Abstract
Among researchers, environmental pollutants and their contribution to male fertility are still being discussed. The use of antioxidants manages to boost the reproductive system with the scavenging of free radicals. This study aimed to investigate the inhibiting function of Vitamin E (VE) on Bisphenol-A (BPA) toxicity in the male rats’ reproductive system. Male rats were divided into 4 groups: control (negative control) group, BPA group treated by 250 mg/kg/day (positive control), VE group treated by 150 mg/kg/day (comparative control) and BPA + VE group that received both doses at the same time (Oral treatment by gavage; 56 days). Sperm parameters, testicular tissue morphometric and biochemical tests were evaluated. Sperm count, motility, viability, normal morphology, sperm tail length, spermatogenesis index and serum testosterone levels significantly decreased in the BPA group compared to the control group. Versus a significant enhancement in the positive-TUNEL germinal cells and serum malondialdehyde (MDA) levels were observed. Moreover, BPA exhibited no effect on sperm maturity and DNA integrity. In the simultaneous treatment group (BPA + EV), VE could improve and regulate all the mentioned parameters within the control group range. As mentioned, there was a significant difference in the results in the positive control group compared to the negative control group. But these data improved significantly in the BPA + VE. It can be concluded that in this group, VE was able to overcome the toxicity caused by positive control in their simultaneous treatment and maintain the data at the negative control group range. Therefore, no significant change was observed in the BPA + VE group compared to the negative control group.
Introduction
Pesticides, industrial chemicals and other synthetic substances release toxic compounds in the environment that can jeopardize wildlife as well as human life. 1 The male reproductive system is very sensitive to chemicals (toxic material even with low dose by oxidative stress induction) and physical factors due to the prompt division of spermatogonia cells. 1 Bisphenol-A (BPA) (CH3)2C(C6H4OH)2 is an environmental pollutant and in the body with its hormonal-like activities can mimic the androgen action 2 that causes disorder (such as spermatogenesis) in the male fertility. 1,2 Usages of this compound that are in direct contact with human life include polycarbonate plastics, epoxy resins (for the inner coating of food cans), plastic bottles, tableware, microwave ovens, sunglasses, building materials, medical devices, dental materials and paper. 3 –5 The human may exposure to BPA through inhalation and skin contact. 1 One of the known reason of BPA toxicity is the induction of oxidative stress, 3 which disrupts the male and female reproductive systems. 6 Many studies have shown that BPA can decreases the levels of antioxidant enzymes 7,8 although, can increases malondialdehyde (MDA) levels 7,8 and other oxidative stress factors, 9,10 In following that, it can degenerate germinal epithelium, 11,12 besides, it causes degradation in the sperm quality and quantity, 11,12 testosterone levels, as well as, spermatogenesis. 13 It is undeniable that the oxidative stress property of BPA is one of the most important reasons for its toxicity, therefore, in this study, Vitamin E (VE) was used as a potent antioxidant to neutralize the free radicals created by BPA.
VE is one of the natural antioxidants found in semen that disrupts the activity of free radicals, prevents the production of lipid peroxides and protecting sperm against reactive oxygen species (ROS) damage and also is a strong (fat-soluble) that deposited in cell membranes, especially mitochondrial membranes and microsomes. 14 –16 VE has the highest concentrations in adrenal cells, pituitary glands, platelets and testicular cells. 17 Shortage of this vitamin in testicular tissue leads to oxidative stress reactions and consequently a decrease in testosterone synthesis and spermatogenesis, in a way that obviously, VE is essential for the maintenance of spermatogenesis in mammals. 13,16,18 VE contributes to inhibiting oxidative stress in the testicular tissue 16,18 and prevents the apoptosis induction in germinal epithelium 18 and plays an essential function in spermiogenesis. 13,16 –18 Besides, This antioxidant be able to return glutathione to its standard levels on the intracellular free radical sweeping and lessens the oxidative stress. 17,18
The current experimental study aimed to assay antioxidant impact of VE on population, DNA fragmentation and maturation, quality, and quantity of spermatocyte, spermatid, Sertoli, Leydig and spermatozoa cells in the
Our observations showed that VE, as an antioxidant, can counteract the adverse effects of BPA in rat testicular tissue and improve spermatogenesis process.
Material and method
Material and treatments
In this experimental study, 24 adult male
Estimation of population of spermatogenesis, Sertoli and Leydig cells
Spermatocytes, long and round spermatids, Sertoli and Leydig cells were counted by optical dissector technique and special frame count. 20,21 This used an unbiased counting frame, a 100× magnification, and a regular random method of 20-micron testicular sections in each rat to count cells. Also, a microcator (ND 221 B, Heidenhain, Germany) was used to count the depth of tissue. Finally, the method of Stereological analysis was used to count the number of cells 19 that details can be obtained in reference. A detailed description of this method is given in Malmir’s study. 20,22
Sperm tail length
In the two-dimensional space, estimation of the sperm tail length (STL) according to the Dehghani et al method (stereological study) was carried out. 23 Eosin-necrosin impregnated slides were used for this purpose. For each rat, 150 sperms were selected using a regular random sampling method and counted by a light microscope with ×40 magnifications. The plan of this grid is such that if the sperm head was in the frame and does not collide with the continuous line (forbidden line), these spermatozoa can be counted. Then, the sperm counts were assayed with the Merz grid. This grid has two rectangular frames that call the counting frame (large frame). Within the basic frame, there is a Merz grid with two equal half circles (a curve). Finally, the following formula was used to calculate the STL: ΣL (total tails) = (π/2). (a/l). (1/asf). ΣI, “L (tail) = ΣL/ΣN. ΣN”: count of sperm count in the counting frame, π”: 3.14, a/l”: Merz grid constant, Asf”: area sampling fraction (Figure 1).
