Abstract
Background
To recover normal functions, remyelination in multiple sclerosis is crucial. Although endogenous remyelination occurs, it is often insufficient, and finding molecules promoting repair of demyelinated lesions is needed.
Objectives
To compare the remyelination potential of evobrutinib, an inhibitor of Bruton's tyrosine kinase and clemastine, an antagonist of M1 muscarinic acetylcholine receptor.
Methods
Remyelination was investigated in lysolecithin demyelinated organotypic mouse cerebellar slices and a transgenic Xenopus model of inducible-demyelination.
Results
Evobrutinib (100 nM) and clemastine (200 nM) potentiated remyelination of mouse cerebellar slices by a factor of 2.9 and 1.76, respectively. In conditionally demyelinated Xenopus, evobrutinib and clemastine increased remyelination by a factor of 1.61 and 1.92, respectively. Evobrutinib targets Bruton's tyrosine kinase expressed by microglia, and we showed that the increase in number of myeloid cells following demyelination is due to an extravasation from nearby vessels of macrophages migrating toward the optic nerve. In contrast, clemastine is expected to antagonize muscarinic receptor 1 expressing cells of the oligodendroglial lineage. We investigated a possible synergistic effect on remyelination by adding simultaneously both molecules. In both experimental models tested no significative improvement on remyelination of co-treatment with evobrutinib plus clemastine was observed.
Discussion
While evobrutinib increased 1.59 fold the number of microglia/macrophages, in the presence of clemastine the number of innate immune cells was decreased by 0.39 fold, therefore counteracting the beneficial effect of microglia/macrophages on remyelination.
Introduction
Patented nearly 60 years ago, clemastine is an over-the-counter antihistamine drug with anticholinergic properties used to relieve hay fever and allergy symptoms. 1 More recently, upon screening 1000 bioactive molecules, clemastine has been identified as a compound enhancing oligodendrocyte differentiation and remyelination. 2 Initially performed in rodent, the promyelinating property of clemastine has been confirmed in other vertebrates such as zebra fish 3 (Zada et al., 2016) or Xenopus. 4 It has been established that treating demyelinated lesions with clemastine to enhance remyelination is associated with functional improvement. 5 Further studies specifically ablating the muscarinic receptor 1 (M1R) in oligodendrocyte precursor cells, corroborated that the pro-remyelinating property of clemastine was by antagonizing M1R expressed by young oligodendrocytes. 6 Based on these experimental findings several clinical studies have been undertaken in optic neuritis supporting a beneficial effect quantified by shortening of P100 latency delay on visual-evoked potentials in human optic neuritis and in EAE mice7,8 as well as myelin water fraction changes evaluated by 3T MRI occurring in normal-appearing white matter of corpus callosum. 9
Evobrutinib, a member of the Tec family of nonreceptor tyrosine kinases is an oral, CNS-penetrant, highly selective covalent inhibitor of Bruton's tyrosine kinase (BTK). It blocks B-cell activation and cytokines release 10 and inhibits the activation of proinflammatory M1 macrophages in vitro. 11 In vivo evobrutinib was efficient in preclinical models of experimental autoimmune encephalomyelitis (EAE) with and without B-cell involvement, 12 suggesting a benefit of BTK inhibitors beyond B-cell inhibition. In line with this observation, BTK inhibition by evobrutinib was shown to reduce proinflammatory activation of microglia when treating chronic EAE or following the adoptive transfer of activated T-cells. 11 In multiple sclerosis, evobrutinib showed efficacy in a Phase 2 trial versus a placebo arm, 13 however, two Phase III clinical trials comparing the efficacy of evobrutinib with teriflunomide did not meet the primary endpoint of a better reduction of annualized relapse rates in people with relapsing multiple sclerosis (RMS). 14
Having shown that in the brain BTK is expressed mostly by microglial cells and to lesser extent by astrocytes 15 we investigated the consequence on remyelination of inhibiting BTK ex vivo in lysophosphatidylcholine (LPC) exposed mouse organotypic cerebellar slices 16 and in vivo in transgenic Xenopus line Tg(Mbp:gfp-ntr) of metronidazole inducible-demyelination.4,17 We showed that, in these two experimental models of demyelination, BTK inhibition significantly promoted remyelination. 15 Here, we first established the dose response curve of evobrutinib and clemastine on remyelination. Since the cellular target of clemastine and evobrutinib is oligodendrocyte and microglia, respectively, we questioned a potential beneficial synergistic effect of simultaneous addition of both molecules to restore demyelinated lesions.
