Abstract
Availability of human medicines is affected by problems associated with both manufacturing and distribution-related issues. The lack of definition of “shortages” or “unavailability” of medicines in European regulation represents an obstacle to the set up of proper preventive and counteracting measures. In this review, we describe how, within this framework, Italy started a program of shared activities, involving central and local authorities, police forces, and private stakeholders’ associations through an “ad hoc technical forum,” that reduced the extent of the problem, at least in the short term, as demonstrated through some indicators defined by considering the key medicines affected by unavailability due to distribution-related issues. These measures should be strengthened and complemented with others so as to ensure their effectiveness in the long term.
Introduction
Short supply of medicines is an important issue in public health and medical care delivery.1–4 The “symptom” of the “disease,” which is frequently reported even in the general press, is quite simple: patients have difficulties in finding the medicines that were prescribed to them, and these difficulties generate delays or uncomfortable situations. The “external appearance” related to this issue is actually related to different root causes, calling for different solutions: using “short supply of medicines” for classifying all those problems is then misleading since it focuses on the symptoms, instead of targeting the disease.
In the European regulation, there is not an harmonized definition of “shortages of medicines”5–7 (manufacturing-related) and of “unavailability of medicines” (distribution-related), nor of “critical and/or irreplaceable medicines”—an element that would better qualify the need for an exceptional intervention in supporting the patients: this is a key issue hindering efforts in setting up any law enforcement strategy against the phenomenon.
The two issues identified at the European level as “main roots for shortages” include two different macro-phenomena: (1) manufacturing shortages, due to drug manufacturers’ problems, and (2) distribution unavailability, due to problems at distribution chain level.
In the European Union (EU), manufacturing-related shortages are mainly faced by fostering the importation of products between Member States (MSs) with different levels of availability, while distribution unavailability, on reverse, is usually causing measures aimed at “controlling” the export (via blacklists of products not to be exported, or to be exported only after notification to the competent authorities, that may object to the request).
The main objective of this study is to evaluate the impact of the Italian approach to the issue, that—in consideration of the difficulties in acting against the free circulation of goods—prioritized the creation of an ad hoc forum of stakeholders (central and local administration; associations of marketing authorization holders (MAHs), distributors, pharmacists, and health professionals) aimed at creating consensus about a stricter interpretation of the existing regulatory framework, allowing a pilot phase of field inspections targeting (and sanctioning) specific distortions that were considered as the main roots of the unavailability of some critical medicines.
Framework: definitions, regulation, and counteracting activities
Manufacturing-related shortages and distribution unavailability of medicines
Manufacturing shortages of medicines
Manufacturing shortages of medicines are often related to the non-profitability of low price/old products.8–10
Short-term manufacturing shortages may be related to technical difficulties at the level of manufacturing sites (e.g. damage to production lines, and short supply of active pharmaceutical ingredients), regulatory framework (e.g. delay in approval of variations to the marketing authorization), or specific temporary issues: but most of the manufacturing-related shortages are connected to economic considerations, considering, for instance, the low prices and the small market size expected for old medicines. 11
In a recent case of worldwide manufacturing shortage, affecting a neurologic product, the involved company published a statement highlighting that the root cause for the problem was the increasing demand caused by the lack of availability of equivalent products from other suppliers: as for the available data, the product accounted for less than 0.1% of the annual revenue of the company, and many of the generic equivalents manufacturing authorizations were revoked upon request of the other MAHs, confirming the expected priority framework related to products with a limited economic impact. 12
Article 81 of the European Commission (EC) Directive 2001/83 (and subsequent amendments) identifies the marketing authorization holder (MAH) as responsible for the continuity of the supply of a given medicinal product on the MS’s territory “within the limits of their own responsibility,” without further specification of these limits.13,14
Trying to address this issue, during the last years, the development of ad hoc specific prevention “shortages risk management plans,” to be included in the documentation of all manufacturing sites’ master files, has been discussed at a technical level.
With respect to manufacturers, the kind of preventive activities that could be put in place may, for instance, include the following:
Managing adequate inventories for critical medicines, allowing full compliance with the provisions laid down in Article 23 of Directive 2001/83/EC (advance notification of deficiencies provided to the competent national authorities).
