Abstract
Renal cell carcinoma (RCC) is the most common kidney tumor in adults. RCC is rare in children, although its incidence increases in the second decade of life. Renal medullary carcinoma (RMC) is a rare and often aggressive RCC subtype characterized by complete loss of SMARCB1/INI1, predominantly diagnosed in patients with sickle cell disease or -trait. Here, we describe a pediatric and a young adult patient with clinicopathological characteristics similar to RMC but without hemoglobinopathies. These tumors are currently classified as “RCC unclassified with medullary phenotype” (RCCU-MP). Both our patients had an infiltrating, rather small kidney tumor with SMARCB1/INI1 deficiency. Remarkably, the aggressiveness and clinical course and treatment differed greatly between these two cases. So far, only nine patients, predominantly adults, have been reported. In a literature overview, most cases were metastatic and progressed or relapsed after varying treatment approaches, with low survival. Whereas the classification of these tumors remains a topic of debate, case reports add to the current knowledge of RMCs without hemoglobinopathies, stressing the importance of sharing experience concerning rare renal neoplasms with often poor outcomes, also concerning younger age categories.
Keywords
Introduction
Renal cell carcinoma (RCC) is the most common malignant kidney tumor in adults, whereas in children it only accounts for 2–6% of patients with malignant kidney tumors.1,2 RCC consists of several histological subtypes, of which the distribution between adults and children differs.3,4 One of those subtypes is renal medullary carcinoma (RMC), a very aggressive and rare entity. RMC accounts for <0.5% of all RCC diagnoses, mainly occurring in the third decade of life.5,6 It is most often diagnosed in black patients, and >95% of the patients have hemoglobinopathies, such as sickle cell disease or -trait.5,7 –9 RMC is characterized by complete loss of the tumor suppressor gene SMARCB1, causing loss of immunohistochemical expression of INI1, which is also distinctive for rhabdoid tumors.9 –11
However, the past years, a subtype of RMC with similar clinicopathological characteristics, but without sickle cell hemoglobinopathies has been described. Until now, these tumors have been provisionally classified as “RCC unclassified with medullary phenotype” (RCCU-MP), but have not been officially recognized by the WHO.5,11 –13 At the moment of presentation of the two patients described here, only nine patients diagnosed with RCCU-MP had been described.10,14 –17 The majority of these cases are adult white males, aged between 24 and 71 years. RMC in adults appears less sensitive to the targeted therapies for other RCCs, resulting in very low survival rates. 16 Knowledge of the clinical course and treatment approach of this recently identified subtype remains limited, posing a question for clinicians as to how to treat these patients.6,7
Here, we describe the clinical characteristics, treatment and course of a child and a young adult with RCCU-MP, and summarize literature-based insights of this rare and yet fairly unexplored renal malignancy, especially in these age categories.
Case presentation
Case report 1
The patient was an 11-year-old boy of African descent, presenting with left abdominal flank pain, macroscopic hematuria, fatigue, and weight loss at a secondary care facility (Table 1). Physical examination showed no abnormalities besides a blood pressure above the 95th percentile. The patient had no relevant medical history. The parents of his mother suffered from malignancies at a later age and his mother’s half-sister was diagnosed with breast cancer at age 52. The patient’s cousin from his father’s side was diagnosed with neuroblastoma at 4 months of age.
Timeline of the clinical course of pediatric and young adult case.
A.: not applicable.
Abdominal ultrasound (US) showed a heterogeneous and ill-defined abnormality in the lower pole of the left kidney with a volume of approximately 40 cm3. Abdominal- and chest computed tomography (CT) confirmed the heterogeneous hypodense lesion in the lower pole of the left kidney, with hypodense lymph nodes and pulmonary lesions. Magnetic resonance imaging (MRI) revealed an unencapsulated heterogeneous mass with central hemorrhagic necrosis and limited diffusion restriction on diffusion-weighted imaging (DWI), with multiple pathological lymph nodes in the renal hilum (Figure 1). DWI measures the random motion of water protons. Highly cellular lesions will show an impeded diffusion restriction. The radiological findings combined with the age of the patient raised suspicion of an RCC, for which US-guided core needle biopsies of the tumor were taken.