Materials and methods
Animals
Mice, either wild type (C57/Bl6) or Tg(Plp:gfp) transgenic line 18 were raised in our animal facility (Agreement No. A75-13-19). In this transgenic line, oligodendrocytes cell bodies and the myelin sheath are identified by the expression of the green fluorescent reporter.
To test in vivo the effect of evobrutinib and clemastine on remyelination we used a conditional demyelination model, Tg(Mbp:gfp-ntr), developed in Xenopus laevis, in which myelinating oligodendrocytes selectively express the GFP reporter fused to the bacterial nitroreductase (NTR) enzyme, under the control of a portion of mouse myelin basic protein (MBP) regulatory sequence. 17 The NTR enzyme converts the nitro radical of prodrugs, such as metronidazole (MTZ), to a highly cytotoxic hydroxylamine derivative; therefore, introduction of MTZ into the swimming water of transgenic Tg(Mbp:gfp-ntr), tadpoles provokes an oligodendrocyte cell-death followed by demyelination. 17 After withdrawal of the demyelinating agent (MTZ), spontaneous remyelination occurs, a process which can be either accelerated 4 or delayed, 19 by addition of molecules tested in the aquarium water. To label the myeloid lineages we used the Tg(mpeg1:mCherry) in which the mCherry reporter is under the control of the zebrafish mpeg1 promoter, which is predominantly expressed in macrophage lineages.20,21 The double transgenic Tg(Mbp:gfp-ntr)//(mpeg1:mCherry) was generated by crossing the two lines. Xenopus tadpoles were staged according to Nieuwkoop and Faber. 22 Transgenic tadpoles were treated between stages NF 50 and 55, corresponding to premetamorphosis, a stage of ongoing myelination. Tadpoles of either sex were anesthetized in 0.05% MS-222 (ethyl-3-aminobenzoate methanesulfonate; Sigma-Aldrich) before quantification of GFP+ cells and returned to normal water to recover. Before brain and spinal cord dissection, tadpoles were euthanized in 0.5% MS-222. Animal care was in accordance with institutional and national guidelines. All animal procedures conformed to the European Community Council 1986 directive (86/609/EEC) as modified in 2010 (2010/603/UE) and have been approved by the ethical committee of the French Ministry of Higher Education and Research (APAFIS#5842-2016101312021965).
Preparation of evobrutinib and clemastine
Stock solution of evobrutinib (Merck KGaA, Darmstadt, Germany) and of clemastine (Sigma-Aldrich) were at 100 mM and 25 mM, respectively in dimethyl sulfoxide (DMSO D8418-50 mL Sigma-Aldrich). Further dilutions were either in culture medium for mouse cerebellar slices or filtered tap water for Xenopus experiments.
Antibodies
Mouse mAb anti-Calbindin (IgG1, Sigma C9848, 1:800), chicken anti-GFP (1:500; Millipore); Chicken mAb anti mCherry (IgY, Abcam 205402, 1:1000); corresponding Alexa Fluor secondary antibodies were from Invitrogen (Thermo-Fisher) and all were used at a dilution of 1:600.
Metronidazole preparation and use
The procedure is described in the supporting information section; see also Kaya et al. 17
Quantification of GFP+ cells
The procedure is described in the supporting information section; see also Mannioui and Zalc. 24
Isolectin B4 staining
The procedure is described in the supporting information section; see also Sekizar et al. 25
Immunolabeling of Xenopus whole-mount optic nerve
The procedure is described in the supporting information section.