Systematic and preventive selection of productive alternatives (suppliers of raw materials, and manufacturing sites for the finished medicinal product) for all essential/critical medicines.
The standard corrective actions applied by the administrations in the case of manufacturing shortages consist in the authorization (granted to MAHs or to local health structures) for the importation of corresponding products from other Member States (MMSS) (or, in exceptional cases, from non-EU countries).
Distribution unavailability of medicines
Distribution unavailability of medicines is due primarily to economic factors correlated with the distribution network; it is often related to “parallel trade,” that is, the sale between wholesalers operating in different markets with different prices, and it is particularly relevant for countries with higher average prices such as Germany, where the parallel trade is fostered through regulatory tools.
Over the years, several strategies for tackling this problem have been proposed and implemented, and even the (definitely unlikely) imposition of an “average European price,” comparable enough to discourage the export to more profitable markets, has been from time to time mentioned in the discussion.
The main potential measure against unavailability is the block of the export of “essential medicines” that as such would not comply with the principle of free movement of goods, but that could be someway realized through “export licensing” ad hoc processes by national authorities. The implementation of the Directive 2011/62/EU created other opportunities for MMSS for developing measures aimed at better “controlling” the export, such as those put in place in France, specifically connecting two concepts from the EC regulation, that is, the wholesalers’ obligation of giving continuity to their “public service” (in particular, with respect to the priority to the supply of the area for which they were authorized) and the shortage-related public health issues.
It is important to note that, in almost all cases, new measures focussing on trade control were specifically objected by the EC, as it happened in 2016 to Portugal and Slovakia. In Portugal, wholesale distributors of medicinal products for human use were requested to notify their intention to export medicines considered “at risk of shortage” by the authorities and to provide information on the export operations that have been carried out. Further to this, in Slovakia, wholesale distributors were also requested to notify exports of all human medicines and to wait 30 days for a tacit clearance by the state authority. The Commission stated that these notification procedures do not set out clear and transparent criteria for determining the medicinal products that may be at risk of a potential shortage due to parallel trade: furthermore, the procedures impose disproportionate reporting obligations on wholesalers, and in Slovakia, the duration of the notification is disproportionately long. The Commission then requested Portugal and Slovakia to change their laws and consider less restrictive measures to intra-EU trade, to avoid being referred to the Court of Justice of the EU. 15
Recently, the EC position on this matter slightly changed. Measures and restrictive measures proposed by some Member States against the parallel trade, with the goal of ensuring the continuous supply of critical medicines to the national market, were considered in compliance with the European rules: in response to complaints before it, against several forms of export restrictions by Member States, the EC took the view that—given the need for a fine balance between the free movement of goods and the access to healthcare by patients—there need to be other ways than infringements to adequately solve the complex situation, in consideration of the fact that parallel trade in medicines may be one of the reasons for the occurrence of shortages of a number of medicinal products for human use. 16 This change of perspective may trigger a change in the strategies of the interested MMSS in facing the phenomenon.
The Italian regulation and activities against manufacturing-related shortages
The Italian regulation is clearly based on the EU one: MAHs are requested to timely inform the administration with respect to possible forthcoming manufacturing shortages, to allow for the set up of counteracting measures, while distribution-related unavailabilities are not openly mentioned in the regulation, even if in the implementation of the Directive 2011/62/EU, a specific article underlining the priority to the continuity of supply to the country calls for possible “specific acts” against the export of critical medicines, to be better defined in the broader framework of the free circulation of goods principle.
In detail, with respect to manufacturing shortages, also Italy has put in place standard counteracting measures on import from EU/third countries, similar to those mentioned above. In addition, Italy applied with success other models against established shortages, such as exceptional manufacturing activities performed by specific sites (e.g. the National Military Manufacturing site) or ad hoc agreements with the involved companies.
This “case by case” management had positive results; however, it cannot be considered as a cost-effective solution to the issue, since developing and testing a full model against a single, specific shortage requires an investment of resources which is often non-proportionate to the case. However, this model could be evaluated at EU level, since the availability of “National manufacturing sites” in some Member States could be considered as a resource for managing specific manufacturing shortages affecting the whole Union, for example, related to old, “non-profitable” products.
