Abstract
The Pyrethrins Stewardship Program (PSP) was established to better understand adverse effects following exposure to pyrethrins-containing insecticide products. Running from April 2010 through December 2016, symptomatic dermal and inhalation exposures were entered into Phase I of the PSP and analyzed for exposure details and nature of the effects reported. Phase II consisted of an in-depth telephone interview using an enhanced questionnaire to investigate additional exposure details. Phase III scored the association between exposure and reported effects. Based on the data collected and analyzed, we conclude that: (1) Both in absolute number and relative to the wide distribution and use by consumers, adverse respiratory or dermal events after product exposure were rare; (2) Most outcomes for the reported events involving either dermal or respiratory effects were of minor severity and self-limiting; (3) None of the data collected and analyzed indicate that pyrethrins-containing products, including those formulated with synthetic pyrethroids and/or synergists, pose a significant risk of serious dermal or respiratory reactions even in cases where the exposed individual reported having allergies or asthma; (4) No additional label warnings or other mitigation techniques are warranted with pyrethrins-containing products formulated with or without synthetic pyrethroids and/or synergists.
Introduction
Origins of the Pyrethrins Stewardship Program
Compositae (or Asteraceae) is a diverse plant family that includes the species Chrysanthemum cinerariifolium. Pyrethrum is the extract that is derived from the flowers of this plant. Subsequent extraction and refinement steps result in pyrethrins, the six insecticidally active components of the extract, which are categorized into two groups, Pyrethrins Type I and Pyrethrins Type II, based on their biologically active esters. The Pyrethrins Stewardship Program (PSP) was established to evaluate adverse events following exposure to insecticidal consumer products containing pyrethrins Chemical Abstracts Service ((CAS) Registration Number 8003-34-7), also known as “Total Pyrethrins.”
Numerous case reports of dermatitis and occasional reports of respiratory reactions associated with pyrethrins have appeared in the medical literature over the last decades.1–8 Several more recent publications reported analyses of aggregated case reports from consumer and/or occupational uses of pyrethrins, often also in the presence of pyrethroids.9–12 Pyrethroids are synthetic esters derived from the naturally occurring pyrethrins and are commonly used in insecticidal consumer products (with and without pyrethrins) and commercial products. Due to their chemical modifications these compounds tend to be more acutely toxic and generally less degradable in the environment. 13
Though some literature reports have suggested an association between exposure to pyrethrins and adverse dermal and respiratory responses, a cause-and-effect relationship has not been established and case reports often lack exposure details that would help elucidate the relationship between exposure to a purported causal agent and reported adverse effects. In those instances where individuals with asthma or other evidence of hyper-reactive airways have reportedly experienced adverse effects, it is possible that other product-related factors such as the delivery system (e.g., aerosol and fogger) or other formulation components may be responsible for or significantly contributed to the responses observed. Similarly, comprehensive evidence-based reviews of these anecdotal reports failed to establish a causal association between pyrethrins and either Type I hypersensitivity—contact urticaria or Type IV hypersensitivity —allergic contact dermatitis.9,14
Further, the U.S. Environmental Protection Agency (EPA) performed a comprehensive review of the available data in 2006 and, based on the weight of the evidence, concluded that: “…there is not a clear relationship between pyrethrins/pyrethroid exposure and asthma and allergies. Therefore, the Agency is not requiring additional warnings or label statements specific to asthmatics on pyrethroids and pyrethrins end-use product labels at this time.”
15
To better understand the potential for adverse dermal and respiratory effects from pyrethrins-containing products, as a condition of the Reregistration Eligibility Decision (RED) for Pyrethrins, EPA requested the Pyrethrin Joint Venture (PJV) 1 to commit to a “product stewardship program that would involve outreach to physicians and Poison Control Centers (PCCs) with better guidance and diagnostic standards, as well as prospective in-depth follow-up of reported cases.” 15
The goal of the Product Stewardship Program (PSP) was to better understand potential predisposing factors that might influence the likelihood of an adverse effect after exposure. Given that both dermal and respiratory symptoms are relatively common findings among individuals who experience adverse clinical effects after exposure, it was important to determine if these individuals would benefit from additional label warnings or other mitigation measures. Assessing the contribution of predisposing factors to the incidence of adverse events was paramount to determining whether label warnings might help prevent serious outcomes in populations with pre-existing conditions (e.g., allergy and asthma).
The PJV tasked SafetyCall International, Limited Liability Corporation (LLC) (“SafetyCall”), 2 an industry PCC and Science Strategies, LLC, a toxicological consultancy, to administer the PSP with SafetyCall to serve as the central medical call center managing incident data involving pyrethrins-containing products. SafetyCall was chosen for this function due to its ongoing relationship with many of the PJV member companies for which SafetyCall provides adverse event management and post-market surveillance support. As such, SafetyCall’s call center was already responding to and managing reports of exposures involving the targeted group of products. SafetyCall also has a long-standing history of involvement in supporting manufacturers in the reporting of adverse events to regulatory agencies including reporting of Federal Insecticide, Fungicide and Rodenticide Act (FIFRA) section 6(a) (2) incident data to the EPA.
Development of the PSP
A five-member Medical Advisory Council was established in 2009 to help create the framework and scientific elements of the PSP. It consisted of independent advisors selected for their expertise in pertinent disciplines such as dermatotoxicology, pharmacovigilance, and/or respiratory toxicology (including asthma and allergy). The PSP formally began in April 2010 (considered a pilot year) and ran continuously through the end of the calendar year 2015.
A follow-up meeting of the Medical Advisory Council was held in October 2016 in San Francisco to review aggregate findings of the PSP and discuss any special analyses to be considered. This discussion led to a refinement of the classification of respiratory symptoms of allergy and asthma and the subsequent statistical analysis.
Nomenclature
We defined the following terms and definitions that are used throughout the PSP: 1. Adverse event: an exposure resulting in unintended or unwanted adverse effects affecting an individual after exposure to a pyrethrins-containing product; 2. Call: contact with an individual reporting an incident involving a pyrethrins-containing product; 3. Caller: an individual reporting an exposure and/or adverse event to a pyrethrins-containing product, either self-exposed or reporting for an exposed individual; 4. Case: an exposure report focused on a single individual exposed to a pyrethrins-containing product during an incident; 5. Completers: Eligible individuals who completed the Enhanced Questionnaire and were included in the detailed case review phase of the PSP; 6. Eligible individuals: individuals who reported at least one dermal or respiratory symptom believed to have resulted in at least a minor or greater outcome; 7. Incident: a single event or exposure scenario involving one or more individuals exposed to a pyrethrins-containing product during the project duration. A single case report was generated for each exposed individual involved in the incident.
Methods
Three Phases of the PSP
The PSP consisted of three phases, each of which had certain requirements for entry and each of which served a different purpose. Pyrethrin Joint Venture member companies agreed to submit all reports of exposure to their pyrethrins-containing products for potential inclusion in the PSP. Such reports are usually made using a toll-free telephone number on product labels and generally came from: 1. Exposed subject and/or reporter directly reported to the company (via online reporting forms, by email, regular mail, or social media); 2. Marketers of sub-registered products who forwarded reports to the company; 3. Exposed subject and/or reporter who contacted a public PCC; 4. Exposed subject and/or reporter who contacted an adverse event management entity such as SafetyCall.
Submitted incidents were promptly evaluated by SafetyCall healthcare professionals (at least 2 years of experience in adverse event management and documentation) to see if they met the inclusion criteria, and if so, were manually entered into a proprietary online database.
Phase I: assessment of eligibility: intake of incidents and initial assessment of outcome
The screening criteria for including a case for the initial evaluation were: 1. SafetyCall’s healthcare professionals established that the subject was exposed to a pesticide product containing pyrethrins (the product formulation was identified by the EPA Registration Number); 2. Sufficient details (e.g., name and telephone number) were available to allow follow-up with the person reporting the incident.
SafetyCall’s healthcare professionals reviewed the details of the case including the nature of the effects reported and the overall quality of the report. All cases of moderate or higher severity were assessed by a clinical toxicologist. The primary criterion for inclusion in the PSP and subsequent follow-up was that the caller reported at least one dermal or respiratory symptom in a human subject believed to have resulted in at least a minor or greater outcome consistent with the American Association of Poison Control Centers (AAPCC) medical outcome criteria for human incidents as defined below
16
: 1. No effect: The patient did not develop any signs or symptoms as a result of the exposure. 2. Minor effect: The patient developed some signs or symptoms as a result of the exposure, but they were minimally bothersome and generally resolved rapidly with no residual disability or disfigurement. A minor effect is often limited to the skin or mucus membranes (e.g., self-limited gastrointestinal symptoms, drowsiness, skin irritation, first-degree dermal burn, sinus tachycardia without hypotension, and transient cough). 3. Moderate effect: The patient exhibited signs or symptoms as a result of the exposure that were more pronounced, more prolonged, or more systemic in nature than minor symptoms. Usually, some form of treatment is indicated. Symptoms were not life-threatening, and the patient had no residual disability or disfigurement (e.g., corneal abrasion, acid-base disturbance, high fever, disorientation, hypotension that is rapidly responsive to treatment, and isolated brief seizures that respond readily to treatment). 4. Major effect: The patient exhibited signs or symptoms as a result of the exposure that were life-threatening or resulted in significant residual disability or disfigurement (e.g., repeated seizures or status epilepticus, respiratory compromise requiring intubation, ventricular tachycardia with hypotension, cardiac or respiratory arrest, esophageal stricture, and disseminated intravascular coagulation). 5. Death: The patient died as a result of the exposure or as a direct complication of the exposure.
Incident reports that met these criteria were collected as part of Phase I. In each case, SafetyCall attempted to contact the caller regarding participation in a more focused interview (Enhanced Questionnaire) and investigation of the reported adverse event(s). The participation in the questionnaire was encouraged by offering financial incentive (initially set at $100 and then increased to $200 by 2012) which was designed to compensate participants for their time in completing the questionnaire. Additionally, the offer of compensation for eligible participants was added to voice mail messages left with the individuals if they could not be reached on the first follow-up call. This resulted in a significantly higher participation rate in later years of the program (2014 and 2015). Upon participation agreement, an informed consent was verbally obtained and the case formally entered into Phase II of the PSP.