Two-photon observation
For in vivo examination of GFP and mCherry expressing cells along the optic nerve, tadpoles were anesthetized in MS222 and placed in a POC-Chamber-System (H. Saur, Reutingen), under a two-photon microscope. Mono-photon or two-photon excitation was performed using Zeiss LSM 710 microscope system. The microscope was equipped with objective (X20, 1 NA). Calculated optical slice thickness was 2 μm. Each image presented is the 3D projection of 18 to 26 stacks of images. The settings (gain and aperture pinhole) were held constant within individual experiments. All images shown were processed with image J software.
Statistical analysis
We used Prism v5 software for statistical analyses. Results were expressed as means ± SEM. For organotypic slices experiments data obtained following molecule to be tested were compared to control using an unpaired two-tailed t-test. Data obtained in the Xenopus were compared using Student's t-test. Statistical significance was set at p < .05.
Data availability statement
Data supporting the findings of the present study are available from the corresponding author on reasonable request.
Results
Dose response of evobrutinib and clemastine on remyelination ex vivo in mouse cerebellar slices
The potential effect of either evobrutinib or clemastine on remyelination was investigated on LPC demyelinated mouse cerebellar organotypic slices from either wild type or transgenic Tg(plp:GFP (Figure 1A–C). After removal of LPC, control slices were left for four days in normal culture medium (controls) (Figure 1A, D) or treated with increasing concentrations of either clemastine (100 nM to 400 nm) (Figure 1A, E) or evobrutinib ranging between 1 nM and 400 nM (Figure 1A, F) before being fixed and immunolabeled. Slices were stained by double-labeling of myelin using anti-PLP (wild type) or anti-GFP (Tg(plp:GFP) and Purkinje cells and axons detected with anti-Calbindin antibodies and the extent of remyelination was measured using an ImageJ macro language that allows a fast and unbiased automated quantification of cerebellar myelinated axons. 23 Clemastine (200 nM) treatment (1.91, n = 8), (95% CI [1.44–2.37]) of demyelinated cerebellar slices significantly promoted remyelination (1.76-fold increase) compared to control condition (1.08, n = 23) (95% CI [0.93–1.23]) (Figure 1E, G). However, clemastine when added at either 100 nM or 400 nM had no detectable effect compared to remyelination levels under control condition (Figure 1G). Addition of evobrutinib (100 nM) (3.08, n = 15), (95% CI [2.33–3.83]) resulted in a 2.9-fold remyelination increase as compared to control (1.03, n = 16), (95% CI [0.95–1.11]) (Figure 1F, H). Similarly to our data observed with clemastine, evobrutinib treatment had no effect when added at lower (1 nM or 10 nM) or higher concentrations (200 nM or 400 nM) (Figure 1H).

Spontaneous and drug-promoted remyelination of lysophosphatidylcholine (LPC) demyelinated mouse cerebellar slices. (A) Flow chart showing the sequence of events tested. Mouse cerebellar slices from Tg(plp:GFP) immunostained with a combination of anti-GFP (green) and anti-Calbindin (white) antibodies. After 6 DIV cerebellar slices are well myelinated (B). Demyelination was induced by LPC exposure for 15 h (C). Four days after removal of LPC, (11 DIV) slices are either spontaneously remyelinated (D) or following treatment with either clemastine (E) or evolbrutinib (F). Extent of remyelination by either clemastine (G) or evobrutinib (H) was evaluated by establishing the myelination index as described in the M&M section.