The Italian regulation and activities against distribution unavailability of medicines
The model that Italy developed for counteracting distribution-related unavailability is definitely easier to share than the “case by case” one described for manufacturing shortages, being focused on sharing practices and cooperation between stakeholders in better enforcing good distribution practices (GDPs): in the Italian framework, distortions in the distribution network leading to massive export of “essential medicines” are only possible through practices not in compliance with GDPs (e.g. “raking”—gathering huge quantities of products through small orders of few packages to many different pharmacies—illegally selling medicines to wholesalers, instead of selling to the patients) that are already subjected to sanctions according to EU and National regulations.
As a first step, in counteracting the phenomenon, Italy, when transposing Directive 2011/62, added a provision similar to the French one mentioned above (Legislative Decree no. 17/2014): “medicines that were subjected to specific measures to prevent or limit states, even temporary, of shortage or unavailability on the market in the absence of viable therapeutic alternatives, cannot be subtracted, distribution and sales to the country”.
According to the legislation, the Italian Medicines Agency (AIFA) and the Italian Ministry of Health (MoH) should periodically publish a list of medicines “under short supply” that cannot be considered for export. At the current state of implementation, AIFA regularly publishes the list of the medicinal products subjected to production shortages; MoH and AIFA may verify through the traceability data, already available for all medicines’ transactions in Italy, all exports related to the products in the list, and therefore highlight possible issues.
Considering the potential misalignment between the full application of the process as defined in the regulation and the EU Treaties, confirmed by the above-mentioned pronouncements against Portugal and the Slovak Republic, Italy started a parallel process of management of the phenomenon. An ad hoc technical forum involving all relevant authorities (in addition to AIFA and MoH: regions responsible for the controls on the distribution network and the Italian specialized police force dealing with health-related matters, Carabinieri NAS) and the associations of manufacturers, distributors, and retailers was activated in 2015.
The first objective of this forum (achieved in June 2016 with an ad hoc inspection campaign and in September 2016 with a formal “Memorandum of Understanding” (MoU) signed by all involved authorities and stakeholders’ associations) was primarily the development, sharing, and enforcing of basic rules on the management of the distribution of medicines, that is,
To get proper enforcement of Legislative Decree no. 17/2014 with respect to the measures for avoiding the subtracting of “essential medicines” from the Italian market;
To develop a shared, operative definition of “shortage,” “unavailability,” and “essential medicines”;
To define harmonized enforcement processes for GDP and traceability rules, to be supported also by ad hoc training programs for the inspectors of local competent authorities;
To start targeted monitoring measures on exports of “essential medicines.”
The difficulties in properly enforcing appropriate checks on distribution may be related to the 2006 regulation which allowed the pharmacy holders to manage the activity of a pharmaceutical wholesaler: this “deregulation” was one of the key factors for the increase in the number of authorized wholesalers (from 150 to over 1000), thus multiplying the burden on local authorities dealing with checks and inspections. Then, the ad hoc technical forum started some specific projects aimed at rationalizing and optimizing the use of resources to be allocated for inspection purposes.
The pilot project on distribution verifications
The ad hoc technical forum verification project: background
In view of a number of potential violations of the GDP/traceability rules by certain categories of exporting wholesalers, the technical forum shared some key standard procedures between all stakeholders, and to promote the control on their proper enforcement. Verifications performed in the field through Carabinieri NAS inspections, in the framework of a pilot project on a specific region of Italy, confirmed the working hypothesis, and also caused the withdrawal of about 20 wholesaler licenses (see section “The ad hoc technical forum verification project: some results”).
The background of the “ad hoc technical forum” project is the operation carried out in 2014–2015 by AIFA and NAS, with the support of the MoH, that allowed the eradication of a specific form of pharmaceutical crime, the thefts from hospitals, through targeted actions on the distribution network that was proven to be the root cause. 17 Investigations showed that the European distribution network had been infiltrated by organized crime and highlighted a number of other distortions, due to apparently unrelated phenomena, such as the distribution unavailability generated by the overlap between the fully legal exports, existing in Italy before 2006, and the “borderline” or irregular practices, often ascribed to the aforementioned deregulation.
