Abstract
Neuronal activity can modulate myelination throughout life, influencing circuit function and behaviour. Pathological changes in neuronal activity have been associated with myelin remodelling in a rat model of idiopathic absence seizures, raising the question of whether this represents a common mechanism underlying absence epilepsy. Here we examined oligodendrocyte populations and myelin architecture in a rat model of GRIN2B neurodevelopmental disorder, a rare and severe condition that is frequently accompanied by absence seizures. Using histology and 3D imaging, we report that despite robust seizure-like activity in this model, there was no evidence of changes in oligodendrocyte lineage cell populations, overall myelin content, or node of Ranvier organization. These results contrast with prior reports of seizure-associated myelin modulation, indicating that seizure-induced myelin plasticity may not be a uniform feature across all models of absence seizures. Our findings indicate that such adaptations may depend on additional factors including seizure burden and timing, highlighting the need for further analysis to determine when and how myelin plasticity contributes to absence epilepsy.
Introduction
Neurodevelopmental disorders comprise a complex spectrum of conditions and are often characterized by altered neuronal connectivity and impaired cognitive functions (Gidziela et al., 2023). Among the various genes implicated in these disorders, GRIN2B encodes the GluN2B subunit of the N-methyl-D-aspartate receptor (NMDAR), which is highly expressed both early in development and into adulthood (Platzer et al., 2017). Pathogenic mutations in GRIN2B cause GRIN2B encephalopathy, a rare but severe condition with a range of neurological symptoms, including intellectual disability, epilepsy, autism, sleep and movement disorders (Platzer et al., 2017). De novo GRIN2B mutations are considered a leading cause of monogenic epileptic encephalopathies, accounting for more than half of GRIN2B neurodevelopmental disorder cases and presenting with a variety of seizure types (McTague et al., 2016; Platzer et al., 2017).
The precise molecular and cellular mechanisms underlying seizure onset are currently unclear. Pathogenic GRIN2B variants can result in either loss or gain of function mutations which have been shown to disrupt NMDA subunit trafficking to the plasma membrane and alter receptor physiology (Amin et al., 2018; Kellner et al., 2021; Santos-Gomez et al., 2021; Sceniak et al., 2019). Given its expression during both prenatal and postnatal development, pathogenic mutations have been shown to dysregulate key neurodevelopmental processes, such as neuronal differentiation, dendritic morphology and synaptic plasticity, which can affect neuronal activity and network synchrony (Bahry et al., 2021; Bell et al., 2018; Kellner et al., 2021; Platzer et al., 2017; Sabo et al., 2022; Sceniak et al., 2019; Shin et al., 2020).
Neuronal activity also modulates myelination, the process of axonal ensheathment by myelin, an insulating membrane produced by oligodendrocytes in the central nervous system that enables fast and energy-efficient transmission of action potentials (Huxley and Stampfli, 1949; Nave and Werner, 2014). Neuronal activity and synaptic vesicle release promote oligodendrocyte progenitor cell (OPC) proliferation, oligodendrocyte differentiation and myelin sheath growth (Barres and Raff, 1993; Demerens et al., 1996; Hines et al., 2015; Mensch et al., 2015; Wake et al., 2011). Oligodendrocyte lineage cells express diverse neurotransmitter receptors and ion channels that enable them to sense and respond to neuronal activity by remodelling myelin sheathes (Hamilton et al., 2017; Karadottir and Attwell, 2007; Lundgaard et al., 2013). Notably, optogenetic or chemogenetic stimulation of cortical projection neurons increased myelin sheath thickness in adult mice (Geraghty et al., 2019; Gibson et al., 2014; Mitew et al., 2018). This activity-dependent myelin plasticity is predicted to tune inter-regional oscillations and synchrony (Kato et al., 2020; Munyeshyaka and Fields, 2022; Noori et al., 2020; Pajevic et al., 2014; Pajevic et al., 2023) and to influence cognition (Bonetto et al., 2021).
In epilepsies, abnormalities in neuronal firing and synchrony co-occur with changes in white matter. Imaging studies in people with focal and generalized epilepsies including absence seizures report changes in white matter integrity and volume (increases or decreases), while histological evaluations reveal altered numbers of myelinated axons and changes in oligodendrocyte lineage cell densities (Deleo et al., 2018; Drenthen et al., 2019; Goldsberry et al., 2011; Sandoval Karamian et al., 2020; Yang et al., 2012). Recently, Knowles et al. (2022) identified seizure-driven hypermyelination in WAG/Rij rats and Scn8a+/mut mice. Specifically, after seizure-onset OPCs, mature oligodendrocytes and myelin thickness increased in the corpus callosum – a myelinated commissural white matter tract mediating interhemispheric cortico-cortical communication. Interestingly, pharmacological blockade of seizures in WAG/Rij rats prevented this myelin increase, while pharmacological or genetic disruption of myelin plasticity in Scn8a+/mut mice reduced the seizure burden linking seizure activity with myelin maladaptation (Knowles et al., 2022). However, whether a similar mechanism is involved in other models of absence seizures remains to be determined.
