Abstract
Background
The term “inflammaging” has been introduced to describe the chronic, low-grade inflammatory process associated with the increase in age and the senescence of the immune system, that confers susceptibility to suffering age-related pathologies. In this context, previous studies have already demonstrated that the regular consumption of mineral water can be a valuable tool for detoxifying the body from reactive oxygen species (ROS) and for mitigating the systemic inflammation.
Objective
Herein, we tested the beneficial effects of a mineral water with human-like electrolytic content (San Carlo® water) in a pre-clinical model of vascular inflammaging and investigated the mechanisms underlying this effect. San Carlo® water was administered ad libitum to both young (10-week old) and aged rats (40-week old) for 3 months.
Methods
Vascular functionality, oxidative stress (fluorescent dye dihydroethidium assay, levels of MDA, and levels of 8-isoprostanes in the urine), and systemic inflammation (levels of IL-1β and TNF) were assessed.
Results
San Carlo® water ingestion mitigated the inflammation by reducing TNF plasma levels and improved endothelial-dependent vasorelaxation, acting against ROS production.
Conclusion
The present study suggests that the use of San Carlo® water can effectively counteract the low-grade inflammation typically associated with age and the related vascular dysfunction, likely through both antioxidant and anti-inflammatory properties.
Introduction
In the early 2000s, the term “inflammaging” was introduced to describe the chronic, sterile, low-grade inflammatory process that develops with age, confers susceptibility to suffering age-related pathologies, and facilitates the spread of the aging effect at the systemic level. 1 During aging, the majority of the cells in our body undergo the process of senescence marked by the acquisition of a dysfunctional state and a senescence-associated secretory phenotype. 2 In particular, immune cells lose their capacity to clean unfunctional cells, leading to a phenomenon called “immunosenescence” which alters the ability of the immune system in responding to noxious stimuli. Therefore, this phenomenon causes the accumulation of senescent cells, thus triggering inflammation and increasing the development of organ damage and aging-related diseases, such as diabetes mellitus, sarcopenia, and cancer.2,3 In this context, pro-inflammatory cytokines such as interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF), IFN-γ, and IL-1RA have been shown to play a pivotal role since they can encourage the expansion of clones of the hematopoietic/immune system depending on the spread of inflammatory signals. 1 In parallel, the increased oxidative stress associated with age, resulting from a reduction in antioxidant defenses and an increase in the production of oxygen free radicals (ROS), especially those with mitochondrial origin, represents an important stimulus for the activation of the inflammatory response. 4 At the vascular level, aging is associated with an unbalanced status consisting in high levels of ROS and low levels of NO that gradually affects the structure and function of blood vessels, thus causing loss of elasticity of the arterial wall, leading to endothelial dysfunction, potentially responsible for age-related chronic disorders, such as the cardiovascular diseases (CVDs), chronic kidney diseases, cancer, depression, dementia, osteoporosis, sarcopenia, physical, and cognitive disability and frailty. 5 Of interest, various studies suggest that regular consumption of mineral water with particular electrolyte contents can be a valuable tool to improve the functionality of the body, detoxifying it from the presence of ROS. 6 Moreover, evidence suggests that bicarbonate mineral water reduces cholesterol and glucose levels, thus playing a preventive effect on CVDs.7,8 Notably, the intake of acidic mineral water with a low concentration of potassium, calcium, and magnesium improves metabolic and redox homeostasis in rats with metabolic syndrome. 9 Additionally, several studies report that natural mineral water consumption can efficiently lower oxidative stress markers, 10 positively influence tissue homeostasis, and counteract the onset of inflammaging and preventing related diseases.
Based on these premises, the present study has been designed to evaluate the putative beneficial effects of mineral water (San Carlo® water) with human-like electrolytic content in a pre-clinical model of vascular inflammaging.
Materials and methods
Reagents
Composition of San Carlo® water and control water.

Abbreviations: Ca, calcium; Cl, chlorine; F, fluorine; K, potassium; Mg, magnesium; Na, sodium; n.d., not detected; NH4, ammonium; NO3, nitrate; Si, silicium; SO4, sulfate.
Concentration of sodium (Na) and potassium (K) in control water, San Carlo® water, and medium concentration in human serum and their respective ratios.