The interim results of this exercise are already visible: the reduction in distribution-related unavailability was confirmed in 2017 by retailers’ associations and regional authorities, and despite some specific situations still requiring ad hoc interventions, the current approach seems to be quite effective. Currently, the “ad hoc technical forum” continues to foster control campaigns and to sanction non-compliant operators, with the goal of optimizing both the quality of auditing and the use of resources at central and local level.
The ad hoc technical forum verification project: methods and targets
The auditing performed by Carabinieri NAS in the field was developed according to some key features of the already known framework:
Market unavailability is not measurable as such: at present time, the only indicator of a problem may be the signal sent by an operator (or even by patients) to the competent authorities, reporting the difficulty in obtaining the supply of a given medicinal product, as for the national regulation released in 2014 and implementing Directive 2011/62/EC. This regulation asks pharmacists to send a signal to regional authorities for any “interruption of supply”: as a consequence, some regional pharmacists’ associations set up systems for gathering the signals, developing periodic lists of “medicines in short supply.” Of note, this indicator may not only refer to patients’ needs but may also signal a specific business opportunity—that is, the operator that was not timely supplied might have been interested in obtaining the product for exporting purposes. Despite this caveat, since there is no other suitable way for measuring the “perceived problem,” it was agreed to consider this indicator as the most reliable reference.
At the time of the exercise, the Latium Regional Pharmacists’ Association (Federfarma Lazio) had the most reliable list of “medicines in short supply” and could represent a reliable proxy of the phenomenon, confirming AIFA’s/MoH’s evaluation that the issue had national features, and that was affecting an extremely limited number of products/brands, despite its large general press coverage.
According to the preliminary evaluation performed by the administrations, the issue was related to some systematic exporting routines, generating local, temporary difficulties in satisfying the demand of the distribution chain. Since neither the traceability data nor industry studies highlighted any “big buyer” dealing with export, the expected model studied for explaining the process was a “raking procedure”: as already proved during the “Volcano Operation” investigation, some exporting wholesalers were “affiliating” double-license operators (i.e. pharmacists with a wholesaler license) buying small quantities (as a pharmacy) to be re-sold (through a wholesaler’s license—held by the same pharmacy) to the exporters. Such a model is a clear infringement of the national distribution rules: the movement of pharmacy goods from the pharmacy to the licensed pharmacy-wholesaler is not in compliance with the EC regulation, since pharmacies may not sell to wholesalers, and wholesalers cannot buy from operators not entitled to sell to them.
The standard “double-license operator” was then an operator mainly dealing with pharmacy activities and holding the wholesaler license only for access the exporting network: such an operator is not interested in properly acting as a wholesaler, then he would only manage a little number of products, using limited storage space, with the smallest possible investment of resources, often not in compliance with GDPs and other distribution rules.
The inspection campaign that was performed during the pilot phase of the project, later repeated at national level, was then focused on operators exporting the “tracing products” from the list of “medicines in short supply” of Federfarma Lazio; these operators were identified through the MoH’s traceability data, and Carabinieri NAS were requested to verify on the field the source of the exported products, tracing them back through the previous suppliers up to the MAH. During the verification process, all illegal transactions and all GDP relevant non-compliances were sanctioned: the 2015–2016 pilot phase led to more than 20 license withdrawals and sanctions for almost 800,000 Euros.
The 2016 formal MoU above mentioned extended the auditing process at a national level, allowing regional authorities to improve their auditing procedures, and giving them full support in case of disputes: the quality of the qualification of the chain was definitely increased due to the growing priority given to the issue.
The ad hoc technical forum verification project: some results
The results of the campaign may be evaluated by checking how the situation of the “tracing products” changed through the years: while the “perception of the issue” did not change so much in terms of number of signals per year (a result that may be explained with the increasing awareness with respect to the “reporting processes,” that are now easier and more efficient than in 2014–2015), the incidence of export—the only confirmed “disruption factor”—is clearly decreasing.
Our analysis was limited to the products covered by more than 100 “reports of local shortage” in 3 years. The majority of the “reports of local shortage” are related to four brand names from three MAHs.
Table 1 presents the situation of the four more relevant products (A, B, C, and D, from two MAHs: 1 and 2), used as “tracing products” during the inspections performed in the pilot project.