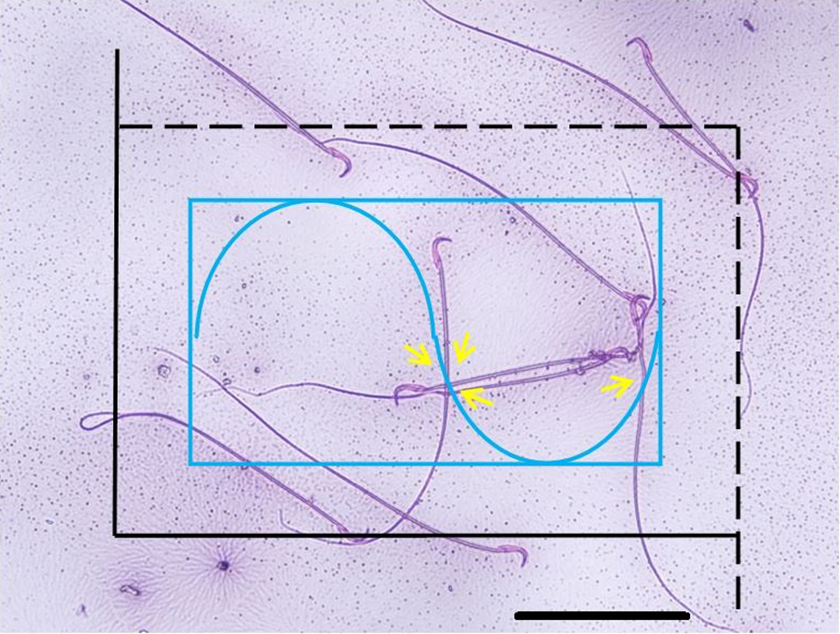
An estimate of sperm tail length in different groups 56 days after treated with Bisphenol-A (250 mg/kg/day) and Vitamin E (150 mg/kg/day), (eosin–necrosin staining; the magnification is ×200). In this figure, the counting frame (Large frame), the Meze grid (Curves within the small rectangle), forbidden lines (continuous), permitted lines (discontinued) and 4 countable sperm (arrow) are shown. For this purpose, if the sperm head lied inside the frame and did not touch the exclusion lines, that was counted (control group).
Sperm count
After testis sampling, the left epididymis tail was removed and immediately (without any time-wasting) inserted into a plate containing culture medium to examine the sperm count used. Hemocytometers were used to sperm counts. 10 µl of diluted sperm solution (The culture medium suspension containing sperm from different groups was diluted 1:9 with a formalin fixative of 2%) placed on the Neobar lam. The sperm with head, middle and tail located in the four corners and center of the square, were counted by light microscope. The number of sperm per each milliliter via n × 50000 × d formula and 400× magnification was counted. “n” is the number of sperm counted in the five squares of the hemocytometer and “d” is the opposite of the dilution of a suspension containing sperm. 24 The count was performed twice for each sample and its mean was reported.
Sperm motility
Sperm motility was assessed according to the WHO protocol. 24 Thus, first 10 µl of culture medium suspension plus sperm was transferred to the Neubauer slide. Five microscopic field of view that at least contains 200 sperm were evaluated for each rats. The percentage of sperm with progressive motility, non-progressive motility and immotile were evaluated. Sperm motility in different groups was examined under a light microscope at 200× magnification.
Sperm viability and morphology
Eosin-nigrosin staining was performed according to WHO protocol 24 to assess sperm viability and to detect morphologically abnormal sperm. Eosin (1%, Merck, Germany) and nigrosin (10%, Merck, Germany) were provided in normal saline. Volume of sperm suspension and eosin (ratio of 1:2) was mixed in micro tube, and after 30 s of incubate in a 37°C, an equal volume of nigrosin solution was added and a thin expansion of the sample was made on the smear. The basis for distinguishing dead sperm from live sperm is damage to the plasma membrane of the sperm, which becomes permeable to this compound. Therefore, those sperms that each part of the head, neck or tail was red were considered as dead sperm. In this regard, sperms with morphological disorders were considered as abnormal sperms. For each sample, 100 spermatozoa with ×100 magnification light microscope were examined and the abnormalities were expressed as percentages 24 (Figure 2).

Some morphological abnormalities of rat sperm by eosin–necrosin staining in different groups 56 days after treated with Bisphenol-A (250 mg/kg/day) and Vitamin E (150 mg/kg/day; ×200 magnification). 1-Evaluation of protamine replacement instead histone in rat sperm: Red-pink sperm represent adult sperm with nuclear protamine in the control group (a), positive control with blue sperm head (b) that collected from immature rats that contain huge of histone in chromatin (×100 magnification). 2-Evaluation of DNA integrity of rats’ sperm by Acridine Orange staining: Positive control (c): Sperm with an orange head that was the result of DNA denaturation by high heat (×100 magnification) and negative control (d): Sperm with a green head (Normal DNA; ×40 magnification). 3-Two kinds of sperm abnormality in Bisphenol-A group: Coil tail with an amorphous head (e) and cytoplasmic droplets (f).
Evaluation of sperm genetic material
Acridine Orange (AO) and aniline blue staining were used to evaluate sperm genetic material. According to Tejada et al. method, AO staining was used to evaluate spermatozoa with damaged chromatin. 25 AO is a fluorescent dye for recognizing two-stranded DNA (healthy) from single-stranded DNA (denatured). At least 100 spermatozoa were counted in each slide to determine the percentage of double-stranded spermatozoa (diploid) and single-stranded spermatozoa (haploid).AO by molecular reaction with the double-stranded, single-stranded and intermediate DNA, alter them dye to bright green, red, and yellow, respectively. Next, Aniline Blue (AB) staining was used to evaluate sperm maturity according to Wong et al method. 26 Histone protein contains a large number of the amino acid lysine, which reacts with AB. Sperms that turned blue were immature sperm with extra histone, which indicates protamine is not replaced in the nucleus and as a result, the sperm chromatin is not sufficiently dense. 100 sperm in each slide were used to determine the percentage of adult and immature spermatozoa (Figure 2).
Histopathological and spermatogenesis index assay
After going through the steps related to tissue processing, the samples were blocked applying molten paraffin. In Next step, 5-micrometer sections of paraffin blocks were prepared by a microtome set. Finally, Heidenhain’s Azan method used for staining samples. 27 In addition to, for this purpose, 100 seminiferous tubules of each testis to evaluate the following index were examined: Tubule Differentiation Index (TDI): To determine this index in the testicular tissue of each rat, the percentage of seminiferous tubes, which had three layers or more of germinal cells differentiated from type A spermatogonia cells, was calculated. 28 Spermatogenic Index (SPI): This index indicates the percentage of seminiferous tubules with normal spermatogenesis and containing sperm. 28 Sertoli Cell Index (SCI): This index was compared to the germinal cells number by the Sertoli cells number (spermatogonia + spermatocyte/Sertoli cells per each seminiferous tube. 22 Meiosis Index (MI): For meiosis Index, the ratio of the round spermatozoa number to primary spermatocytes was calculated.