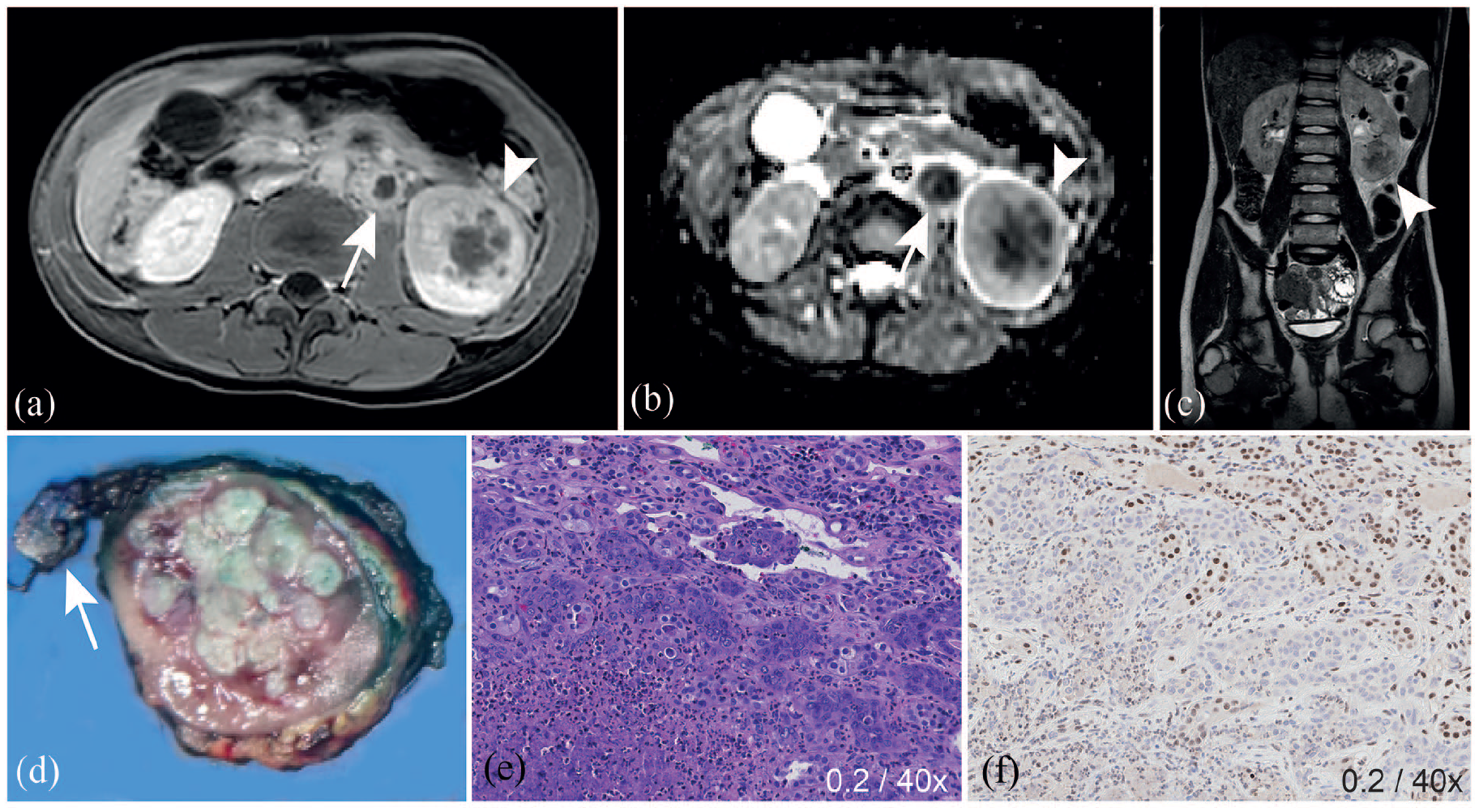
Imaging of the pediatric RCCU-MP case at diagnosis with direct correlation to histopathological findings after total nephrectomy. Imaging and histopathology of the 11-year-old male with RCCU-MP. Abdominal contrast-enhanced axial T1-weighted- (a) and coronal T2-weighted (c) imaging showed an infiltrating non-encapsulated mass in the lower pole of the left kidney, with central areas of low T2 signal intensity and limited enhancement (
The biopsy material revealed a high-grade malignant epithelial tumor of the kidney with a biallelic somatic mutation in SMARCB1, compatible with immunohistochemical loss of INI1 expression. Sickle cell trait or other hemoglobinopathies were excluded by hemoglobin electrophoresis. Based on morphological findings most similar to RMCs, the tumor was therefore suspected to be an RCCU-MP. Upfront total nephrectomy of the left kidney was performed, and histopathology confirmed the biopsy findings, including revealed immunopositivity for cytokeratin 7 (a protein specifically expressed in simple epithelial lining) and PAX8 (a transcription factor involved in regulating genes essential for kidney function), together with OCT4 (a transcription factor involved in self-renewal of undifferentiated stem cells) and INI1 negativity. No clinically relevant germline mutations were identified by whole exome sequencing (WES). The histopathology was directly correlated to the MRI using a patient-specific 3D-printed cutting guide (Figure 1). 18
The patient started with a 21-day cycle of cisplatin, paclitaxel, and gemcitabine (Supplemental Table 1). No additional treatment targets were identified from individualized therapy (iTHER) analysis.