In vivo dose response of evobrutinib and clemastine on remyelination in X. laevis, a model of conditional demyelination
To test the effect of clemastine or evobrutinib on remyelination in vivo, we used a conditional demyelination model, Tg(mbp:GFP-NTR), developed in X. laevis, in which demyelination is achieved by 10 days of exposure to metronidazole (MTZ). After withdrawal of the demyelinating agent (MTZ), spontaneous remyelination occurs (Figure 2A). Thanks to the transparency of Xenopus tadpoles, we have previously shown that counting the number of oligodendrocytes (GFP + cells) per optic nerve in living animal is a reliable quantitative index of myelination, demyelination and remyelination 4 (Mannioui et al., 2018) (Figure 2B, C, D). Addition of clemastine in the swimming water for three days after stopping the MTZ exposure increased remyelination by 1.92 ± 0.05 (200 nM; n = 5) (95% CI [1.78–2.06]) and 1.8 ± 0.2 (400 nM; n = 3), (95% CI [0.82–2.85]), but had no significant effect at lower concentration (10 nM or 100 nM) (Figure 2E, G). Similarly, addition into the swimming water of evobrutinib (100 nM) for three days at the end of demyelination, resulted in a 1.61 ± 0.1 (n = 6, (95% CI [1.27–1.95]), increased remyelination. As observed in organotypic slices no significant effect of evobrutinib was recorded at lower (10 nM) or higher concentrations (1 µM or 10 µM) (Figure 2F, H).

Confocal imaging of Xenopus laevis Tg(mbp:GFP-ntr) optic nerve before demyelination, at the peak of demyelination and during recovery. (A) Flow chart showing the sequence of events tested. GFP reporter is expressed in myelinating oligodendrocytes before demyelination (B), at the peak of demyelination after 10 days of MTZ treatment (C) and during spontaneous recovery three days after the end of MTZ exposure (D) or treatment with either clemastine (E) or evobrutinib (F). Remyelination is evaluated by counting the number of oligodendrocytes (GFP+ cells) per optic nerve following either clemastine (G) or evobrutinib (H) treatment.
No additive effect of clemastine and evobrutinib on remyelination
In a previous study, we have shown that in the brain BTK was detected mainly in Iba1+ microglia/macrophages (nearly 75%), and to a lesser extent in S100ß+ astrocytes (about 25%), but not in CC1+ oligodendrocytes, NeuN+ neurons, nor Olig2+/CC1− oligodendrocyte precursor cells 15 (Martin et al., 2020). In contrast, the clemastine-induced remyelination has been proposed to be mediated by its anticholinergic properties against muscarinic M1 receptor (M1R) expressed by oligodendrocytes. We therefore reasoned that inhibiting BTK in microglia/macrophage and antagonizing oligodendrocyte muscarinic M1 receptor by cotreatment of evobrutinib and clemastine may have an additive or synergistic proremyelinating effect. However, this was not the case: when LPC-induced demyelinated cerebellar slices were treated with evobrutinib (100 nM) and clemastine (200 nM) we did not observe any statistically significant difference in the level of remyelination compared to either evobrutinib (100 nM) or clemastine (200 nM) given alone (Figure 3A). Similarly, simultaneous addition of clemastine and evobrutinib in the swimming water of tadpoles at the end of demyelination period had no synergistic effect on remyelination (Figure 3B).

Remyelination is not improved by co-treatment with clemastine and evobrutinib. (A) Spontaneous remyelination (CTRL) of lysophosphatidylcholine-demyelinated cerebellar slices was improved by treatment with either clemastine (200 nM) or evobrutinib (100 nM). Remyelination was not increased by simultaneous treatment with both molecules at the same concentration. No further improvement was observed by decreasing clemastine concentration. (B) In vivo, in MTZ-demyelinated Tg(mbp:GFP-ntr) optic nerve, remyelination was promoted by single administration of either clemastine (200 nM) or evobrutinib (100 nM), but no improvement by cotreatment with the two drugs and lowering clemastine concentration had no significative effect.