In a new rat model of GRIN2B-related neurodevelopmental disorder, Grin2b heterozygous knockout rats exhibited more frequent and longer generalized, 5–10 Hz spike wave discharges (SWDs), the electrographical correlate of absence seizures, than wildtype littermates (Hristova et al., 2025). Given the potential link between aberrant neuronal firing and maladaptive myelination, we predicted that Grin2b-related absence seizures would induce excessive oligodendrogenesis and abnormal myelination within the seizure network.
To investigate potential changes in myelin and in oligodendrocyte populations, we focused on the body of the corpus callosum at the level of the somatosensory cortex, an area previously shown to exhibit SWDs in the somatosensory cortex absence seizure models (Crunelli et al., 2020; Meeren et al., 2002; Polack et al., 2007; Studer et al., 2019). We report that seizure-like activity in this area does not result in changes in oligodendrocyte populations or myelination in a rat model of Grin2b haploinsufficiency. Our findings underscore the complexity of neurodevelopmental disorders and highlight the necessity of further investigation to determine how Grin2b haploinsufficiency may alter neuronal and circuit function without affecting oligodendrocytes or myelin properties in this model.
Materials and methods
Animals
The rats were housed and used in accordance with the guidelines established by the Animal Care (Scientific Procedures) Act 1986 and under the authority of Home Office Licences in the UK. Long–Evans Grin2b heterozygous knockout rats (hereafter referred to as Grin2b+/− and Grin2b+/+ refers to wildtype rats) were generated by the Medical College of Wisconsin gene Editing Rat Resource Centre with support from the Simons Foundation Autism Research Initiative (LE-Grin2bem1Mcwi, RRID: RGD_14394515). Rats were bred in-house and kept on a 12 h/12 h light/dark cycle with ad libitum access to food and water. Following weaning on postnatal day 21, rats were group housed in mixed-genotype cages of 2–6 rats per cage. Both male and female rats were used for this analysis.
Immunohistochemistry
Grin2b+/+ and Grin2b+/− rats that had not undergone prior electrographical recordings were perfused with 4% paraformaldehyde (PFA, 252549, Sigma-Aldrich) and the brain tissue was harvested and post-fixed with 4% PFA overnight at 4oC before transferring to 1xPBS solution. The brain was sectioned using a Leica VT1000S vibratome. Coronal vibratome sections measuring 100 μm in thickness were briefly washed in 1xPBS before blocking with 10% normal horse serum (26050088, Thermo Scientific), 0.3% Triton-X (X100, Sigma-Aldrich) in 1xPBS for 2 hours at room temperature. Primary antibodies were diluted in blocking solution and sections were incubated for 48 hours at 4°C on an orbital shaker. After the primary antibody incubation, sections were washed in 1xPBS (3 × 1 h each), incubated with Alexa Fluor secondary antibodies (Thermo Fischer Scientific, 1:1000) for further 16 hours and counterstained with Hoechst 33342 solution (62249, Thermo Fischer Scientific) for nuclear visualization. All slides were mounted using Fluoromount-G® mounting medium (0100-01, SouthernBiotech).
The primary antibodies used were as follows: against MBP (rat monoclonal, MCA409S, BioRad, 1:300), OLIG2 (rabbit polyclonal, AB9610, Sigma-Aldrich, 1:100), PDGFRα (goat polyclonal, AF1062-SP, Novus Biologicals, 1:200), CC1 (mouse monoclonal, OP80| Anti-APC (Ab-7) Mouse mAb (CC1), OP80, EMD Millipore 1:300) and CASPR (rabbit polyclonal, ab34151, Abcam, 1:500).
Image analysis
For cell density quantification, three z-stacks of 20-μm thickness (63x objective, 211.30 × 211.30 μm each; 2048 × 2048, step size: 0.5 μm) were obtained from the body of the corpus callosum (CC) at the level of the somatosensory cortex using the Leica THUNDER Imager with instant computational clearing and adaptive deconvolution. The total number of cells was quantified automatically using IMARIS 3D imaging software (RRID: SCR_007370, spots module) and represented either as cells/mm3 where the number of each population was divided by the total z-stack volume, or as the percentage of a specific cell population over the total number of cells (labelled with Hoechst) or oligodendrocyte lineage cells in the z-stack. The density estimates are not extrapolated to the total cell numbers of the corpus callosum. From each rat, three sections between Bregma −3.30 mm to −3.60 mm (Paxinos and Watson, 1998) were analysed, and the numbers were averaged for each animal. ROIs, stack dimensions, thresholds, and sampling locations were matched across animals.
We assessed the callosal myelination by measuring the total volume of the MBP-positive and CASPR-positive signals in the CC. Three confocal z-stacks of 5 μm thickness (63x objective, 184.70 × 184.70 μm each; pixel size: 180.38 × 180.38 nm, step size: 0.5 μm) and three sections per rat were obtained using the Leica TCS SP8 confocal microscope. The surface rendering module of the IMARIS 3D imaging software with absolute intensity thresholding was used to determine the respective signal volumes, and values were normalized to the total volume of the z-stack.