Abbreviations: K, potassium; Na, sodium; n.a. not applicable.
ammol/L concentration of Na and K in control water and in San Carlo® water was calculated by dividing Na and K mg/L concentration (Table 1) with their molar mass (22,99 g/mol and 39,10 g/mol, respectively).
Animals
Experiments were performed on young (10-week old) and aged (40-week old) male Wistar rats. The animals were fed standard laboratory chow (Envigo, Udine, Italy) and control water ad libitum, and they were not employed for at least 1 week after their delivery to the laboratory. They were housed in solid-bottomed cages, equipped with wire-mesh bottom inserts to prevent coprophagy, and located in temperature-controlled rooms (at 22°C–24°C and 50%–60% humidity) under a 12-h light cycle (06:00–18:00 h). Animal care and handling were in accordance with the provisions of the European Community Council Directive 210/63/UE, recognized and adopted by the Italian Government. The experiments have been approved by the Ethical Committee for Animal Experimentation of the University of Pisa and by the Italian Ministry of Health (authorization n D4133.N.RU6).
After 1 week of acclimatization, control water was replaced by San Carlo® water in one group of young rats and in one group of aged rats. San Carlo® water was kindly provided by San Carlo S.p.A.
The detailed division of groups is as follow (10 animals/group): (a) Young rats administered with San Carlo® water for 3 months. (b) Aged rats administered with San Carlo® water for 3 months. (c) Young rats administered with control water for 3 months. (d) Aged rats administered with control water for 3 months.
Preparation of small mesenteric arteries for functional experiments
After dissection, the first branch of mesenteric artery was placed in cold (4°C) physiological salt solution (PSS) containing (in mmol/L) NaCl 120, NaHCO3 25, KCl 4.7, KH2PO4 1.18, MgSO4 1.18, CaCl2 2.5, EDTA 0.026, and glucose 5.5, as previously published. 13 A second-order branch of the mesenteric arterial tree (≈2 mm in length) was dissected and mounted on 2-glass microcannule in a pressurized myograph, as previously described. 13 Vessels were equilibrated for 60 min under constant intraluminal pressure (45 mmHg) in warmed (37°C) and bubbled (95% air and 5% CO2) PSS, at pH 7.4. Vessels were considered viable and used if they constricted >70% of their resting lumen diameter in response to an extra-luminal application of high-potassium solution (125 mmol/L of KCl) containing 100 μmol/L of noradrenaline (NA).
Vascular remodeling and endothelial function
Endothelial-dependent and independent vasorelaxation in all experimental groups were defined by the vasodilatory response to cumulative concentrations of Ach (0.001–100 μM) and sodium nitroprusside (SNP) (0.01–100 μM), respectively. All experiments were performed in vessels pre-contracted with NA (10 μM). To ensure an equal NA-induced contractility in vessels from young and aged rats, we performed preliminary experiments to assess the amount of vasoconstriction induced by increasing NA concentrations (from 1 nM to 100 μM). The results of these experiments showed that NA 10 μmol/L was able to elicit similar contractions among different groups (data not shown), thus such a concentration was selected for functional experiments.
To evaluate the proportion of endothelial-dependent vasodilation due to NO availability, concentration-response curves ACh were constructed after 30 min pre-incubation with the NOS inhibitor Nw-nitro-L-arginine methylester ([L-NAME] 100 μM, Sigma Co., St. Louis, MO, United States). To assess the influence of ROS on NO availability, concentration-response curves to ACh were repeated in the presence of the antioxidant ascorbic acid (100 μM, Sigma, 30-min pre-incubation). Finally, to estimate whether ROS generation could impair NO-mediated endothelium-dependent relaxation, ACh was infused during simultaneous incubation with L-NAME and ascorbic acid. Vessels were then deactivated by perfusion with Ca2+-free PSS containing 10 mmol/L EGTA for 30 min. Media thickness and lumen diameter were measured in three different points from each small artery to obtain the media-lumen ratio (M/L), with intraluminal pressure at 45 mmHg. 13 Media cross-sectional area (MCSA) was obtained by subtraction of the internal from the external cross-sectional areas using external plus lumen diameters, as previously described. 14
Detection of vascular superoxide anion (O−2) generation
The in situ production of superoxide anion (O −2 ) from 30 mm frozen mesenteric vessel sections was evaluated at the confocal microscope by means of the fluorescent dye dihydroethidium ([DHE] Sigma), as previously described. 13 Three slides per segment were analyzed simultaneously after incubation with Krebs solution at 37°C for 30 min. Krebs-HEPES buffer containing 2 μM DHE was then applied to each section and evaluated under fluorescence microscopy. In the presence of O −2 , DHE undergoes oxidation and intercalates in cell DNA, thus staining the nucleus with red fluorescence (excitation at 488 nm and emission 610 nm). The percentage of arterial wall area stained with the red signal was normalized to the total area examined and quantified using an imaging analysis software (McBiophotonics Image J; National Institutes of Health, Bethesda, MD, United States).