Phase II: in-depth review and follow-up of Phase I incidents
Phase IIa: administration of the Enhanced Questionnaire
SafetyCall conducted telephone interviews with the callers using the PSP Enhanced Questionnaire (see supplemental online Appendix 1). The questionnaire was developed with input from the Medical Advisory Council and with the intent to collect pertinent quantitative and qualitative information on exposure details including potential confounding factors such as exposure to cosmetics, household or other pesticide products. Callers were asked open-ended and open response-option questions. The interviewers were SafetyCall healthcare professionals with at least 2 years of working experience in the PCC setting and were trained to administer the questionnaire in a consistent fashion and able to answer requests for clarification if they occurred. Exposed individuals who completed the Enhanced Questionnaire formed the group of so-called “completers” and were included in Phase III.
Phase IIb: diagnostic evaluation of reported dermal effects
Following completion of the Enhanced Questionnaire, we evaluated the strength of association between product exposure and reported adverse effects. Because of their long history working with pyrethrins, members of the PJV are in a unique position to provide diagnostic guidance to physicians treating an individual with dermal symptoms. This diagnostic guidance is important to address some of the inconsistencies noted in diagnostic procedures associated with cases reported in the literature and to better understand the relationship between exposure and the reported dermal effects. Thus, a test kit containing a pyrethrins sample, guidance for patch testing (for diagnosis of allergic contact dermatitis) as well as prick testing (for diagnosis of contact urticaria) was available from the PJV for testing at the discretion of the caller’s physician.
The primary information used in assessing respiratory effects came from the Enhanced Questionnaire. We did not recommend nor provide diagnostic guidance for the conduct of controlled inhalation exposures with callers reporting respiratory issues. For those individuals who in consultation with their healthcare practitioner would have elected to undergo the skin prick test, it was expected that the test would have detected the presence of IgE-mediated allergens including those affecting the upper respiratory tract.
Phase III: assessment of association between exposure and reported effects
Phase IIIa: review of completer cases
The narrative (incident history) of each completer case was reviewed to ensure that reported symptoms were consistent with symptoms typically associated with the conditions of respiratory (asthmatic) and allergic reactions (see supplemental online Appendix 2—based on personal communication with Dr D Bernstein).
Phase IIIb: Association Score
Incidents that are more likely than others to potentially represent either a causal relationship or association must be separated from reports of injury or illness that may be more representative of “background noise” (loosely defined as coincidental or temporal occurrences or allegations of unrelated illness). This ultimately requires the stratification and scoring of individual incidents to determine potential association between product exposure and reported adverse events. Once identified, these incidents can then be subjected to more intense investigation and comparison to other similarly scored events in an effort to prevent injury and mitigate risk. No other direct-from-consumer system of surveillance currently in place provides a framework for differentiating the potential validity of one spontaneously reported adverse event report from another. Without the ability to differentiate reports, all reports are given equal weight which reduces the likelihood of identifying actionable post-market surveillance trends.
To initiate a process of report differentiation, individual incidents must be evaluated to assess the strength of standard factors for association of product and adverse event including 1.temporality; 2.biological, physiological, or pharmacological plausibility; 3.de-challenge; 4.re-challenge; and 5.confounding factors.
Establishment of a post-market surveillance and scoring algorithm for evaluation of cases was the subject of intense effort prior to the beginning of the program in 2010 and involved consultation with a variety of experts including those in the Office of Pesticide Programs at the EPA.
The “Association Score” was developed by SafetyCall to aid in the evaluation of spontaneously reported data from multiple consumer product market sectors. This is discussed in greater detail elsewhere. 17 Briefly, it embodies many of the elements contained in foundational publications by Kramer et al. (1979, 1980)18,19 and Naranjo et al. (1981) 20 that set out basic principles for the assessment of adverse drug reactions or the Bradford Hill criteria (1965) 21 that identify factors for assessing causation/association between the environment and disease as determined by trained medical observers. Our Association Score is a tool designed to provide a simple, standardized method to score individual incidents with the purpose of assessing the correlation or strength of potential associations between product exposure and reported adverse effects. It allows the identification of incidents of special interest that may require further investigation, such as a dermal diagnostic testing, to more definitively assess potential association between product use and reported symptoms, and as such, is a component vital to the PSP. As with most, if not all assessment tools used in assessing spontaneously reported events, it is typically not able to determine causation given the limitations of self-reporting.
Association scoring
The criteria are evaluated using six questions, one for each criterion (see supplemental online Appendix 3). The resulting numerical total score is referred to as Association Score. Association Scores were determined by a clinical toxicologist on an ongoing basis following the conclusion of Phase IIIa.
The six item scores are categorized into three groups assessing the possibility that the incident described is associated with the use of and/or exposure to a pyrethrins-containing product. Incidents receiving scores from 0 to 2 are considered unlikely and having weak associations, those receiving scores of 3–4 are possible associations, and those receiving scores of 5–6 were adverse events probably associated. 1. 0–2 Points: Unlikely Association: Incident details suggest that an association between a properly manufactured product and the reported effect(s) is considered unlikely, or nonexistent. 2. 3–4 Points: Possible Association: An association between the product use and the effect cannot be ruled out but the remains ill-defined or questionable. 3. 5–6 Points: Probable Association: Incident details and circumstances as reported suggest that an association likely exists between the product exposure and reported effect. In such cases, continued assessment of the association between product and event is warranted.
Phase IIIc: statistical analysis
Most of the data reported in the PSP are descriptive in nature and stand without the need for statistical analysis. However, a few questions are central to the understanding of the data and are informed by statistical analysis. These include: 1. Are exposed individuals with known asthma more likely to show respiratory symptoms than those without asthma? 2. Are exposed individuals with known respiratory allergy more likely to show respiratory symptoms than those without known respiratory allergy?
We were interested in testing the following associations: 1. Asthma status versus presence of asthma-associated symptoms; 2. Asthma status versus outcome severity of case; 3. Allergy status versus presence of allergy-associated symptoms; 4. Allergy status versus outcome severity of case; 5. Smoking status versus outcome severity of case; 6. Presence of pyrethroid in addition to pyrethrins versus outcome severity of case.
The chi-square test 22 was used to determine if statistically significant associations exist between selected demographic and case characteristic variables and the self-report of asthma, allergy, or being a smoker. Individuals reporting an asthma or allergy status of “unknown” were excluded from the chi-square analysis. A p-value of p < 0.05 was chosen a priori to indicate significance for all tested variables.
A multivariate analysis technique, logistic regression, was also used to examine the relationship between case severity and (a) the self-report of allergies and (b) the self-report of asthma. For each model, independent variables included demographic variables (age and sex), smoking status, a seasonal indicator related to call date, and product use. A p-value of < 0.05 was chosen a priori to indicate significance for any independent variables.
Results
A total of 4558 cases (i.e., reports of human exposure to a pyrethrins-containing insecticide) that were received during the years 2010–2015 were referred to the PSP for evaluation. These cases represented 1919 exposed individuals of which 1905 individuals were eligible (with at least one dermal or respiratory symptom believed to have resulted in at least a minor or greater outcome) for inclusion in the PSP. As mentioned earlier, the program was initiated in April 2010 and the lower number of exposed individuals for the year 2010 compared to the year 2011 reflects this. We are not certain for the reasons for the fluctuation in the number of exposed individuals during the subsequent program years. Nothing remarkable can be said about qualitative or quantitative changes in responses over time and this aspect was not considered further.
Cases by severity of exposure (n = 4118) a by organ system for 1905 eligible individuals.
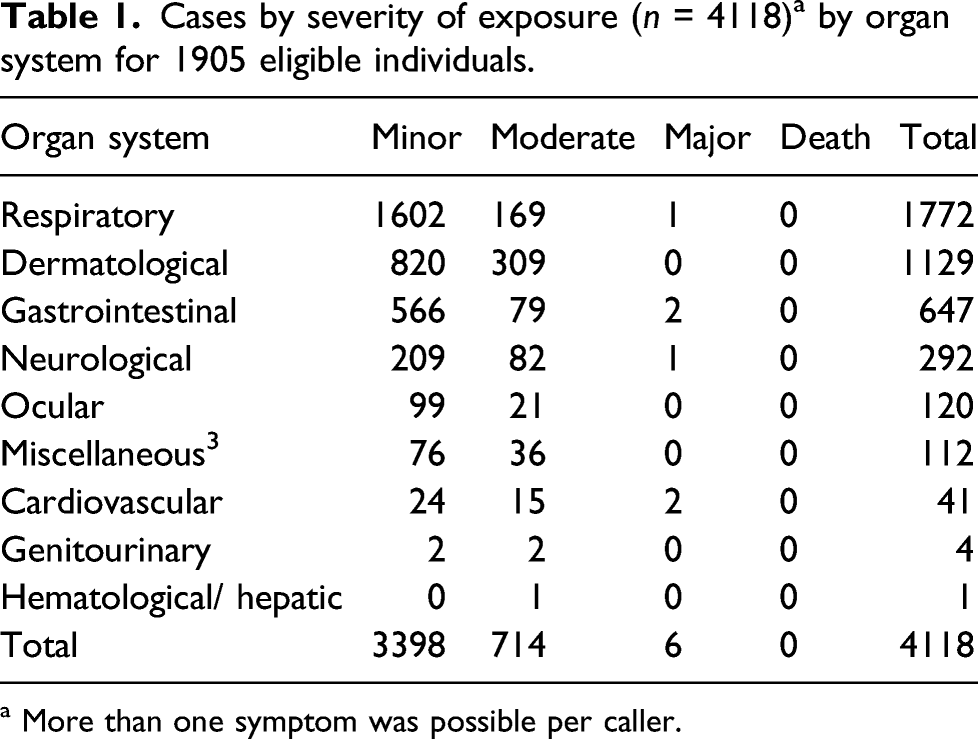
a More than one symptom was possible per caller.
Severity of outcome for eligible individuals (n = 1905).