For node of Ranvier density and length analyses, three confocal z-stacks of 5 μm thickness (63x objective, 184.70 × 184.70 μm each; pixel size 180.38 × 180.38 nm, step size: 0.5 μm) were obtained from the same area, and three sections per rat were analysed. For the quantification of the nodal densities, four regions of interest with an area of 50 × 50μm2 and 5 μm depth were assessed from each individual z-stack. Only nodes flanked with two CASPR-positive paranodes within a single 0.5-μm optical slice were considered for the analysis. The densities were manually quantified using Fiji image analysis software (Fiji, RRID: SCR_002285, cell counter plugin), averaged per rat and represented as number of nodes/mm3. For the quantification of the nodal length, maximum intensity projections of z-stacks were used (Fiji), and overlapping CASPR signals were avoided. Individual lines spanning both CASPR-positive paranodes and with adjusted width to the thickness of the staining were drawn to plot CASPR intensity profiles. 120-150 nodes per z-stack and three sections per rat were quantified. The length of each node was automatically calculated using a previously published MATLAB (The MathWorks, Inc.) script which measures the distance between the half maximum intensity points for each paranode (Arancibia-Carcamo et al., 2017; Smith et al., 2024). The values were averaged per rat.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 10 (RRID: SCR_002798), JASP (RRID: SCR_015823- Version 0.95.3) and R Project for Statistical Computing (RRID: SCR_001905). Data were tested for normal distribution using the D’Agostino–Pearson test or the Shapiro–Wilk test and depending on data distribution, comparisons between genotypes were assessed using an unpaired t-test with or without Welch’s correction or a Mann–Whitney test. A difference was considered statistically significant if p < 0.05.
When a t-test for our primary outcomes was non-significant (p > 0.05) and the data were normally distributed, we additionally performed equivalence testing using the Two One-Sided Tests (TOST) procedure to examine equivalence between groups. Equivalence bounds for TOST were prespecified as −30% (lower bound) and +30% (upper bound) of the mean value of Grin2b+/+ rats, based on biologically meaningful differences reported by (Knowles et al., 2022). For nodal length, the equivalence bounds were prespecified as −20% (lower bound) and +20% (upper bound) of the Grin2b+/+ mean, based on (Miyata et al., 2016).
Power analysis and minimal detectable effect (MDE) quantification were conducted using G*Power (RRID: SCR_013726) or the R function power.t.test as appropriate. For each primary outcome, we report the absolute MDE, the MDE as percentage of the Grin2b+/+ mean and the observed percent change between genotypes. When the observed percent change is smaller than the MDE, non-significant results (p > 0.05) cannot exclude true effects that are smaller than the MDE.
For all primary outcomes, the sexes were initially pooled, as no effect of sex had been previously documented (Knowles et al., 2022). However, because we did not observe significant genotype differences, we additionally performed two-way ANOVAs to assess the main effects of genotype, sex and their interaction, to account for potential sex-specific effects.
Data are shown as mean ± SD in which the biological replicate (n) is the rat, depicted as individual points on the graphs. The experimenter was blind to genotype and sex throughout the duration of the experiment and until the end of the analysis. Details of statistical tests used are provided in Tables 1 and 2, Table S1, and in Supplemental Information File. Illustrations were created in BioRender.com.
Descriptive statistics of all t-test comparisons performed in this study.

Descriptive table of two-way ANOVA statistical analyses in this study assessing the effects of sex end genotype.

Results
Oligodendrocyte lineage cell densities are not altered in Grin2b+/− rats
We have previously recorded electroencephalograms and local field potentials from Grin2b+/− and Grin2b+/+ rats (Hristova et al., 2025). We found higher frequency and duration of SWDs in 90% of Grin2b+/− animals compared to wildtype littermates at approximatively 20 weeks of age (Hristova et al., 2025). To investigate changes associated with seizure activity, we focused on the body of the corpus callosum (CC) at the level of the somatosensory cortex, as one of the myelinated tracts involved in seizure propagation and generalization across cortices (Chen et al., 2020; Knowles et al., 2022; Meeren et al., 2004; Musgrave and Gloor, 1980; Peng and Hsin, 2017) (Figure 1(a)), and measured the proportion and density of oligodendrocyte lineage cells (Figure 1(b)), 7 weeks later. We reasoned that this time interval would allow for both cellular and structural changes in myelination associated with increased SWDs (Bacmeister et al., 2022; Hughes et al., 2018; McKenzie et al., 2014; Pan et al., 2020; Xiao et al., 2016).
