Abstract
Background:
The potential impact of an adverse drug reaction (ADR) is multidimensional, and includes prolonged length of hospitalization which significantly increased morbidity, mortality, and economic burden with additional intense medical care.
Aim:
This study was designed to characterize and evaluate the potential effect of ADRs on the length of hospital stay among adult medical inpatients in an academic teaching hospital in a large West African nation. The overarching goal of the study is to highlight approaches toward mitigating the sequelae of ADRs in this setting, and improving their recognition and timely optimal management.
Methods:
The patients admitted into the adult medical wards of a Nigerian University Teaching Hospital over a 9-month period from December 2013 to August 2014 were prospectively recruited for the study and followed up till discharge.
Results:
A total of 507 patients were evaluated during the study, out of which 269 (53.1%) of them were males and 238 (46.9%) were females. The mean age of the study population was 48.9 ± 17.8 years (median of 46 years). In this study, the mean duration of hospital stay for all patients was 11.9 ± 11.3 days (median of 9 days). Females had a mean duration stay of 12.9 ± 12.3 days (median of 10 days) and stayed significantly longer than males who stayed a mean duration of 10.9 ± 10.2 days with a median of 7 days (t = −1.985, df = 505, p = 0.048). Furthermore, patients admitted solely because of ADRs (ADR-out) had a significantly shorter duration of stay with a mean duration stay of 6.1 ± 3.7 days (median duration stay of 7 days) when compared to those without ADRs whose mean duration of stay was 11.6 ± 11.0 days (median duration stay of 8 days), and this was found to be statistically significant (t = 2.110, df = 472, p = 0.035). Those patients who developed ADRs during admission (ADR-in) had a mean duration of stay of 18.3 ± 14.8 (median of 15 days). This was significantly longer than the duration of stay for patients without ADRs (t = −3.398, df = 487, p = 0.001) and also significantly longer than the duration for those who were admitted solely because of ADRs (t =3.432, df = 49, p = 0.001).
Conclusion:
In this study, ADRs were associated with a significantly prolonged length of hospital stay for adult medical inpatients in this Nigerian University Teaching Hospital.
Keywords
Introduction
An adverse drug reaction (ADR) is associated with a significantly prolonged length of hospital stay (duration of hospitalization), increased economic cost burden of treatment, and almost twofold increased risk of mortality. 1 ADR in hospital patients can be divided into two broad categories: those that result in admission to hospital (ADR-out), and those that occur during inpatient care following hospital admission for other reasons (ADR-in). 2 –4 The potential effect of an ADR to cause a significantly prolonged length of hospital stay has been associated with negative outcomes in the course and recovery of adult medical inpatients. 4 –6 The management of ADRs requires huge amounts of funds, 7 expensive medical facilities for emergency care, 8 and significant dedicated time. 9 Globally, approximately 5% (range from 2% to 20%) of reported hospitalizations are triggered by an ADR, and at least one ADR has been reported to occur in 10–20% of hospitalized patients. 10 –12 An adverse drug event (ADE) refers to any injury occurring at the time a drug is being used, whether or not it is identified as a cause of the injury. 1,2,13 An ADR is a special form of ADE in which a causative relationship can be shown or established and is attributable to the offensive/precipitating medication. 14 –16 It is important to recognize when a patient is experiencing an ADR, or the oversight may lead to subsequent prescribing of additional drugs (with their own side effect profile) to correct the drug-induced toxicity disorder. Regardless of clinical setting, a serious “fog-of-war” challenge may be that it is difficult to distinguish an ADR from an exacerbation of an existing disease or a new medical problem because the clinical picture can be so complex and interwoven. 17 –19 When empirical methods fail or produce cloudy causation results, invoking the more formal process of using an ADR probability assessment tool, such as the Naranjo algorithm scale 15,16,20 or the World Health Organization (WHO) causality assessment rating criteria, 15,16,21 may produce clearer results. Whenever a patient experiences what looks like an exacerbation of an existing condition, or when a patient develops what seems like a new medical problem while being treated for something else, the possibility of an ADR must be added to the differential diagnosis. 22 –24 Despite the extensive study and attention given to ADRs, they still represent a clinically significant problem and burden with high incidence and prevalence. 25 –27 A difficult reality of clinical toxicology is that pharmacological treatment sometimes results in ADEs either due to inherent ADRs 28 or from inappropriate medicine use 29 or medication errors. 30 In developing countries, including Nigeria, there is minimal information on the in-hospital assessment of ADRs, associated length of hospital stay, and the responsible culprit medications. 31 –33 The development of these data is clearly important and useful for health-care planning, 34,35 management, 36 budgeting, 37 policy formulation, 13 and development of treatment protocols to enable appropriate, optimal, and timely patient care. 38
The present study was designed to characterize and evaluate the potential effect of ADRs on the length of hospital stay among adult medical inpatients at the University of Benin Teaching Hospital (UBTH), Benin City, Edo State, Nigeria, with the goal of enhancing recognition and improving treatment outcome.
