Abstract
Managed entry agreements (MEAs)—a type of formal institutional arrangement between pharmaceutical companies and payers for sharing the risk with respect to the introduction of new pharmaceutical technologies—may expand access to new pharmaceutical technologies for non-communicable diseases (NCDs). Although common in highincome countries (HICs), there is limited evidence of their use in low- and middle-income countries (LMICs). This article aims to document international experiences of countries implementing MEAs and potential barriers and facilitators for their use in LMICs. We reviewed published literature sources on MEAs over the past 10 years considering peer-reviewed publications and gray literature data. We took into consideration the MEAs taxonomy presented by Kanavos and Ferrario et al. to categorize our findings, and extract information on factors for their implementation. We retrieved 285 MEAs documented in the literature, mostly from HICs and for a broad spectrum of NCDs. Financial schemes were slightly more prominent than performance-based agreements. Identified factors that could potentially facilitate or hinder the implementation of MEAs included the presence of quality administrative and information systems to track their implementation; availability of quality data and evidence of positive outcomes; uncertainty of drug efficacy/effectiveness, safety, and financial impact; and cultural factors, namely country’s preference for certain type of agreement and trust among payers and manufacturers. The increased availability of publications in recent years suggests a growing interest among policy-makers and researchers in the implementation of MEAs. While the use of MEAs in LMICs is very limited, this could be the result of limited empirical evidence on its use and possibly due to the use of a different taxonomy for describing MEAs in these settings. As any other policy option, the implementation and use of MEAs come with advantages and challenges. Since there is limited evidence on their use in LMICs, the identified cases of implementation in HICs may serve to inform the interest on MEAs in resource limited settings. Therefore, further research in this field especially in the context of LMICs may be of value for the global community as all countries are embarking into fairer and sustainable Universal Health Coverage (UHC).
Keywords
Background
The growing burden of non-communicable diseases (NCDs) in low- and middle-income countries (LMICs) poses a considerable challenge to advancing progress toward Universal Health Coverage (UHC) and reaching health-related sustainable development goals (SDGs) by 2030. 1 Nearly 40 million people die annually from NCDs globally, with cardiovascular disease (CVD), cancers, strokes, chronic respiratory diseases, and diabetes being responsible for 70% of NCD-related mortality.2,3 An estimated 75% of these deaths occur in resource-constrained settings, where access to diagnostics and treatment for NCDs are still limited. 4
In LMICs, the high costs of new patented NCDs therapies and the lack of information on their effectiveness at the early stages of entering the market, has contributed to delays in reimbursement decisions and ultimately limited patient access to (in many cases) life-saving medicines and other health-related technologies. 5 To enable access to innovation and improve the allocation of scarce resources for health, numerous access strategies at various levels of the health system have been utilized. These include, but are not limited to, national pharmaceutical policies and regulations, focused on priority setting, procurement, and distribution of medicines for NCDs, as well as interventions aimed at defining how they are financed, priced, and used.
Despite these policies, effective, access to NCDs medicines remains limited in LMICs due to the countries’ different levels of development, their wide range of health systems constraints, and resource limitations, as well as fragmentation of their sources of funding which many times heavily rely on donor assistance or out-of-pocket (OOP) payments. Even the limited segment of the most affluent population potentially able to purchase medicines through OOP payments may face financial constraints over time, given the strain imposed due to NCDs chronicity. 6
Another increasingly implemented access strategy involves negotiated agreements between pharmaceutical companies and payers (either public or private) or donors. These arrangements take the form of risk-sharing agreements and are commonly referred to as managed entry agreements (MEAs), managed access schemes, performance-based agreements, or patient access schemes. MEAs are a type of formal institutional arrangement between pharmaceutical companies (manufacturers) and payers, aimed at sharing the financial risk with respect to the introduction of new pharmaceutical technologies. 7 MEAs are designed to promote access to medicines for which there is still uncertainty with respect to their cost-effectiveness, real-life clinical benefit and potential impact on health sector expenditure. 8 Increasing pressure to identify alternative access solutions for NCDs has led manufacturers, governments, and health insurers to consider the use of MEAs and look at the international evidence of their implementation, which predominantly has been the case of high-income countries (HICs).