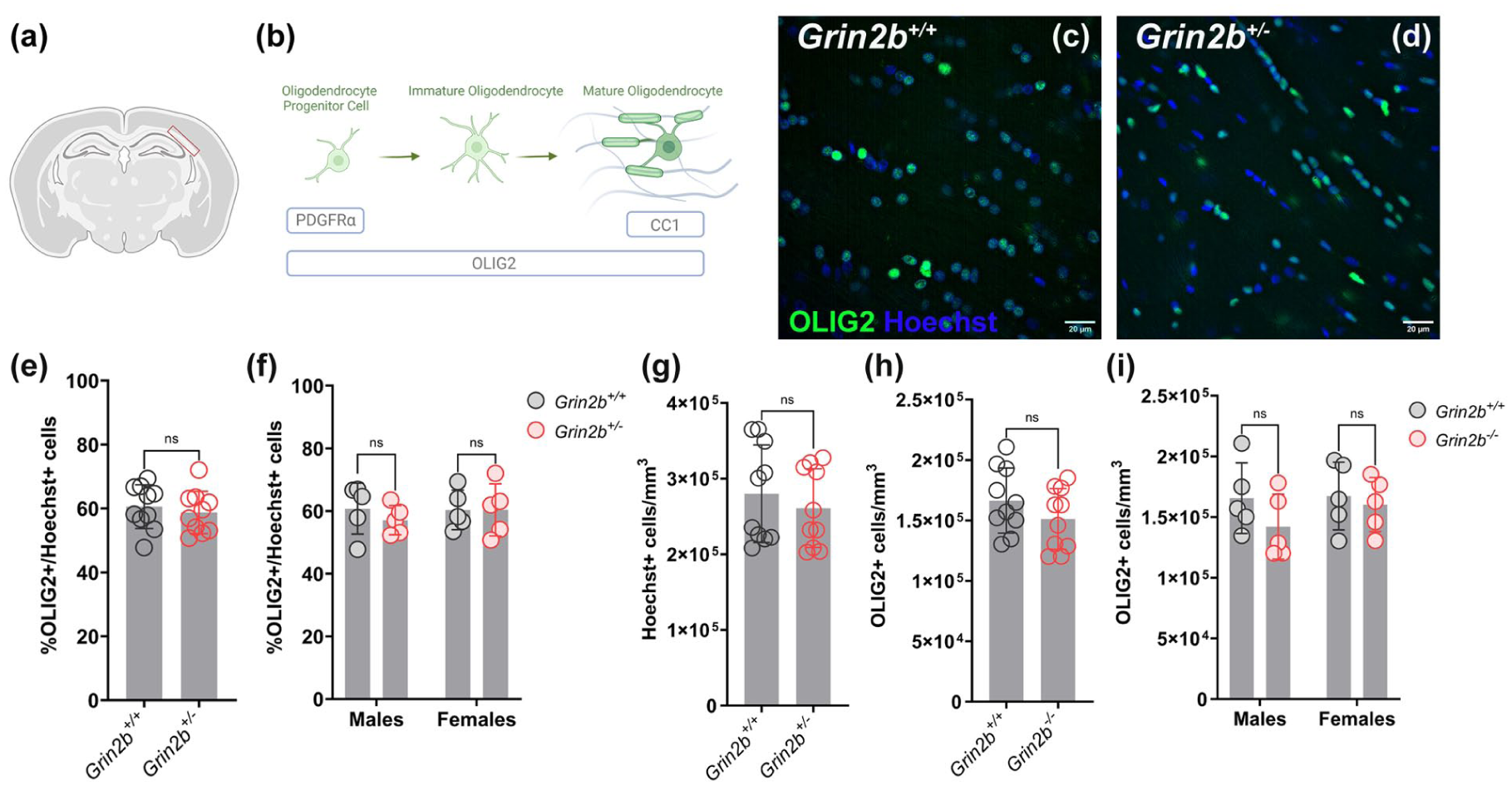
No difference in oligodendrocyte lineage cell populations in adult Grin2b+/− rats. (a) Scheme of a coronal rat section indicating the CC area used for all analyses in this study. (b) Diagram of oligodendrocyte lineage cell differentiation with respective molecular markers indicative for each oligodendrocyte stage analysed. (c) and (d) Representative images of adult Grin2b+/+ and Grin2b+/− CC sections immunostained for OLIG2 (green) and counterstained cell nuclei with Hoechst (blue). (e) Percent of OLIG2 expressing oligodendrocyte lineage cells relative to the total cell number across genotypes. (f) Percent of OLIG2 expressing oligodendrocyte lineage cells relative to the total cell number across genotypes in male and female rats (g) Total cell density in the CC across genotypes. (h) Density of OLIG2 expressing cells in the CC across genotypes. (i) Density of OLIG2 expressing cells in the CC across genotypes in male and female rats.
To assess changes in oligodendrocyte lineage cells, we performed immunohistochemistry for the transcription factor OLIG2 (Figures 1(b)-(d)). We first quantified the percentage of oligodendrocyte lineage cells relative to the total cell population marked by Hoechst in the regions of interest within the CC and found that it was not significantly different between genotypes (Figure 1(e), Table 1), but was equivalent within ±30% (Supplemental File), suggesting that there is no meaningful difference between genotypes. We next asked whether the two sexes were affected differently and found no significant differences (Figure 1(f), Table 2, Table S1). We also compared the total cell density (Hoechst/mm3) between genotypes to detect potential changes in callosal volume within our regions of interest and found no differences (Figure 1(g), Table 1). Equivalence analysis showed that the total cell density of Grin2b+/− and wildtype rats was equivalent within ±30% (Supplemental File), suggesting no meaningful difference between genotypes. Similarly, the density of oligodendrocyte lineage cells was not statistically different and equivalent within ±30% (Supplemental Figure) across Grin2b+/− and wildtype and comparable between male and female rats (Figure 1(i), Table 2, Table S1). These results suggest that despite the presence of absence seizure-like neuronal activity in Grin2b+/− rats, we observed no overt changes in the proportion or density of oligodendrocyte lineage cells in the CC of Grin2b+/+ and Grin2b+/− rats.
