Abstract
Huddling, a tactile, thermoregulatory and affiliative social interaction, is a predominant and conserved behaviour for mammalian and bird species. Huddling is particularly important in early life, when thermoregulation, touch and bonding are influential for survival, brain and behavioural development. Our study aims to understand how tactile, vocal and kinship dynamics develop in rat pups. We designed a huddling apparatus where we synchronise huddle formation with ultrasonic vocalisations (USVs). With development, we see that groups (P6–8 vs older pups) stay longer in triad aggregon (pup huddle) configurations in the huddle trial period. Older pups (P18–20) switch huddle states more often; in all ages, join transitions were preferred to breaking. The spectral characteristics of rat pup USVs change in development, with higher peak frequency in P18–20 pups. In all age-groups, we observe vocal quieting as aggregons form. We hypothesised that kinship is a determinant of huddling dynamics but findings reveal otherwise. When comparing kin versus non-kin groups, we found no differences in aggregon durations or switches. In the youngest age (P6–8), USVs were reduced in kin versus non-kin groups, though not in older pups. To address the role of social touch in quieting, we integrated dividers in the huddle arena. Without skin/fur contact, USVs increased in P6–8, P11–14, but not in P18–20 kin groups. We suggest that rat pups have a strong internal drive towards huddling regardless of whether partners are related by kinship. USV analysis suggests that huddling has a calming effect, where related sibling young pups show less USVs overall; absence of social touch is associated with increased vocalisation in young huddle groups. Thus, huddling is a natural social behaviour shared between both related siblings and unrelated conspecifics. Huddling has calming effects–indicated by USVs–that depend on kinship and tactile social contact during the early stages of development.
Keywords
Introduction
Social contact and warmth, through thermoregulation or emotional support, are important factors in mammalian development which influence later-life mental health and survival (Alberts and Decsy, 1990; Chen and Baram, 2016). The importance of conspecific contact in development has been emphasised by severe consequences that lack of social touch has on well-being and health (Cascio et al., 2019; Harlow, 1958; Hasenack et al., 2023; Head, 2014).
Huddling behaviour features predominantly in mammalian and bird species, where huddling can reduce metabolic costs for individuals and enhance survival of the group. Emperor penguins, for example, utilise huddling to survive prolong fasts and ensure the survival of offspring (Ancel et al., 1997; Haig, 2008). Maternal touch has been identified as an important early life stimulus, however sibling touch can be as prevalent, if not more prevalent, in early life experience. Rabbit pups which huddle with siblings conserve more energy than individually reared animals, leading to more conserved energy for nursing and milk consumption (Gilbert et al., 2007; Haig, 2008).
In rodents, huddle preferences show species diversity, where Prairie voles, but not mice show preference for huddling with social partners and familiar individuals (Beery et al., 2018, 2021). Rat pup huddling functions in early (infant) development for thermoregulation and later becomes an olfactory-based affiliative activity, indicating a role in social bonding beyond metabolic conservation (Alberts, 1978, 2007; Schweinfurth, 2020; Sokoloff and Blumberg, 2001). Previous work indicates that rat pups may show sibling preference in huddling (Hepper, 1983a); however, confirmatory studies are lacking. Studies in Prairie voles indicate partner and familiarity preference in huddling (Beery et al., 2018, 2021). In sibling-preference and familiarity-preference experiments, rat and mouse pups show preference for sibling-familiar pups until around 15 days of age, where sibling and familiar preference changes to non-sibling, unfamiliar social preference (Clemens et al., 2020; De León Reyes et al., 2023; Hepper, 1983b; Hepper, 1987b). In-group biases in rats are also observed, where rats show preferential helping to free a trapped rat from their in-group. If raised with an unfamiliar strain of rat, however, rats will learn to help other rats from the unfamiliar strain (Ben-Ami Bartal et al., 2014). Huddling may be an altruistic act, where sharing of warmth and touch selectively could be a form of kin-selection, thus enhancing survival of the huddling partners. Classical conditioning in young mice (6–10 days old) has shown thermal preference learning where pups will prefer odours paired with warm stimuli (Bollen et al., 2012) and rat pups show learned preference for odours associated with siblings over their mother (Hepper, 1987a).
Rodents elicit ultrasonic vocalisations (USVs) which express the internal state of the animal (Knutson et al., 2002). Rat pups elicit distress calls with separation, temperature discomfort or anxiety (Brudzynski et al., 1999; Hofer, 1996; Oswalt and Meier, 1975). In older pups, USVs may indicate positive emotional states such as in play behaviour (Burgdorf et al., 2011). Calling interactions between the mother and pup have been well characterised, and the neural circuits described (Marlin et al., 2015); however, vocalisation between peer and sibling animals in development, specifically in the context of pup huddling, is less understood.
To address the hypothesis that huddling and vocal dynamics of rat pups are regulated by development, sex and kinship, we designed a setup for recording and quantitatively analysis of rat pup tactile and vocal interactions in natural huddling behaviour.