Plasma IL-1β and TNF levels
Plasma IL-1β and TNF levels were measured using commercially available enzyme-linked immunosorbent assay (ELISA) kit (Biosciences), according to the manufacturer’s guide. Aliquots (100 μL) of plasma were used for the procedure. IL-1β and TNF levels were expressed as pg/mL of plasma.
Plasma malondialdehyde (MDA) levels
Malondialdehyde (MDA) levels in plasma were assessed to investigate lipid peroxidation, an index of oxidative stress. Plasma MDA concentrations were determined with a colorimetric assay (Calbiochem, San Diego, CA, USA), and the results were expressed as μmol of MDA/ml of plasma.
Urinary levels of 8-isoprostanes and pH
Urine samples were collected at four different times point during the study: T0 (the first day of the experiment), T1 (1 month after the first day of the study), T2 (2 months after the first day of the study), and T3 (3 months after the first day of the study, immediately before the sacrifice). After each time points, urine pH was measured using a glass electrode on a Beckman f390 pH meter. Then, aliquots (100 µl) of urine were used to assess 8-isoprostanes levels, a marker of oxidative stress, using a competitive ELISA assay (Abcam ab175819 Cambridge, United Kingdom).
Statistical analysis
Data are expressed as mean ± SEM. The significance of differences was evaluated for raw data, before percentage normalization, by one-way ANOVA followed by the appropriate post hoc test. p < 0.05 was considered significant. The colonic preparations included in each test group were obtained from different animals, and therefore the number of trials was always the same as the number of animals allocated to the group. Calculations and analyses were performed using GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA). A power analysis was used to estimate sample sizes for each experiment.
Results
Endothelium-dependent vascular relaxation and remodeling
In mesenteric vessels from young rats, relaxation to ACh was significantly attenuated by L-NAME (100 μM) (Figure 1(a)). Treatment of young rats with control or San Carlo® water did not modify the patterns of responses to ACh in mesenteric vessels, in the absence or in the presence of L-NAME (Figure 1(a)). Mesenteric vessels from aged rats administered with control water showed a reduced vasorelaxation to ACh as compared to young animals (Figure 1(b)). The impaired vasorelaxation observed in aged rats was rescued by the incubation of the vessel with the ascorbic acid (ROS scavenger), suggesting that the endothelial dysfunction observed in aged animals was primarily due to an increased ROS production within the vascular wall (Figure 1(c)). By contrast, San Carlo® water administration enhanced the relaxation to ACh in mesenteric vessels from aged rats (Figure 1(b)). Incubation of the vessels with sodium nitroprusside (SNP) in combination with or without San Carlo® water restored a complete vasodilatory response, suggesting that endothelial-independent pathways did contribute to the improved Ach-vasorelaxation induced by the San Carlo® water (Figure 1(d)). No differences were observed in media-lumen ratio of young or aged rats administered with control or San Carlo® water (Figure 1(e)). Endothelial-dependent relaxations of mesenteric resistance arteries to acetylcholine in (a) young and (b) aged rats treated with control water or San Carlo® water for 3 months. (c) Rescuing of the Ach-dependent vasodilation in aged rats following vessel incubation with ascorbic acid (ROS scavenger). (d) Vasodilatory response induced by pre-incubation of the vessels with sodium nitroprusside (SNP). (e) Media-lumen ratio of mesenteric arteries from young and aged rats treated with control water or San Carlo® water for 3 months. Each column represents the mean ± SEM of eight animals. Statistics: one-way analysis of variance followed by Newman–Keuls test. 