The densities of oligodendrocyte progenitor cells and mature oligodendrocytes are unaffected in Grin2b+/− rats
The fact that the overall population of oligodendrocyte lineage cells was unaffected does not rule out the possibility of specific changes occurring in oligodendrocyte progenitor cells (OPCs) and/or mature oligodendrocytes that could account for the preserved total number of oligodendrocyte lineage cells (Figure 1(b)). To detect changes in oligodendrocyte progenitor cells, we performed immunohistochemistry for the cell surface marker platelet-derived growth factor receptor alpha (PDGFRα; Figure 2(a) and (b)). We quantified both the percentage of OPCs over the population of Hoechst + cells in the CC and their density per mm3 (Figure 1(a)) between genotypes and found that they were not statistically different (Figure 2(c) and (e), Table 1) although they were equivalent within ±30% (Supplemental File), suggesting no meaningful difference. Further analysis indicated no effect of sex, genotype, or their interaction on the percentage (Figure 2(d), Table 2, Table S1) or density (Figure 2(f), Table 2, Table S1) of OPCs in the CC. These results suggest that despite the presence of absence seizure-like neuronal activity in Grin2b+/− rats, we observed no changes in the proportion or density of OPCs in the CC of Grin2b+/+ and Grin2b+/− rats.

No difference in oligodendrocyte progenitor cells in adult Grin2b+/− rats. (a)–(b) Representative images of adult Grin2b+/+ and Grin2b+/− CC sections immunostained for PDGFRα (green) and counterstained cell nuclei with Hoechst (blue). (c) Percent of PDGFRα expressing OPCs relative to the total cell number across genotypes. (d) Percent of PDGFRα expressing OPCs relative to the total cell number across genotypes in male and female rats. (e) Density of PDGFRα expressing cells in the CC across genotypes. (f) Density of PDGFRα expressing cells in the CC relative to the total cell number across genotypes in male and female rats.
To determine potential changes associated with SWDs in mature oligodendrocytes, we performed immunohistochemistry in the CC of adult Grin2b+/+ and Grin2b+/− rats using the mature oligodendrocyte marker APC (CC1), which labels both pre-myelinating and myelinating oligodendrocytes, in combination with the transcription factor OLIG2 (Figure 3(a) and (b)). We quantified the percentage of CC1+ mature oligodendrocytes within the total population of OLIG2 expressing oligodendrocyte lineage cells across genotypes and found that they were not statistically different (Figure 3(c), Table 1) despite the groups being equivalent within ±30% (Supplemental File), suggesting no meaningful difference. Further analysis indicated no effect of sex, genotype or their interaction on the percentage of mature oligodendrocytes between the groups (Figure 3(d), Table 2, Table S1).

No difference in mature oligodendrocytes in adult Grin2b+/− rats. (a)–(b) Representative images of adult Grin2b+/+ and Grin2b+/− CC sections immunostained for CC1 (red), OLIG2 (green) and counterstained with Hoechst (blue). (c) Percent of CC1/OLIG2 expressing mature oligodendrocytes relative to the total OLIG2 expressing oligodendrocyte lineage cells across genotypes. (d) Percent of CC1/OLIG2 expressing mature oligodendrocytes relative to the total OLIG2 expressing oligodendrocyte lineage cells across genotypes in male and female rats. (e) Density of CC1/OLIG2 expressing mature oligodendrocytes relative to the total OLIG2 expressing oligodendrocyte lineage cells in the CC across genotypes. (f) Density of PDGFRα expressing cells in the CC across genotypes in male and female rats.
We also quantified the callosal density of CC1+ oligodendrocytes (number of CC1+ cells/ mm3) in the regions of interest within the CC (Figure 1(a)) across genotypes and found that they were not statistically different (Figure 3(e), Table 1), and that they were equivalent within ±30% (Supplemental File), suggesting no meaningful difference. In addition, we found that sex, genotype and their interaction have no effect on the density of these cells in the CC (Figure 3(f), Table 2, Table S1). These results indicate that despite the presence of absence seizure-like neuronal activity in Grin2b+/− rats, we observed no changes in the percentage or density of mature oligodendrocytes in the CC of Grin2b+/+ and Grin2b+/− rats.
No difference in callosal myelination between Grin2b+/+ and Grin2b+/− rats
Myelin adaptations in response to changes in neuronal activity involve changes in the amount of myelin (specifically changes in myelin sheath number, length and thickness) and alterations in the area of and around the nodes of Ranvier, where voltage-gated sodium channels cluster and action potentials are regenerated (Xin and Chan, 2020).