19
On the first day of post-operative chemotherapy the patient experienced an anaphylactoid event (i.e. nonimmunologic anaphylaxis) during starting up the paclitaxel infusion. Therefore, paclitaxel was replaced with
The chest CT-scan after four courses of post-operative chemotherapy showed decrease in the size of the pulmonary lesions. Furthermore, the abdominal US showed no signs of local relapse. Based on the imaging findings, surgical resection of the pulmonary lesions did not seem possible, and they were not considered to be radio-sensitive. In the third week of the sixth course, treatment was continued without cisplatin given a reduced kidney function, classified as chronic kidney disease (CKD) stage 2. Approximately 1 year after diagnosis there were no signs of malignancy. It was decided to stop therapy and continue with surveillance through abdominal US and chest CT-scan every 6 weeks.
Approximately 8 weeks after the end of therapy, the patient presented with a persistent headache and vomiting, also showing a right hemiparesis. The cerebral MRI showed a hemorrhagic metastasis in the pons with extensive edema and without restricted diffusion expanding to the mesencephalon and cerebellum, for which the patient was treated with a radical radiotherapy dose of 35.0 Gy in 10 daily fractions of 3.5 Gy. Nine weeks after start of radiotherapy, the symptoms had mostly resolved and imaging showed a decrease in size of the metastasis and edema (Figure 2). Follow-up after 2 months showed no changes on cerebral MRI, and ongoing complete remission on chest CT and abdominal US.
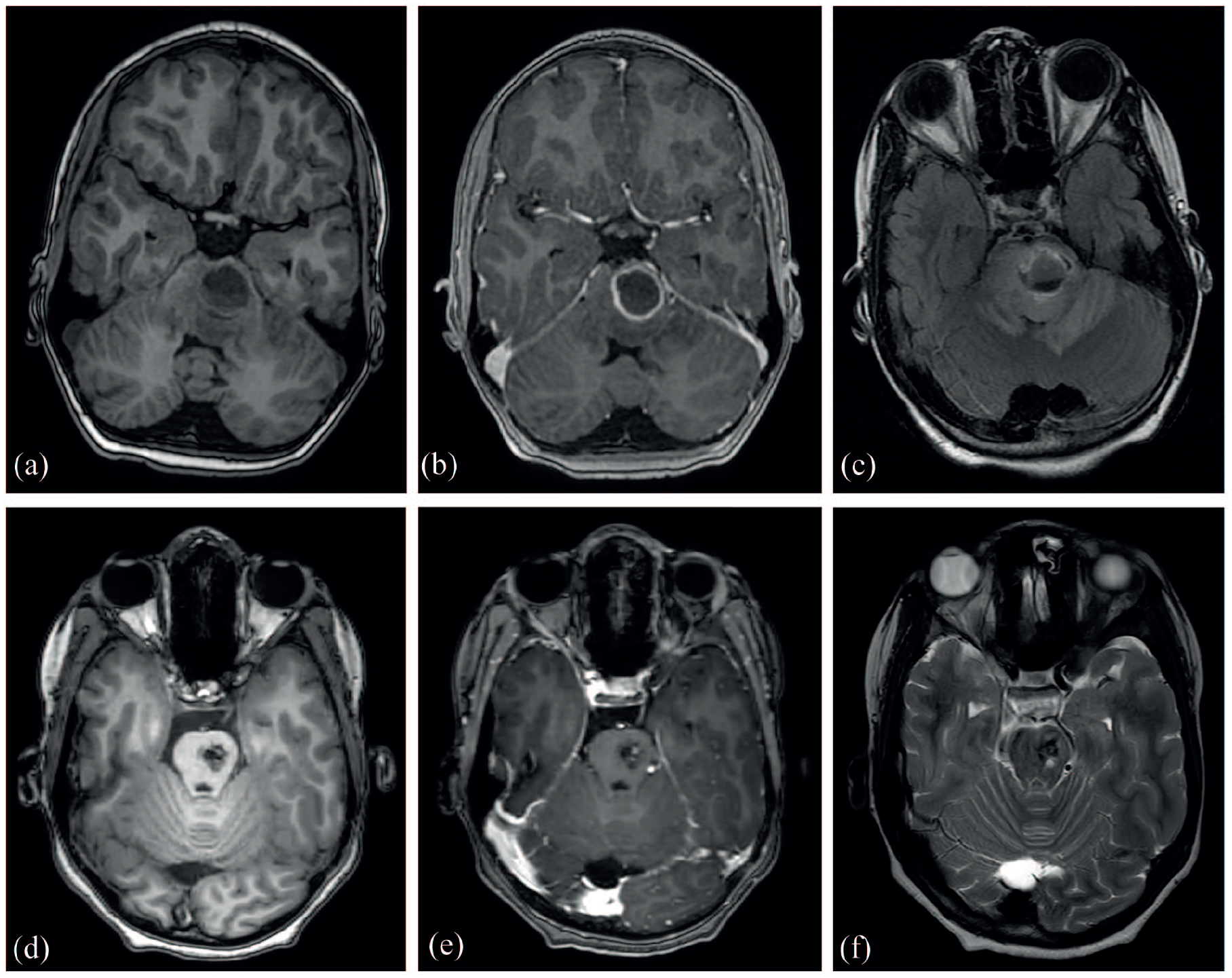