Vascular O−2 generation
DHE assay revealed a significant increase in O
−2
production in vessels from aged rats administered with control water as compared with young animals (Figure 2). Young rats administered with San Carlo® water did not modify O
−2
generation (Figure 2) while the ingestion of San Carlo® water in aged rats counteracted significantly such an increment (Figure 2). Quantitative analysis (bar graph) and representative dihydroethidium (DHE) staining and of the red signal in mesenteric arteries (magnification ×40) from young and aged rats treated with control water or San Carlo® water for 3 months. Each column represents the mean ± SEM of eight animals. Statistics: one-way analysis of variance followed by Newman–Keuls test.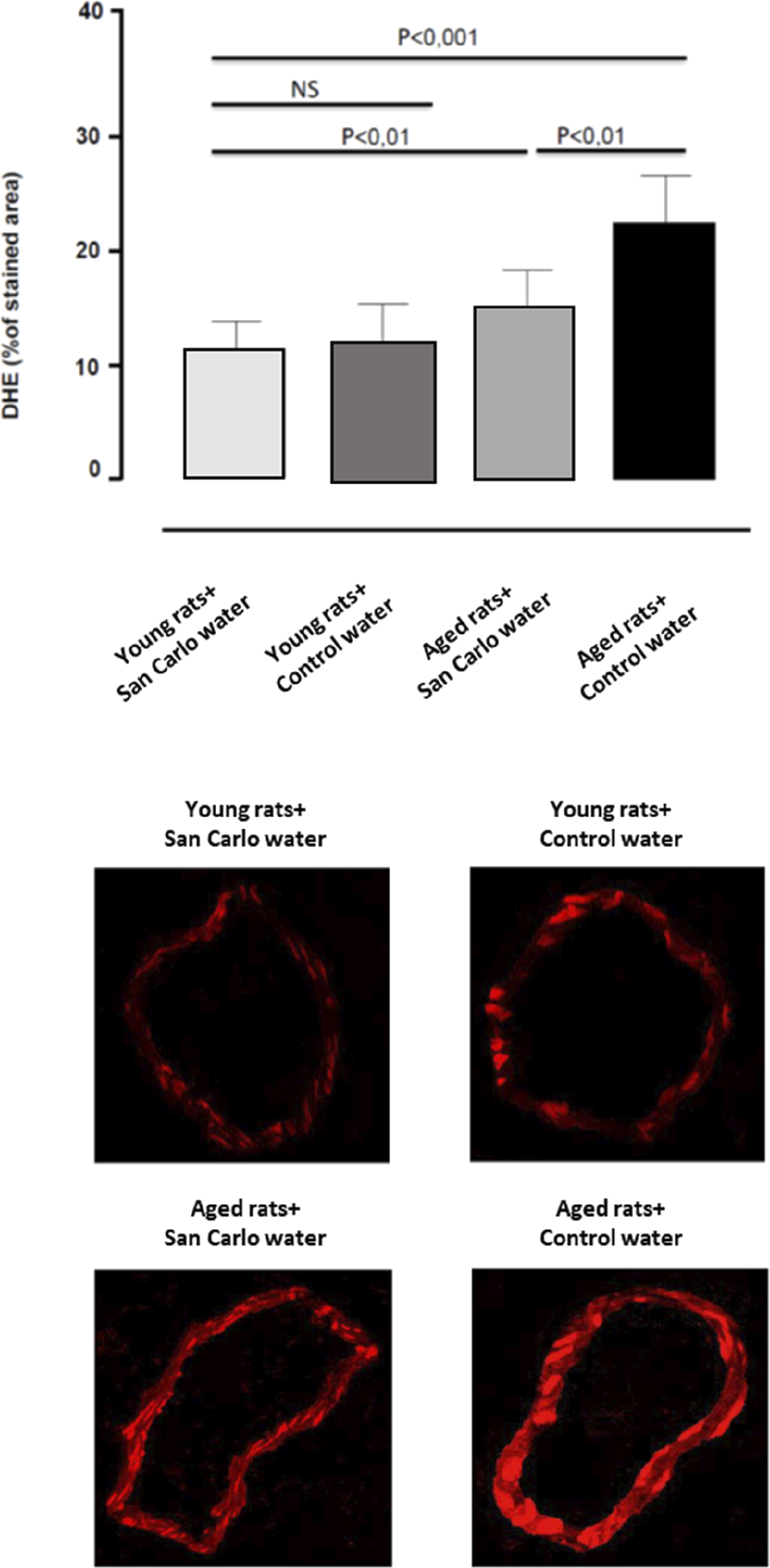
IL-β, TNF, and MDA levels in plasma
IL-1β and TNF levels in plasma from aged animals administered with control water were significantly increased, as compared with young animals (Figure 3). Administration of aged animals with San Carlo® water decreased TNF but not IL-1β levels toward control values, while it did not produce any significant effect in young rats (Figure 3). No significant changes were observed in MDA levels in young and aged rats administered with control or San Carlo® water (Figure 3). IL-1β, TNF, and MDA levels in plasma obtained from young and aged rats treated with control water or San Carlo® water for 3 months. Each column represents the mean ± SEM of eight animals. ap < 0.05, significant difference versus young rat + Control water; ∗p < 0.05, significant difference versus aged rats + Control water. Statistics: one-way analysis of variance followed by Newman–Keuls test.