Methods
This is a descriptive, prospective study with serial entry points for the patients admitted into the internal medicine wards of UBTH over a 9-month period from December 2013 to August 2014. They were all adult medical patients whose ages were above 17 years and were fulfilling the inclusion criteria for recruitment, evaluation, and follow-up. Medications prescribed for the admitted patients were supplied by the hospital pharmacy or, however, patients may occasionally be required to purchase some medications from retail outlets outside the hospital when these drugs are not available in hospital pharmacy. Patients are generally admitted through the accident and emergency unit where they are reviewed by various cadres of medical physicians until they are transferred to the wards under unit consultants. Some patients are admitted directly from the outpatient clinics into the wards, while a few may be transferred in from other nonmedical wards. The patients are then reviewed daily in the various units and managed till discharge.
The inclusion criteria for evaluation were all the patients admitted to the medical wards after commencing the study, provided they granted their informed consents to participate in the study.
The exclusion criteria were patients already on admission before commencing the study, patients admitted from other wards after initial management for nonmedical condition(s), patients diagnosed and subsequently managed for nonmedical condition(s) after initial medical diagnosis and management, and patients who did not grant their informed consents to participate in the study.
Information about all the recruited patients were obtained and entered into a data collection form modified from the World Health Organization-International Network for the Rational Use of Drugs (WHO-INRUD) prescribing indicator form. 3 Patients were evaluated with respect to medications used on days 0 (admission day), 1, 3, 7, 10, 14, 21, and weekly thereafter till day of discharge or death. An encounter was regarded as a patient studied on admission on such specified days. Initial sociodemographic data stating age, sex, religion, occupation among others were obtained at admission. Thereafter, the records of all prescribed medications including the dates, route, doses, and frequencies were all noted for these inpatients during admission. The reviews and changes made in patients’ medications between the days of evaluation were also noted to enable the estimation of number of drugs taken during admission.
Information concerning ADR occurrence and its nature was sought from attending physicians, patients, their relatives, and nursing staff. Charts and case notes were screened for records of ADRs. Clinical evaluation and assessment of laboratory results were also carried out. Repeated admission of the same patient was regarded as two separate admissions when separated by an interval of at least 1 month, otherwise such admission was considered as a single admission and the interval excluded from the duration of hospital stay. 16
The definition of ADRs used in the study was that of WHO: “-any noxious and unintended response to a drug that occurs at doses used in humans for the prophylaxis, diagnosis or therapy of disease.” 3 ADRs were classified as mild (laboratory abnormality or symptoms not requiring treatment), moderate (laboratory abnormality or symptoms requiring treatment/admission to hospital or resulting in nonpermanent disability), severe (laboratory abnormality or symptoms that were life-threatening or resulted in permanent disability), and fatal (any ADR that resulted in patient’s death regardless and irrespective of the initial severity grading assessment). 13,14 The ADRs were classified according to their severity grading assessment 4,5,10 and using the System-Organ Classification. 11
Data collected were encoded and analyzed using the Statistical Package for Social Sciences (SPSS) version 17 (released 2008; SPSS Incorporation, Chicago, Illinois, USA). Results were expressed as mean ± standard deviation or using frequency and percentage values where necessary. The t-test and χ 2 were used to compare means and proportions, respectively. The level of statistical significance was set at p value less than 0.05.
Ethical clearance was obtained from UBTH Ethical Research Committee before commencing this study. The Ethical Clearance/Protocol Research Number issued for the study was ADM/E.22 A/VOL. VII/104. In addition, a verbal informed consent was obtained from each of the patients whose medical records were used, while the medical records for those who did not grant their informed consent were excluded from the study. Consent was sought from patient’s relative where patient had impaired level of consciousness. Participants’ confidentiality was respected and maintained by ensuring that no unauthorized person had access to the information on the data information sheets, no information can be traced to the subjects (as coding system was used for the data information sheets instead of writing the patients’ names on them), and no unauthorized use of information was made.