Materials and methods
Animals
Long Evans outbred rats were bred and maintained on a 12-hour/12-hour reverse light-dark cycle and provided ad libitum access to food and water. All animal procedures adhered to the regulations outlined by the United Kingdom Home Office. For each experiment, three animals were randomly selected per group (male kin, male non-kin, female kin, female non-kin), and animals were only used once for behavioural experiments to eliminate the possibility of adaptation or stress effects for the mother or pup with multiple handling sessions. Litters were housed in individually ventilated caging (IVC) to minimise olfactory familiarisation. Care was taken to have one experimenter handling the litters at a time. Two experimenters conducted the experiments independently; detailed methodology was maintained between experimenters. The total number of groups of animals was 30 groups for post-natal days P6–P8; 29 groups for P11–P14; and 31 groups for P18–P20. In aggregon analyses, data consisted of: P6–8 (23 groups total): N kin = 15 groups (9 male, 6 female), N non-kin = 8 groups (4 male, 4 female); P11–14 (23 groups total): N kin = 14 groups (7 male, 7 female), N non-kin = 9 groups (5 male, 4 female); P18–20 (24 groups total): N kin = 15 groups (8 male, 7 female), N non-kin = 9 groups (4 male, 5 female). We observed no sex differences between groups throughout all analyses. Some groups lacked the alignment signal or vocal recordings due to technical error (5 groups P6–8, 4 groups 11–14, 2 groups 18–20) so analyses with vocal data consisted of fewer groups (P6–8 (18 groups total): N kin = 11 groups, N non-kin = 7 groups; P11–14 (19 groups total): N kin = 10 groups, N non-kin = 9 groups; P18–20 (22 groups total): N kin = 13 groups, N non-kin = 9 groups). Experiments for the non-kin condition were limited due to variability in the timing of birth across timed-mated dams (i.e. if timed mated dams gave birth more than 3 days apart, the pups could not be used for the experiment because of difficulty in interpretation with comparing behaviour of pup groups with age-differences). The dividers test experiment was conducted with a between-subject design to avoid confounds with adaptation or stress in multiple huddling trials within subjects. Outbreeding of rats was maintained locally by tracking genealogies and by importing novel animals from the supplier to refresh stock (Charles River, Italy).
Huddling behaviour setup
An open arena made of flexible plastic, measuring 22 cm in diameter and 10 cm in height, was utilised for P6–8 and P11–14 sessions, while for P18–20, the arena dimensions were increased to 33 cm in diameter and 31 cm in height. Experimental conditions were maintained in a dimly lit room with controlled temperature (approximately 18°C–21°C). To address heat loss in the trial, we performed thermal camera imaging and found that temperature was maintained (within ~3°C). Video recordings and USVs were captured using IC Capture software (Imaging Source monochrome camera) and Avisoft USV recording (USV) software, respectively.
Video analysis
Video analysis was conducted blinded to group conditions using Elan software (version 6.3). Analyses were performed by two scorers initially, re-analysed and reliability was confirmed. Vocalisations were analysed using DeepSqueak (Coffey et al., 2019) and alignment of vocalisations and behavioural scoring was done with Matlab. Aggregon number shows the quantity of animals huddling. Aggregon 0 represents complete separation of all three animals, while aggregon 2 indicates two animals huddling together, and aggregon 3 signifies three animals huddling together.
Aggregons and Calls Per minute
Videos were scored continuously to determine whether each pup was in physical contact with others, allowing for the assignment of aggregon scores (0, 2, or 3) throughout the 20-min session. The continuous scoring was converted to an array of aggregon scores per second. From this array, the number of switches and identity of switches (ex: 0 to 2, 3 to 2 etc.) were extracted over the 20-min session. In datasets which examine aggregon score per minute, aggregon of 0 was assigned a 1, aggregon 2 assigned 2, and aggregon 3 assigned 3. Then the aggregon score for each minute was taken as the mean of the preceding 60 s. The aggregon score across groups was then taken as the average for all groups per minute. The join-break index was calculated: (N joins - N breaks)/(N joins + N breaks). Vocalisations were aligned to video and for each minute, the number of vocalisations were summed and divided by time (calls/min).
Statistical analysis
Groups were compared without assumptions of normal distribution, thus non-parametric testing was applied (Mann–Whitney, Kruskal–Wallis tests). Data are presented as mean ± standard error of the mean (SEM) unless stated otherwise. To create the probability matrix, probability for each state was calculated per group. The matrices show the mean probability across groups for each state transition.
Results
Experimental setup for examination of huddling behaviour development in rat pups
To address the organisation, development, vocal, and kinship interactions which occur throughout development, we designed a huddling apparatus in which we monitored interaction in triads of rats (male kin, male non-kin, female kin, female non-kin) at three pre-weaning developmental stages (P6–8, 11–14, 18–20). The size of the arena was modified to accommodate the size and mobility of the rat pups. The arena was illuminated with infrared illumination and filmed with a monochrome camera which was synchronised to the USV recording (Figure 1(a)). In each developmental stage, rat pup groups were placed in the arena in a separated state in a triangular configuration (aggregon 0) at the start of the trial and pups were allowed to behave freely. The trial period was limited to 20 min. Kin groups consisted of three male or female siblings or non-kin groups (one from each litter of a timed-mated female mother). Rat pups were used for only one trial and were not re-used to reduce adaptation and stress of repeated handling or separation (Kaidbey et al., 2019; Kosten et al., 2012). We scored contact behaviour as aggregons where an aggregon of 0 consisted of no pups huddling, aggregon of two consisted of two pups in contact and aggregon of three, where all pups were in a huddle (triad) (Figure 1(b)). In the example trial, we found that rat pups alternated between aggregon 0 and aggregon 2 before forming a triad (aggregon 3) and largely remaining in an aggregon of three with few alternations back to aggregon 2. Aggregons for the entire session were scored and aligned to vocalisations using synchronisation methods (Figure 1(c)). Vocalisations occurred more frequently at the start of the trial which then reduced over time (Figure 1(d)).