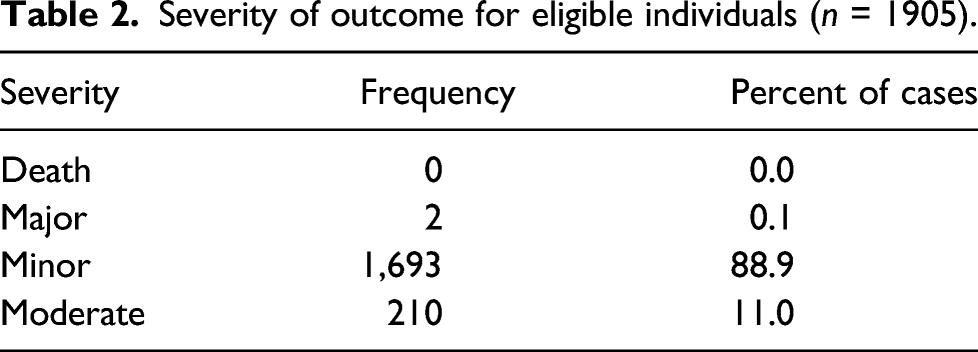
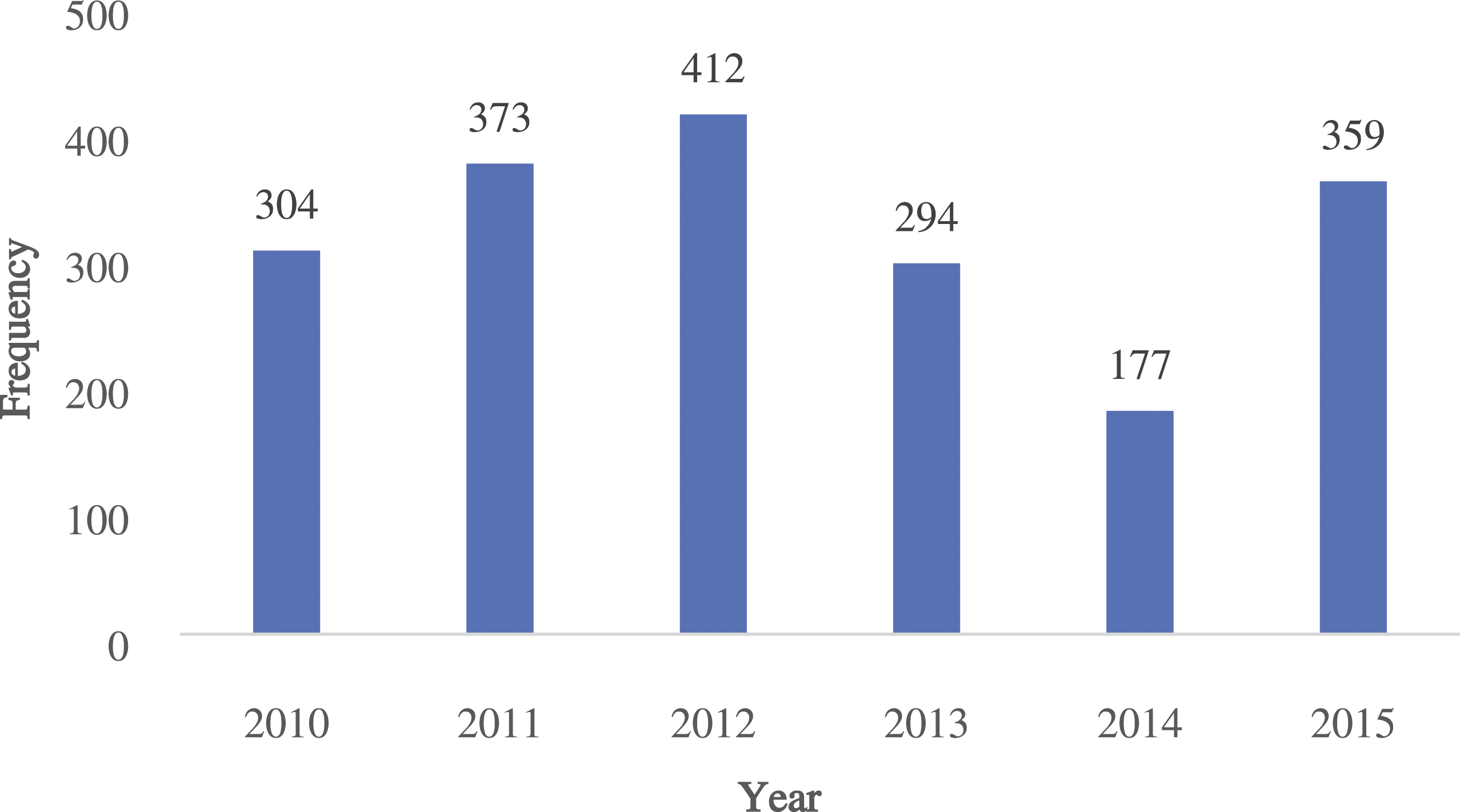
Number of exposed individuals (n = 1919).
Participation in Phase II (i.e., participation in the Enhanced Questionnaire) increased significantly over the years of the PSP (Figure 2). This reflected efforts to reach potential participants earlier after eligibility was determined and by increasing the financial incentive offered to participants. Percentage of Phase I (exposed individuals) moving to Phase II (completers).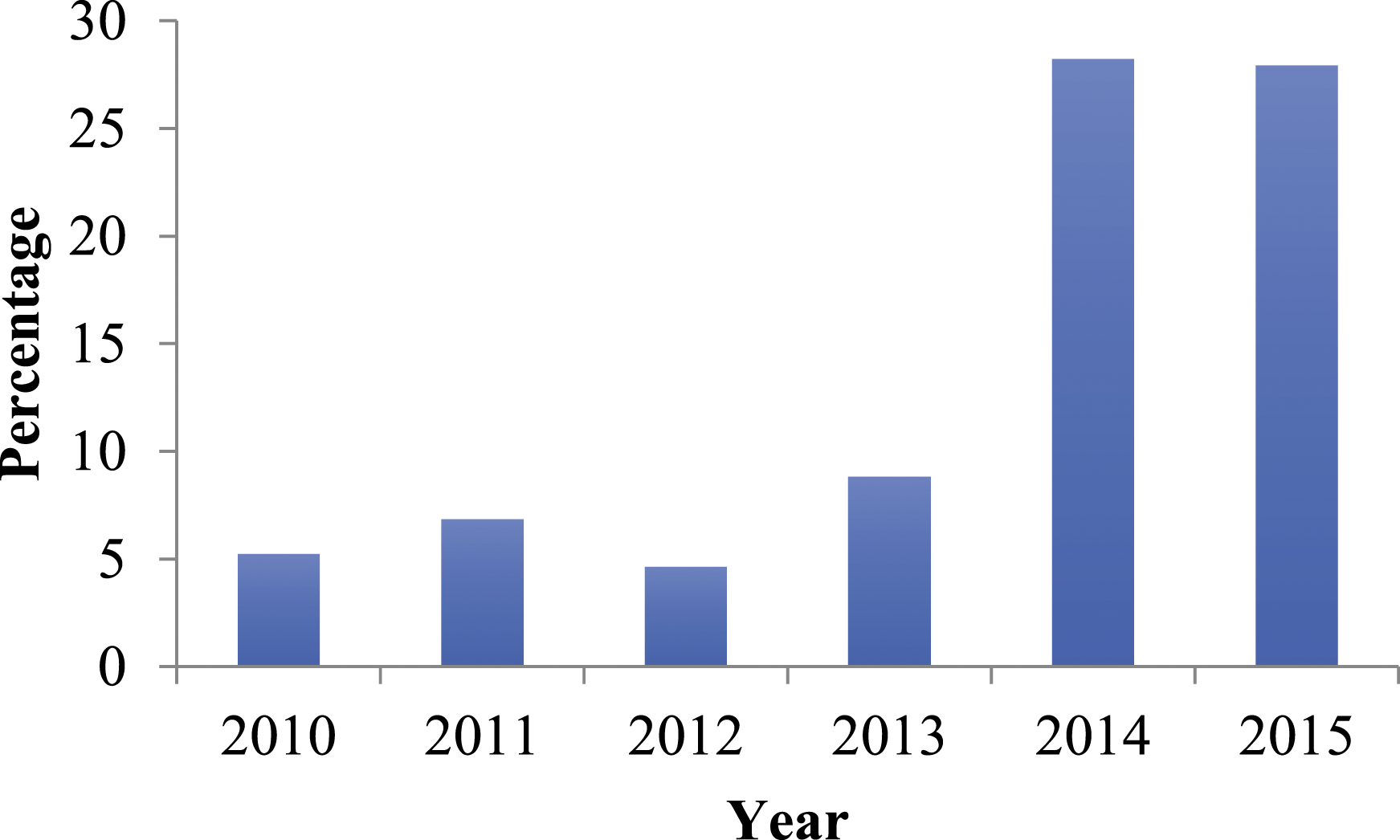
Completers by allergy, asthma, and smoking status.
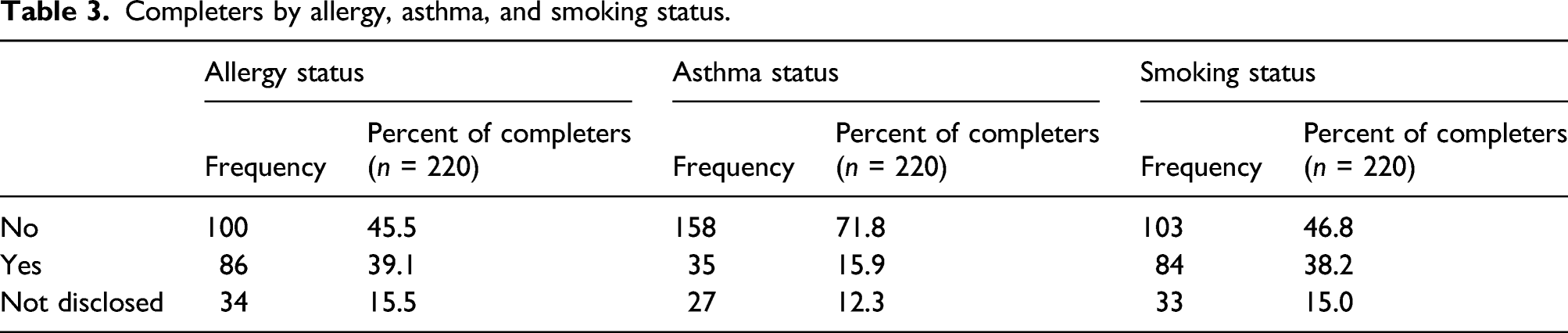
As expected, characteristics of Phase II completers were comparable to the characteristics of all exposed individuals (Phase I) in that the clear majority of completers (>66%) reported “unintentional” exposures characterized by exposures managed on site (85.5% of completers), symptoms lasting 24 h and less (>35% of completers) and had exposures with outcomes of minor severity (84.5% of completers; no major outcomes or deaths).