Imaging of the pediatric RCCU-MP case showing a cerebral metastasis after discontinuation of treatment and follow-up imaging after treatment with radiotherapy. Imaging of the 11-year-old male with RCCU-MP with a cerebral metastasis. Cerebral T1-weighted imaging (a) showed a hemorrhagic metastasis in the pons with a diameter of 22 mm, containing sedimentation and a fluid level. On contrast-enhanced T1-weighted imaging (b) the lesion showed ring enhancement without a nodular component, whereas on T2-weighted imaging (c) a hyperintensity consistent with edema was seen. After radiotherapy (d–f), cerebral T1-weighted imaging (d) showed an evident decrease in size of the solitary metastasis, with only limited residual enhancement on the contrast-enhanced T1-weighted scan (e).
Case report 2
The patient was a 20-year-old white male, presenting with macroscopic hematuria for approximately 6 months (Table 1). Family history revealed kidney failure of unknown cause for both paternal grandparents for which dialysis was needed. The patient’s maternal grandfather suffered from prostate cancer above the age of 60.
A single CT urography with combined nephrogenic and excretory phase showed a solid filling defect with caliceal dilatation of the upper pole of the left kidney, suspicious for upper tract urothelial carcinoma (Figure 3). An ureterorenoscopy (URS) reported a multi-cystic tumor, however the biopsy results of the URS were inconclusive. A second URS showed a bullous and pedunculated tumor with calcifications of about 3 cm. The tumor was partly vaporized with a laser and biopsies were taken, revealing no malignant cells. Therefore, it was decided to repeat URS with laser treatment after 1 month.