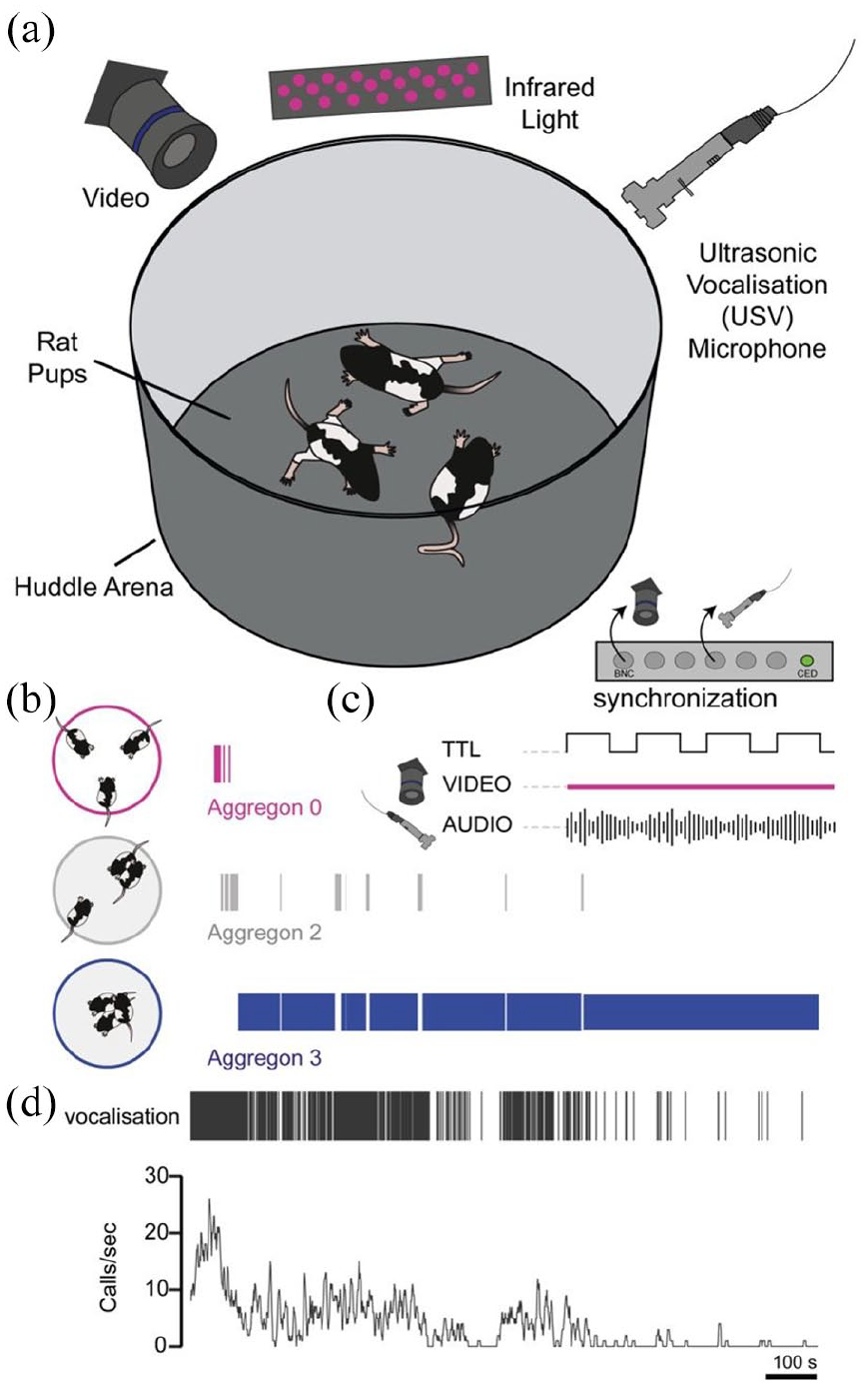
Experimental setup for examination of huddling behaviour development in rat pups. (a) The huddling setup consisted of a circular arena which was placed upon a washable base inside a behavioural cabinet which was sound-insulated and dark. Video recording was performed with infrared illumination and recording of ultrasonic vocalisations with one microphone. Temperature was monitored and maintained at 18–21°C. (b) Three rats were placed in a triangular configuration at the start of the experiment. A typical progression of aggregons is displayed, where separated rats (aggregon 0) transitioned to an aggregon of two (two out of three rats touching) and then progressed further to all rats huddling (aggregon 3). Aggregon states fluctuated throughout the experimental time (20 min total). (c) Video recording was synchronised with ultrasonic vocalisations (USV) with a Cambridge Electronic Design (CED) board which triggered camera frames and elicited an electronic (TTL) time stamp data which were encoded in the USV audio file via Spike2 software (CED). (d) Example USV events (Calls/sec) which were detected using DeepSqueak, deep-learning-based analysis package for USV spectrograms (Coffey et al., 2019). Vocalisation frequency was initially high in the huddle trial and reduced in number as the huddle trial progressed.