Although MEAs have been less commonly used in LMICs, they have increasingly seized the attention of decision-makers in these settings. 9 MEAs may bring an opportunity for policy action, allowing stakeholders to explore a wide range of different types of instruments to address context-specific needs. For pharmaceutical companies, MEAs may provide some predictability about initial price conditions and potential market size estimations and could also help create a more collaborative environment between payers and pharmaceutical companies.
This article illustrates salient features from existing MEAs—namely financial schemes and performance-based agreements—aimed at informing their potential implementation in LMICs to advance early and affordable access to on-patent innovative medicines for NCDs in these contexts. Firstly it presents the methods of the literature search, followed by results and a discussion on the challenges of implementing MEAs in LMICs; finally it draws some conclusions to inform future policy action and research agendas.
Methods
We conducted a search of the literature between December 2017 and February 2018 to identify studies documenting international experiences on the implementation of MEAs. The review looked at peer-reviewed publications and gray literature sources.
To summarize our findings, we adapted and applied the taxonomy published by Kanavos et al. 7 which defines two main categories of MEAs: financial schemes and performance-based agreements. Financial schemes focus on targeting the financial impact of new drugs to patients and/or health systems and leverage instruments such as discounts, price/volume agreements, patient/dose-dependent discounts, and utilization-based price capping. 7 Conversely, performance-based agreements address the uncertainty with respect to evidence on clinical outcomes or eligibility of patient populations. Instruments of performance-based agreements include outcome guarantees, patient eligibility requirements/registries, and coverage with evidence development. Other models that combined financial- and performance-based instruments were categorized as “hybrid” in our summary (Figure 1).
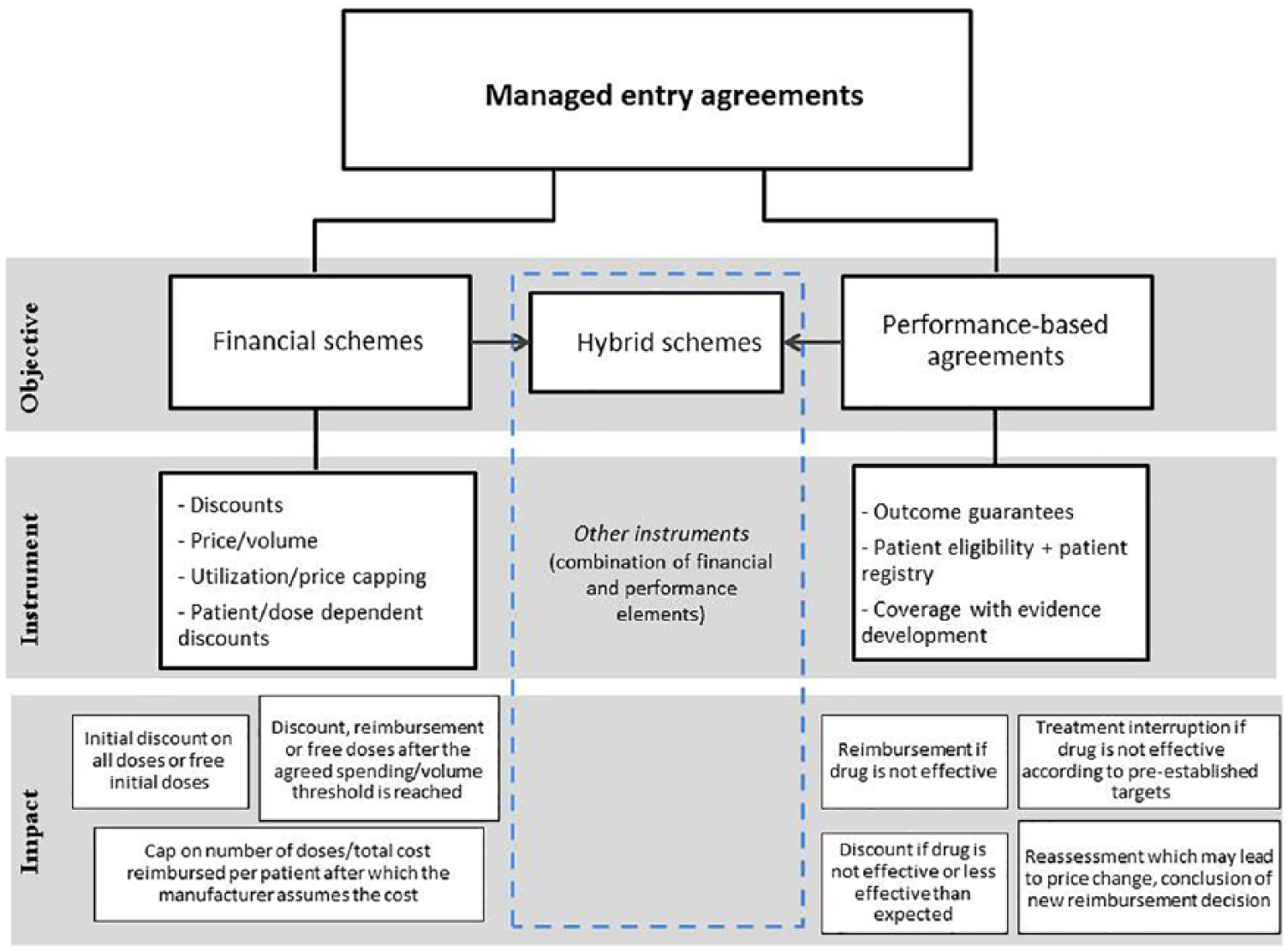
Taxonomy for managed access schemes (adapted to include “hybrid” approaches).