To test whether the irregular neuronal activity associated with absence seizures leads to activity-dependent myelin adaptations within the seizure network in Grin2b+/− rats, we first measured the myelin volume in the regions of interest within the CC (Figure 1(a)) and compared it to that of Grin2b+/+ rats (Figures 4(a)-(e)). Specifically, we used confocal microscopy and 3D image analysis to quantify the total volume occupied by myelin basic protein (MBP) staining, indicative of compact myelin sheaths within the area analysed (Figures 4(a)-(c); Figure S1(a) and (d)). In addition, we calculated the total volume of the paranodal protein CASPR as a proxy for myelin sheathes flanking both nodes of Ranvier and immature heminodal structures in the region (Figures 4(a)-(c); Figure S1(b) and (c)). We did not detect any significant differences across genotypes and sexes in either of these analyses (Figures 4(d)-(g)). For both analyses (Figure 4(d) and (f), Table 1) there was no difference between Grin2b+/+ and Grin2b+/− rats, while the groups were equivalent within ±30% (Supplemental File), suggesting no meaningful difference. Furthermore, there was no effect of sex, genotype or interaction between the two on the total MBP (Figure 4(e), Table 2, Table S1) or CASPR volumes (Figure 4(g), Table 2, Table S1) across the groups.

Preserved myelination in the corpus callosum of adult Grin2b+/− rats. (a) Schematic showing the areas of the node of Ranvier and the paranode along a myelinated fibre with the respective protein markers (MBP, green and CASPR, red) used for this analysis. (b) and (c) Representative images of adult Grin2b+/+ and Grin2b+/− CC sections immunostained for MBP (green), CASPR (red) and counterstained with Hoechst (blue). (d) Ratio of the volume of myelin basic protein (MBP) signal relative to the total volume of the z-stack across genotypes (e) and between male and female rats. (f) Ratio of the volume of paranodal protein CASPR signal relative to the total volume of the z-stack across genotypes (g) and between male and female rats.
Changes in the nodal length (Figures 5(a), (d)-(f)) and nodal densities (Figure 5(b) and (c)) have previously been shown to regulate conduction speed and action potential synchrony (Arancibia-Carcamo et al., 2017; Ford et al., 2015; Smith et al., 2024). To identify potential changes at the level of the nodes of Ranvier, we measured the nodal densities in the CC of adult Grin2b+/+ and Grin2b+/− rats of both sexes (Figure 5(g) and (h)). Only nodes flanked by two paranodes were included in this analysis (Figures 5(d)-(f)). We observed comparable nodal density values across genotypes and sexes (Figure 5(g) and (h), Tables 1 and 2, Table S1). Using confocal microscopy and nodal intensity profiles, we also automatically quantified the average nodal length (Figures 5(d)-(f)) across genotypes and sexes (Figure 5(i) and (j), Tables 1 and Table 2, Table S1). The average nodal length in the adult rat corpus callosum ranged between 1 and 2 μm, with no significant differences between Grin2b+/+ and Grin2b+/− animals of either sex (Figure 5(i), Table 1). However, in this case equivalence analysis was inconclusive within ±20% (Supplemental File), while no effect of sex, genotype or their interaction was observed between groups (Figure 5(j), Table 2, Table S1). These results suggest that the myelination properties of the corpus callosum analysed here remain largely unaffected by any irregularities in network activity in Grin2b+/− rats.

Comparable nodal densities and lengths in the corpus callosum of adult Grin2b+/− rats. (a) Schematic showing the changes in nodal length. (b) and (c): Representative images of adult Grin2b+/+ and Grin2b+/− CC sections immunostained for CASPR (red) and counterstained with Hoechst (blue). (d)–(f): 60x confocal images showing nodes of three different lengths flanked by two CASPR+ paranodes. Dashed lines highlight the nodal length (Scale bar: 2 μm). (g) Average nodal densities in the CC of Grin2b+/+ and Grin2b+/− rats. (h) Average nodal densities in the CC of Grin2b+/+ and Grin2b+/− rats and between male and female rats. (i) Average nodal length in the CC across genotypes. (j) Average nodal length in the CC across genotypes and between male and female rats.
Collectively, these findings demonstrate that despite the previously observed seizure-like activity, Grin2b haploinsufficiency did not lead to measurable alterations in oligodendrocyte populations or myelination in adult rats.
Discussion
Absence seizures represent a form of generalized nonconvulsive epilepsy, typically arising from the hypersynchronous firing of deep-layer pyramidal neurons of the somatosensory cortex (Coenen and Van Luijtelaar, 2003; Hristova et al., 2025; Meeren et al., 2002; Polack et al., 2007, 2009; Studer et al., 2019). It is thought that these pathological oscillations then propagate to the thalamus by engaging corticothalamic circuits and subsequently generalize bilaterally, involving the interhemispheric myelinated fibres of the corpus callosum (Brodovskaya et al., 2022; Liang et al., 2016; Meeren et al., 2004; Musgrave and Gloor, 1980).