Aggregon configurations increase in complexity as rat pups develop
We next addressed the progression of aggregons across 20-min trials with developmental days. When placed in the triangular, separated start formation, pups transitioned between aggregons 0, 2, and 3 (Figure 2(a)). Huddle progression was visualised across groups (top P6–8, middle P11–14, bottom P18–20) for the 20-min trial (Figure 2(b)). All aggregon configurations were found to vary with development (aggregon 0 durations, p < 0.0001; aggregon 2 durations, p = 0.0032; aggregon 3 durations, p < 0.0001, Kruskal–Wallis test). Triad (aggregon 3) duration was lowest at P6–8 compared to developmental ages P11–14 and P18–20 (P6–8 vs P11–14, p = 0.0003; P6–8 vs P18–20, p < 0.0001; P11–14 vs P18–20, p > 0.999; Figure 2(c)). Thus, as pups develop, aggregon complexity increases.

Aggregon configurations increase in complexity as rat pups develop. (a) Depiction of aggregon 0, 2, and 3. Transitions can occur between all aggregon states. All groups consisted of 3 animals. (b) Rasters of aggregon groups. Aggregon 0 is depicted in magenta, aggregon 2 in grey and aggregon 3 in blue. Each row represents the raster behaviour of a triad group (N = 23, P6–8, N = 23 P11–14, N = 24 P18–20). Total time displayed is 20 min, scale bar 200 s. (c) Quantification of total time spent in aggregon state throughout development. Individual data points represent groups of three rat pups. Magenta: Aggregon 0 durations vary with development (Kruskal–Wallis test, p < 0.0001). Time spent in aggregon 0 is highest P6–8 (P6–8 vs P11–14, p < 0.0001; P6–8 vs P18–20, p = 0.0004), without a significant difference between P11–14 and P18–20 (p = 0.9365). Grey: Aggregon 2 durations vary with development (Kruskal–Wallis test, p = 0.0032). Time spent in aggregon 2 is highest P6–8 and reduces with development (P6–8 vs P11–14, p = 0.0285; P6–8 vs P18–20, p = 0.0018), without a significant difference between P11–14 and P18–20 (p > 0.999). Blue: Aggregon 3 durations vary with development (Kruskal–Wallis test, p < 0.0001). Time spent in aggregon 3 is lowest P6–8 and increased with development (P6–8 vs P11–14, p = 0.0003; P6–8 vs P18–20, p < 0.0001), without a significant difference between P11–14 and P18–20 (p > 0.999). Kruskal–Wallis followed by Dunn’s multiple comparisons test (N = 23, P6–8, N = 23 P11–14, N = 24 P18–20). Left axis corresponds to all bars. Bars indicate mean ± SEM.
Huddle transitions are regulated by developmental age
We observed that rat pup groups occupied three states (aggregon 0, 2, 3) and alternated between these three states in all developmental stages. To examine whether the number of transitions between aggregon states was developmentally regulated, we summed the number of aggregon switches throughout the 20-min trial. We found that the number of switches varied across age groups (p < 0.0001, Kruskal–Wallis test) with significant increases between P6–8 and P18–20 (p < 0.0001, N = 23 P6–8, N = 24 P18–20) and P11–14 versus P18–20 (p < 0.0001, N = 23 P11–14, N = 24 P18–20). Number of switches was not different for P6–8 versus P11–24 (p > 0.999, N = 23 P6–8, N = 23, P11–14) (Figure 3(a) and (b)).

Aggregon switches increase with development and favour joining over breaking in all age-groups. (a) The number of aggregon switches across the 20-min session was summed. A switch was defined as a transition between aggregon 0, aggregon 2, or aggregon 3, in any direction. A join included a transition from 0 to 2, 2 to 3 or 0 to 3. Breaks included transitions from 2 to 0, 3 to 0 or 3 to 2. (b) The number of total aggregon switches across all groups (P6–8, 22.61 ± 4.40; P11–14, 28.83 ± 5.017; P18–20, 94.88 ± 11.16 switches Mean ± SEM) increased with developmental age. Number of switches varied with developmental age (p < 0.0001, Kruskal–Wallis test). Switches for group comparisons was not significantly different between P6–8 and P11–14 (p > 0.999, N = 23 P6–8, N = 23, P11–14) and was significantly different in P6–8 vs P18–20 (p < 0.0001, N = 23 P6–8, N = 24 P18–20) and P11–14 vs P18–20 (p < 0.0001, N = 23 P11–14, N = 24 P18–20) comparisons (Dunn’s multiple comparisons test). (c) A Join-Break index was calculated for each group over the 20-min session ((N Join - N Break)/(n Join + n Break)). The Join-break index favoured joining in all age-groups (Wilcoxon Signed Rank Test, against theoretical median of 0, P6–8 p < 0.0001, P11–14 p < 0.0001, P18–20 p = 0.0006) and was reduced in P18–20 vs P11–14 pups (Kruskal–Wallis test, p = 0.027, Dunn’s multiple comparisons P18–20 vs P11–14 p = 0.024, P6–8 vs P11–14 p > 0.999, P6–8 vs P18–20, p = 0.255). Bars represent mean ± SEM and individual data points are the total switches or Join-Break index for 20 mins for each rat pup group.