Products associated with the ten greatest number of reports of completers.
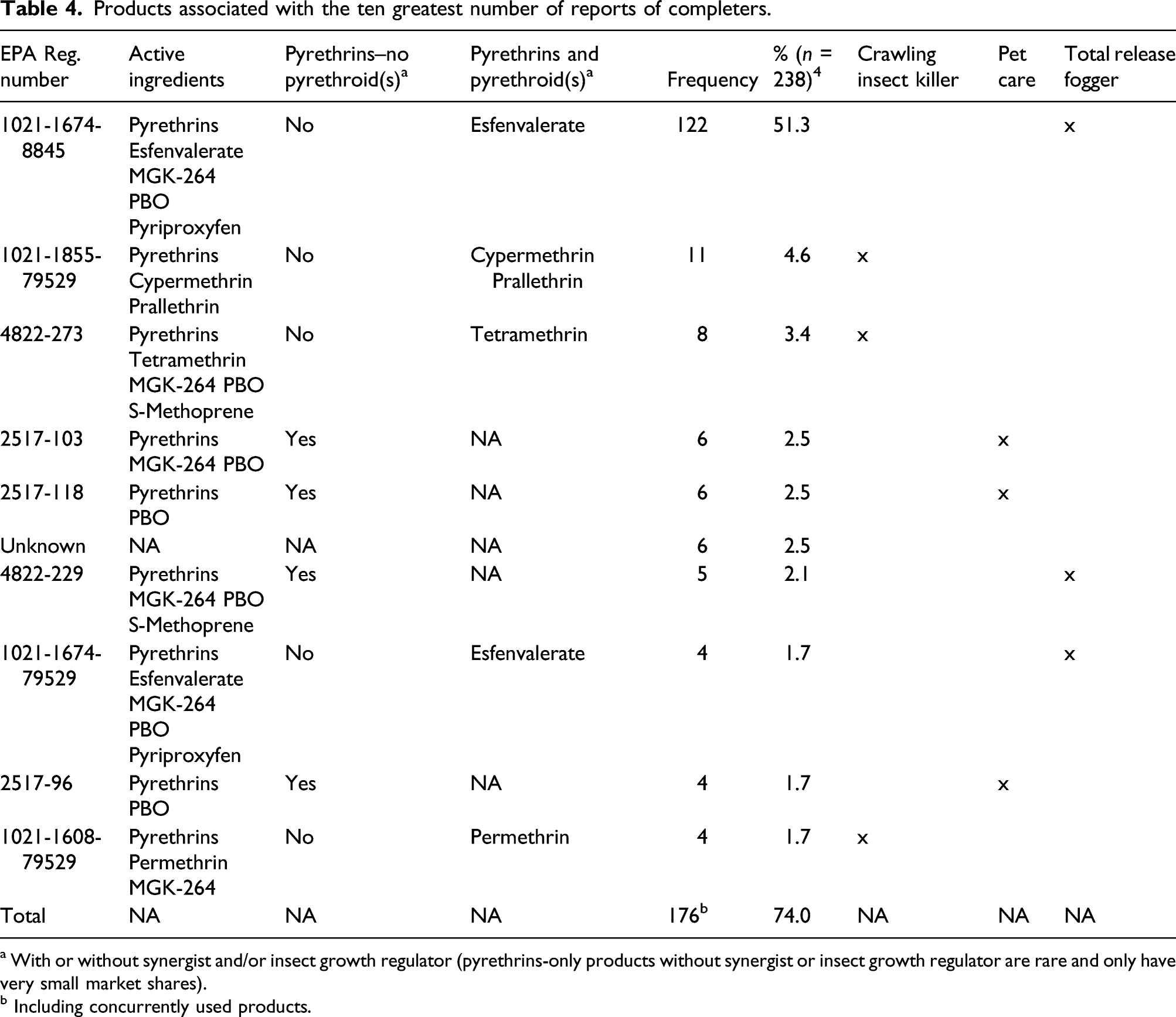
a With or without synergist and/or insect growth regulator (pyrethrins-only products without synergist or insect growth regulator are rare and only have very small market shares).
b Including concurrently used products.
The manual review of incident narratives of completer cases (Phase IIIa) indicated that individuals reporting exposures and symptoms were typically not familiar with medical diagnoses and coding and were calling in high-stress situations and reporting symptoms not always consistent with respiratory, dermal and/or allergic reactions. For example, some individuals reported emesis and cough following inhalation exposures to respiratory irritants. Emesis as a symptom is not consistent with respiratory (asthmatic) and allergic adverse effects. The detailed review of the narratives indicated that emesis typically was described as a symptom secondary to cough. Based on this understanding, it was decided to include cases such as these in the subsequent Phase IIIb (determination of the Association Score). This conservative approach resulted in the inclusion of all 220 completer cases in Phase IIIb.
Association Scores for all completer cases.
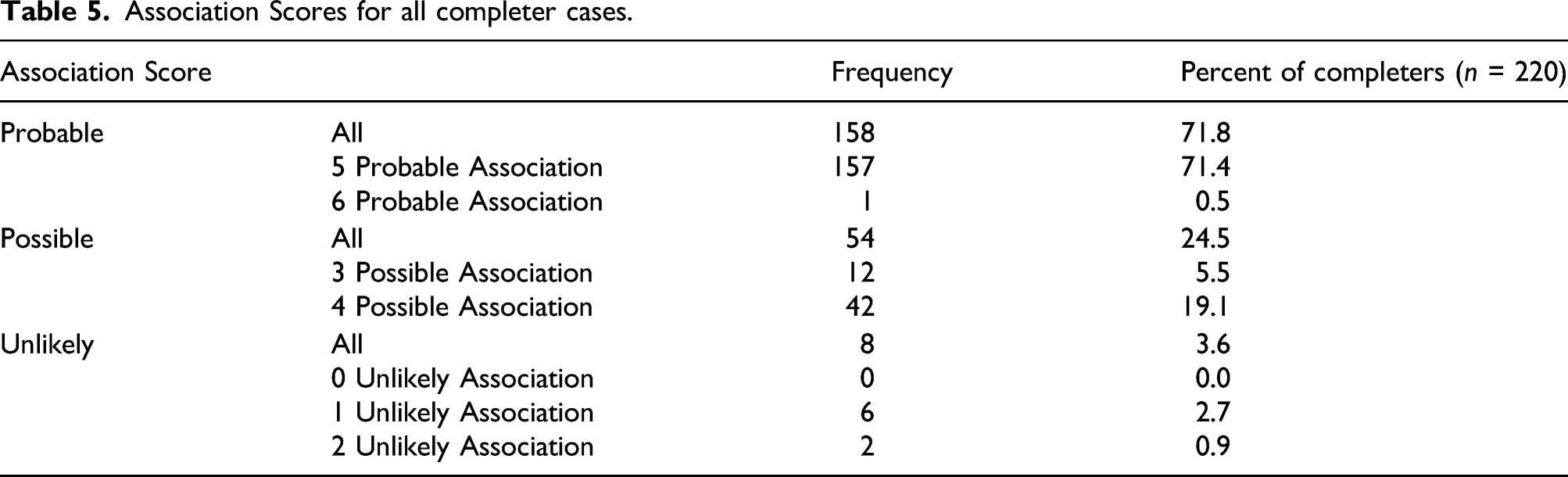
The association scores in a year-by-year comparison are presented in supplemental online Appendix 4.
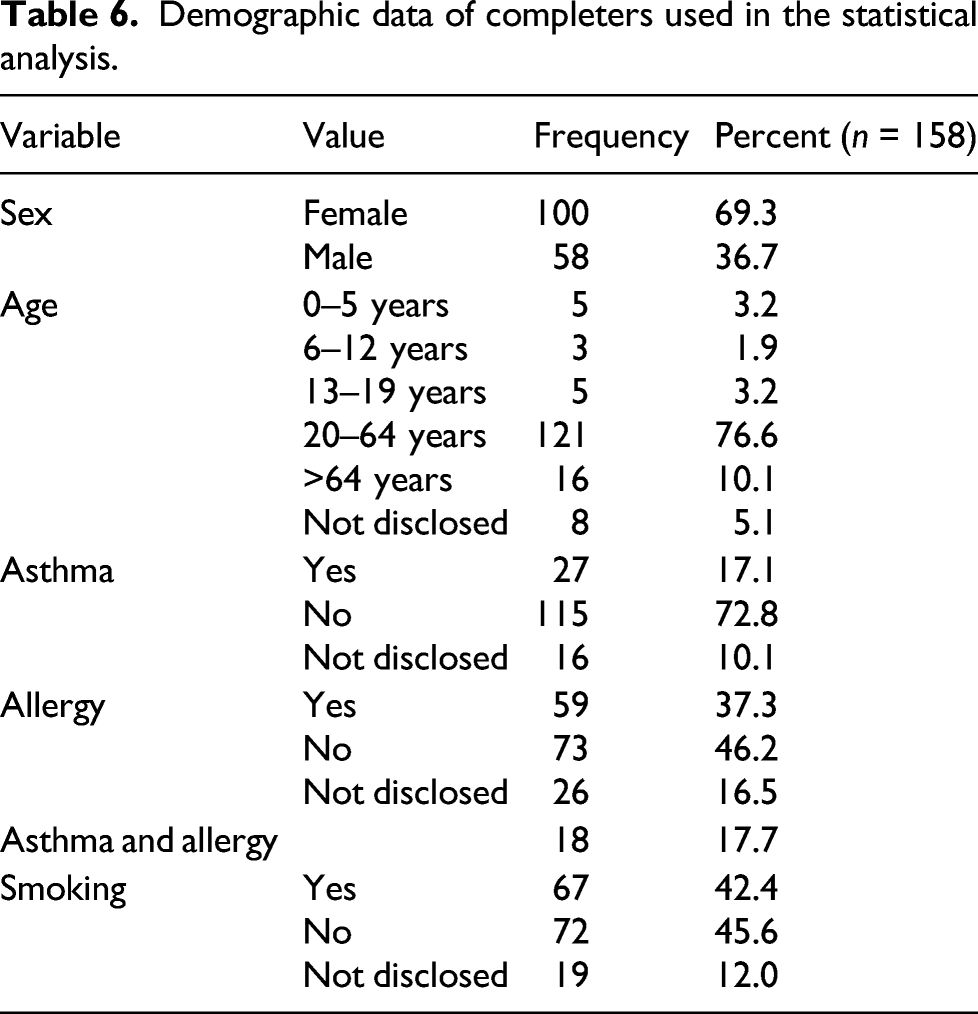
Demographic data of completers used in the statistical analysis.