Urinary oxidative stress and pH
Young and aged rats displayed comparable levels of 8-isoprostanes in the urine (Figure 4). Administration with San Carlo®, but not with control water, significantly reduced the levels of this oxidative stress marker in both animal groups (Figure 4). By evaluating the urinary pH values at four different time points (T0, T1, T2, and T3), we observed that San Carlo® water reduced, although not significantly, the urinary pH in both young and aged rats (Figure 4) in comparison with rats administered with control water (Figure 4). 8-Isoprostane analysis and pH levels in urines obtained from young and aged rats treated with control water or San Carlo® water for 3 months. Each column represents the mean ± SEM of eight animals. ∗p < 0.05, significant difference versus their respective T0. Statistics: one-way analysis of variance followed by Newman–Keuls test.
Discussion
During physiological aging, a chronic sterile inflammation affects the innate immune system, known as “inflammaging” has been observed. 15 This condition is driven by several age-related mechanisms, inducing cell senescence, such as altered mitochondrial function, DNA damage, telomere shortening, and oxidative stress along with the over-activation of pro-inflammatory mediators. 16 In particular, oxidative stress and inflammation have interdependent mechanisms that, in turn, induce and exacerbate each other’s. 17 Indeed, aging is characterized by an increased production of ROS and a concomitant impairment of endogenous antioxidant systems. 18 This chronic low-grade inflammation level, characterizing inflammaging, has been recognized as a crucial aspect of different CVDs, contributing to the pathogenesis and outcomes of these age-related diseases. 19 It is worth noting that CVDs are the main cause of death worldwide, with increasing incidence in aged individuals. 5 Indeed, vascular aging is associated with microvascular dysfunctions, followed by pathological vascular remodeling and vascular stiffness, predisposing to severe atherosclerosis. 20 Interestingly, it has been observed that vascular aging starts during childhood with a gradual change in the vascular structure and endothelial dysfunction, leading to a reduced vascular compliance and increased arterial stiffness in the elderly. 21 The present study demonstrated that the intake of San Carlo® water significantly reduced systemic TNF levels and in situ oxidative stress in vascular wall, with a consequent amelioration of the endothelial-dependent vasorelaxation impairment in aged rats.