Histopathological and tubule diameter, lumen diameter and germinal epithelium thickness assay
The rats were weighed after the end of the treatment period (56 days). Next, after euthanasia and opening the scrotum, the right testis was taken and underwent histopathological examinations. Next, after weighing, testes were transferred to a container containing Modified Davidson’s fluid fixative (MDF) and kept at room temperature for 1 week. 29 It should be noted that all health principles in animal care and extermination were based on ethical protocol. After confirmation, isotropic uniform random sampling (IUR) was performed. Some organs like testes have a heterogeneous structure. Therefore, for preparing a uniform random slice of testes IUR method was applied. 30 After going through the steps related to tissue processing, the samples were molded applying molten paraffin. Next step, 5-micrometer sections of paraffin blocks were prepared by a microtome machine. Finally, Heidenhain’s Azan method applied for staining samples 27 (Figure 3). Next, the method of Malmir was used for tubule diameter, lumen diameter and germinal epithelium thickness assay. 22 First, using a microscope equipped with a photo camera and Olisya with Objective Software 100 µm of all 5 µm of the section were selected using a randomized, 25 field of view were taken using a probe and motic image software were measured. Next, Harmonic mean thickness layer was obtained. Details can be obtained in reference.
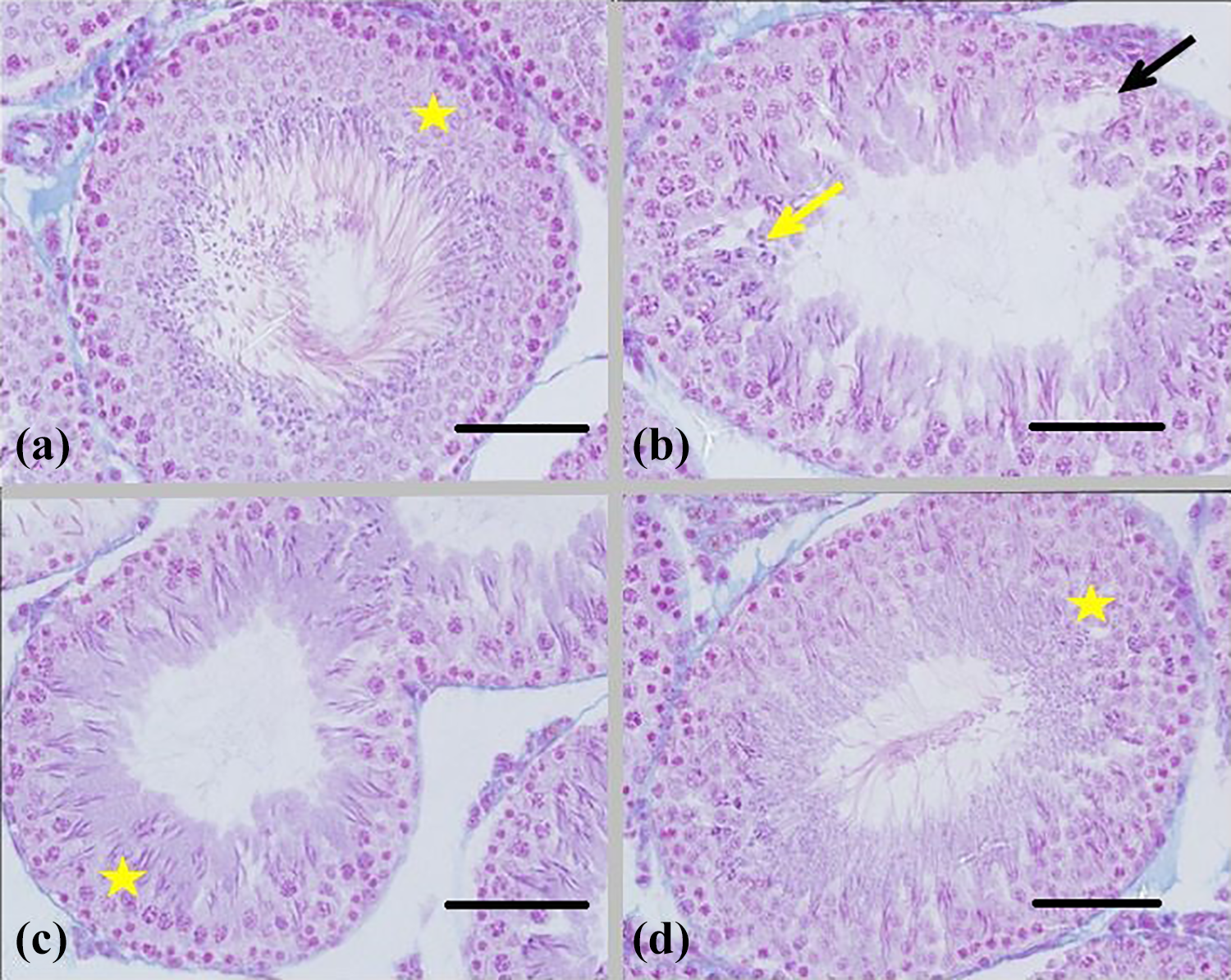
Microscopic images of the testicular tissue of adult rats in different groups, treated with Bisphenol-A (250 mg/kg/day) and Vitamin E (150mg/kg/day) and Bisphenol-A + Vitamin E (for 56 days). The thickness of the incisions is 5 microns, the hematoxylin-eosin is staining and the magnification is 400×. Negative control: The natural structure of seminiferous tubules in the testicular tissue of the control group with normal germinal epithelial formation (star; a). Positive control: Degeneration and vacuolation (arrow black) of the seminiferous tubule, spermatogenesis destruction and immature cell shedding into the lumen space (yellow arrow) in the group treated with Bisphenol-A (b). The almost normal arrangement of germinal epithelium and normal spermatogenesis (star) in the group treated with Bisphenol-A + Vitamin E (c). Comparative control: View of seminiferous tubule similar to the control group with increasing thickness of germinal epithelium (star) in the group treated with Vitamin E (d).
Biochemical assay
TUNEL assay
To evaluate apoptosis in spermatogenesis cells in the testicular tissue slices, a TUNEL Kit (Roche in situ cell death detection kit, Fluorescein) applied according to the manufacturer’s instructions. The basis for the diagnosis in the TUNEL test is the detection of DNA fracture by the terminal enzyme and the deoxynucleotidyl transferase marker, which marks the free 3.-OH in the end sections of nucleic acids. TUNEL is capable of detecting two groups of apoptotic cells with damaged DNA (positive-TUNEL) and non-apoptotic cells with healthy DNA (Negative-TUNEL).