Clemastine inhibits innate immune cells
We completed this initial in vivo evaluation by a double labeling for GFP (oligodendrocytes) and IB4 (microglia/macrophages) and examination under confocal microscope after fixation of the tadpoles treated with either evobrutinib (100 nM) or clemastine (200 nM). Evobrutinib treatment (1.59, n = 4), (95% CI [1.07–2.12]) resulted in an increase in the number of IB4+ cells compared to control treated tadpoles (1, n = 7), (95% CI [0.77–1.24]) (Figure 4 A, B). In contrast, under similar conditions, treatment with clemastine (n = 5), (95% CI [−0.03–0.82]) resulted in a 0.39 fold decrease in the number of IB4+ cells (Figure 4C, D). The negative effect of clemastine on the microglia/macrophage population, may account for the absence of additive effect on remyelination by simultaneous addition of clemastine and evobrutinib. We then questioned whether lowering clemastine concentration to limit clemastine-induced inhibition of microglia may potentiate evobrutinib remyelination effect. Evobrutinib was maintained at 100 nM (its highest efficiency concentration) and clemastine lowered at 100 nM, 50 nM, and 10 nM. Whether tested on mouse organotypic cerebellar slices (Figure 3A) or on live demyelinated tadpoles (Figure 3B) lowering clemastine concentration had no significative improvement on remyelination.
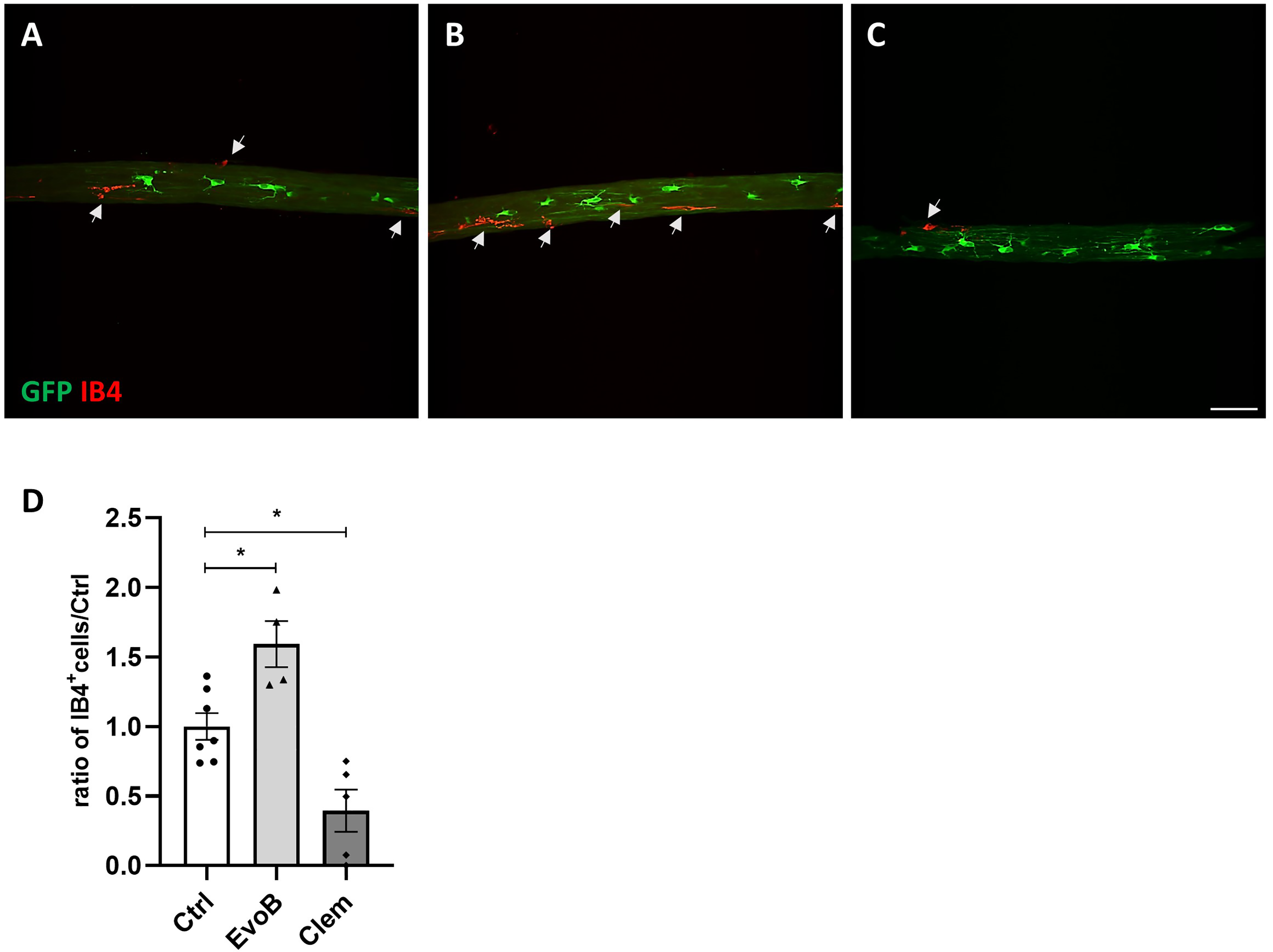
Clemastine-inhibited microglia/macrophage. Tg(mbp:GFP-ntr) Xenopus laevis were exposed to MTZ and optic nerve stained with Alexa Fluor 647-conjugated isolectin IB4 (red microglia/macrophages) before being immunolabeled with anti-GFP (green oligodendrocyte). (A) before demyelination. (B, C) MTZ exposure is stopped after 10 days and animals are treated with either evobrutinib (B) or clemastine (C). (D) The number of IBA+ cells is increased by evobrutinib but dramatically decreased by clemastine.