Differences in the incidence of export for the key “tracing products” used as a reference during the inspections of the Italian distributors during the pilot project.
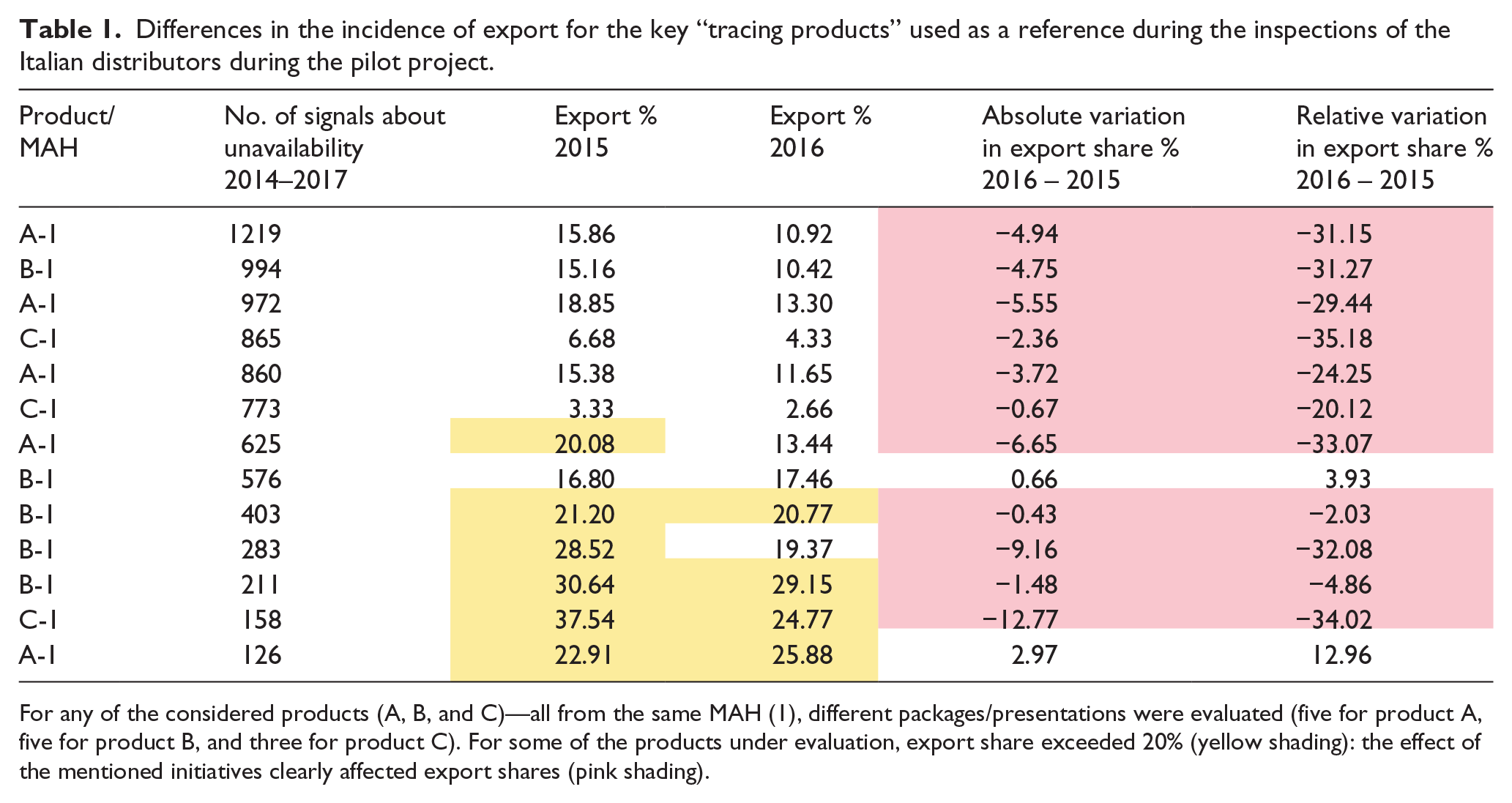
For any of the considered products (A, B, and C)—all from the same MAH (1), different packages/presentations were evaluated (five for product A, five for product B, and three for product C). For some of the products under evaluation, export share exceeded 20% (yellow shading): the effect of the mentioned initiatives clearly affected export shares (pink shading).