Peer-reviewed literature search
Our search of peer-reviewed publications considered a 10-year time frame (1 January 2007–31 December 2017). We searched the following data sources: Biomedical Reference Collection, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, Embase, Medline, and PubMed. The search terms were grouped into three main categories (i.e. risk-sharing agreements, patented medicines, and improved access) and then combined using the Boolean operator “AND”.
The following search terms were used: (financing mechanism* OR financial mechanism* OR risk sharing OR managed entry agreement* OR performance based agreement* OR patient assistance program* OR patient access program*) AND (patented medicines OR on-patent OR on patent OR on-patent medicin* OR on patent drug* OR patented drug* OR generic medicine* OR generic drug* OR off-patent OR off patent OR off-patent medicin* OR off patent drug* OR patent*) AND (improve access OR improving access OR improved access OR market access OR improve outcome* OR improved outcome* OR improve result* OR improved result*).
Search terms were translated into French and Spanish to capture non-English language publications. The literature search findings were reported according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) standards.
Only primary studies using original data, literature reviews, editorials, and author commentaries were included. We also considered additional papers suggested by international matter experts. The inclusion criteria used are as follows: (1) articles describing financial or performance-based mechanisms for granting access to innovative patented medicines and generics globally and (2) articles generally describing lessons learned and previous experiences with MEAs. We deliberately excluded conference abstracts because of their potential limited representation of MEAs features and studies that described other market access mechanisms beyond MEAs, such as those focused on community based, management strengthening, health-care delivery, therapeutic substitution (e.g. introduction of generics), supply chain or production related (manufacturing or research and development), and licensing agreements or Trade-Related Aspects of Intellectual Property Rights (TRIPS).
Literature review data extraction, synthesis, and analysis
We undertook a two-phase screening approach to first, control for duplicates and remove studies that did not meet the inclusion criteria (based on abstracts screening), and second, to review full-text articles and make final selection of studies based on the inclusion criteria. For each article, we extracted key information related to the country where MEAs were implemented, the main category of MEAs presented (financial scheme, performance-based agreement, or hybrid), the type of instrument used (e.g. discounts, utilization/price cap, and coverage with evidence development), as well as the disease and medicines involved under such agreement. Two reviewers extracted information on the factors potentially facilitating or hindering the implementation of MEAs. Schemes that did not fit into the Kanavos and Ferrario et.al. taxonomy and other models that combined two or more types of a financial scheme and a performance-based agreement were categorized as “hybrid”.
Gray literature sources
A review of the gray literature from targeted sources recently involved in access solutions for innovative medicines for NCDs was simultaneously carried out by two independent reviewers to complement the findings. The review entailed gathering relevant information from the sources listed in Box 1.
List of targeted sources reviewed on the gray literature.

Results
Number of MEAs publications and trends
The literature search considered 330 publications, of which 296 records were retrieved through database searching, and 34 were added publications identified through other sources (Figure 2). After controlling for duplicates, 253 records remained for consideration. After this stage, 186 records were excluded after title and abstract screening, and 67 full-text articles were assessed for eligibility. A total of 34 publications were ultimately considered for the qualitative analysis: 18 primary publications (using original data), 12 literature reviews, and 4 editorial or author’s commentaries. Additional Information in the Supplemental material provides the list of 34 publications considered for the qualitative analysis.

