveness of some cancer treatments, they might also offer benefits in certain contexts by improving treatment tolerability and duration.
Therapeutic interventions and other medications
The GM has been shown to influence a wide range of human processes, from homeostasis to antigen presentation and immune function to medication and treatment efficacy.34,47–49 As such, the GM often plays a role in cancer outcomes, including RCC, through interactions with immune processes. Therefore, substances that modify the GM could potentially alter the treatment outcomes for RCC. While antibiotics have been discussed for their ability to affect cancer therapy responses, other substances, including probiotics and corticosteroids, have also been explored in this context (Figure 2).

Summary of microbiome studies and RCC.
Since prior studies have shown the detriments of a diminished microbiome on cancer survival, efforts have been made to identify therapeutics that may promote improved outcomes through microbiome modulation.50–53 Probiotics have been investigated in this regard, and Dizman et al. conducted a randomised trial assessing the impact of probiotic supplementation on the gut and its potential influence on the clinical outcomes of targeted therapy in metastatic RCC. Given the observed deficiency of Bifidobacterium species in RCC patients, supplemental intake of this bacterial species via yogurt consumption may affect the outcomes of patients treated with VEGF-TKI therapy through normal flora reconstitution. They randomised patients to an intervention group that consumed probiotic yogurt containing Bifidobacterium or to a control group that did not consume yogurt, and the gut microbiota was assessed using whole-genome shotgun sequencing. Although they found it possible to modulate the microbiome via yogurt supplementation, with 67% of the intervention but only 0.2% of the control group harbouring the bacterial species of interest, they failed to show any clinical benefit from the intervention. Both the intervention and control group had similar rates of response to anti-cancer therapy (70% vs. 80%, p = 0.606) (Table 4). The study by Dizman et al. was constrained by a small sample size and slow participant enrollment. Additionally, the research focused exclusively on patients undergoing VEGF-TKI therapy. This choice of medication is notable because its effectiveness is not typically linked to immune system modulation, unlike treatments involving ICIs. 54
Summary of studies included about therapeutic interventions that target the RCC microbiome.

However, Dizman performed another experiment on patients treated with ICI therapy. In their study, 30 treatment-naïve patients with metastatic RCC were given combination nivolumab-ipilimumab with or without bacterial supplementation. Unlike the prior study, they were unable to elucidate a change in gut Bifidobacterium species composition. However, they did observe difference in survival metrics. Namely, patients treated with bacterial supplementation in addition to nivolumab-ipilimumab had longer PFS (12.7 months vs. 2.5 months, HR 0.15, 95%CI 0.05–0.47, p = 0.001) (Table 4). Furthermore, although not statistically significant, a trend towards increased response rate was also observed (58% vs. 20%, p = 0.06). 55
Buti et al. further investigated other therapeutics and their indirect effects on advanced cancer patients treated with ICIs. Although they did not directly investigate the effects of these medications on the microbiome, nor did they separately analyze RCC patients, all medications included in the analysis have been shown to modulate the human microbiome in some capacity. 44 For example, corticosteroids affect mucous production and shift bacterial composition, and antibiotics have a direct effect on bacterial diversity.35,56 The authors assigned a simple scoring method to patients taking corticosteroids (2 points), antibiotics (1 point), or PPIs (1 point) based on the magnitude of the effect these medications had on overall patient survival. A patient was assigned to either a good prognosis (0 points, i.e., not taking any of the medications), intermediate prognosis (1 or 2 points), or poor prognosis (3 or 4 points) group based on their total score. This scoring system was then correlated with outcomes such as ORR, PFS, and OS in both a training and validation cohort, revealing that a higher prognostic score was associated with poorer outcomes. Although the authors did not specifically analyze RCC as a separate entity, the results shed light on the negative effects that common medications can have on the microbiota. These results, were echoed in a study by Spakowitz et al. who concluded that the timing and strength of both antibiotics and corticosteroids were consistent with microbiome modulation and ultimately associated with lower OS survival when taken concomitantly with ICI therapy.39