Origin of innate immune cells following demyelination
To monitor in vivo myeloid cells we used the X. laevis transgenic line Tg(mpeg1:mCherry) expressing mCherry reporter (red) under the control of the zebrafish mpeg1 promoter, which is predominantly expressed in macrophage lineages.20,21 We had previously shown that in Tg(Mbp:GFP-ntr) transgenic Xenopus after 10 days of MTZ exposure (i.e., at the end of demyelination treatment) the number of IB4+ cells per optic nerve had increased 2.6 fold independent of an active mitotic process (evaluated by BrdU incorporation) 26 (Sekizar et al., 2015). To address the origin of the observed increase in the number of IB4+ cells we generated a double transgenic line by crossing Tg(Mbp:GFP-ntr) with Tg(mpeg1:mCherry). In the resulting line Tg(Mbp:GFP-ntr)//(mpeg1:mCherry) we performed a double labeling for IB4 and mCherry showing that cells expressing the mCherry reporter were also IB4+ (Figure 5A). After five to seven days of ongoing MTZ-induced demyelination mCherry positive cells were monitored on live embryos by successive examinations of the optic nerve and surrounding area of the same animal under a two-photon microscope. In the optic nerve we were able to catch over a 4 h time-lapse mCherry+ cells in the process of phagocyting GFP+ debris (Figure 5B, C, movie #1). During the same period, in the neighborhood of the optic nerve we observed mCherry+ cells extravasating from blood vessels and migrating toward the optic nerve (Figure 5D). These results suggest that in response to a demyelination insult microglia are not dividing, but circulating macrophages are attracted toward the demyelinated optic nerve, where they adopt phagocytic properties.