Diagram of reporting items of the literature search according to PRISMA standards.
Of the 34 selected publications, 13 (38%) were published between 2007 and 2012 and 21 (62%) between 2013 and 2017, demonstrating a 38% increase over the last 5 years (Figure 3).

Publication trends on MEAs.
Geographic distribution and diversity of MEAs
We identified 285 MEAs from the literature review, with more than 95% of these being implemented in HICs. A total of 34 countries were implementing one or more type of MEAs—23 European countries, 6 Asian countries, 2 countries in North America, 2 in Oceania, and 1 in Africa (Figure 4). Beyond Bulgaria, China, India, Serbia, and Thailand, the level of documentation on MEAs in LMICs was very limited. There was no record of MEAs for NCDs in Latin America or in Africa retrieved through our search, apart from one financial scheme in Rwanda for human papillomavirus (HPV) vaccines which falls into a mixed category of a health technology aimed at preventing a chronic infectious disease that may lead to a NCD. Identified MEAs in LMICs can be found in Table 1.8,21–24

Implementation of MEAs by region.
Examples of MEAs implemented in LMICs.

Financial schemes were slightly more prominent than outcome-based schemes at 50.2% versus 44.9%, respectively. The remaining 4.9% of the agreements covered hybrid schemes which included elements of both financial schemes and performance-based schemes (e.g. price and volume + patient eligibility and registry requirements). Generally, financial-based schemes were more prominent in Europe, Asia, and Oceania, 25 while performance-based agreements were more frequent in North America.
Among financial schemes, price/volume, and discounts were the most frequently used instruments at 18.9% and 17.5%, respectively. Utilization and price capping schemes accounted for 7.7% of the financial schemes, and 56% of the remaining financial schemes were either not specified in the literature or had a mix of financing components. Among performance-based agreements, coverage with evidence development was the most frequent type of scheme (39.1%), followed by outcome guarantees (32.8%) and patient eligibility based on patient registry (5.5%). The remaining performance-based agreements (22.7%) were either not specified or contained more than one performance-based component (Figure 5).

Participation (%) of sub-scheme by type of agreement (financial and performance based).
Most of the financial or outcome-based agreements retrieved covered a wide range of NCDs, predominantly different types of cancer (e.g. breast cancer, chronic myelogenous leukemia, colon, and rectal cancer). Other NCDs covered included multiple sclerosis, osteoporosis, diabetes, CVD, and Alzheimer’s.
Factors enabling or hindering the implementation of MEAs
Through our study, we also identified a set of factors derived from the experience of countries implementing MEAs that could facilitate or hinder their use in LMICs.
Overall, MEAs seemed more likely to be successful if they were simple, supported by robust clinical data, and easily monitored. 26 Other factors that seemed to facilitate the successful implementation and effective design of MEAs included the existence of legal requirements supporting patient enrollment; clear contract terms and definitions; evaluations of performance metrics and goals linked to specific pricing and reimbursement decisions; and clear, measureable and realistic objective outcomes.
Despite some of the advantages and opportunities of implementing MEAs, their implementation is often characterized as a labor and resource-intensive activity. Well-implemented agreements required service provision that can meet the operational, administrative, and financial requirements of the schemes and appropriate data systems capable of monitoring them. A general trend over the past decades has been toward MEAs with less administrative burden.9,27
Aspects related to the quality of data and evidence were also reported in the literature; the need for data on pricing and real-world evidence on clinical outcomes should be considered when pursuing MEAs. For example, the evidence of positive performance on clinical outcomes and the availability of patient information in administrative claims databases seem to favored MEAs implementation. Findings suggest that limited evidence on the effectiveness of new treatments may discourage payers from pursuing agreements that could potentially benefit patients with diseases that lack strong “evidence based” outcome data.
Moreover, issues about uncertainty regarding data (availability and quality), financial impact, or other patient and institutional factors that may affect patient outcomes could influence the perception of MEAs as potential policy options for improving access. The published data also indicated that the presence of quality administrative and information systems was key to tracking the implementation of coverage with evidence schemes. The use of health technology assessment (HTA) or comparative safety and effectiveness reports could enable the implementation of MEAs by reducing the uncertainty of clinical benefits.