Case characteristics of completers that were included in statistical analysis.
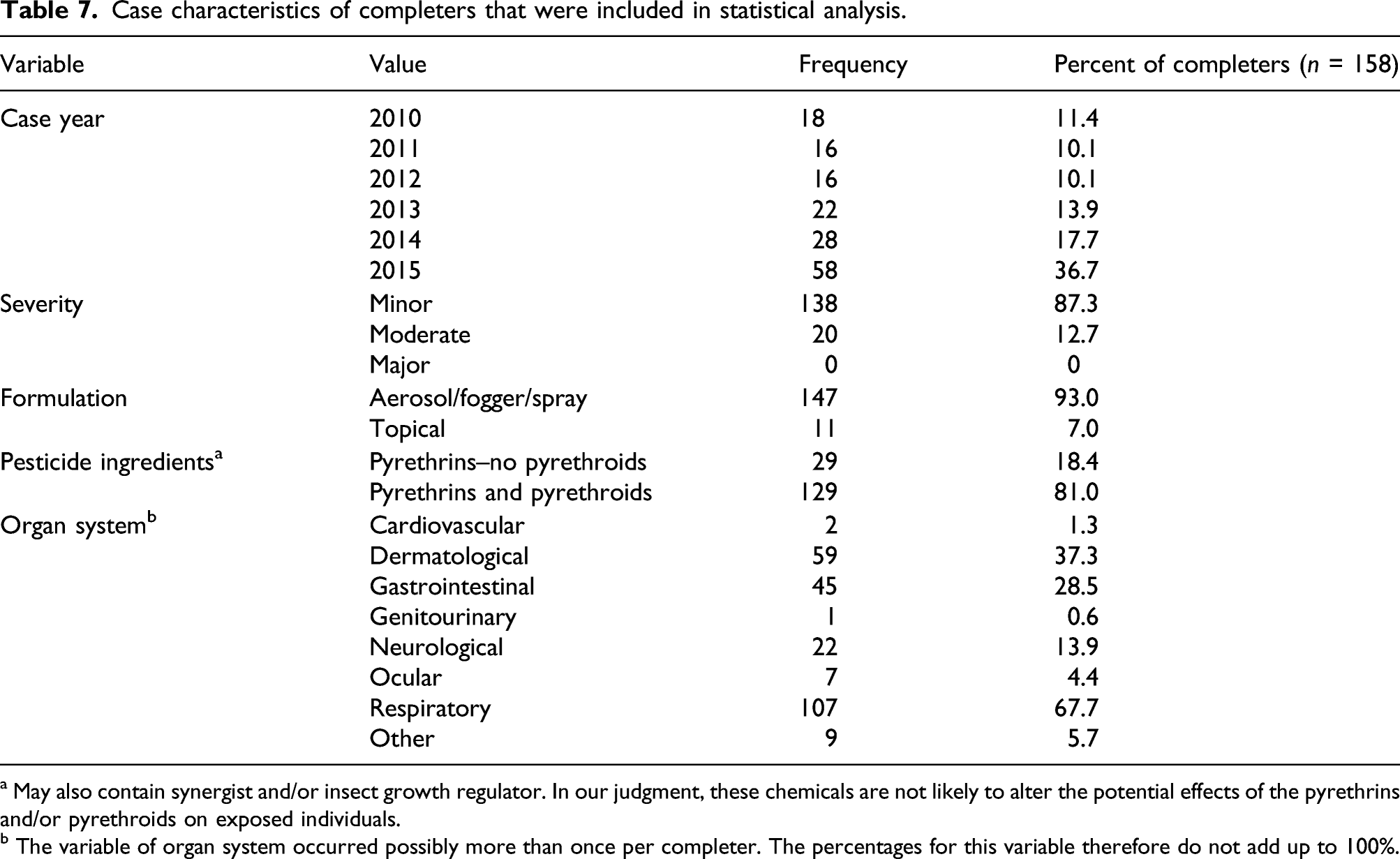
a May also contain synergist and/or insect growth regulator. In our judgment, these chemicals are not likely to alter the potential effects of the pyrethrins and/or pyrethroids on exposed individuals.
b The variable of organ system occurred possibly more than once per completer. The percentages for this variable therefore do not add up to 100%.
Severity of outcome for pesticide ingredients.
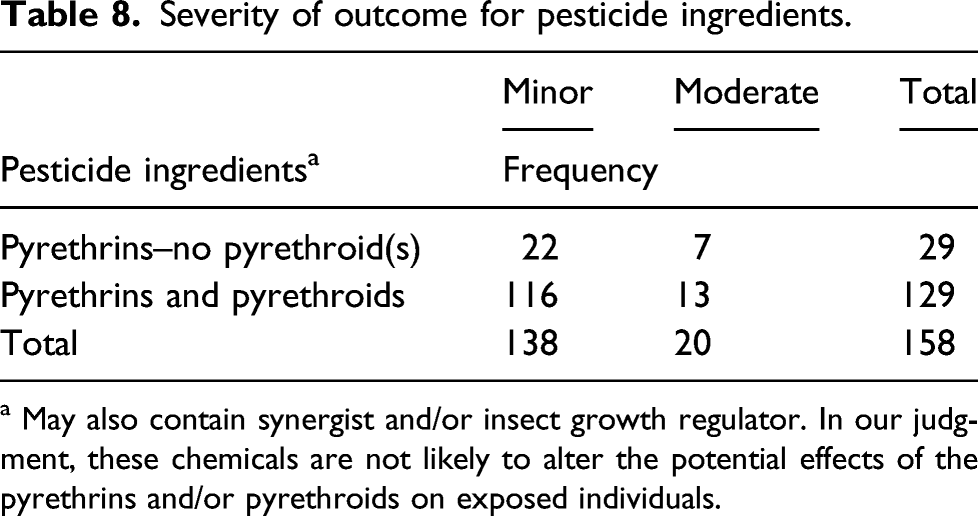
a May also contain synergist and/or insect growth regulator. In our judgment, these chemicals are not likely to alter the potential effects of the pyrethrins and/or pyrethroids on exposed individuals.
It is important to note that for both asthma and allergy status, we had to rely on the information provided by the caller over the phone. We were unable to confirm their report of their medical status and diagnoses by a physician. A substantial proportion of the callers reported not knowing their asthma and/or allergy status. The data associated with these individuals were not included in the statistical analyses pertaining to asthma and/or allergy status.
Statistical analysis of completer data
Asthma status and respiratory symptoms
The chi-square test was used to determine if there was a significant relationship between the two nominal variables of a completer’s asthma status (yes: having physician-diagnosed asthma; no: not having physician-diagnosed asthma) and reported respiratory symptoms (yes: reporting respiratory symptoms; no: not reporting respiratory symptoms).
Asthma status and respiratory symptoms.
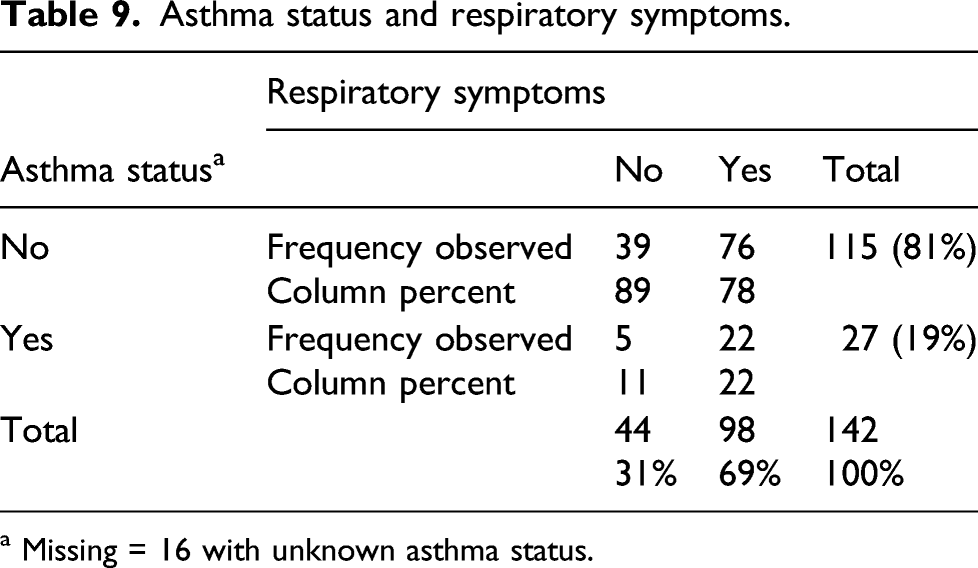
a Missing = 16 with unknown asthma status.
Asthma status and severity of outcome
Asthma status and severity of outcome.