To account for the identity of the transitions underlying switches, we calculated probabilities for each transition as well as the probability of staying in a state (Figure S1A). We then took the mean for each state across groups at each developmental state to compile a probability matrix. Across all developmental stages, we see that the probabilities of staying in each aggregon state were the highest per second events (diagonal of matrix) and the probabilities of transitions between states were lower (Figure S1B–C). To gain insight into the transitions between states, we summed the number of state joins (0 to 2, 2 to 3 or 0 to 3) and state breaks (2 to 0, 3 to 0 or 3 to 2). We computed a Join-Break index per group and found that joining was on average higher in all developmental ages, and there was a reduction of the Join-Break index between P11–14 and P18–20 (Kruskal–Wallis test, p = 0.027, Dunn’s multiple comparisons P18–20 vs P11–14 p = 0.024, P6–8 vs P11–14 p > 0.999, P6–8 vs P18–20 p = 0.255) (Figure 3(c)).
Thus, we find that huddling behaviour can be described with state probabilities that are changed in development. Staying in each state is more common per second than transitioning; transitions between states (switches) increase in development. Over all ages, joining transitions were more common than breaking transitions.
Aggregon durations and switches are not influenced by kinship
We hypothesised that huddling dynamics would be influenced by kinship, thus we analysed data while separating kin and non-kin group data. We found no kinship differences in aggregon durations across developmental ages (Figure 4(a)). We additionally analysed the number of aggregon switches between kin and non-kin groups and found no statistical differences (Figure 4(b)). Thus, huddling dynamics are consistent in kin and non-kin groups, without kinship differences, rejecting our hypothesis. We additionally examined whether huddle dynamics differed in male and female animals and found no sex-differences in huddling behaviour. Thus, the drive to huddle seems to override preferences based on sex and kinship.

Huddle aggregon times and switches are not different for kin and non-kin huddle groups. (a) Time spent in aggregons did not differ between kin and non-kin huddle groups. Aggregon 0 (P6–8, N kin = 15, N non-kin = 8, p = 0.7763; P11–14, N kin = 14, N non-kin = 9, p = 0.557; P18–20, N kin = 15, N non-kin = 9, p = 0.096), aggregon 2 (P6–8, N kin = 15, N non-kin = 8, p = 0.825; P11–14, N kin = 14, N non-kin = 9, p = 0.403; P18–20, N kin = 15, N non-kin = 9, p = 0.907), aggregon 3 (P6–8, N kin = 15, N non-kin = 8, p = 0.374; P11–14, N kin = 14, N non-kin = 9, p = 0.344; P18–20, N kin = 15, N non-kin = 9, p = 0.770). (b) Number of aggregon switches did not differ between kin and non-kin huddle groups P6–8 (N kin = 15, N non-kin = 8, p = 0.602), P11–14 (N kin = 14, N non-kin = 9, p = 0.865), P18–20 (N kin = 15, N non-kin = 9, p = 0.221). Comparisons made with Mann–Whitney test. Bars represent mean ± SEM.
Aggregon complexity is inversely related to the amount of calls
To examine the interaction between aggregons and USVs, we synchronised vocalisations to align to the video recording of huddling. The timing of each vocalisation was detected with DeepSqueak (Coffey et al., 2019) and manually curated. When rat pups formed aggregons of increasing size, the number of vocalisations reduced, in all age-groups (Figure 5(a) and (b)). We observed that aggregon score per minute and the number of calls per minute followed a linear trend where calls reduced as aggregon number increased, with the most negative slope found in P6–8 and P11–14 groups (Figure 5(c)). Across development, the spectral characteristics of calls changed from being uniform in principal frequency to more complex with age, particularly in P18–20 groups (Figure S2A). The peak frequency of the vocalisation spectrogram was significantly higher when comparing P11–14 and P18–20 groups, likely reflecting the emergence of higher frequency calls in P18–20 animals (Figure S2B). The effect of vocal quieting with huddling was most pronounced in youngest age groups (R2 = 0.93, slope = −98.28; P11–14 R2 = 0.91, slope = −103.4; P18–20 R2 = 0.58, slope −17.09), thus the quieting effects may reflect lower spectral frequency calls in young pups.

Vocal quieting occurs throughout the duration of huddle observation and is inversely related to aggregon score, in all pre-weaning age-groups. (a) Examples of aggregon events aligned to vocalisations at P6–8 (top), P11–14 (middle) and P18–20 (bottom). Magenta bars indicate aggregon 0, grey indicates aggregon 2 and blue aggregon of 3. (b) Ultrasonic vocalisation (USV) rate calculated per minute for each group at P6–8 (left), P11–14 (middle) and P18–20 (right). Squares indicate the mean aggregon score per minute across all groups, dotted line on either side indicates the mean ± SEM. Circles indicate the mean number of calls for each minute across all groups, shaded area indicates the mean ± SEM. (c) Mean call rate for each minute versus mean aggregon score per minute (P6–8 N = 18, R2 = 0.93, slope = −98.28; P11–14 N = 19 R2 = 0.91, slope = −103.4; P18–20 N = 22, R2 = 0.58, slope −17.09). Group data only included if both video and USV recordings collected with TTL alignment signal intact.