Serum testosterone assay
In order to test the serum testosterone levels, blood samples (for each rat) were taken from the animal’s heart, then plasma was isolated from the serum by centrifuging the blood samples for 10 minutes and at a rate of 3000 g. The serum testosterone evaluation in this study was evaluated by the ELISA method and in accordance with the instructions of the manufacturer kit (Testosterone ELISA kit, Cat No (EIA-1559), DRG International, Inc., USA).
MDA levels assay
According to Buege and Aust’s method, the MDA test was performed to the evaluation of lipid peroxidation. In this way, MDA reacts with thiobarbituric acid (TBA) and they produce a combination of orange color that set the stage for observing rays with a wavelength of about 532–535 nm. The first, TCA (Trichloroacetic acid)-TBA-HCL (Hydrochloric acid) reagent (15% w/v TCA, 0.375% w/v TBA and 0.25 N HCl; Merck KGaA) was blended (2:1) with sperm suspension. Then, the samples were placed in a boiling water bath for 15 min. Then, after taking the samples from the bath and cooling, they were centrifuged for 10 min. The next, after the supernatant removing, their adsorption were read via spectrophotometry in the presence of Blank. Finally, MDA concentration was calculated by the extraction coefficient that is 1.56 × 105 M−1 cm−1. 31,32
Statistical analysis
Results were expressed as mean ± standard deviation (SD) for an animal per group. Via SPSS 2016 and one-way ANOVA analysis of variance followed by Tukey’s test was applied to determine the statistical differences among means (p < 0.05) was considered significant.
Results
Body weight and testis and morphometric assay
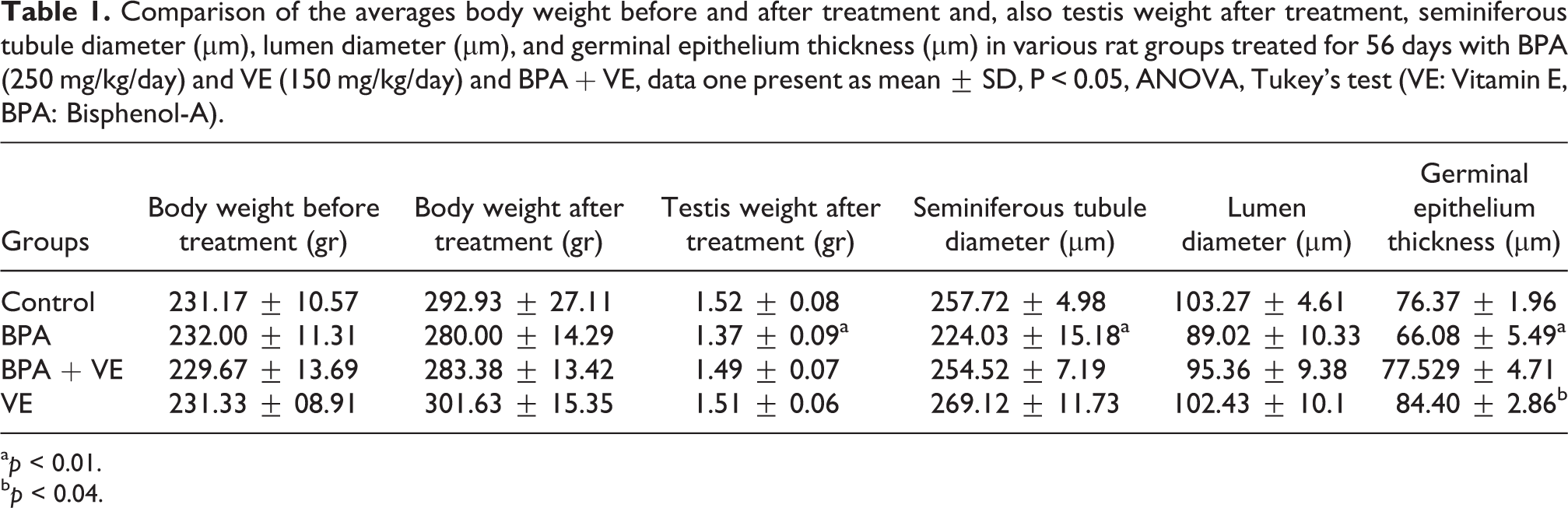
After treatment with BPA, the mean body weight of the rats, there are not significantly differ between any of the groups (P > 0.05), while the mean testis weight in the BPA group was significantly reduced compared to the control group (P < 0.01). Besides, in the VE + BPA group, this decrease was normalized in the control group (P < 0.04; Table 1). Also, the mean seminiferous tubule diameter (µm) and germinal epithelium thickness (µm) in the BPA group was significantly reduced compared to the control group (P < 0.01), although in the VE + BPA group, this decrease was normalized in the control group (P < 0.01; Table 1).
Comparison of the averages body weight before and after treatment and, also testis weight after treatment, seminiferous tubule diameter (µm), lumen diameter (µm), and germinal epithelium thickness (µm) in various rat groups treated for 56 days with BPA (250 mg/kg/day) and VE (150 mg/kg/day) and BPA + VE, data one present as mean ± SD, P < 0.05, ANOVA, Tukey’s test (VE: Vitamin E, BPA: Bisphenol-A).