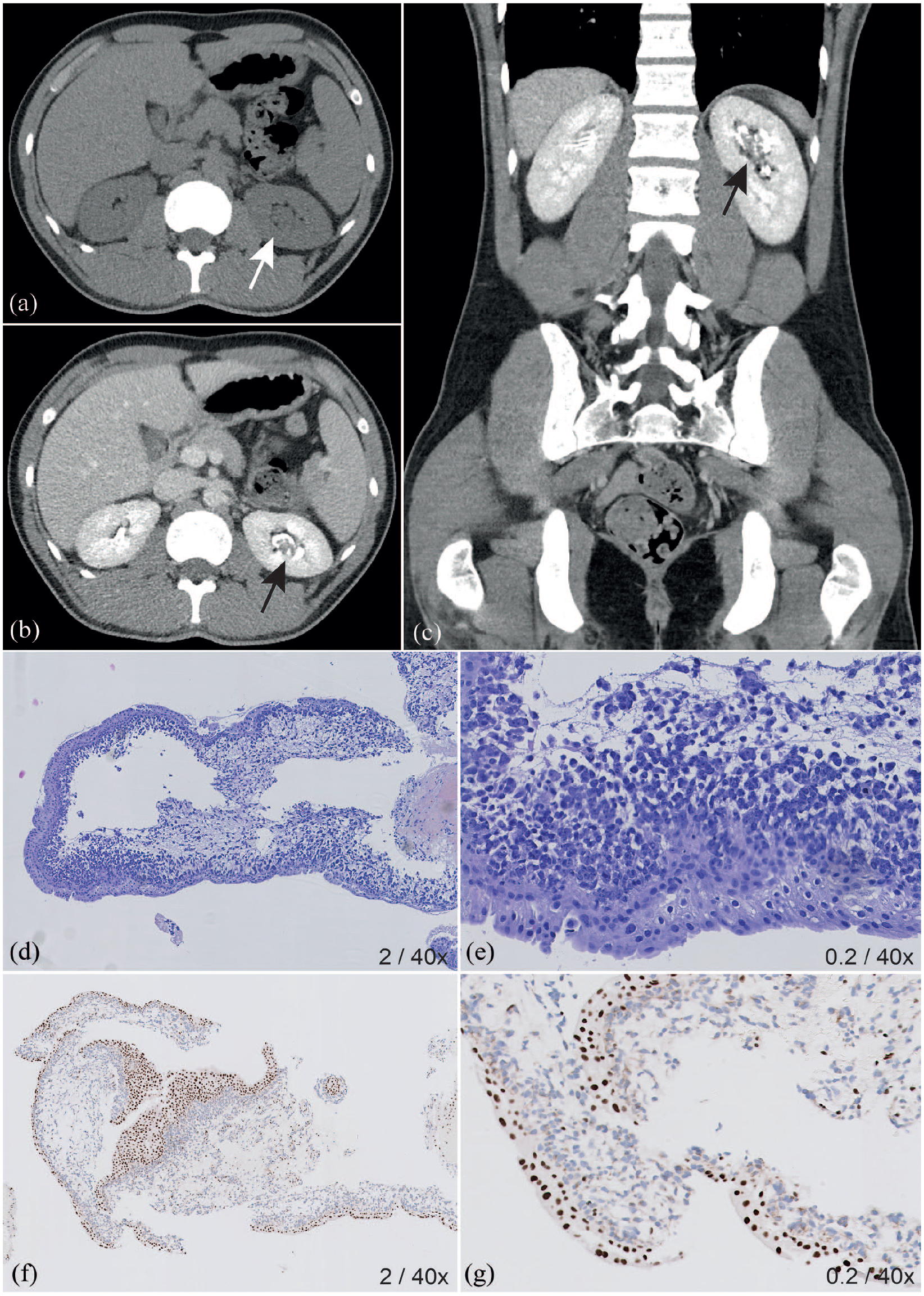
Imaging of the young adult RCCU-MP case at diagnosis with histopathological findings of the biopsy at diagnosis. Imaging and histopathology of the 20-year-old male with RCCU-MP. On the axial abdominal CT scan without intravenous contrast (a) no calcified stones were visible, but the collecting system of the left kidney showed a calyceal dilatation (
The reviewed biopsy results of the URS performed at first presentation showed a high-grade malignant epithelial SMARCB1-deficient tumor, differential diagnostically most likely representing an RMC or RCC not otherwise specified (RCC-NOS) (Figure 3). Subsequent biopsies confirmed the same histopathological presentation after review. Two somatic SMARCB1 mutations were found, and germline WES revealed no sickle cell disease or -trait and no pathogenic variants of 155 known cancer predisposition genes. Given the absence of hemoglobinopathies, the patient was diagnosed with RCCU-MP.