USVs are reduced in young (P6–8), but not older kin groups
To address whether kinship differences could be observed in the huddle task, we examined the mean number of calls per minute for kin and non-kin groups. We observed that the mean number of calls per minute was reduced in youngest kin groups (P6–8). The number of calls per minute did not differ in kin vs non-kin groups in older (P11–14 and P18–20) groups (Figure 6).
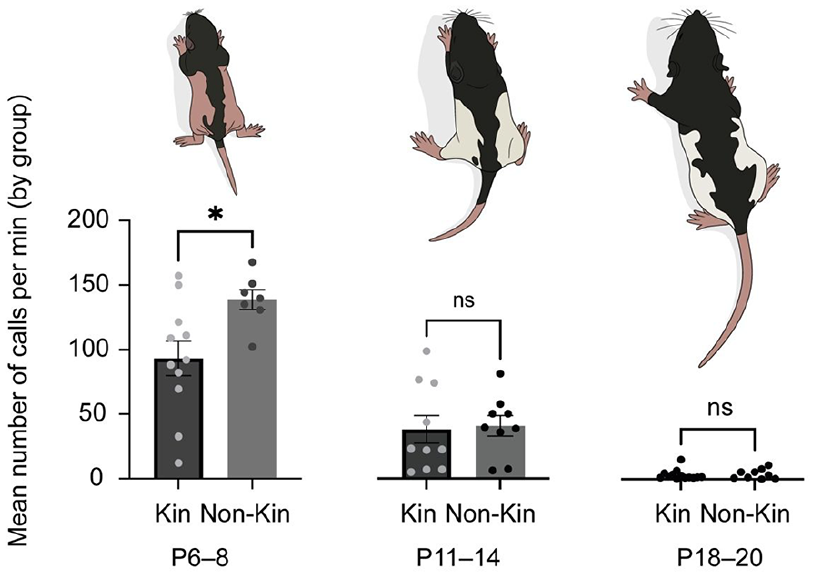
Ultrasonic vocalisations (USV) in the huddle trial are reduced in young age (P6–8) kin vs non-kin groups, but not older pre-weaning age groups. The average amount of calls over the 20-min recording was calculated for each group across developmental age. Left: Kin (N = 11 groups) vs Non-kin (N = 7 groups), p = 0.0268. Middle: Kin (N = 10 groups) vs Non-kin (N = 9 groups), p = 0.6038. Right: Kin (N = 13 groups) versus Non-kin (N = 9 groups), p = 0.948. Mann–Whitney test. Left axis corresponds to all graphs. Bars indicate mean ± SEM.
Huddle touch absence increases vocalisations in early-mid pre-weaning kin groups
We finally examined the role of touch in vocal quieting and kin-dependent huddle vocalisations. Thus, we designed a divider which separated direct contact (skin-to-skin and fur-to-fur contact) between the three pups in the huddle trial, though they could obtain touch and contact with the divider. Airflow was not contained; thus pups maintained abilities to smell, hear and see each other, where applicable. Pups were only tested once to reduce possible stress. The huddle trials were carried out with the same protocol as in previous experiments, with maintained temperature and huddling protocol (Figure 7(a)). When sibling (kin) groups without direct huddle touch (divider intact) were compared with kin groups without divider, we observed increased vocalisations without sibling huddling touch in P6–8 and P11–14 groups, but not in P18–20 groups (Figure 7(b)). Thus, social touch and skin/fur contact appears to be an important regulator of internal, vocal state in early pre-weaning huddling behaviour.