Live imaging of microglia/macrophages during demyelination. (A, A’) Brain tissue section of the transgenic Tg(mpeg1:mCherry) line (stage 50 tadpole) doubly stained for mCherry (A) and Alexa Fluor 488-conjugated isolectin IB4 (A’), showing the overlap of the two markers on microglia. (B) Movie No. 1 showing a mCherry+ microglial cells phagocyting GFP+ debris in the optic nerve of a stage 50 double transgenic Tg(Mbp:gfp-ntr)//(mpeg1:mCherry) undergoing demyelination due to a five days exposure to MTZ. Note the morphological change of microglia in comparison to A. (C, C’, C’’) GFP debris (white arrow) being phagocyted by a round shape mCherry microglia in the optic nerve of Tg(Mbp:gfp-ntr)//(mpeg1:mCherry). (D) extravasation of an mCherry+ macrophage (white arrow) from a blood vessel in the immediate vicinity of the NO. Successive images of an mCherry+ cell leaving a blood vessel and heading toward the optic nerve at different times T = 0s, T = 90s, T = 360s, T = 630s; the GFP+ oligodendrocyte is taken as a reference point (two perpendicular dotted white lines) which is immobile.
Discussion
Using two experimental models of inducible-demyelination, ex vivo in the mouse and in vivo in Xenopus, we have investigated the remyelination potential of clemastine acting on oligodendrocyte M1R muscarinic receptor and evobrutinib activating microglia cells. We show that in each demyelinated condition both compounds promoted remyelination with a sharp bell-shaped dose–response. However, no additive or synergistic effect was observed by simultaneous addition of both molecules.
In these two demyelination models, the dose–response of evobrutinib and clemastine showed a sharp bell-shaped curve, with a maximum effect observed at 100 nM and 200 nM, respectively, and no effect at either lower or higher concentrations. It is interesting that the higher doses failed to promote remyelination rather than showing a plateau. One possible interpretation of this effect could be related to the fact that evobrutinib as well as clemastine are first solubilized in DMSO before being diluted in the water vehicle. At higher concentration, it is likely that evobrutinib and clemastine are under a micelle form, which may mask the real concentration, a phenomenon well described by lipid enzymologists.27,28 A similar effect has been reported for some anticancer drugs, which colloidal formulations exhibited bell-shaped curves, losing activity at higher concentrations, while monomeric formulations gave typical sigmoidal curves, sustaining a plateau of maximum activity. 29
Efficacious drug concentrations in complex in vitro cultures might not always translate to the in vivo human disease situation. How does the total efficacious Evobrutinib concentration of 44 ng/mL (100 nM) used in the in vitro remyelination models translate to the observed free CSF concentrations (3.3 ng/mL) in RMS patients? Since we have not measured the free evobrutinib levels under in vitro culture conditions, we used the data of total (115 ng/mL) and free (5.5 ng/ml) evobrutinib plasma concentrations in MS patients, to determine the approximate free culture medium concentration of evobrutinib (2.1 ng/mL). 30 In summary, the total and approximate free efficacious concentration of Evobrutinib used in the in vitro remyelination models translates well to the total and free plasma and the free CSF concentrations reached MS patients.
It has been shown that the zebrafish mpeg1 gene promoter driving the reporter mCherry has been successfully used in Xenopus transgenic Tg(mpeg1:mCherry) to label cells with monocytic/macrophage characteristics. 21 Here we show that in addition to circulating monocytes/macrophages mpeg1 regulatory sequence also labeled IB4+ microglial cells. Thanks to the transparency of Xenopus tadpoles, in the double transgenic Tg(Mbp:GFP-ntr)//(mpeg1:mCherry) we were able to monitor in vivo during demyelination microglial/macrophage cells engulfing and phagocytizing GFP+ myelin debris and illustrate mCherry+ macrophages migrating out from blood vessels toward the optic nerve during the demyelinating process. In a previous study, we had shown that the increased number of microglia/macrophages observed during demyelination could not be explained by a mitotic activity since the index of BrdU incorporation did not exceed a factor of 1.2, which was not enough to account for the 2.6 increase in the number of myeloid cells. 25 In this respect, our data are in agreement with the functions of microglia/macrophages illustrating their early evolutionary implication and their conserved function for myelin health and regeneration.31,32 In this respect, our double transgenic is a useful tool to monitor simultaneously innate immune cells and oligodendrocytes in living animals.