Chest- and abdominal CT-scan did not identify metastases or lymphadenopathy. A radical nephrectomy was performed and histopathology review revealed no remaining malignant cells. Therefore, no further post-operative treatment was administered. Follow-up imaging showed no signs of relapse 15 months after surgery.
Discussion
In this study, we report on two cases of RCCU-MP in young patients, and adding to the overview by Sirohi et al. and Valeri et al., we show the clinical characteristics of all reported RCCU-MP cases to date (Table 2).10,14 –17 RMC was defined as a distinct histopathologic entity in 1995, regarded to be the “seventh sickle cell nephropathy.”20,21 In absence of hemoglobinopathies, these tumors may have been misdiagnosed as other types of RCC or kidney tumors in the past, based on their immunohistochemical and molecular characteristics.5,22,23 Despite the fact that the diagnostic terminology of RCCU-MP is still debated, our cases seem to meet the currently proposed definition of RMCs without sickle cell disease and -trait.5,11,12,16 Nevertheless, reports of pediatric RCCU-MP are rare (Table 2). Sarkar et al. 17 recently reported a case of RCCU-MP with pulmonary metastases in a 15-year old female with neurofibromatosis type 2. Interestingly, in 1997 Kalyanpur et al. 24 described a case of a white 13-year-old female patient diagnosed with RMC, without sickle cell disease or -trait, however this case was not officially diagnosed as RCCU-MP.
Literature overview of clinical data of all known RCCU-MP cases.
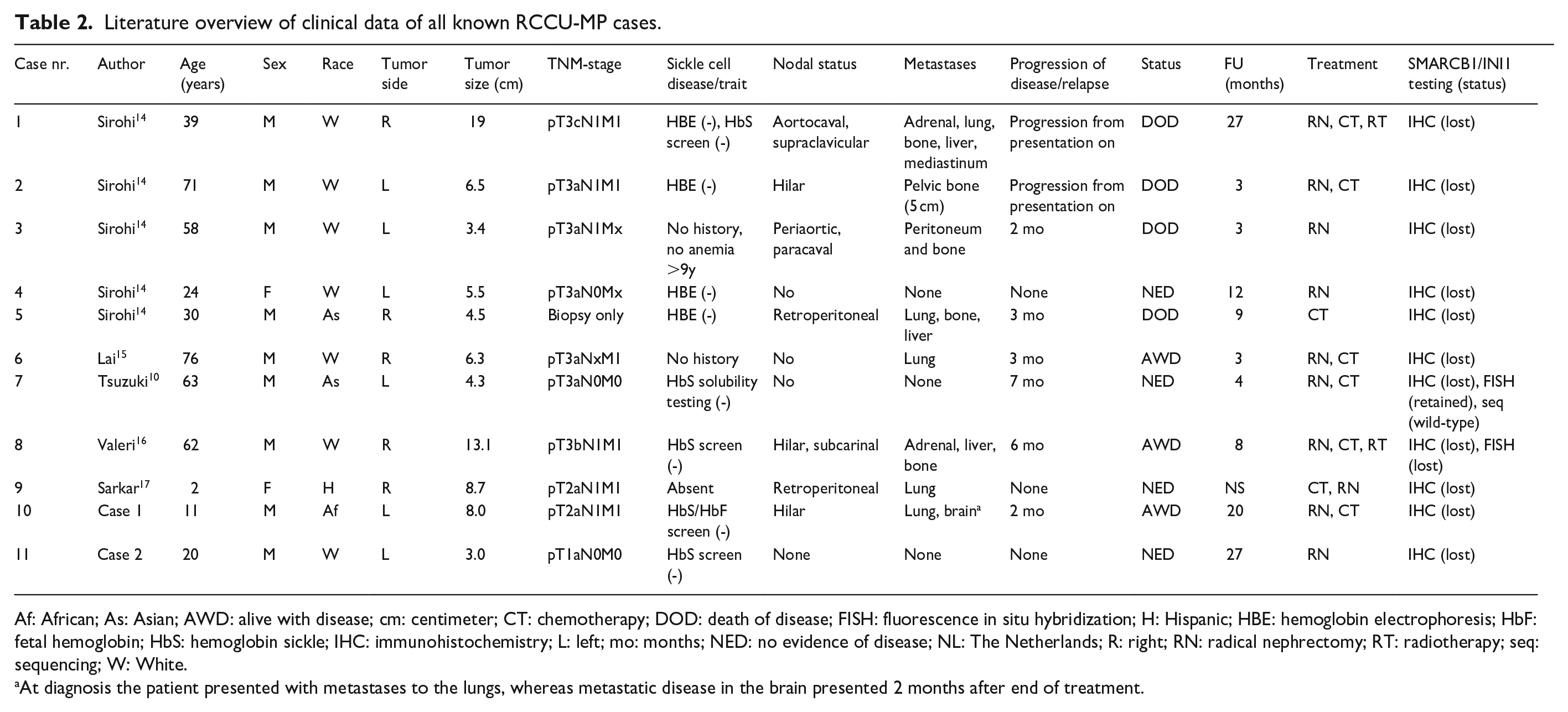
Af: African; As: Asian; AWD: alive with disease; cm: centimeter; CT: chemotherapy; DOD: death of disease; FISH: fluorescence in situ hybridization; H: Hispanic; HBE: hemoglobin electrophoresis; HbF: fetal hemoglobin; HbS: hemoglobin sickle; IHC: immunohistochemistry; L: left; mo: months; NED: no evidence of disease; NL: The Netherlands; R: right; RN: radical nephrectomy; RT: radiotherapy; seq: sequencing; W: White.
At diagnosis the patient presented with metastases to the lungs, whereas metastatic disease in the brain presented 2 months after end of treatment.
In adults, imaging of kidney tumors is often performed using a CT-scan, whereas MRI is preferred in children given its lack of ionizing radiation and possibility for non-invasive semi-quantitative measurements (i.e. DWI). 25 Previously described cases of RCCU-MP seem to appear aggressive on imaging, with a bulky and ill-defined appearance, often invading the kidney and surrounding tissue.14,16,17,24 In contrast, the more frequently occurring Wilms tumor in children is a large, round lesion with a tumor pseudo-capsule. 26 The MRI-findings of our reported pediatric case raised the suspicion for RCC because of the relatively small size, bulky lymphadenopathy and the lack of pseudo-capsule.27,28 The same ill-defined appearance and invading nature of the tumor was seen on imaging of our adult case. 29