Some types of MEAs appeared to be more culturally accepted than others. For example, performance-based agreements (preferred schemes in the United States) are often used for innovative and expensive new medicines, for treating conditions with a high clinical unmet need (i.e. orphan diseases). In countries where there is no enforcement of price control of new medicines like it is the case of the U.S, performance-based or coverage with evidence development may be more feasible than financial discounts.26,28 Level of trust among payers and pharmaceutical companies and willingness to dialogue were also mentioned as factors that could facilitate or hinder the implementation of MEAs.
Finally, issues about transparency of pricing, confidentiality of agreements, or competition regulation, as well as information on rebates and discounts, were widely identified throughout the revision of the literature as important contextual factors that may prevent MEAs implementation.8,9,23 For example, one of the studies noted that the lack of transparency with rebates and discounts precluded physician associates from accurately monitoring drug budgets. Other studies mentioned several issues regarding the confidentiality of agreements, including the difficulty to analyze trends and impact of programs and use them to inform future decisions; duplication of data collection efforts was also a concern.
Gray literature
The targeted gray literature search retrieved few relevant sources. For example, the World Health Organization (WHO) toolbox on NCDs contains a range of policy statements and technical documents, including those related to pricing, financing, and intellectual property issues. The independent tools on the Medicines Law and Policy website focused on licensing agreements and how to navigate TRIPS flexibilities. Likewise, the World Health Assembly (WHA) meetings (2011–2017) discussed five main mechanisms to improve access to medicines: TRIPS flexibilities and the Doha agreement, voluntary pooled funding and procurement, sin taxes (alcohol and tobacco) to boost government revenue, public–private partnerships (shared risk), and grant schemes. All of the non-governmental organizations (NGOs) that we identified, except for one, focused on patent opposition to lower drug prices and were therefore excluded from the narrative report since this was not the main purpose of our search.
The work presented by the Organisation for Economic Co-operation and Development (OECD) 29 and the European Commission reports30,31 essentially provided reviews of peer-reviewed publications that were included in the review of the literature, and no additional distilling of information from these sources was necessary. The findings reflect initiatives undertaken by organizations whose focus is to increase access to medicines and health technologies for specific NCDs.
A total of 36 access initiatives for NCDs were identified and examined from the International Federation of Pharmaceutical Manufacturers & Associations (IFPMAs) Health Partnership Directory and Access Accelerated Initiative (AAI), none of which were considered within the narrowly defined categories of MEAs. The NCDs that the access programs are targeting include chronic conditions such as epilepsy and hypertension, cancers, and some conditions that go beyond NCDs (i.e. infectious diseases, including vaccine-preventable conditions; neglected tropical diseases; and maternal, newborn, and child health concerns, including contraceptives and pediatric medicines for a broad range of conditions).11,12
Of the access initiatives reviewed from these sources, 35 programs included differential pricing—10 focused exclusively on it, and 25 paired differential pricing with supplemental efforts such as capacity-building activities, general financial support, product donations, and technology transfer, all of which we classified as “other”. The geographical reach of access programs initiated by pharmaceutical companies suggests the critical “bottom-up” role of the industry in improving access to medicines (Figure 6).

Geographical reach of medicines access programs initiated by pharmaceutical companies.
In addition to the reach of access programs for NCDs, we identified the Drugs for Neglected Diseases initiative (DNDi)’s which aimed to ensure consistent and affordable access to medicines that treat neglected diseases. This comprehensive approach for accessing to medicines was of interest because it includes (1) entering into agreements with manufacturers to secure long-term treatment and/or active pharmaceutical ingredient production; (2) accelerating regulatory approvals and/or updating WHO and in-country guidelines by working closely with stakeholders (e.g. WHO, National Regulatory Authorities, and Ministries of Health); (3) getting accurate demand forecasts to ensure realistic supply planning by working closely with stakeholders (manufacturing and program); (4) in partnership with manufacturers and users, facilitating the extension of medicine shelf lives and improving the product profile and packaging so that it is relevant for targeted health systems; and (5) simplifying procurement and supply chain in cooperation with national control programs, WHO, distributors, and other partners; all of which seemed also potentially applicable for NCDs. 19
Discussion
Increasing documentation of MEAs over the last decade suggests growing interest from policy-makers and researchers. Most of the retrieved publications report the experience of HICs, with European countries being the most prominent settings. As such, the majority of schemes reported in HICs covered treatments for NCDs, predominantly different types of cancer.