Results
A total of 507 admitted patients were evaluated during this study. Regarding sex distribution, 269 (53.1%) were males while 238 (46.9%) were females. The mean age for all patients was 48.9 ± 17.8 years (median of 46 years; range of 17–89 years). Though the mean age for females (49.5 ± 17.7 years) was older than that of the males (48.3 ± 17.9 years), the difference was not statistically significant (t = −0.771, df = 505, p = 0.44). Table 1 shows the age and sex distribution of the patients. Table 1 also reveals that those patients under 45 years of age were the most predominant with a frequency of 236 (46.5%) patients, followed by those within the age-group of 45–64 years with a frequency of 146 (28.8%) patients. While the elderly age-group (65 years and above) constitutes 125 (24.7%) of the admitted patients.
Age and sex distribution of medical inpatients evaluated for ADRs in a Nigerian Teaching Hospital from December 2013 to August 2014.
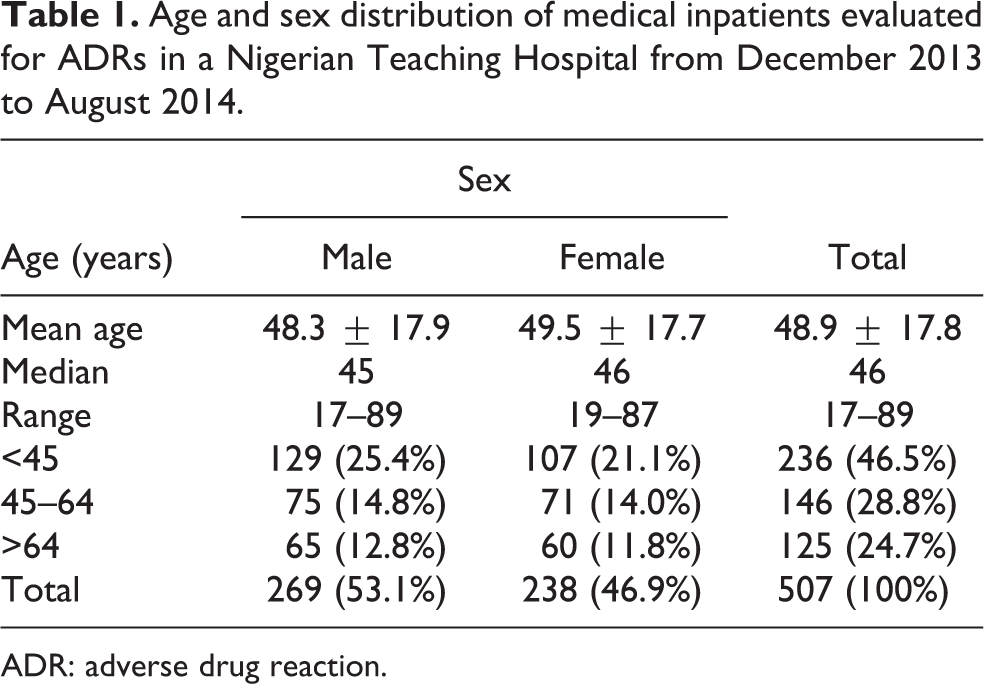
ADR: adverse drug reaction.
Table 2 shows the summary for duration of hospital stay by different categories of patients. The mean duration of hospital stay for all patients was 11.9 ± 11.3 days (median of 9 days). Females had a mean duration stay of 12.9 ± 12.3 days (median of 10 days) and stayed significantly longer than males who stayed a mean duration of 10.9 ± 10.2 days with a median of 7 days (t = −1.985, df = 505, p = 0.048). The young age-group (i.e. those less than 45 years old) stayed a mean duration of 11.3 ± 9.3 days (median of 9 days), while the middle age-group had a mean duration stay of 11.7 ± 9.7 days (median of 10 days). While the elderly age-group (65 years and above) had a mean duration stay of 13.0 ± 15.7 days (median of 8 days), they appeared to have stayed longer than patients below 65 years of age (i.e. the young and middle age groups). However, this was not found to be statistically significant (t = −1.299, df = 505, p = 0.195). Furthermore, patients admitted solely because of ADRs (ADR-out) had a significantly shorter duration of stay with a mean duration stay of 6.1 ± 3.7 days (median duration stay of 7 days) when compared to those without ADRs whose mean duration of stay was 11.6 ± 11.0 days (median duration stay of 8 days), and this was found to be statistically significant (t = 2.110, df = 472, p = 0.035). Those patients who developed ADRs during admission (ADR-in) had a mean duration of stay of 18.3 ± 14.8 (median of 15 days). This was significantly longer than the duration of stay for patients without ADRs (t = −3.398, df = 487, p = 0.001) and also significantly longer than the duration for those who were admitted solely because of ADRs (t =3.432, df = 49, p = 0.001).