Imaging studies in patients with generalized SWDs have reported structural alterations in white matter, including in the corpus callosum (Hatton et al., 2020; Liang et al., 2016; Moeller et al., 2008; Yang et al., 2012). Similar abnormalities have been observed in animal models of absence seizures. For example, Diffusion Tensor Imaging (DTI) in two well-characterized rat models of absence seizures–WAG/Rij and genetic absence epilepsy rats of Strasbourg (GAERS)–has identified microstructural changes in the corpus callosum which correlate with SWD activity (Chahboune et al., 2009). More recently, seizure-induced alterations in OPC proliferation, oligodendrocyte differentiation and increased myelination have been documented in the corpus callosum of WAG/Rij rats and of an additional model of generalized epilepsy with absence seizures, the Scn8a+/mut mice (Knowles et al., 2022). Blockage of seizures with anti-seizure drug ethosuximide rescued the callosal hypermyelination phenotype in WAG/Rij rats. Conditional deletion of brain-derived neurotrophic factor (BDNF) receptor, TrkB from oligodendrocyte progenitors in Scn8a +/mut mice blocked activity-dependent myelination in response to seizures and reduced seizure burden. Similar results were also obtained after pharmacological inhibition of activity-dependent myelination in Scn8a+/mut mice. Collectively these data suggest that maladaptive changes in myelination after the establishment of absence seizures contribute to seizure progression in these rodent models (Knowles et al., 2022).
In this study, we investigated the oligodendrocyte lineage cells and myelinated callosal fibres in a new model of absence seizures, the Grin2b+/− rat, which exhibits nonconvulsive, bilaterally generalized SWDs of comparable frequency and duration to those observed in WAG/Rij rats (Chahboune et al., 2009; Coenen and Van Luijtelaar, 2003; Hristova et al., 2025; Knowles et al., 2022). We hypothesized that the seizure-induced alterations in myelination or in oligodendrocyte cell populations are a common feature across different models of absence epilepsy and therefore we predicted that comparable changes would be present in the Grin2b+/− brains.
Our findings indicate that despite the presence of ictal SWDs reported in these rats (Hristova et al., 2025), there were no significant differences in the density or proportion of oligodendrocyte progenitor cells, mature oligodendrocytes, or the overall oligodendrocyte lineage population in the corpus callosum. In addition, we found no detectable changes in callosal myelination, nodal density or nodal architecture. Given that sex can influence oligodendroglia and myelin development (Bourisly et al., 2017; Cerghet et al., 2006; Schmied et al., 2020; Seeker et al., 2023), we also assessed potential sex differences across genotypes. However, our analyses revealed no significant differences between male and female rats in any of the examined parameters. Our findings raise important questions about the molecular and cellular mechanisms underlying oligodendrocyte adaptations in this model and suggest that the presence of SWD activity alone may be insufficient to drive changes in oligodendrocyte dynamics or in myelination in Grin2b neurodevelopmental disorder.
An important consideration is the timing of seizure onset and the overall seizure burden in Grin2b+/− rats at the time of the analysis. In WAG/Rij rats, absence seizures emerge spontaneously during adolescence and progressively increase with age (Coenen and Van Luijtelaar, 2003; Lazarini-Lopes et al., 2021). Consequently, the myelin alterations observed in this model may reflect cumulative effects of seizure activity over time (Knowles et al., 2022). In contrast, absence seizures in Grin2b+/− rats were first documented in 5-month-old animals (Hristova et al., 2025), and our analysis was conducted in adult animals approximately 7 weeks later. We did not perform longitudinal, within-subject comparisons before and after seizure onset in Grin2b+/− rats, which may well limit the sensitivity to detect seizure-driven changes in oligodendrocyte populations or myelin architecture. Since the precise time of seizure onset in this model is currently unknown, it is possible that although the seizure activity is chronic, the overall seizure burden may differ from that of WAG/Rij rats and is therefore insufficient to induce detectable changes in oligodendrocyte populations or subtle alterations in myelin architecture.
Differences in seizure burden between models may also reflect differences in background strain (Wistar WAG/Rij versus Long-Evans Grin2b+/− rats) (Hristova et al., 2025; Taylor et al., 2017, 2019). Alternatively, transient changes may have occurred earlier during a more developmentally plastic window and subsequently resolved by adulthood, leaving no lasting structural differences in the corpus callosum detectable at the time of analysis.
It is also possible that seizure-induced changes in myelination may be regionally restricted. Our analysis was focused on the corpus callosum as one of the major white matter tracts implicated in seizure propagation (Brodovskaya et al., 2022; Chen et al., 2020). However, the thalamus, known to play a central role in seizure generalization and maintenance in this model (Hristova et al., 2025), may be more directly affected. Similarly, other brain regions such as the somatosensory cortex, may be more susceptible to seizure-induced myelin plasticity compared to the CC. Notably, the existence of regional heterogeneity in oligodendrocyte lineage dynamics and vulnerability (Foerster et al., 2024; Hughes and Appel, 2019; Marisca et al., 2020; Marques et al., 2016; Seeker et al., 2023; Viganò et al., 2013) underscores the need for region-specific analyses to more comprehensively assess seizure-related myelin adaptations.
Methodological differences should also be considered. We used fluorescent microscopy with typical molecular markers to label oligodendrocyte lineage cell populations and quantified their densities in defined regions of the corpus callosum (Figure 1(a)), on sections spanning Bregma −3.30 mm to −3.60 mm (see also Materials and Methods). This differs from (Knowles et al., 2022) that used unbiased stereology. Since our rats shared the same genetic background, we reasoned that group changes in oligodendrocyte populations would be detectable with our approach. We did not, however, assess OPC proliferation or cell death and therefore cannot exclude genotype-dependent differences in OPC proliferation or OPC apoptosis rates. Finally, in the absence of electron microscopy, we evaluated callosal myelination by quantifying myelin basic protein staining and assessing the density and geometry of nodes and paranodes in this region across genotypes. Future work combining precise cellular and ultrastructural analyses before and after seizure onset could provide further insight into the potential myelin changes in Grin2b+/− rats.