Over the years, increasing attention has been paid to the putative beneficial effects of crenotherapy in the management of rheumatologic, respiratory, or dermatologic diseases characterized by inflammation.22–24 In particular, thermal waters reportedly produce a protective effect on cell membranes and genomic DNA, in virtue of their antioxidant and anti-inflammatory properties. 25 Indeed, a comparative study demonstrated an inhibitory effect of La Roche Posay and Avène thermal waters on IL-6 and ROS formation in a human keratinocyte cell line irradiated with UVB. 26 Recently, Sun et al. described the ability of a water treatment to suppress pro-inflammatory cytokines including TNF, IL-6, IL-1β, and NF-κB p65, with a final anti-hypertensive effect. 27 From our functional experiments, we observed that mesenteric small arteries from aged rats showed altered endothelium-dependent relaxations due to a reduced NO availability. Such a deficiency is likely to depend on an increased intravascular ROS generation, as documented by the increased DHE fluorescence in the vascular wall from aged animals as compared to young rats. In this regard, it has been widely demonstrated that the overproduction of O−2 plays a significant role in the development of endothelial dysfunction, through the reaction with NO and subsequent production of peroxynitrite (ONOO−). 13 In line with our results, previous evidence showed a marked endothelial dysfunction associated with a significant vascular pro-oxidant condition in aged animals.28,29 Moreover, evidence from clinical studies highlighted that endothelial dysfunction occurs during the human aging process, triggering the onset of many vasculopathies. 30 The authors observed that the age-dependent endothelial dysfunction in human vessels is due to the combined effect of oxidative stress and vascular wall inflammation. 30 In this regard, it has been widely demonstrated that circulating TNF levels are higher in carotid arteries, aortic wall, and heart of aged rodents and older humans. 31 These studies demonstrated that administration of exogenous TNF can induce oxidative stress by upregulating/activating NADPH oxidase, enhancing endothelial dysfunction and apoptosis. 31 With regard to the latter aspect, our data on TNF levels in mesenteric arteries from aged rats confirmed a condition of age-related low-grade inflammation that may lead to an increase in oxidative stress, thus exerting detrimental effect on endothelial function. 31 The administration of San Carlo® water markedly reduced circulating TNF levels, thus ameliorating endothelial dysfunction in aged rats. Moreover, it has been observed that TNF serves as one of the key mediators involved in the intracellular ROS formation. 32 Indeed, in endothelial cells, NO is responsible for the maintenance of vascular homeostasis and so a reduction in NO bioavailability, resulting from reduced NO production and/or increased NO degradation by O−2, marks the onset of endothelial dysfunction. 33 Specifically, O−2 reacts with NO, leading to the formation of ONOO−, which causes protein nitration and contributes to dysfunction and death of endothelial cells. 33 In parallel with the increase of plasma TNF levels, our DHE assay revealed a significant increase in O−2 production in vessels from aged rats, that was significantly reduced by San Carlo® water administration. Such inhibitory effect on ROS production could be ascribable to the ability of San Carlo® water to reduce the circulating TNF level. Indeed, as previously described, TNF is emerging as a regulator of ROS and reactive nitrogen species generation via NF-kB activation. 34 However, we cannot rule out an antioxidant effect of San Carlo® water related the presence of ions relevantly involved in the correct functioning of endogenous antioxidant systems, including SOD, CAT, and GPx. In line with this view, a marked antioxidant effect of the San Carlo® water has been observed not only in aged but also young rats at urinary level. Indeed, the administration of San Carlo® water determined a marked reduction of 8-isoprostanes, a recognized urinary biomarker of oxidative stress. 35 For this reason, further studies will be designed to better evaluate a putative direct effect of San Carlo® water on the endogenous antioxidant systems.
Of note, San Carlo® water is characterized by an enhanced concentration of silicium (Si) in comparison with control water. This is an intriguing point since it has been widely demonstrated that Si holds a pivotal role in the homeostasis of connective tissues and bones. In particular, a reduction of Si levels has been observed in aortic connective tissues of aged patients, with consequent alterations of vessel reactivity. 36 Based on this premise, we can speculate that the ameliorative effect of San Carlo® water intake, but not of control water, on the vascular reactivity in aged animals could be, at least in part, ascribable to the major presence of Si.
In conclusion, the present results suggest that the use of San Carlo® water can effectively counteract the low-grade inflammation typically associated with age and the related vascular dysfunction, likely through both antioxidant and anti-inflammatory properties.
Statements and declarations
Footnotes
Author contributions
Conception, L.A.; performance of work: V.D., L.B., C.D.S., G.V., S.M, A.M., and E.D; interpretation of data, E.D., L.B., A.M. and C.D.S.; writing the article, L.A. and M.F. All authors have read and agreed to the published version of the manuscript.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from San Carlo S.p.A. (539901_2020_ANTONIOLI_STUDIO_S.CARLO), which had no direct involvement in study design, data acquisition, data interpretation, or the decision to submit this work for publication.