Duration of hospital stay (in days) by different categories of inpatients in a Nigerian Teaching Hospital from December 2013 to August 2014.
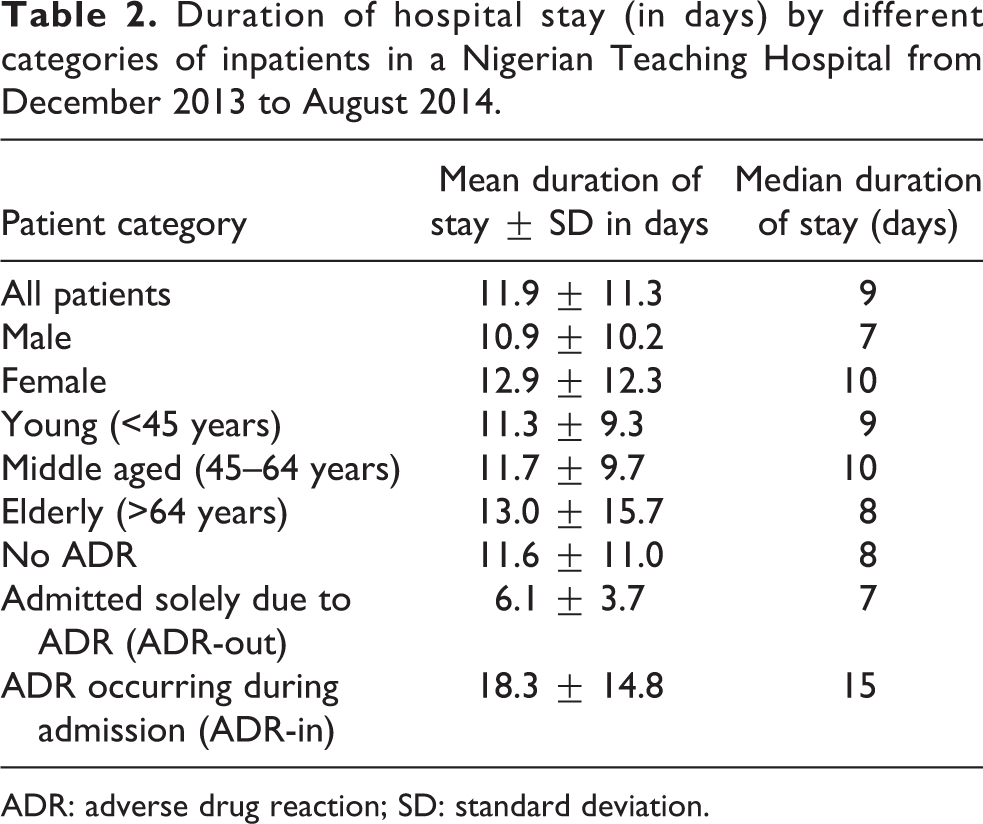
ADR: adverse drug reaction; SD: standard deviation.
Furthermore, Table 3 shows the details of the medications causing ADRs. The drugs causing the most frequent ADRs were insulin in 14 (27.5%), nonsteroidal anti-inflammatory drugs (NSAIDs) in 10 (19.6%), antihypertensives in 8 (15.7%), and antimalarials in 5 (9.8%). In addition, herbal medications and antibacterials caused ADRs in four (7.8%) and three (5.9%) patients, respectively. The other medications involved in ADRs are shown in Table 3. In addition, four fatal ADRs were observed during the study; in which herbal medications caused two deaths, sulfadoxine/pyrimethamine combination (maloxine®) caused one death, and iron dextran caused one death.
Individual medicines causing ADRs among inpatients in a Nigerian Teaching Hospital from December 2013 to August 2014.