Although the level of documentation on MEAs appears to be very limited in LMICs, few financing schemes were reported in these settings. One example was the financial scheme between Merck and the Rwandan Ministry of Health that guaranteed free Gardasil® HPV vaccine for 3 years, with future doses at concessional pricing. 22 Another example was the establishment of the Glivec International Patient Assistance Program, through which over 99% of Indian patients received imatinib (for chronic myeloid leukemia) at no cost. 21
Performance-based agreements have primarily been implemented in HICs, as they require more capacities for data collection and monitoring and evaluation. These schemes were most used for innovative and expensive medicines that require more evidence. Payers in many HICs seemed willing to pay high prices for innovative medicines if they are able to provide ‘good value’ for money. However, for some new products, the evidence remains limited as to how these medicines will perform in real-world circumstances. 32 In addition, these schemes were most effective for products that have simpler methods for measuring clinical effects and well-defined relevant outcomes.9,26 Evidence from the literature review showed only one LMIC (Bulgaria), used this type of agreement although detailed information about the scheme was not provided. 8
MEAs have the advantage of including a range of different types of instruments to address different contextual needs; financial schemes can improve budget uncertainties for innovative medicines, and performance-based mechanisms can assist with the collection of information on real-world effectiveness. In addition, they could potentially help mitigate the impact of increasing costs and create incentives for all involved parties (patients, payer, and manufacturer) to work in more collaborative environments. For instance, for patients, they represent an opportunity of earlier access to innovative therapies; for payers, a tool for bargaining and better allocating finite resources; and for producers, a means to forecast the size of the market and potentially reduce their investment on advertising or marketing to reach their population targets.
However, as with any other intervention, the use of MEAs also comes with caveats. For example, key stakeholders may attempt to “game the system” when they are used in countries that utilize HTA for informing decisions about coverage, and in such cases, the cost-effectiveness thresholds become the targets to bit in order to get reimbursed, leaving limited space for seeking higher levels of efficiency. 7 In such cases, pharmaceutical companies may have an incentive to offer high initial prices during their first submission for drug approval knowing that they will be refused, as this will be revised in future rounds of confidential negotiations. Equally, payers are more likely to turn down an initial price offered by pharmaceutical companies, aware that a lower price will be offered and negotiated through a MEA. This process can result in significant transaction costs for both payers and pharmaceutical companies.7,33
There is a perception that while tiered pricing may lead to equitable prices, there is no guarantee that tiered prices may be sustainable in the long run, since the approach may have anti-competitive effects; if the price for a drug is too low, it may discourage market entry of by potential competitors. 34 Although consumers may benefit in the short term from tiered prices set below production costs, the resultant lack of competition and absence of dynamic price reductions could result in consumers paying higher prices in the long term. 34
MEAs are also perceived as quick fixes to barriers to access, and there is criticism about fairness for those patients with diseases that are not on the policy agenda as funding priorities. Price capping, for example, may undercut prices but would require to implement resource- and labor-intensive mechanisms to monitor compliance to these levels of capping, nor would be the case shall local competition arises. Taking into consideration the supply and demand dynamics within the market, if more patients require longer courses of treatment (higher demand), payers may challenge pre-agreed prices due to production cost increase, since there would be higher volumes than expected. Finally, the lack of compliance with clinical protocols and the inability to monitor and evaluate prescribed dosages once the patients are granted access to treatment in many LMIC where there is no endorsement of evidence-informed clinical practice guidelines or protocols, may lead to supplier-induced demand and higher drug expenditures, hence jeopardizing trust among payers and manufacturers.
Given the limited use of MEAs in LMICs, there is an opportunity to extend access to life-saving medicines and advance progress toward UHC and achieving SDG.3 by disseminating our results. The previous experience of many LMICs in granting access to drugs for infectious diseases may also pave the road for incrementally embarking on MEAs for NCDs. However, MEAs should not been seen as a “stand-alone” approach to improve access to medicines. MEAs might be used in combination with other price strategies (e.g. donation programs for certain population groups) as well as with health delivery and health sector strategies. There is a potential greater opportunity to use MEAs in LMICs, particularly in those contexts with political stability, commitment to UHC, an enabling legal frameworks, HTA initiatives, and less-fragmented sources of funding. Based on experience from HICs, MEAs are particularly suitable where there is a more centralized system (i.e. one predominant purchaser), which is able to negotiate and develop schemes in line with local health care needs, or in countries where resources are pooled (domestically or by donors) in order to better negotiate prices and aggregate demand. However, it is worth noticing that the feasibility of implementing financial schemes versus performance-based agreements in LMICs will depend not only on local capacities to monitor and evaluate population outcomes and prices but also cultural preferences, attitudes toward transparency, and regulation in place.