a
b
Population of spermatogenic, Sertoli and Leydig cells assay
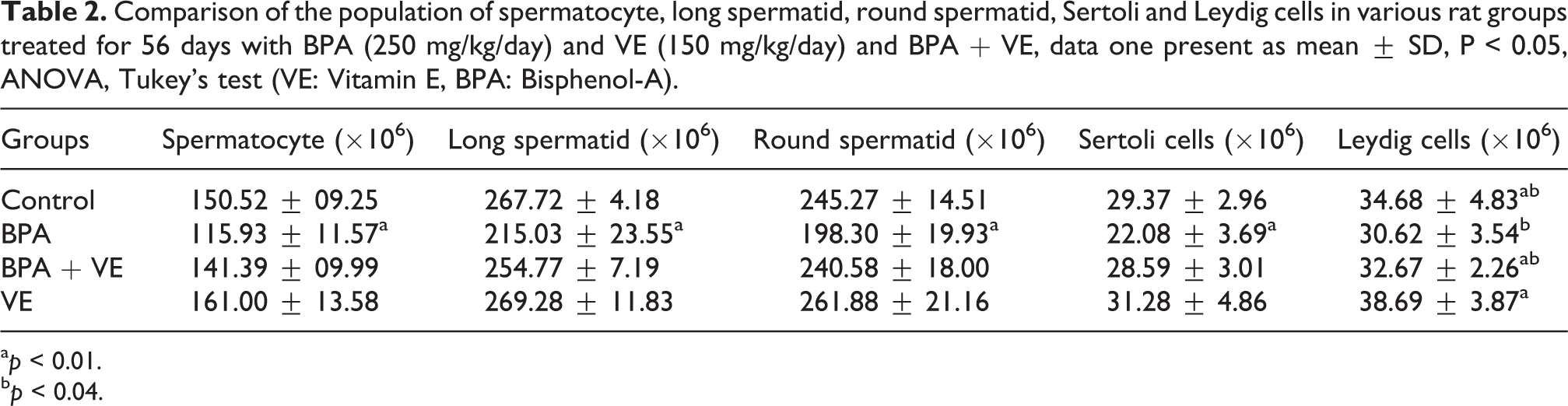
As illustrated in Table 2, the BPA group was showed a significant reduction in the population of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells compared with the control group (P < 0.01). However, in the simultaneous treatment group, VE was able to significantly upregulate population of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells compared to the BPA group (P < 0.01; Table 2).
Comparison of the population of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells in various rat groups treated for 56 days with BPA (250 mg/kg/day) and VE (150 mg/kg/day) and BPA + VE, data one present as mean ± SD, P < 0.05, ANOVA, Tukey’s test (VE: Vitamin E, BPA: Bisphenol-A).
a
b
Positive-TUNEL population of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells assay
As illustrated in Table 3, the BPA group was showed a significant increase in positive-TUNEL population of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells compared with the control group (P < 0.02). However, in the simultaneous treatment group, VE was able to significantly downregulate positive-TUNEL population of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells compared to the BPA group (P < 0.01; Table 3).
Comparison of the TUNEL-positive of spermatocyte, long spermatid, round spermatid, Sertoli and Leydig cells in various rat groups treated for 56 days with Bisphenol-A (250 mg/kg/day) and VE (150 mg/kg/day) and Bisphenol-A + VE, data one present as mean ± SD, P < 0.05, ANOVA, Tukey’s test (VE: Vitamin E).
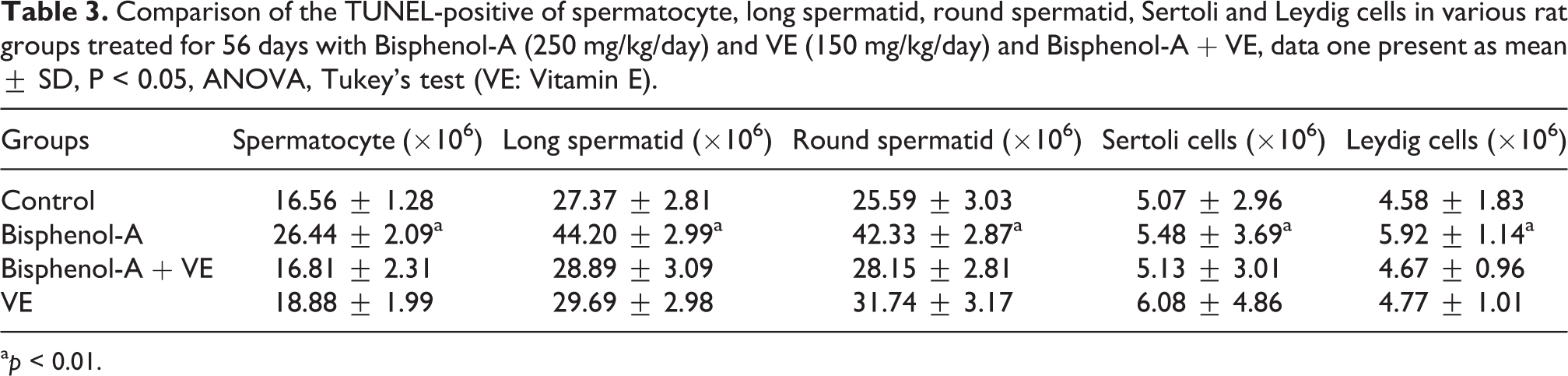
a
Evaluation of spermatogenesis index
A statistical evaluation comparing the values of spermatogenesis indexes in different groups exhibited that BPA leads to the elimination of germinal cells in the process of spermatogenesis, which this leads to a significant reduction in TDI (P < 0.001), SPI (P < 0.001) and MI (P < 0.002) compared to the control group. However, treatment with VE was increased significantly (P < 0.001) in the mentioned parameters compared to the group that received only BPA (Table 4).
Comparison of the Tubule Differentiation Index (TDI), Spermatogenic Index (SPI), Sertoli Cell Index (SCI), and Meiosis Index (MI) in various rat groups treated for 56 days with BPA (250 mg/kg/day) and VE (150 mg/kg/day) and BPA + VE, data one present as mean ± SD, P < 0.05, ANOVA, Tukey’s test (VE: Vitamin E, BPA: Bisphenol-A).