ADR: adverse drug reaction; NSAIDs: nonsteroidal anti-inflammatory drugs.
aMedicines causing fatal ADRs.
In addition, Table 4 presents the statistical evaluations of some known risk factors for ADRs, taking into consideration their gender, age, number of medications used, and number of comorbidities. Gender was not found to be a statistically significant risk factor for ADRs in this study (χ
2 = 0.371, p = 0.542). Age was found to be a statistically significant risk factor as the elderly age-group (age ≥65 years) was more at risk compared to those patients less than 65 years (χ
2 = 10.152, p = 0.001). Number of medications used was also a significant risk factor for developing ADRs (χ
2 = 13.174, p = 0.018). Number of comorbidities was also found to be strongly associated with ADRs (χ
2 = 21.962, p
Assessment of some known risk factors for ADRs among medical inpatients in a Nigerian Teaching Hospital from December 2013 to August 2014.
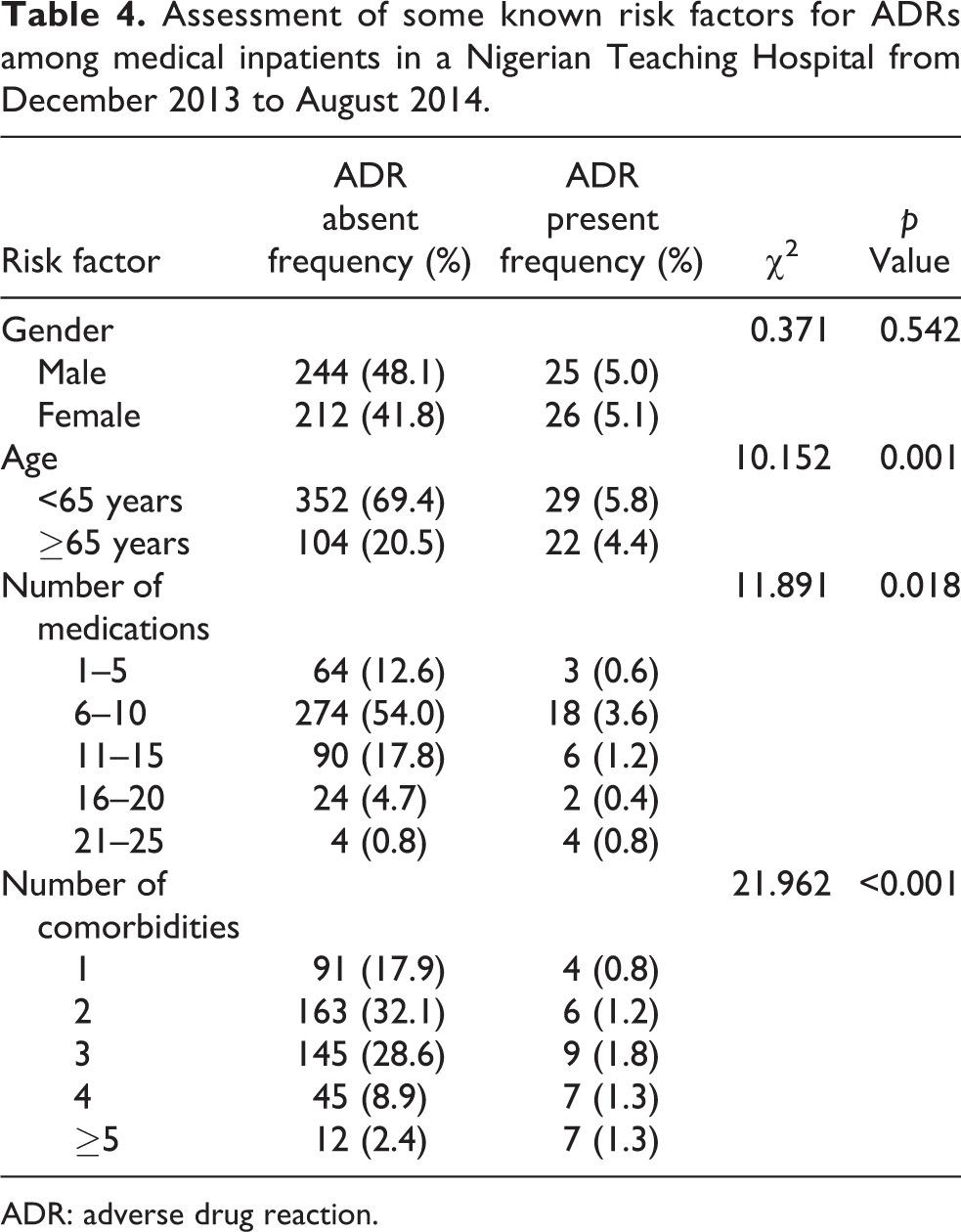
ADR: adverse drug reaction.