Anticancer therapeutics have also been noted to effect the gut microbiome. Derosa et al. characterised the GM of patients treated with VEGF-TKI therapy and noted distinct differences between them and patients who were not treated with VEGF-TKI therapy. They observed increased abundance of A. senegalensis and A. muciniphila as a result of VEGF-TKI therapy. Although their analysis was limited by their inability to collect stool samples prior to VEG-TKI therapy, they further explored the association GM shifts with VEGF-TKI therapy in mouse models. They collected longitudinal stool samples from mice, both before and after administration of tumoricidal antiangiogenic doses of TKIs, and determined that all medications caused changes in GM diversity. 22 Salgia et al. found similar results in terms of ICI therapy's ability to alter the GM, with increased Akkermansia muciniphila levels in patients who experienced clinical benefit from ICI therapy. 20
It is worth considering based on the above mentioned studies, how prebiotics and other alimentary factors could also influence the microbiome and provide therapeutic benefits for RCC patients receiving checkpoint inhibitors. Prebiotics, such as dietary fibres, can promote the growth of beneficial gut bacteria, which may enhance the efficacy of immune-based therapies by fostering a more favourable gut microbiome. These alimentary factors could potentially amplify the immune system's response, suggesting an avenue for future studies to explore the combined impact of probiotics and prebiotics in optimising therapeutic outcomes for RCC patients.
Conclusion
The identification of the microbiome's role in RCC pathogenesis has gained attention recently. Various local and distant mechanisms are at play, from bacterial and viral influences to concurrent drug usage. Many studies have investigated which bacterial species are most guilty for influencing RCC, and while there is little consensus in the literature, Ruminococcus torques group has been identified on multiple occasions. Furthermore, viral influence from HCV and HPV, as well as the interplay of various other viral taxonomies, has been implicated in RCC development. Some of these bacterial and viral components have been investigated for use as tumor biomarkers, disease prognostication, or even as indicators of drug response.
Still, further research into the RCC microbiome has investigated the effects of concurrent medication use on therapeutic efficacy. Antibiotics, which have a profound on the natural microbiome, have been repeatedly shown to diminish the effectiveness of immune-modulating therapy. Other medications, such as corticosteroids, have also shown detrimental effects on the microbiome, although these relationships have not been elucidated as thoroughly. Additional studies have evaluated the efficacy of microbiome modulation on therapy effectiveness. While these studies are still highly investigational, there has been some positive results in terms of microbiome modulation and future studies may provide further modalities for increasing the effectiveness of medical therapy.
Footnotes
Abbreviations
Acknowledgements
The authors would like to thank librarian at Fox Chase Cancer Center Beth Lewis for her assistance with meta-analysis and systematic review.
Author contributions
Conceptualisation: Laura Bukavina, Philip Abbosh;
Data Curation: Francesca Montanaro, Zev Leopold;
Funding Acquisition: None;
Methodology: Laura Bukavina, Steven Leonard;
Supervision: Laura Bukavina;
Validation: Emma Helstrom, Francesca Montanaro;
Visualisation: Steven Leonard;
Writing – Original Draft: Steven Leonard; Laura Bukavina
Writing – Review & Editing: All authors
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
Dr Barata reported financial relationships with AstraZeneca, AVEO, Bayer, Bristol Myers Squibb, Caris Life Science, Eisai, EMD Serono, Exelixis, Merck, and Pfizer; institutional relationships with Dendreon, AstraZeneca, Bayer, Caris Life Sciences, Merck, and Pfizer/Astellas; and has received institutional research funding from AVEO, Blue Earth Diagnostics, Exelixis, Merck, and Pfizer.
L.Bukavina: consulting for Charite, UroGen Pharma. Associate Editor of European Urology, Editorial Board Member Urology Times. She has received funding from Bladder Cancer Advocacy Network.
Dr Nizam has received consulting fees from Aptitude Health, IntegrityCE, Targeted Oncology, MECC Global Meetings, ASCO.
Drs Leonard, Montanaro, Leopold, Helstrom, Abbosh, Fulmes, Correa and Weight have no conflict of interest to report.
Data availability
There is no data relevant to the analysis.