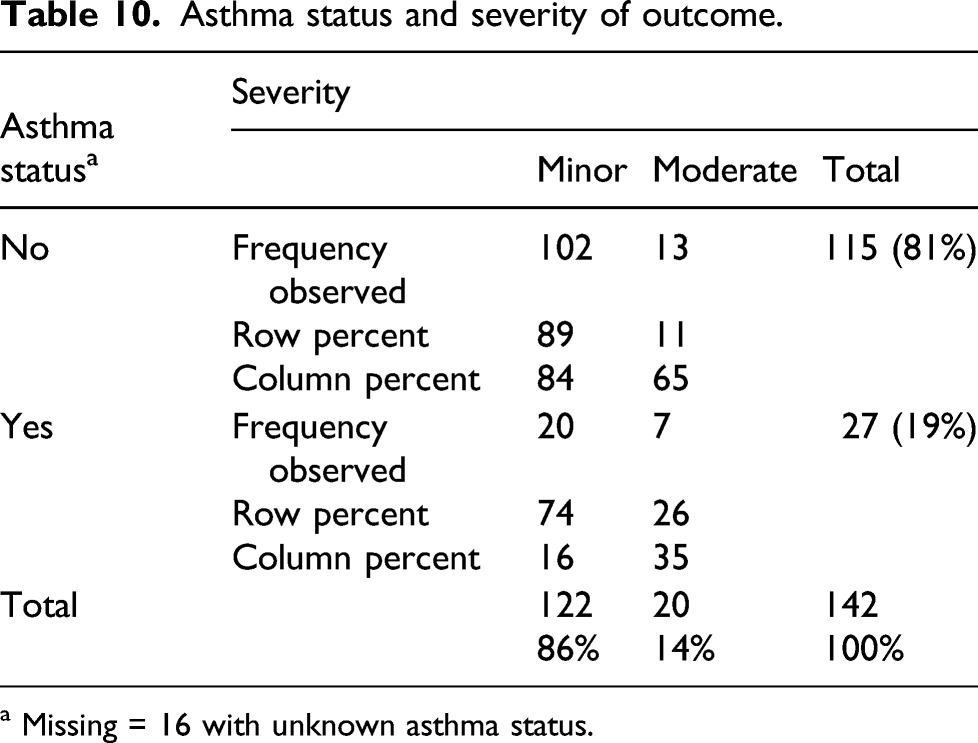
a Missing = 16 with unknown asthma status.
Allergy status and respiratory symptoms
Allergy Status and Presence of Respiratory Symptoms.
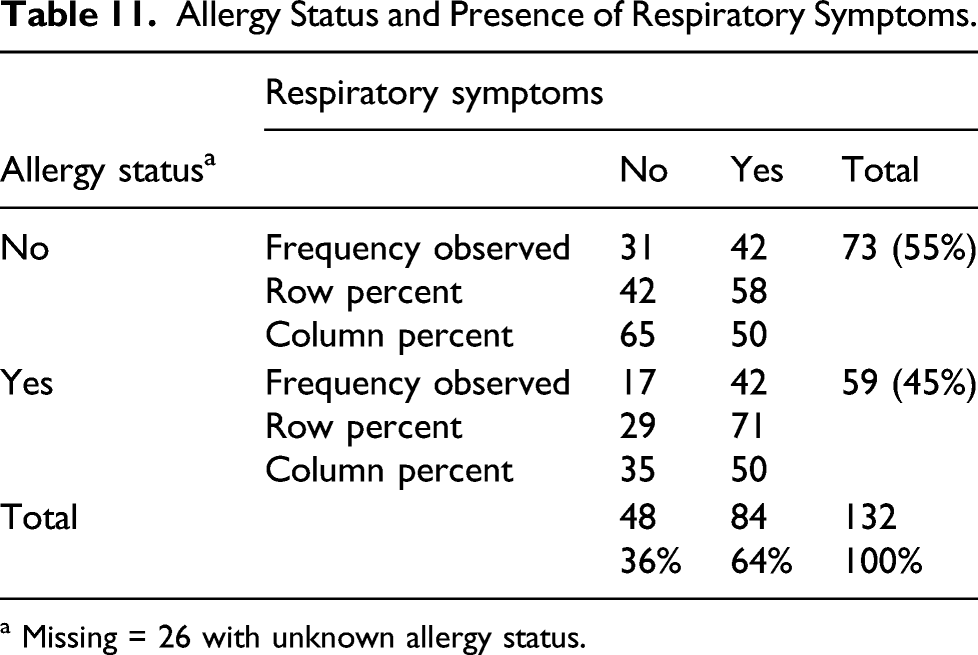
a Missing = 26 with unknown allergy status.
Chi-square test of allergy status and severity
Allergy Status and Severity of Respiratory Symptoms.
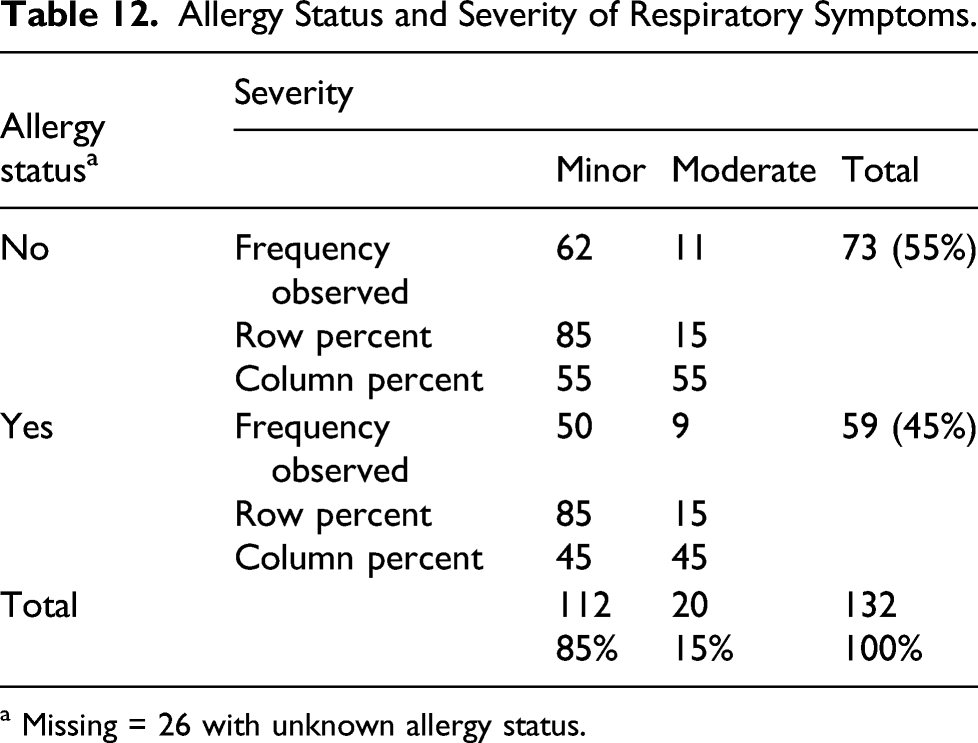
a Missing = 26 with unknown allergy status.
Chi-square test of smoking status and severity
Smoking Status and Severity of Respiratory Symptoms.
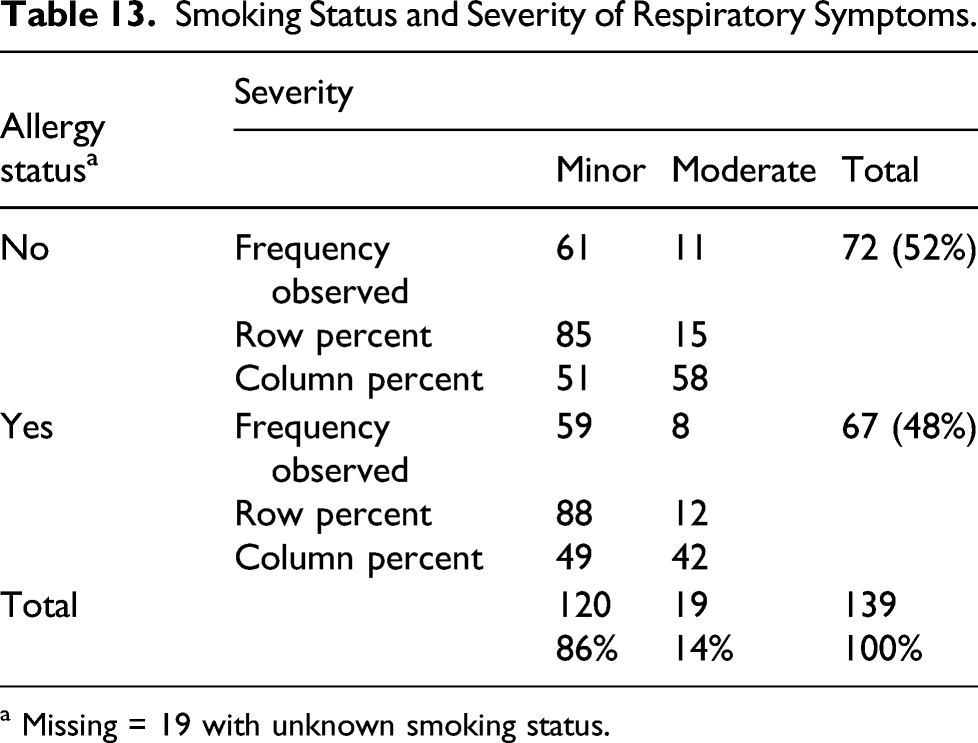
a Missing = 19 with unknown smoking status.