Using severity grading assessment, ADRs were classified as mild (laboratory abnormality or symptoms not requiring treatment), moderate (laboratory abnormality or symptoms requiring treatment/admission to hospital or resulting in nonpermanent disability), severe (laboratory abnormality or symptoms that were life-threatening or resulted in permanent disability), and fatal (any ADR that resulted in patient’s death regardless and irrespective of the initial severity grading assessment). 13,14 Furthermore, Table 5 reveals that most of the ADRs were mild and moderate in 21 (41.2%) cases and 24 (47.1%) cases, respectively. Severe ADRs occurred in two (3.9%) cases, while four (7.8%) cases were fatal ADRs.
Severity grading for the observed ADRs among medical inpatients in a Nigerian Teaching Hospital from December 2013 to August 2014.
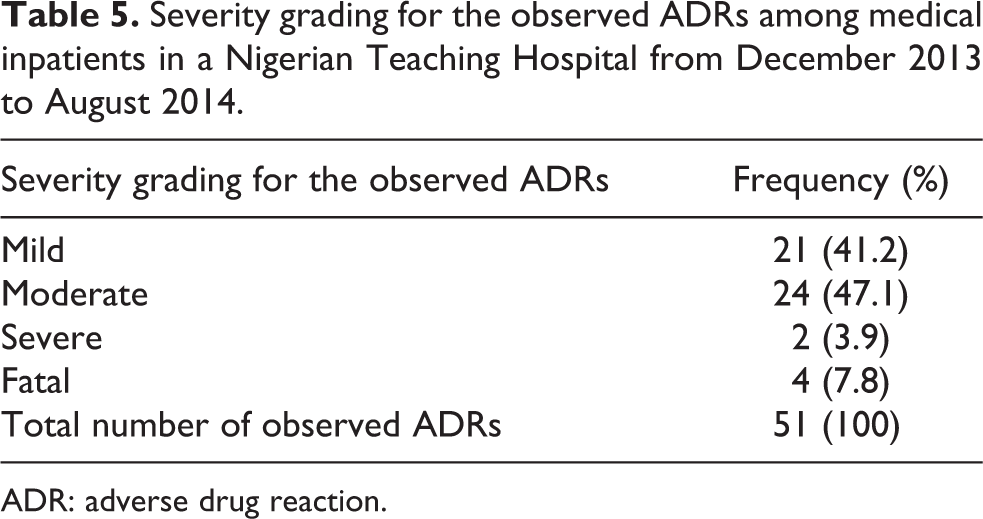
ADR: adverse drug reaction.
Moreover, the body systems and organs involved in ADRs are shown in Figure 1. The most frequently involved body system was the central nervous system (neurological) in 169 (33.3%) patients. This was followed by the gastrointestinal system in 110 (21.6%) patients, the skin (dermatological) in 89 (17.6%) patients, and cardiovascular system in 40 (7.8%) patients. The endocrine, respiratory, and renal systems were equally affected in 20 (3.9%) patients each. In this study, it was observed that ADRs often affect multiple body systems/organs in a patient.