It appears very clear that the activities developed by public and private stakeholders since 2015 caused a remarkable decrease in the incidence of export, measured as percentage of export with respect to the total number of marketed units, for all evaluated products.
A similar decrease was not observed neither in general for the exporting activities, since the number of exported packages was quite unvaried between 2014 and 2016—with a 2% change at its best and with a small increase in 2016 with respect to 2015 (28.5 million in 2014, 28 million in 2015, and 28.3 million in 2016), nor specifically for other similar products, such as the one labeled “D” in this study.
MAH 1 and MAH 2 products are quite different in terms of turnover and main distribution channels: the two situations may then better be evaluated separately.
The counteracting measures were extremely efficient for the products of MAH 1 (having a relevant distribution to pharmacies) but had no impact on the products of MAH 2, mainly distributed to hospitals, for which “short supply” was mainly a perceived effect, only marginally related to export (Table 2).
Differences in the incidence of export for the fourth key “tracing products” used as a reference during the inspections of the Italian distributors during the pilot project.

Also, for product D—from a different MAH 2, three different packages/presentations were evaluated.
These data confirm then that the perception of the phenomenon gives a blurred image of the real framework: apparently, reported problems are related to a very small number of products, which are exported in such a relevant share, that considering them “in short supply” is not proper.
“Short supply reporters” will be further investigated in the future studies, to understand how much reporting may be related to business reasons or to patients’ needs.
Conclusion
The “ad hoc technical forum” represents a good model for sharing activities against distortions in the distribution of medicines, often overlooked by relevant local authorities, for lack of knowledge. Italy considers this project as a “good practice” that may be easily shared with other Member States. The positive results obtained through a shared verification project such as the one described above confirm the desirability of the development of similar “soft law tools,” with the goal of supporting policies aimed at counteracting any kind of short supply of medicines.
With respect to proper legislative activities, it would be appropriate to focus on manufacturing shortages (a key issue for many critical products, with respect to whom there are ongoing initiatives at the EU level), clarifying what the MAH should do “within the limits of their own responsibility” with respect to the prevention of the interruption of the supply.
The recent developments in the counteracting activities performed at the level of the European Medicines Agency (EMA), 18 such as the web-based platforms and databases on manufacturing shortages, may also be considered as part of the same “soft law approach” followed by the Italian authorities: such instruments allow all Member States to get an overview of the phenomenon that surely would help address the supply disruptions of critical medicines.
With respect to “unavailability of essential medicines” related to distribution, proper enforcement of existing GDPs would be a good start for addressing the problem, for example, by removing possible distortions in the exporting activities.
In Italy, the above-described model approach was apparently successful in addressing the issue: an “ad hoc technical forum” such as the one established in Italy could be the right working group for sharing the interpretation and the enforcement of the rules, and the “good practices” already developed at the national level, with the goal of developing proper national measures to be shared between all stakeholders, without infringing EC rules.
Footnotes
Acknowledgements
This article was developed by the Italian Medicines Agency (AIFA) in the framework of the activities of the above-mentioned “ad hoc technical forum” (Tavolo tecnico indisponibilità), and it is therefore signed by the representants of the involved associations of manufacturers (Associazione Italiana Parallel Traders—AIP, Assogenerici, Farmindustria), distributors (Associazione Distributori Farmaceutici—ADF, Federfarma Servizi), retailers (Assofarm, Farmacie Unite, Federfarma), and service providers (Assoram), and scientific societies and orders (Federation of the Orders of Italian Pharmacists—FOFI, Italian Society of Hospital Pharmacy—SIFO). The authors thank the colleagues representing the administration in the “ad hoc technical forum”—in particular, T.Col. Erasmo Fontana (Carabinieri NAS), Claudia Biffoli (Ministry of Health, traceability systems) for providing data and advice for this study; Giampiero Camera (Ministry of Health, pharmaceutical distribution) for supporting and liaising with the regional authorities; and Paolo Crenna, Veronica Marangon, and Andrea Angelo Nisic (Regional GDP inspectorates) for all the practical hints.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