Chi-square test of exposure to pyrethrins with and without exposure to pyrethroids and outcome severity
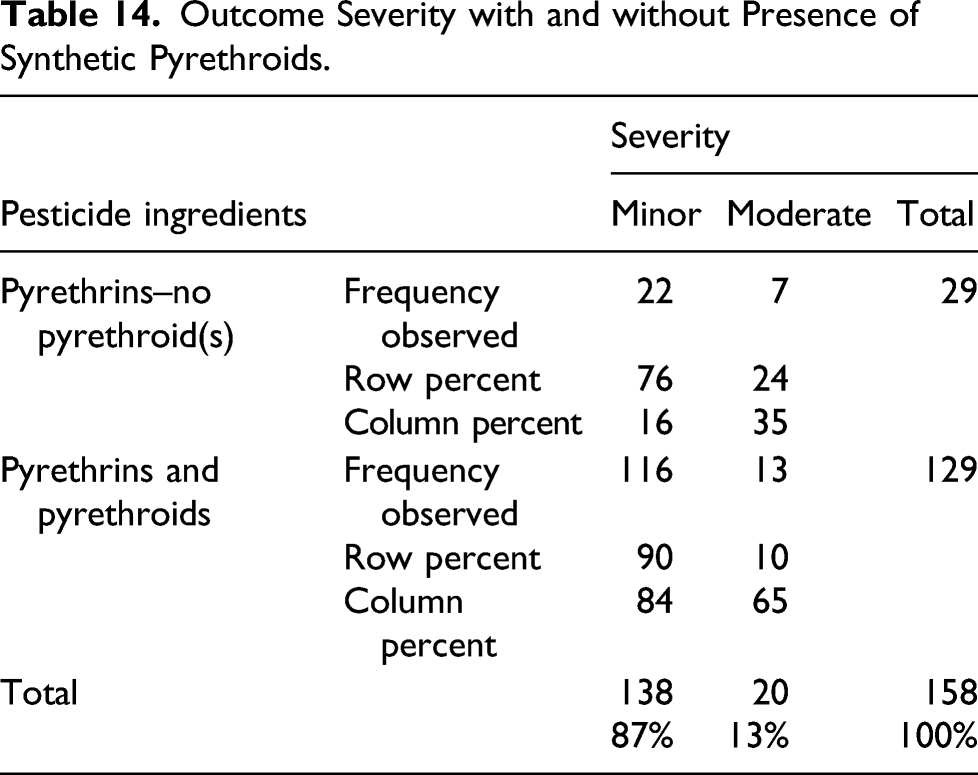
Outcome Severity with and without Presence of Synthetic Pyrethroids.
Multivariate analysis
Two logistic regression models were completed.
Model 1
This model addressed the relationship between case severity and the self-report of allergies. The dependent variable was case severity: moderate versus minor.
The independent variables included allergy status (yes/no), sex (male/female), age, smoking status, product mix (concurrent use of pyrethrins product, pyrethroid product, combination of pyrethrins/pyrethroid or products that contained pyrethrins or pyrethroid and other chemicals), and a seasonal indicator.
The model indicated no significant relationships between case severity, allergy status, or any of the other independent variables.
Model 2
This model addressed the relationship between case severity and the self-report of asthma. The dependent variable was case severity: moderate versus minor.
The independent variables included asthma status (yes/no), sex (male/female), age, smoking status, product mix (pyrethrins product, pyrethroid product, combination of pyrethrins/pyrethroid or products that contained pyrethrins or pyrethroid and other chemicals), organ system with reported symptoms, and a seasonal indicator.
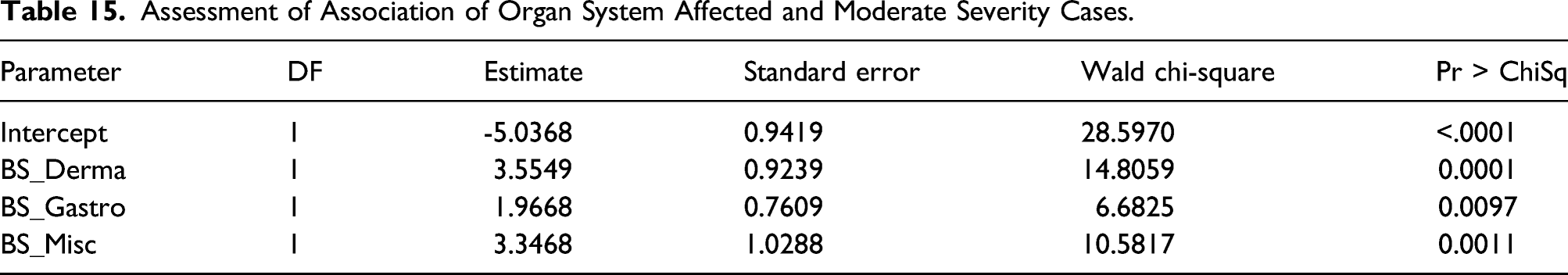
Assessment of Association of Organ System Affected and Moderate Severity Cases.
All statistical analyses were performed using SAS v9.4 (SAS Institute, Cary, NC, USA).
Discussion
Phase I
The reports collected and reviewed reflect the broad variety of products containing pyrethrins including the following residential product formulations in the marketplace: sprays (trigger and aerosol), powder, pet care products, and total release foggers. The results of Phase I show that both dermal and respiratory exposures to pyrethrins-containing products were the most common types of exposure reported to PJV member companies and accounted for 20.6% and 68.8% of all cases, respectively.
5
The most frequent reason for exposure was “unintentional” constituting 70.2% of the reported events. Despite the relative number of incidents involving both dermal and respiratory symptoms, severity outcomes related to all cases indicate that for the majority of incidents, outcomes of a minor nature were the norm with 88.9% of exposures in the PSP reporting minor symptoms.
6
This compares to a minor outcome rate of 84.2% for all symptomatic single pyrethrins exposures with known outcomes reported to poison centers that include all age groups and exposure reasons for the years 2010–2015.23–28 Walters et al. (2009) showed similar findings for 407 cases involving pyrethrins/pyrethroid exposures in Washington and Oregon. Most exposures were of low severity (92%), defined as “Low severity illness or injury usually results in three or fewer missed days, and consists of health effects that likely do not require treatment,” and most were classified as possible cases “in which the affected individuals had health effects that were consistent with the current toxicological information on the pesticide” (73%). The most common category of clinical signs and symptoms was respiratory (52%), followed by neurological (40%).
11
These results are also consistent with the findings of Power and Sudakin
10
who analyzed pyrethrin and pyrethroid insecticide exposure data from the AAPCC Toxic Exposure Surveillance System (TESS) annual reports from 2000 to 2005 and concluded: “The majority of pyrethrin and pyrethroid exposures in this study resulted in minor or no symptoms. The fact that the majority of incidents were accidental and not intentional is a possible explanation for the low severity of clinical outcomes. The total number of pyrethrin and pyrethroid cases with major medical outcomes and mortality were very low and consistent with the low toxicity of pyrethrins and pyrethroids to humans. These findings suggest that the increase in incidents has not resulted in a significant public health impact in terms of major morbidity and mortality.”
10
Hudson et al. (2014) reviewed incident data on pyrethrins and pyrethroids from the Sentinel Event Notification System for Occupational Risk (SENSOR)–Pesticides Program and the California Department of Pesticide Regulation from 2000 to 2008 and reported that 85% were of low severity. 12 Low severity here was defined as “illness/injury usually resolves without treatment and there are fewer than 3 days lost from work” based on standardized criteria for state-based surveillance programs.
Phase II
Participation rate
One constant challenge we faced was to maximize the proportion of individuals captured in Phase I that consented to participate in the Enhanced Questionnaire (Phase IIa). Research shows that response rates for other types of telephone surveys have dropped significantly over the last two decades. The Pew Research Center reports that the response rate (percent of households called that yielded an interview) for opinion surveys dropped from 36% in 1997 to 15% in 2009 to just 9% in 2015. 29 The low response rate was therefore not unexpected. Our assumption was that individuals experiencing either significant or prolonged adverse effects secondary to product exposure would be motivated to learn about possible contributing factors so that they might avoid similar reactions in the future. The low response rate may reflect the fact that individuals’ experiences have been unremarkable and that agreeing to a follow-up interview to complete an extensive questionnaire for such minor incidents apparently did not seem worthwhile. We tried to incentivize participation by offering compensation for their participation. Compensation was initially set at $100 and was increased to $200 by 2012 where it remained for the remainder of the project. Additionally, the offer of compensation for eligible participants was added to voice mail messages left with the individuals if they could not be reached on the first follow-up call. This resulted in a significantly higher participation rate in 2014 and 2015 (>20%) than in prior years (<10%).
Product characteristics
Only 43 of 238 products that 220 completers were exposed to were products with pyrethrins as the only insecticide present in the formulation. This is not surprising, as pyrethrins-only products are uncommon in the marketplace. Of the 238 products, the majority (186) contained a synthetic pyrethroid, most commonly the alpha-cyano-containing esfenvalerate (142 cases). This is also reflected in the products that were associated with the 10 greatest number of reports among completers; that is, the majority of the cases (131) were a result of exposure to total release foggers with one single pyrethrins/esfenvalerate/MGK-264/PBO/pyriproxyfen-containing product (two EPA subregistrant numbers) accounting for 126 cases. Thus, many of the incidents that were reported cannot necessarily be attributed to pyrethrins. For example, the preponderance of cases associated with products containing pyrethrins with the synthetic pyrethroid esfenvalerate is notable.
The EPA recently reviewed human incident data for products containing esfenvalerate. While a “moderately high number of esfenvalerate incidents” were collected by their Incident Data System (IDS), “the substantial majority (94%) of these incidents reported to IDS were either classified as minor severity or had no or unknown effects.” Minor severity was defined as “a person alleged or exhibited some symptoms, but they were minimally traumatic, the symptoms resolved rapidly and usually involved skin, eye or respiratory irritation.” 30 Also of interest, the Evercide® Total Release Aerosol 2614 (EPA Registration Number 1021-1674) was associated with 56% of the esfenvalerate incidents reported to IDS (n = l404). Of these 1404 incidents, 1322 were minor in severity and 82 were moderate in severity. Similarly, 82% of esfenvalerate cases identified in the SENSOR-Pesticides Program were of low severity. Based on the EPA’s assessment of all of the available data, no mitigation measures for esfenvalerate-containing products (e.g., additional precautionary labeling) were proposed.
Characteristics of adverse effects
As was the case for the initial collection of cases in Phase I, the overwhelming proportion of exposures resulted in minor effects. This confirmed our past review of PCC incident reports that showed the majority of cases evaluated were of minor severity. 31 This is not unexpected, as with dermal exposure, for example, it is routine to observe a temporal relationship between direct skin contact with a product not intended for direct skin contact and dermal irritation and/or rash. In contrast, persistent, debilitating or otherwise serious dermal effects (outcome of major or death) after pyrethrins-containing product exposure were nonexistent in the PSP population.
Put into context with the many product applications per year, the overall incidence rate of either dermal or respiratory effects is low. It was previously estimated that there were 50,000,000 applications of pyrethrins-containing products each year from 2001 to 2003. 32 It should also be noted that the database of adverse events reported directly to the manufacturer appears to be capturing the largest percentage of such reported exposures resulting in either dermal or respiratory effects. Therefore, if individuals were experiencing either dermal or respiratory effects in a disproportionately high number of routine product applications, it would be represented in this dataset. Additionally, if individuals were experiencing more serious or debilitating respiratory or dermal effects, it would be reflected in the relative distribution of severity outcomes in the population identified here.