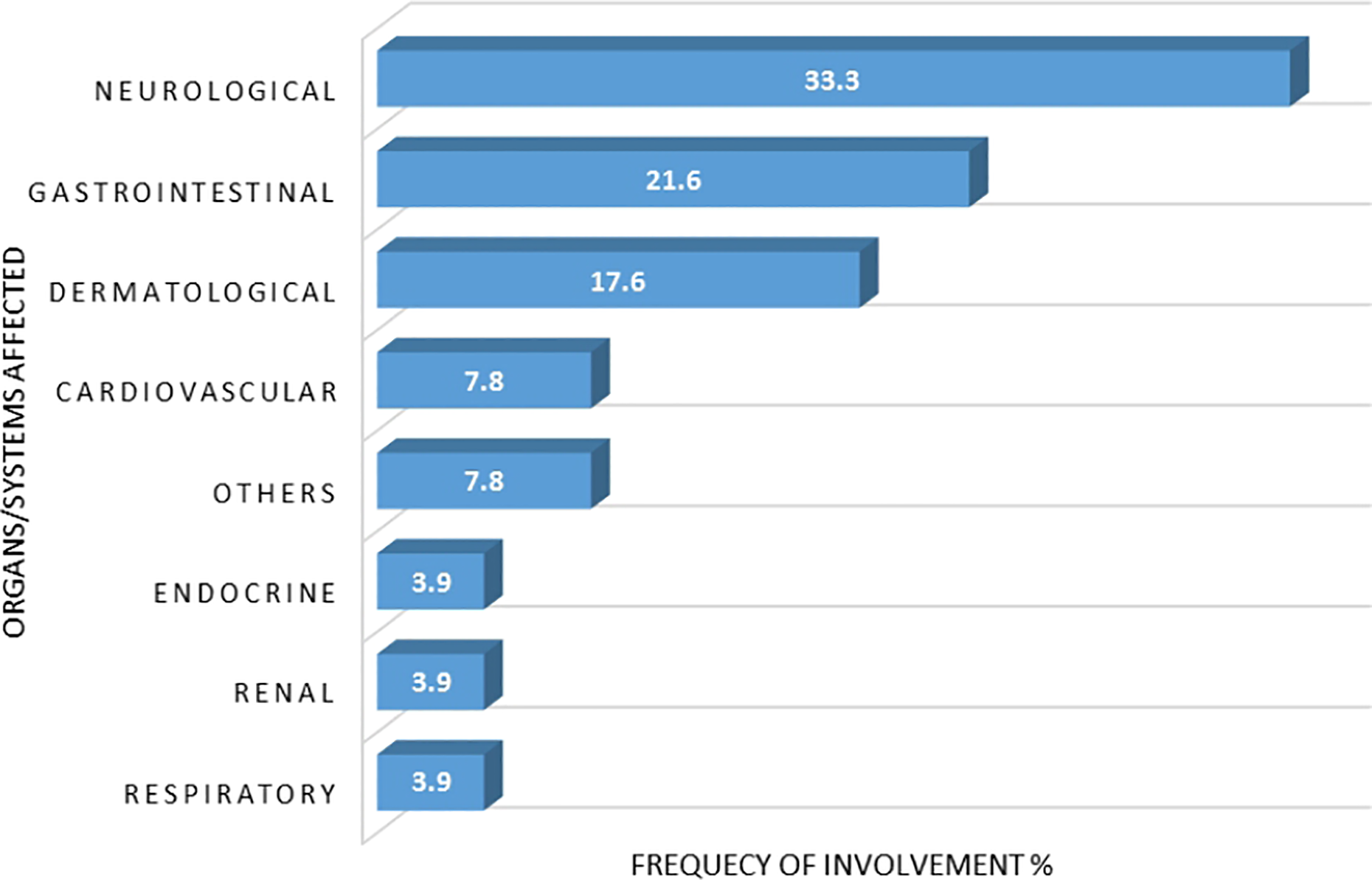
Organ/system affected by ADRs among medical inpatients in a Nigerian Teaching Hospital from December 2013 to August 2014. ADR: adverse drug reaction.
Finally, the number of patients admitted solely because of ADRs (ADR-out) was 18 patients, which accounts for 3.6% of admissions, while those who experienced ADRs during admission (ADR-in) were 33 (6.5%) patients. The total number of patients who had ADRs was 51 patients, giving an ADR prevalence of 10.1%. The incidence of ADRs was 6.5% in this study. The case fatality rate for ADRs was 7.8% (4/51), while the ADR-associated mortality rate was 0.8% (4/507).
Discussion
This study was designed to evaluate the potential effect of ADRs on the length of hospital stay among adult medical inpatients in a Nigerian University Teaching Hospital clinical practice setting with the goal of improving diagnosis and treatment outcomes.