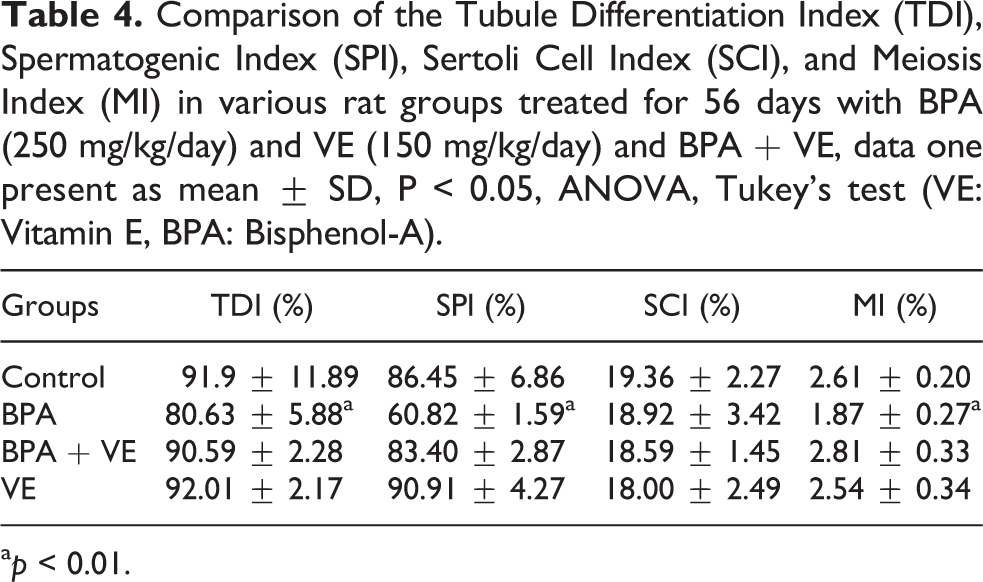
a
Evaluation of sperm parameters
As illustrated in Table 5, the BPA group was showed a significant reduction in sperm motility (Progressive, Non-progressive motility; P < 0.01), count (P < 0.001), viability (P < 0.01), normal morphology (P < 0.02), and tail length (P < 0.04) compared with the control group. However, in the simultaneous treatment group, VE was able to upregulate sperm parameters significantly compared to the BPA group (P < 0.01; Table 5).
Comparison of the sperm motility (progressive, non-progressive, and immotile %), count, viability, normal morphology, and tail length in various rat groups treated for 56 days with BPA (250 mg/kg/day), VE (150 mg/kg/day), and BPA + VE, data one present as mean ± SD, P < 0.05, ANOVA, Tukey’s test (VE: Vitamin E, BPA: Bisphenol-A).
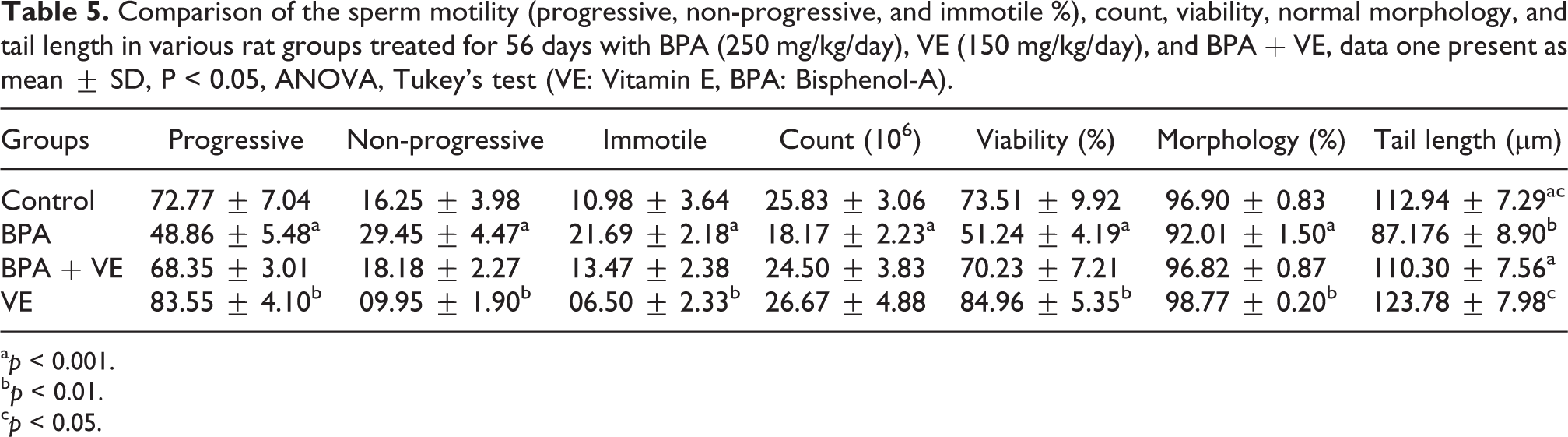
a
b
c
Evaluation of sperm genetic material
Sperm staining by AO in different groups demonstrated that BPA did not affect the denaturation of sperm DNA (double-stranded structure) significantly compared to the control group. As well as, the sperm staining by AB indicated that the treatment of rats with BPA had no effect on histone replacement with protamine in the nucleus significantly compared to the control group.
Biochemical assay
Serum testosterone levels assay
As shown in Figure 4, the BPA group was showed a significant reduction in serum testosterone levels compared with the control group (P < 0.02). However, in the BPA + VE group, there was no change in testosterone concentration compared to the BPA group and the control group (P < 0.05; Figure 4).