In line with the distinctive histopathological characteristics of RMC, both patients presented with an epithelial tumor that was at places largely obscured by microscopic inflammatory characteristics, together with features of hemorrhage and extensive necrosis. 30 Both tumors showed bi-allelic SMARCB1 inactivation, characteristic for RMC, although this molecular event is not specific, as it also occurs in almost all rhabdoid tumors of the kidney and in approximately 15% of collecting duct carcinoma.10,30 –32 Loss of INI1 in these tumors eliminates potential differential diagnoses such as upper tract urothelial carcinoma and anaplastic lymphoma kinase (ALK)-rearranged- or other RCC subtypes.5,16 Immunohistochemically, our findings largely concur with those of Sirohi et al., 14 Lai et al., 15 and Sarkar et al., 17 showing immunostaining for PAX8 and pan-cytokeratin. In contrast to the previous adult reports, our pediatric case showed OCT4 negativity, in line with the pediatric case by Sarkar et al. 17 This has also been observed in fumarate-hydratase deficient RCC, collecting duct carcinoma and upper tract urothelial carcinoma.10,14 –16,30
The pediatric and young adult cases described here highlight how RCCU-MP may differ in aggressiveness and treatment course. The pediatric case had metastasized disease at diagnosis, despite early recognition. After nephrectomy, an intensive chemotherapy regimen was started, based on previous good initial responses to cisplatin or carboplatin combined with paclitaxel and gemcitabine in children with RMC.8,11,33 The patient has responded well to this regimen, which also accounts for the pediatric case reported by Sarkar et al. 17 after pre- and post-operative treatment with cisplatin, carboplatin, gemcitabine, and paclitaxel. Nevertheless, the patient showed a metastasis to the pons shortly after therapy was ended. This is similar to reported adult RCCU-MP patients, which, to date, show no cure in metastasized cases despite systemic therapy.10,14,16,24 In contrast, in the young adult case, confirmation of the RCCU-MP diagnosis was delayed, without occurrence of progression. The patient is alive without evidence of disease, without need for further post-operative treatment. In the two cases with localized disease in the literature, also no relapse after nephrectomy was seen.10,14
In the pediatric case, it was decided to replace paclitaxel with