Asthma and allergy status of completers
As reported above, about 39% and 16% of the completers reported being diagnosed with allergies or asthma, respectively (Table 3). National prevalence rates for asthma and allergies as determined in the 2016 National Health Interview Survey 7 (NHIS) through personal household interviews were 8.3% for current 8 asthma and 6.2% for hay fever (allergic rhinitis diagnosed in the past 12 months) for adults aged 18 and over. 33 Caution must be exercised as the questions asked and the methods of the actual review likely differ, affecting the nature of results. This limits the ability to statistically test for differences in the asthma and allergy status of the completers versus the general population as the reported symptoms of allergy and asthma may be overestimates of true prevalence confirmed by physicians’ diagnoses. Nonetheless, it is plausible that this population would tend to respond to inhalation exposures regardless of product type (e.g., insecticide, air freshener, and cleaning products). Most important, none of the exposures documented in the PSP resulted in significant adverse effects.
Phase IIb
Diagnostic assistance or pyrethrins standards were routinely offered to all completers, but none of the completers were willing to undergo diagnostic testing. Data from post-market surveillance support provided by SafetyCall for other product categories show that it is not unusual for physicians to contact the manufacturer to request assistance for allergy testing in patients suspected of developing an allergy to a given product. That no one requested such assistance with pyrethrins-containing products suggests that none of the cases were sufficiently bothersome or persistent to warrant such a request.
Phase III
Manual review of Phase II completer cases
The manual review of the 220 completer cases (Phase IIIa) highlighted the limitations of consumer-reported adverse events including the inability to describe symptoms correctly, reporting of symptoms considered inconsistent with respiratory and/or allergic reactions, etc. A detailed evaluation of the incident narratives indicated that symptoms considered inconsistent with product exposure were typically described as occurring secondary to reported respiratory symptoms (i.e., emesis secondary to cough). We therefore took a conservative approach and included all cases in the subsequent determination of the Association Score (Phase IIIb).
Phase IIIb: Association Score
The Phase II Enhanced Questionnaire provided additional insight into 220 cases. The majority of these cases (158) received a high Association Score of 5 or 6 indicating a probable relationship between product exposure and reported symptoms. This was to be expected as many completers had experienced respiratory symptoms after being inadvertently sprayed in the face during product application. Typically, the symptoms were minor and self-limiting. Only 29 out of 158 cases involved products where pyrethrins were the only insecticide present in the formulation. Pyrethrins and pyrethroid(s) combined were present in the remaining 129 cases. This is not surprising, as pyrethrins-only products are uncommon in the marketplace.
Discussion of statistical analysis
The data from completers that showed a probable relationship between product exposure and reported symptoms were analyzed using chi square and logistic regression models.
No statistically significant association was observed between 1. asthma, allergy, or smoking status and presence of respiratory symptoms and 2. asthma or allergy status and severity of outcome.
These findings are in contrast to inferences made by Hudson et al. (2014).
12
Because asthma status was not collected by pesticide poisoning surveillance programs during the period of data review, Hudson et al. were unable to directly assess the association between asthma and exposure to pyrethrins and pyrethroids. Nonetheless, they reported that symptoms which were “suggestive for the presence of asthma (i.e., wheeze, dyspnea and cough” were: “significantly more likely to be present in individuals with acute pyrethrin and pyrethroid related illness compared to individuals with acute pesticide related illness not involving pyrethrin/pyrethroid/disinfectant exposures.”
A comparison to incidents not involving pyrethrin/pyrethroid/disinfectant exposures seems inappropriate assuming that this group also includes rodenticides which are sold in solid or liquid forms for which inhalation exposure is most unlikely.
Logistic regression indicated no significant relationships between case severity, allergy status, or independent variables including season and product mix (concurrent use of pyrethrins product, pyrethroid product, combination of pyrethrins/pyrethroid or products that contained pyrethrins or pyrethroid and other chemicals). Another model addressed the relationship between case severity and the self-report of asthma and indicated that symptoms occurring within three organ systems (dermatological, gastrointestinal, and miscellaneous) were associated with an increased likelihood of the case considered to be moderate. No significant relationships were found between case severity and asthma status with product mix as an independent variable.
Overall, although persons who were exposed to pyrethrins-only products appeared to be more likely to have an outcome of moderate severity (7 out of 29) compared to persons exposed to both pyrethrins and pyrethroid(s) (13 out 129), the total number of moderate cases (20 out of 158) over the 6-year length of the program was very low. Thus, caution must be exercised not to overstate the significance of this finding.
Discussion of overall limitations and strengths of dataset
Limitations
This PSP has several limitations, most of which stem from the reliance upon self-reports of adverse effects.
Inability to Confirm Exposure: Acute effects associated with pesticides are non-specific and not pathognomonic. No specific diagnostic tests are available for pyrethrins and pyrethroids.
Misidentification of product an individual was exposed to less likely as callers were asked to identify the product by its unique EPA Registration Number. But concomitant exposure to additional products was likely underreported.
Misrepresentation of exposure: Due to the potentially stressful nature of an exposure, self-reports of exposure might lack important details of the circumstances of product use and exposure. Additionally, exposed individuals might be biased towards “wanting to blame the product for unrelated symptoms they are experiencing” and thus might misrepresent their product use and the exposure and underrepresent potentially confounding factors.
Misrepresentation of medical history: We relied upon self-reports of medical history. Because of potential lack of medical knowledge and/or subjectively attributing symptoms to product and product exposure on the caller’s part, details of medical history were potentially misrepresented (including under- as well as overrepresentation).
Misattribution of adverse effects: Reports were taken at face value. No independent verification of actual effects seen and identification of other potential causes for symptoms were possible.
Underreporting of minor effects: Persons who did not seek medical treatment or advice from PCCs were not captured as cases in our dataset. Because the intent of the PSP was to focus on the more serious cases, such as those leading to risk mitigation, this is not considered to be an important limitation.
Strengths
Details of Follow-up Questioning of Caller: Our dataset of exposures to pyrethrins-containing products is unique in that it includes all exposure incidents of a certain year regardless if they were reported to SafetyCall or PCCs contracted with PJV member companies. Furthermore, our dataset includes data on the medical history (asthma, allergies, etc.) of the exposed individuals.
Ability to Assess Association Between Exposure and Reported Effects: The robust case documentation and the in-depth review based on the Enhanced Questionnaire and Association Score allowed for a statistical analysis of the likelihood a relationship between exposure to a purported causal agent and reported adverse effects.
Likelihood of Exposures Resulting in Adverse Effects Being Represented in the Dataset: The PSP tapped into the most likely sources of adverse event data involving pyrethrin product exposures. If serious adverse events involving pyrethrin-containing products were occurring, the data collection and surveillance systems used in the PSP would be likely to collect at least some of such incidents.
Conclusion
Given the self-reporting origin of the data, the limitations noted above cannot be completely eliminated. Nonetheless, the PSP Enhanced Questionnaire and subsequent association scoring provide more confidence in the data and resulting conclusions than present in previous analyses.
Based on the data collected and analyzed during the 6 years of the PSP, we conclude that: 1. Both in absolute number and relative to the wide distribution and use by consumers, adverse respiratory or dermal events after product exposure were rare; 2. Most outcomes for the reported events involving either dermal or respiratory effects were of minor severity and self-limiting; 3. None of the data collected and analyzed indicate that pyrethrins-containing products, including those formulated with synthetic pyrethroids and/or synergists, pose a significant risk of serious dermal or respiratory reactions even in cases where the exposed individual reported having allergies or asthma; 4. No additional label warnings or other mitigation techniques are warranted with pyrethrins-containing products formulated with or without synthetic pyrethroids and/or synergists.
Supplemental Material
sj-pdf-1-tor-10.1177_23978473211052700 – Supplemental Material for Retrospective analysis of adverse effects associated with pyrethrins-containing products
Supplemental Material, sj-pdf-1-tor-10.1177_23978473211052700 for Retrospective analysis of adverse effects associated with pyrethrins-containing products by Thomas G Osimitz, Kelly Sioris, John Gualtieri, Dean Filandrinos, Ryan Seaverson, Angeline M Carlson, Wiebke Droege and Rick Kingston in Toxicology Research and Application
Footnotes
Acknowledgments
We thank the following individuals for their participation in the Medical Advisory Council and their contributions in designing the program and reviewing the resulting data: D. Bernstein, MD, University of Cincinnati, College of Medicine, Cincinnati, OH, USA; A.W. Hayes, PhD, University of South Florida, School of Public Health, Tampa, FL, USA; M. Karol, PhD, University of Pittsburgh, Department of Environmental and Occupational Health, Pittsburg, PA, USA (participant in initial meeting only); H.I. Maibach, MD, University of California-San Francisco, School of Medicine, San Francisco, CA, USA; and DL Sudakin, MD, MPH, Medicaltox Consulting, Portland, OR, USA.
Declaration of conflicting interests
The authors declare the following financial interests/personal relationships which may be considered potential competing interests.
TG Osimitz and W Droege are employed by Science Strategies, LLC, a health and environmental sciences consulting firm, and do occasional paid scientific analysis for the Pyrethrin Joint Venture. R Kingston, K Sioris, J Gualtieri, D Filandrinos, and R Seaverson are employees of SafetyCall International, LLC, a multidisciplinary healthcare practice that provides manufacturers with adverse event management, regulatory reporting, post-market surveillance, and consulting services. AM Carlson is employed by Data Intelligence Consultants LLC, a company that provides consulting regarding social, behavioral, and health economics research to companies including SafetyCall International, LLC. Note: AW Hayes, Editor for Toxicology Research and Application, was on the Medical Advisory Council for the PSP. However, he was not involved in the decision of where the manuscript would be submitted nor in reviewing the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development of this paper was funded by the Pyrethrin Joint Venture, operating under the auspices of the Household and Commercial Products Association, Inc. TG Osimitz, W Droege, and R Kingston were responsible for the primary authorship of the manuscript. The sponsor has reviewed the manuscript, but the authors had sole authority for all editorial matters.
Supplementary material
Supplementary material for the article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