A significant number of international NGOs and global advocacy groups are working on raising awareness of TRIPS flexibilities to promote access to on-patent medicines for NCDs as a short-term access solution, however these are fully embedded in political complexities. Nonetheless, the level of involvement in the use of MEAs by international organizations and donors seems to be very limited. This may represent an opportunity for these institutions and manufacturers to seize global attention for using MEAs as an alternative to access challenges. Besides potential solutions to sustainable access to on-patent medicines for NCDs, MEAs may also bring a means for health systems strengthening in the long run. Further research to understand stakeholders’ perceptions/positions on MEAs as access solutions for NCDs in LMICs may help inform a roadmap for their future implementation.
More comprehensive approaches used in the past, such as those for neglected infectious diseases, are worth considering for NCDs. Since the level of awareness of and experience with MEAs in LMICs seems limited and the overall geographical reach of medicines access programs initiated by pharmaceutical companies is quasi-global, the importance of market-driven approaches ought be considered; reshaping the balance of top-down versus bottom-up policy initiatives in LMICs could serve the case of access for on-patent medicines for NCDs.
Limitations
Given the exploratory nature of this narrative review, only articles containing original data and literature reviews were included, this could result in an overestimation of the number of existing agreements due to double counting. In addition, when comparing the number of countries implementing MEAs identified through our review and the geographical reach of medicines access programs initiated by pharmaceutical companies, the number of countries does not necessarily overlap. This could be explained by the difference of reporting from peer-reviewed data and the gray literature publicly available in pharmaceutical companies’ websites. Nevertheless, our findings are consistent with other published data.7,9
Confidentiality is at the core of MEA agreements, and thus the details of negotiations and terms are not made public. Since our study relied only on publicly available and published data, it is likely that some schemes remain unpublished or are not in the public domain and were therefore not captured in this analysis. The confidentiality aspect of the negotiations may also prevent learning across countries about what works well and what does not, in regards to the implementation of MEAs, and makes it difficult to assess with certainty what populations have gained access to medicines through MEA initiatives and validate the benefits of the agreements.7,8,23
Conclusions
Our preliminary findings indicate that interest in MEAs has increased exponentially in recent years; however, there is little information on the use of MEAs as policy options for granting access to on-patent medicines for NCDs in LMICs.
The terms surrounding MEAs have been gaining popularity in LMICs only within the past decade, and some countries may still be using different taxonomies to describe such agreements. Therefore, such studies may not have been included in this review. Finally, the findings suggest that there have been slightly more implemented financial schemes than performance-based schemes which seemed potentially less complex to implement; however, due to the confidential nature of many agreements, final proportions of one versus the other, the emergence of hybrid combinations or trend shifts may still be unknown.
As any other policy option, the implementation and use of MEAs come with advantages and challenges. Since there is limited evidence on the use of MEAs in LMICs, the identified cases of implementation in HICs may serve to inform the interest on MEAs in resource limited settings. Therefore, further research in this field especially in the context of LMICs may be of value for the global community as all countries are embarking into fairer and sustainable UHC.
Supplemental Material
Supplemental_Material – Supplemental material for Sharing knowledge for policy action in low- and middle-income countries: A literature review of managed entry agreements
Supplemental material, Supplemental_Material for Sharing knowledge for policy action in low- and middle-income countries: A literature review of managed entry agreements by Hector E. Castro, Tanya Malpica-Llanos, Ruth Musila, Niranjan Konduri, Ana Amaris, Jennifer Sullivan and Colin Gilmartin in Medicine Access @ Point of Care
Footnotes
Acknowledgements
The findings, opinions, and conclusions in this article are those of the authors alone. H.C. led the design and conceptualization of work presented on the manuscript, critically revised the manuscript, and was a key contributor in writing the manuscript. T.M.-L. led the literature review, analyzed the findings, and wrote the first draft of the manuscript. All authors contributed at different moments during the data collection process and read and approved the final version.
Availability of data and materials
Data supporting the manuscript findings are available as additional files. Additional information on data generated is available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was made possible by the financial support of the World Bank through the initiative for Strengthening Global Knowledge on Access Solutions for Innovative Medicines.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