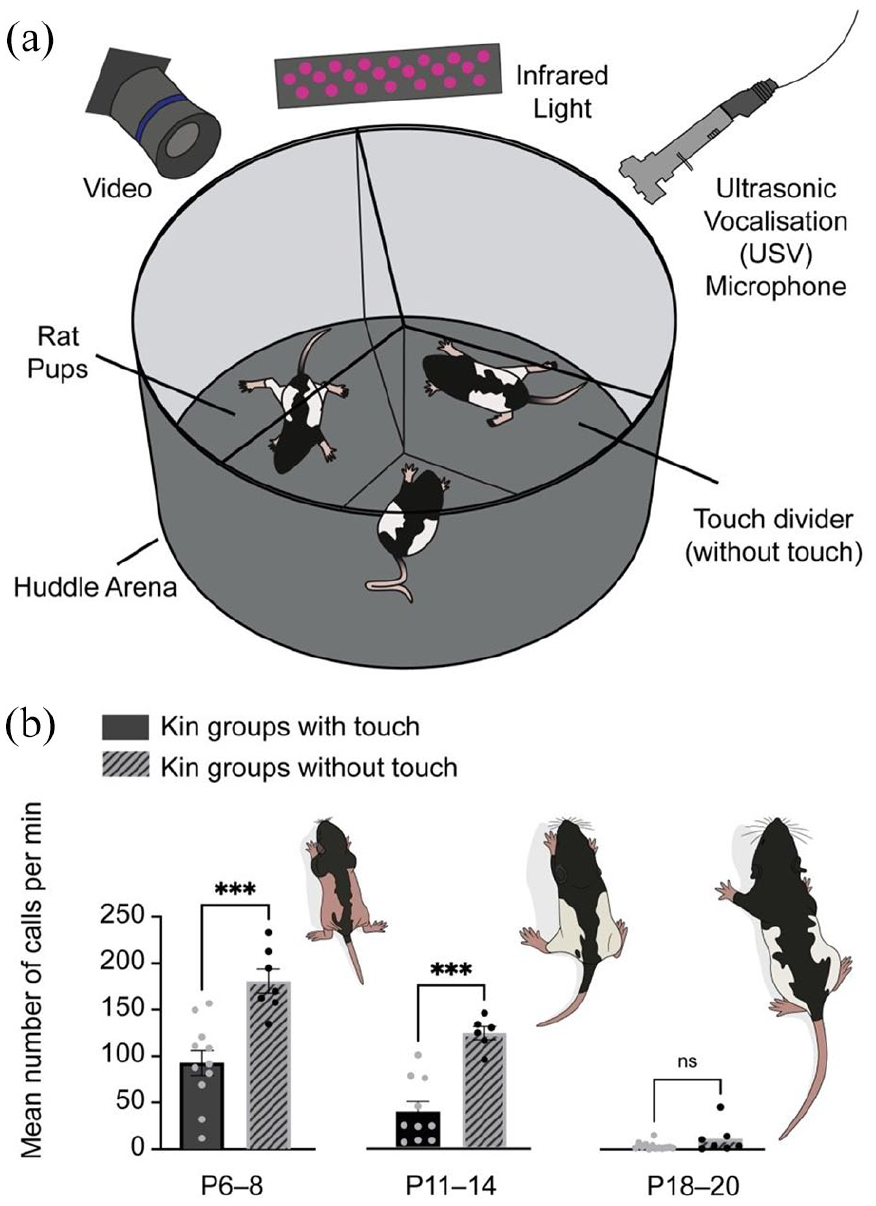
Huddle touch absence increases vocalisations in early and mid pre-weaning development of kin groups. (a) Sibling rat pups were placed in the huddling arena with a three-chamber divider separating direct huddling touch. The experiment was conducted for 20 min, as in previous huddling tests. (b) The average amount of calls per minute, per group was higher in kin groups with the touch divider present (no-sib-touch) compared with kin groups which were free to touch. P6–8 (p = 0.0003, N kin touch = 11, N kin no-sib-touch = 7), and P11–14 (p = 0.0005, N kin touch = 10, N kin no-sib-touch = 6) but not at P18–20 (p = 0.249, N kin touch = 13, N kin no-sib-touch = 7). Bars indicate mean ± SEM.
Discussion
We report that huddling behaviour (1) shows age-dependent changes in the dynamics of huddle formation, (2) huddle aggregon dynamics are not dependent on kinship, (3) aggregon complexity increases with vocal quieting, (4) USV reduction in kin-groups at the youngest age suggest a more calm internal state in the huddle task, (5) social touch absence in the huddle task increases USVs in P6–8, P11–14 but not P18–20 groups.
Huddle aggregon complexity and dynamics with development
We observed that rat pups spontaneously form huddles without training. In the earliest stage of development (P6–8), the time spent in lower aggregon states may be due to under-developed motor abilities. Thus, rat pups of this age may rely more on the parent to move them together in the nest. Interestingly, although thermo-regulatory need for huddling decreases in development (Alberts, 2007), rat pups continue to form huddles with increasing speed (Figure 2). In the examined age-periods, rat pups undergo multiple changes including development of exploratory and motor activity. Our study examines how these aspects of behavioural development contribute to the natural behaviour of huddling in rats. Furthermore, we observe vocal quieting in all ages of pups, indicating that the motivation to huddle may be a long-lasting mechanism for social bonding which is calming.
Huddle drive overrides kinship selectivity
Despite previous reports of kinship differences in huddling behaviour (Hepper, 1983a), our specific measures of huddling aggregon duration and switching did not reveal kinship differences (Figure 4). Similarly, although indications in the literature pointed towards sex-differences in huddling, our setup did not reveal this. One possibility may be the design of the huddling test. In Hepper’s work, groups of kin and non-kin rat pups were mixed and scored based on fur-markings, thus kin differences in huddling may arise when presented with the simultaneous choice of huddling with kin or non-kin. We chose a simpler design, where we could be certain of the composition of the rats being studied. Furthermore, with mixed kin and non-kin groups, the assessment of vocal behaviour would not be distinguishable between kin and non-kin. Further studies could implement Hepper’s approach of mixing groups, wherein the pups would be given the choice of kin and non-kin to associate within the huddle group. Alternatively, it may be that the internal drive to huddle, share warmth and touch may be sufficiently strong such that the behavioural dynamics are indistinguishable for related and unrelated conspecifics, overriding kinship selectivity. A recent study shows that adult mice will give up their preferred warm sleeping location to co-huddle and sleep with other mice, indicating that social touch may override the desire for warmth. Furthermore, post-huddle sleep synchrony was observed in male-sibling mice but not in female or unfamiliar mice (Sotelo et al., 2024). Although we did not measure sleep or wakefulness in our experiments, it would be interesting to explore how neurophysiological features are related to vocal quieting and if this is kin-dependent.