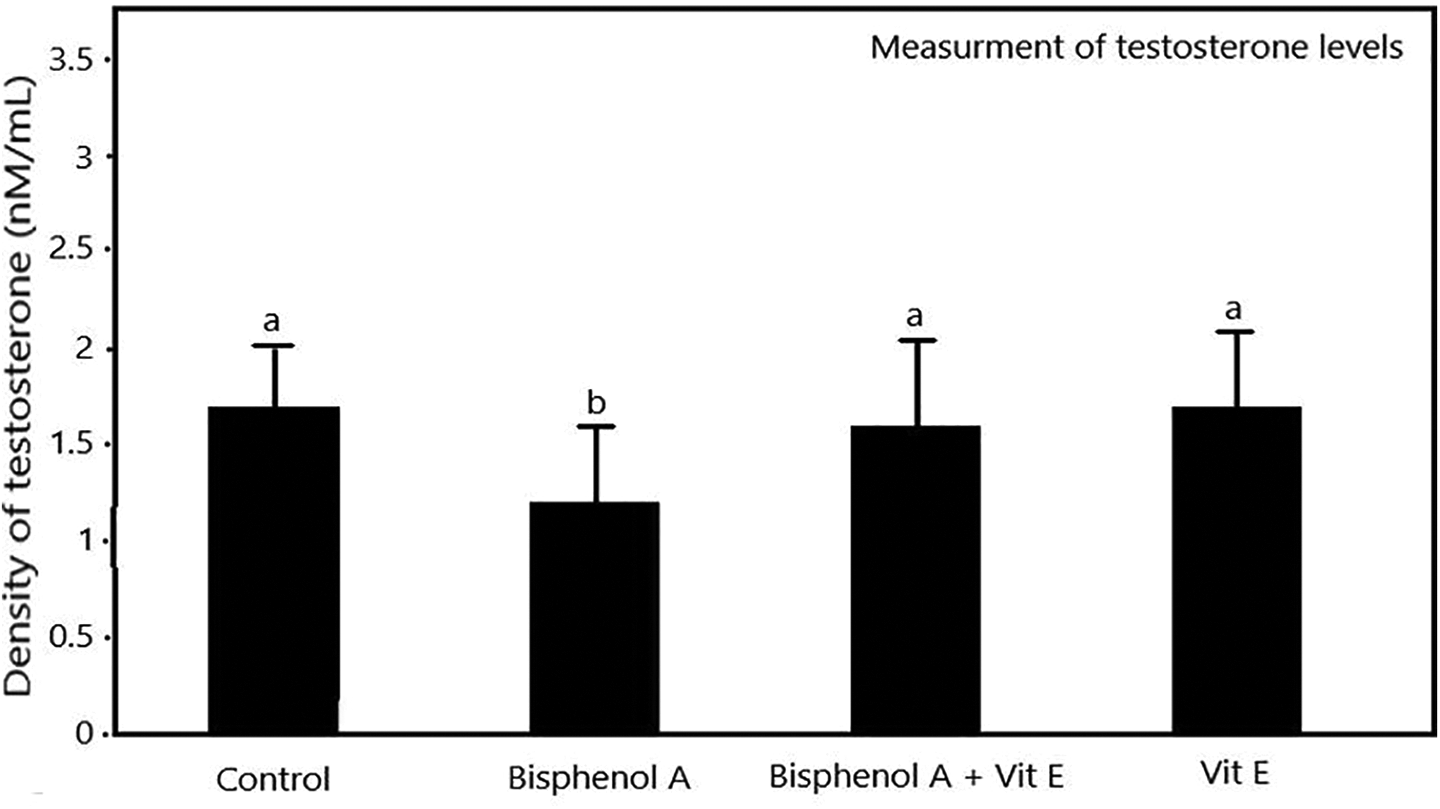
Comparison of serum testosterone (ng/mol), in various rat groups treated (for 56 days) with Bisphenol-A (BPA) (250 mg/kg/day) and Vitamin E (VE) (150mg/kg/day), data is presented as mean ± SD, ANOVA, Tukey’s test, the mean with the same letters do not differ significantly.
Lipid peroxidation by MDA levels assay
MDA concentration in the BPA group exhibited a significant increase compared to the control group (P < 0.03). However, in the BPA + VE group, the MDA level decreased significantly compared to the group BPA (P < 0.04; Figure 5).
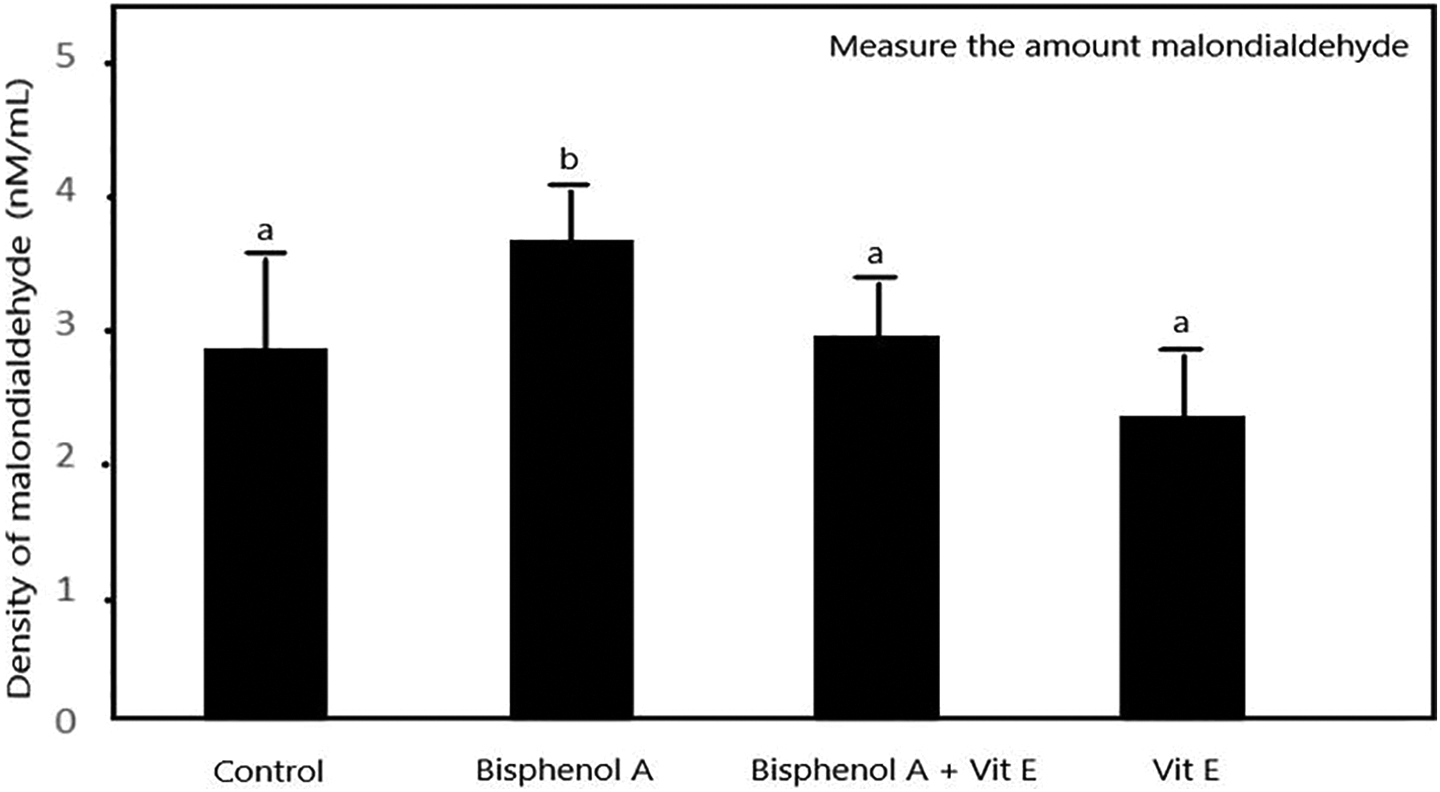
Comparison of serum malondialdehyde (nM/mL) levels, in various rat groups treated (for 56 days) whit Bisphenol-A (BPA) (250 mg/kg/day) and Vitamin E (VE) (150mg/kg/day), date one present as mean ± SD, ANOVA, Tukey’s test, the mean with the same letters do not differ significantly.