Whereas the classification of RCCU-MP remains a topic of debate, expanding our knowledge on RMCs without sickle cell disease or -trait may lead to better and earlier recognition of this tumor.5,11 –13,16 To be able to adequately diagnose and describe patients with this rare tumor, it is important that hemoglobin status is assessed in patients with RMC, ideally with electrophoresis or genetic testing. The challenges of RCCU-MP diagnosis and -treatment stress the importance of increasing knowledge concerning its characteristics and potentially unknown associated hemoglobinopathies. The current case reports contribute to the awareness of RCCU-MP in young adults as well as in children; they show the differences in tumor aggressiveness between patients and elaborate on different treatment strategies. Previous research stresses the necessity of collaboration and sharing experience, especially concerning the poor prognosis of metastasized RCCU-MP.
Supplemental Material
sj-docx-1-jnp-10.1177_23993693241248495 – Supplemental material for A pediatric and young adult case of unclassified renal cell carcinoma with medullary phenotype (RCCU-MP): Clinical course and treatment
Supplemental material, sj-docx-1-jnp-10.1177_23993693241248495 for A pediatric and young adult case of unclassified renal cell carcinoma with medullary phenotype (RCCU-MP): Clinical course and treatment by Justine N van der Beek, Aniek Uittenboogaard, Ronald R de Krijger, Floor AM Duijkers, Marieke JM Meijs, Joyce Baard, Marijn A Vermeulen, Germaine Liebrechts-Akkerman, Geert O Janssens, J. Patrick van der Voorn, Marry M van den Heuvel-Eibrink, Annemieke S Littooij and Annelies MC Mavinkurve-Groothuis in Journal of Onco-Nephrology
Footnotes
Acknowledgements
The authors thank the patients and patient’s family for participating in this study. The authors would like to acknowledge Dr. J.W. Gratama from the Gelre Hospital (Apeldoorn, The Netherlands) for his permission to use the images made at their hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant (grant number 341) from the Stichting Kinderen Kankervrij (KiKa).
Ethical approval (include full name of committee approving the research and if available mention reference number of that approval)
NA.
Informed consent
Written informed consent for scientific publication of anonymized clinical characteristics and imaging was obtained from both patients and parents.
Trial registration (where applicable)
NA.
Guarantor
JvdB.
Contributorship
All authors contributed significantly to this manuscript.
Abbreviations
ADC Apparent diffusion coefficient
ALK Anaplastic lymphoma kinase
CKD Chronic kidney disease
CT Computed tomography
DWI Diffusion-weighted imaging
iTHER Individualized therapy
MRI Magnetic resonance imaging
RCC Renal cell carcinoma
RCCU-MP Renal cell carcinoma unclassified with medullary phenotype
RMC Renal medullary carcinoma
URS Ureterorenoscopy
US Ultrasound
WES Whole exome sequencing
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