Patients experiencing ADR during hospitalization (ADR-in) stayed significantly longer than those without ADRs (median stay of 15 days vs. 8 days, respectively). ADRs have been found to prolong the duration of hospital stay in several studies. 6 –9,39 The median length of stay obtained for patients with ADR-in and those without ADR (15 days vs. 8 days, respectively) is similar to results obtained in an inpatient study in 2006 by Davies et al. (14.5 days vs. 8 days, respectively). It must be acknowledged however that this was a small pilot study. In their large 2009 study, Davies’ group found that ADRs increased the length of hospital stay by an average of 0.25 days per patient admission episode. The discrepancy between these data and those reported in the present study is quite remarkable. In an early landmark study by Classen et al., 40 it was reported that the extra length of hospital stay attributable to an ADE was 1.74 days (p < 0.001). It may well be that the challenges of health care in the developing world context explain the significant difference between these results and those of the present Nigerian data. In any event, further exploration of the underlying factors is of critical importance. In the present study, patients admitted solely because of ADRs (ADR-out) had a statistically and clinically significant shorter duration of hospital stay (median of 7 days) than those patients without ADR who stayed a median duration of 8 days and also much shorter than those patients who developed ADR during hospitalization (median duration of 15 days). The finding of shorter duration of hospital stay for patients with ADR causing admission (occurring outside the hospital) compared to those without ADR and those with ADR occurring during admission was observed in a French study. This shorter duration of hospital stay is likely related to the fact that there is a shorter diagnostic time for ADRs, in effect a shorter time was spent looking for the cause of morbidity since this was obvious from initial clinical evaluation, especially as most of the ADRs were mild (41.2%) or moderate (47.1%). Furthermore, their clinical management was essentially observational care and measures such as stopping or adjusting drug dosage. 17 However, a few of the patients required more intense treatment with prolonged stay.
The most commonly affected system by ADRs in this present study was the central nervous system, probably because two out of the first three topmost implicated classes of medications causing ADRs manifest with symptoms referable to the central nervous system. For example, insulin leads to impaired consciousness due to hypoglycemia, while antihypertensives manifest mainly with postural or orthostatic changes and headache. The next most common system involved was the gastrointestinal tract, and this may be related to the fact that NSAIDs which constituted the second most common class of medications involved in ADRs usually manifest with problems referable to the gastrointestinal mucosa. In fact, the systems largely affected followed the pattern of medications causing ADRs. Fattinger et al. found the gastrointestinal and the hematological systems as the first and second most commonly affected, respectively, among inpatients. The pattern of morbidity and thus medication used differ in both settings, therefore the pattern of ADRs will be different. Mild (41.2%) and moderate (47.1%) ADR cases were far more frequent when compared to severe and fatal ADR cases in this study. This pattern is similar to findings in other studies. 13,14,39
Gender was not found to be a statistically significant risk factor associated with ADR in this study. This finding here differs from what has been observed in other several studies. 6,11,17,18,39 The association of gender and ADRs like other aspects of ADRs is not well characterized among Nigerians and may differ. Some other known risk factors for ADR were however found to be significantly associated with ADRs in this study. In summary, the elderly age-group (≥65 years), presence of polypharmacy (taking more than five different pharmacologically active medications by a particular patient at the same time), and the presence of multiple comorbidities in a particular patient are clinically proven and significant risk factors found to be associated with and predisposed adult medical inpatients to ADRs in this study.
The principal limitation of this study was that it considered only consented adult medical inpatients; while unconsented adult medical inpatients, pediatric unit patients, and adult surgical unit patients were completely excluded from the study. Patients admitted solely for management of ADRs did not serve to illuminate the hypothesis or quality of the observations. More meaningful data and associated statistical analysis might track intervening variables such as specific admitting diagnosis, number of medications, dosages, frequency of administration, route of administration, patient cohort, and as noted, a fully representative survey of admitting wards. Patients presenting with an ADR from the emergency setting could be studied in a separate protocol. Future research should document to the extent possible, drug–drug interactions versus primary toxicity of a single agent. The specific nature of reactions should be logged, that is, cutaneous [Stevens–Johnson syndrome, toxic epidermal necrolysis, drug reactions with eosinophilia and systemic symptoms, idiosyncratic and hypersensitivity syndrome]; or neurologic, that is, mental status changes (delirium); or gastrointestinal symptoms, that is, diarrheal illness, constipation, obstipation; or cardiotoxicity, that is, atrial fibrillation, cardiac electroconduction abnormalities, electrolyte disturbances etcetera.
Conclusion
In this study, ADRs were associated with a significantly, even dramatically prolonged length of hospital stay for adult medical inpatients in this Nigerian University Teaching Hospital. This undoubtedly is further associated with increased morbidity, mortality, and economic burden. The results of this report call for urgent and focused exploration of the specific variables most responsible for ADRs and for delays in diagnosis and effective management.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