Our finding that clemastine affect the number of microglial/macrophage cells is in agreement with previous report. The inhibition of microglia/macrophage population is likely due to clemastine antagonizing M1R expressed on microglia, in consonance with RNAseq analysis showing that in addition to oligodendrocyte M1R is also expressed by microglial cells 33 ; it has been shown that the population of microglia cells, necessary for myelination was decreased in clemastine-treated mice. 34 It has also been reported that in an experimental model of intracerebral hemorrhage clemastine inhibited microglia-induced inflammatory response. 35
Our findings that evobrutinib increases the number of potential antiinflammatory, phagocytic myeloid cells following demyelination in the Xenopus model is in line with observation of Gelaradis et al. 11 in human monocytes and a toxic de- and remyelination model in mice. Evobrutinib-mediated BTK inhibition promoted an antiinflammatory monocyte/macrophage phenotype in vitro and the clearance of myelin debris by microglia in vivo, leading to accelerated remyelination. A recent publication studying another BTKi in MOG petide EAE and the cuprizone de- and remyelination models 36 revealed the modulation of neuroinflammatory pathways in microglia by tolebrutinib. The BTK-dependent transcriptional signature identified following EAE induction showed several immune pathways including neuroinflammation, FcγR-mediated phagocytosis in macrophages and monocytes, phagosome formation, and BCR signaling. While the cuprizone data suggest an alteration in the immune profile in the brain using a BBB penetrant inhibitor, during demyelination BTK inhibition did not alter the course of remyelination following cuprizone treatment at an early time-point. Finally, although the clinical trials investigating the efficacy and safety of evobrutinib did not meet the primary endpoint of better reducing annualized relapse rates significantly versus teriflunomide in people with RMS, 14 a small reduction of slowly expanding lesions and “black holes” was reported. In addition recent data from a Phase 3 study of another BTK inhibitor, tolebrutinib, demonstrated a significative delay in time to onset of disability progression in nonrelapsing secondary progressive multiple sclerosis when tested against a placebo arm. 37 Therefore BTK inhibition could show increased efficacy on readouts which are influenced by macrophages and microglia, additional cellular targets of BTK inhibition to B-cells. Our data suggest that when used alone, evobrutinib as well as clemastine, could promote remyelination and therefore be beneficial to regenerate demyelinated lesions in MS. Therefore, clinical proof of concept trials evaluating BTK inhibitors, instead of quantifying annualized relapse rates, should favor as primary endpoint evaluating myelin content changes, for instance combining magnetization transfer imaging and positron emission tomography in patients to demonstrate the beneficial therapeutic effect on the long term of repairing demyelinated lesion in MS.38,39 In addition, such trials should not aim at evaluating the combination of both drugs as no additive or synergistic effect could be expected.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251326913 - Supplemental material for Positive effect of evobrutinib in CNS remyelination models and lack of synergy with clemastine—A dose response study
Supplemental material, sj-docx-1-mso-10.1177_20552173251326913 for Positive effect of evobrutinib in CNS remyelination models and lack of synergy with clemastine—A dose response study by E. Martin, M.S. Aigrot, N. Frère, R. Lepaulmier, B. Stankoff, U. Boschert, C. Lubetzki and B. Zalc in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
We thank Dr Jacques Robert (University of Rochester Medical Center, USA) for the generous gift of Tg(mpeg1:mCherry) X. laevis transgenic line.
Funding
The author(s) disclosed receipt of the following financialsupport for the research, authorship, and/or publication ofthis article: This work was supported by Inserm, CNRS, Sorbonne-University, the program “Investissements d’Avenir” ANR-10-IAIHU-06 and NeurATRIS, and ANR grant BRECOMY to BZ. The study was partially funded by a research grant from Merck-Serono to BZ.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: U. Boschert is an employee of Merck Serono S.A., Eysin, Switzerland.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