Vocal quieting with aggregon formation
We observed consistent reduction in the number of vocalisations as rat pups huddled more (Figure 5). Vocal quieting was consistent although the spectral characteristics of the calls showed developmental changes (Figure S2). Thus, regardless of the vocalisation type, the overall tone of vocalising or not vocalising reduces and is associated with increased contact. Contact quieting has been observed in other social behavioural paradigms (Hofer, 1996; Hofer et al., 2001) and may underly our observations. Many studies report contact quieting between the pup and caregiver (Hofer, 1996; Hofer et al., 2001; Marlin et al., 2015); our study confirms that siblings provide potent calming and quieting social stimuli as well.
Young-pup kin vocal quieting and dependence on social touch
We found that despite indistinguishable huddling dynamics in kin and non-kin pup groups, calls were reduced in sibling huddle groups in the youngest developmental age (P6–8), but not in older pup groups. Thus kinship, familiarity, or a combination of the two factors may calm sibling pups in the huddle task. Indeed, it is known that anxiolytics reduce USVs in infant rats (Muller et al., 2010). Although the recognition of kin is likely to be regulated by olfactory stimuli (Clemens and Brecht, 2021; Clemens et al., 2020; Logan et al., 2012), when we removed direct sibling contact in the huddling task, the amount of calls was no longer reduced. The touch-dependence of vocal quieting was observed in the P6–8 and P11–14, but not in older (P18–20) pups (Figure 7). Thus, although recognition of siblings may be calming, the intact ability to touch and huddle with social contact between skin and fur is necessary for this potentially anxiolytic stimulus to be maintained. The lack of social touch-dependence in older pups (P18–20) might reflect a shift from huddling as physiological necessity to an olfactory-driven bonding activity (Alberts, 2007; Brunjes and Alberts, 1979).
Neural mechanisms and social homeostasis
Recent work has elegantly shown that vocal quieting with social reunion occurs in adult mice and the overall level of ongoing vocalisation is heightened in groups which have undergone social isolation. The group further shows that tactile stimulation is key to social rebound and that neurons of the medial pre-optic nucleus (MPN) of the hypothalamus underly tactile social homeostasis (Liu et al., 2025). Another recent analysis of huddling in mice, suggests a role for the dorsomedial prefrontal cortex (dmPFC) in active and passive huddling decisions (Raam et al., 2024). Work in Prairie voles implicates corticostrial circuits in affiliative partner bonding (Amadei et al., 2017). Future studies in a variety of rodent species will address if the underlying brain mechanisms are conserved.
Research on the development of kin preference in rat pups point to the lateral septum (LS) as a target area for examination of huddling behaviour (Clemens et al., 2020; De León Reyes et al., 2023). Recent findings in mice demonstrate that neurons of cortex which project to the LS underly the development of social novelty preference (De León Reyes et al., 2023). Work in Spiny mice similarly implicate cortical to LS circuitry as a mediator of group size preference (Fricker et al., 2024). Thus, the LS may constitute an evolutionarily conserved hub which orchestrates social group preference and the underlying emotional states.
Social tactile sensitivity in vocal quieting during huddling suggests sensory transmission from the skin to the brain. Furthermore, there may be a role for the fur and specific skin regions in contact quieting, possibly mediated by softness sensing. Recent studies have identified specific sensory neurons (Piezo2-containing and Mrgprb4-lineage) in the skin which are activated in social interactions including pleasurable touch (Elias et al., 2023), the rat pup transport response (Ni et al., 2024), and passive touch stimuli (Villarino et al., 2023). Identification of specific skin sensory neurons and the downstream circuits activated in huddling and vocal quieting may lead to a comprehensive picture of the neural mechanisms at-play.
Conclusion
Overall, we find that huddling behaviour is a natural, self-organising behaviour in rat pups. Unique dynamics of contact are observed with development; vocal quieting with aggregon complexity occurs in all age-groups. Internal state, indicated by USV analysis, indicate that kinship and sibling touch are unique in the youngest developmental time periods. Thus, huddling is a calming activity shared among siblings and un-related conspecifics through an interplay of tactile and vocal dynamics.
Supplemental Material
sj-docx-1-bna-10.1177_23982128251354936 – Supplemental material for Ontogeny of tactile, vocal and kinship dynamics in rat pup huddling
Supplemental material, sj-docx-1-bna-10.1177_23982128251354936 for Ontogeny of tactile, vocal and kinship dynamics in rat pup huddling by Florbela da Rocha-Almeida, Hugh Takemoto and Ann M. Clemens in Brain and Neuroscience Advances
Footnotes
Acknowledgements
The authors thank Lynn Morrison, Neil Odey, and Callum Davidson for technical support. Thanks to Connie Yung, Grace Chattey, Zuzanna Baran, and Isabel Ibeson for helpful discussion and to Víctor Angulo for rat drawings.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Simons Initiative for the Developing Brain (SIDB), the University of Edinburgh MSc programme in Integrative Neuroscience (HT) and a Simons ESAT fellowship (AC).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