The GluN2B subunit encoded by Grin2b is a critical component of NMDA receptors, which are diverse sodium, potassium and calcium permeable, ligand-gated ion channels that regulate excitatory synaptic transmission and plasticity (Paoletti et al., 2013; Yashiro and Philpot, 2008). The GluN2B subunit is highly expressed early in development and persists into adulthood. Although primarily expressed in excitatory neurons, low levels of Grin2b transcripts have been detected in oligodendrocyte lineage cells. However, its role in oligodendrocytes is poorly understood (Marques et al., 2016; Zhang et al., 2014). Pathogenic mutations have been shown to affect key developmental processes including neuronal migration, dendritic morphology, synaptic turnover and plasticity, potentially contributing to circuit dysfunction and seizure susceptibility (Bahry et al., 2021; Hristova et al., 2025; Kellner et al., 2021; Sceniak et al., 2019; Shin et al., 2020). Although these abnormalities are documented in GRIN2B mutation models, convergent structural and plasticity changes are also found in GAERS and WAG/Rij rats. Specifically, dendritic length, arborization and spine density are increased in somatosensory and hippocampal pyramidal neurons of GAERS (Yazi et al., 2024) and somatosensory pyramidal neurons of WAG/Rij rats (Ilbay et al., 2022; Karpova et al., 2005). Elevated plasma membrane AMPAR expression in somatosensory excitatory neurons is also reported in GAERS which modulates regional plasticity (Derkach et al., 2007; Kennard et al., 2011; Malinow and Malenka, 2002; Shepherd and Huganir, 2007). These structural and synaptic plasticity changes are therefore shared across some absence seizure models and may be relevant to broader features such as myelination.
Finally, given the role of GRIN2B in human neurodevelopmental disorders, understanding the interplay between seizures, white matter development and cognitive outcomes in this model could provide translational insights. Understanding how genetic disruptions in glutamatergic signalling can affect brain connectivity may help clarify the mechanisms linking specific genetic mutations to functional deficits in these disorders. The relative stability of oligodendrocyte populations in this model highlights the importance of further investigating the cellular mechanisms that preserve white matter integrity in the context of genetically induced, chronic absence seizures.
Supplemental Material
sj-docx-1-bna-10.1177_23982128251405032 – Supplemental material for Assessing oligodendrocyte and myelin alterations following absence seizures in a rat model of GRIN2B neurodevelopmental disorder
Supplemental material, sj-docx-1-bna-10.1177_23982128251405032 for Assessing oligodendrocyte and myelin alterations following absence seizures in a rat model of GRIN2B neurodevelopmental disorder by Zita Francsics, Eleni Tsoukala, Katerina Hristova, Alfredo Gonzalez-Sulser and Lida Zoupi in Brain and Neuroscience Advances
Supplemental Material
sj-tif-2-bna-10.1177_23982128251405032 – Supplemental material for Assessing oligodendrocyte and myelin alterations following absence seizures in a rat model of GRIN2B neurodevelopmental disorder
Supplemental material, sj-tif-2-bna-10.1177_23982128251405032 for Assessing oligodendrocyte and myelin alterations following absence seizures in a rat model of GRIN2B neurodevelopmental disorder by Zita Francsics, Eleni Tsoukala, Katerina Hristova, Alfredo Gonzalez-Sulser and Lida Zoupi in Brain and Neuroscience Advances
Supplemental Material
sj-xlsx-3-bna-10.1177_23982128251405032 – Supplemental material for Assessing oligodendrocyte and myelin alterations following absence seizures in a rat model of GRIN2B neurodevelopmental disorder
Supplemental material, sj-xlsx-3-bna-10.1177_23982128251405032 for Assessing oligodendrocyte and myelin alterations following absence seizures in a rat model of GRIN2B neurodevelopmental disorder by Zita Francsics, Eleni Tsoukala, Katerina Hristova, Alfredo Gonzalez-Sulser and Lida Zoupi in Brain and Neuroscience Advances
Footnotes
Acknowledgements
We would like to thank the members of Zoupi lab for their feedback. We would also like to thank Dr Owen Dando for his advice on statistical tests, the Bioresearch & Veterinary Services facilities (University of Edinburgh, UK), Lynsey Dunsmore for the animal care, Nick Dandoulakis for assisting us with script writing for nodal analysis and Dr Adrian Garcia-Burgos and Dr Iain Porter from the IMPACT facility for the technical assistance.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: L.Z. is supported by a Chancellor’s Fellowship (College of Medicine & Veterinary Medicine, University of Edinburgh) and by the Simons Initiative for the Developing Brain. Z.F. was supported by the University of Edinburgh MSc programme in Integrative Neuroscience. E.T. is supported by the Simons Initiative for the Developing Brain. K.H. and A.G.S. are funded by Simons Initiative for the Developing Brain, The Carnegie Trust Award RIG01249 and Royal Society Seed Corn RGS\R1\221112.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