Discussion
The current experimental study aimed to investigate the inhibiting role of VE on BPA toxicity in the reproductive system of adult rats. The outcome of this research indicates a significant reduction in population of spermatogenic, Sertoli and Leydig cells, TDI, SI, MI, sperm count, viability, normal morphology, sperm tail length, motility, and testosterone levels and also however vice versa indicates a significant increase in positive-TUNEL spermatogenesis cells and MDA levels in the BPA group compared to the control group.
Numerous studies were reported a significant reduction on sperm parameters such as the count, 9,12,33 viability, 12 motility, 10,33 normal morphology 9,12,33 in the exposed by BPA in different doses. Increased oxidative stress causes damage to sperm membranes, proteins and DNA that reason for a reduction in sperm viability. 10 With the peroxidation of unsaturated fatty acids in the plasma membrane, the fluidity of the sperm membrane and finally its morphology changes. 12 Along with lipid peroxidation, there is a rapid decrease in intracellular ATP, which leads to a decrease in axonal protein phosphorylation (the middle region of the sperm rich of mitochondria) and thus causes sperm immobilization. 34 BPA can also disrupt spermatogenesis by significantly reducing the expression of genes involved in spermatogenesis, such as ODF1. 35 ODF1 is a prominent cytoskeletal element of the sperm tail that is involved in sperm maturation and motility. 35,36 The decrease in sperm tail length may be due to reduced expression of tail skeletal proteins. 35 Careful consideration of this justification requires further investigation. Sperm motility is highly dependent on the energetic components located in the middle part of the tail and also on the sperm tail length. 23 Apoptosis induction in germinal cells can be a factor in sperm count reduction. 12 On the other hand, BPA disrupts intercellular connections 37 and trigger induces apoptosis in Sertoli cells (through the signaling pathway Fas/FasL and trigger the mitochondrial pathway of apoptosis) and since these cells have a vital impact in spermatogenesis, that may be a reason to serious damage to the germinal cells. 35,38 According to previous study and current observations, BPA makes a contribution to death in germinal, Sertoli, and Leydig cells by oxidative stress and apoptosis inducting. 35,38 Likewise, membrane lipid peroxidation induced by BPA can fragment and disorganize spermatogenesis, Sertoli, and Leydig cells in the germinal epithelium, which causes them to be released into the lumen. This may be due to a decrease in the germinal epithelium thickness. 3,35,38,39 These can be a justification for germinal epithelium thickness reduction. Consequently, sperm count reduction may be Sertoli cell roles that are the main determinant of sperm output and efficiency in the testis. 39 In the following, Vilela in an experimental study by evaluating the effect of BPA (40, 80, and 200 mg/kg/day) on sperm DNA integrity in mice for 70 days and did not report changes in different groups, 40 which was following our results. In addition to, BPA is an endocrine disruptor and has been reported to reduce serum testosterone and testosterone in mice and rats. 40,41 Decreased serum testosterone levels by BPA may result in decreased expression of steroidogenic enzymes and cholesterol-carrying proteins “StAR,” which is involved in testosterone synthesis. 42,43 On the other hand, with a note that testosterone is essential for maintaining spermatogenesis and inhibiting apoptosis in germinal cells. 44 Testosterone shortage in BPA-treated rats can prevent the progression of cell division and spermatogenesis and in the opposite is able to provoke genital cell degeneration and atrophic. 45,46 In Eshak and Osman study decreased testosterone levels were associated with increased levels of MDA in BPA-treated rats. 47 Oxidation of unsaturated fatty acids is called lipid peroxidation (LPO). MDA as one of the final products of LPO and is often the first known parameter to prove the intervention of radical free damage. 31,32 In some studies, BPA was shown a reduction in the activity of the testicular antioxidant enzymes (glutathione reductase, glutathione peroxidase, superoxide dismutase and catalase) and opposite an enhance in LPO significantly. 7 –10
In this study, VE was able to significantly improve all the mentioned parameters in VE + BPA group compared to the BPA group. Antioxidant therapy improves sperm parameters 48 and testicular tissue 22 against cytotoxic damage and can be effective in reducing BPA toxicity. 16,46,49 According to the studies, the protective effect of VE against the toxicity of formaldehyde, 50 sodium valproate, 51 chromium, 49 and sodium aside 46 on the testis action of adult rats is undeniable. VE by increased the activity of antioxidant enzymes and reduced lipid peroxidation 52 compensate for the adverse changes caused by BPA on the testicular tissue, sperm parameters, testosterone and MDA levels. 16,52 On the other side, VE can reduce LPO and increase serum testosterone levels in rats. 13 The main function of VE is inhibiting the phospholipid membrane peroxidation and protect cell membranes through its antioxidant action. 52,53 The lipophilic properties of tocopherol cause it to be located inside the cell membrane. 53 Also, tocopherols-OH can transfer their hydrogen atom to a single electron from a free radical and eliminating it before the free radical interacts with the cell membrane. 53 Therefore, VE can cause stability membranes and prevent lipid peroxidation. 53 Besides, VE is a non-enzymatic defense system in testicular mitochondria. 16 This vitamin can inhibit oxidative damage in the testis 16,49 and prevents the apoptosis induction in spermatogenic, Sertoli, and Leydig cells 18 which, it plays an important role in their count, as well as, sperm viability. 22,49 Additionally, VE can return glutathione to its normal levels on the system intracellular free radical scavenging and reduces the oxidative stress levels. 18,53 VE may improve spermatogenesis in BPA-treated rats by reducing peroxidation and oxidative stress, stabilizing cell membranes, and modulating testosterone levels.
Conclusion
In this experimental study, BPA has disordered in the seminiferous tubules’ germinal epithelium and sperm parameters. Likewise, VE has downregulated toxicity of BPA in testicular tissue and spermatogenesis process. According to what has been mentioned, may the use of VE can be a benefit strategy against the adverse effects of this toxin on the male reproductive system, hence molecular study is advised to more clear VE role against BPA toxicity on the testis.
Highlights
Evaluation of the effect of high dosage bisphenol-A on spermatogenesis and spermiogenesis.
The effect of Vitamin E on preventing the toxicity of high dosage bisphenol-A toxicity.
Evaluation of spermatogenesis, Sertoli and Leydig cell populations and the extent of apoptosis in them.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
