Abstract
Previous studies of the biocompatibility of β-tricalcium phosphate (β-TCP) focused on bulk-sized β-TCP, and little is known about the biocompatibility of nano β-TCP particles (nβ-TCP). The objectives of this study were to synthesize nβ-TCP particles and determine their efficacy in a rabbit tibial defect model. The nβ-TCP particles were first synthesized using a wet chemical precipitation process. The particles were then implanted in the left tibia of New Zealand white rabbits, and the defect site healing was evaluated for a period of 16 weeks using radiography, computed tomography, and histology. Data were compared with those of a sham (empty) control. Results showed that the defect site treated with nβ-TCP particles did not heal completely after 16 weeks, whereas full cortical bone recovery was observed in the sham control group of rabbits. Histopathological examination showed that the nβ-TCP particles caused an excessive and prolonged inflammatory response by the host. The nano-scaled size and biodegradability of the synthesized nβ-TCP particles may have been responsible for this progressive and extended inflammatory response, which delayed the bone healing process. The underlying mechanism for this effect remains unclear and warrants further investigation.
Introduction
β-Tricalcium phosphate (β-TCP) is one of the most important classes of synthetic bone graft materials used in bone regeneration. β-TCP mimics the major natural inorganic component of bone 1,2 and is bioactive, biodegradable, and osteoconductive. 3 –9 It serves as a template for bone cell attachment, proliferation, migration, and phenotypic expression, leading to the formation of new bone. 10 –12 Moreover, toxicity assessment has shown that β-TCP is a safe and biocompatible biomaterial. 13 –16
Previous studies of the biocompatibility of β-TCP focused on bulk-sized β-TCP products such as granules, blocks, or wedges with size ≥100 nm. Little is known about the biocompatibility of nano-sized β-TCP (nβ-TCP) particles, which may be an alternative to bulk-sized β-TCP. The use of nβ-TCP may dramatically increase surface area, surface roughness, and surface area to volume ratios, leading to superior physicochemical properties. Lin et al. reported that scaffolds fabricated from nβ-TCP had more than two times the Vickers hardness and compressive strength compared to the scaffold fabricated from micro-sized β-TCP. 17 The size of nβ-TCP may better mimic the nanocomposite of the natural bone, which consists mainly of nanocrystalline hydroxyapatite (Ca5(PO4)3), with the length of approximately 100 nm, width of 20–30 nm, and thickness of 3–6 nm, and is embedded between the nanofibrous collagens with diameter of 50–500 nm. 18 There is a possibility that these biomimetic features and physiochemical properties of nβ-TCP might play a key role in stimulating cell growth and tissue regeneration. The nβ-TCP might also promote more specific protein interactions, which could lead to more efficient osteointegration between the synthetic implant and the bone tissues and to successful bone regeneration. 19
However, the effect of nβ-TCP on human health is poorly understood, and the extent to which nβ-TCP will react in in vivo models as compared to bulk β-TCP has yet to be investigated. There is a possibility that the nanoparticles may provide better cellular targeting and uptake, but there is still a risk of adverse cellular responses to these particles. Nanoparticles might be endocytosed more readily due to their smaller size and might subsequently cause cytotoxicity. 20 Thus, the objectives of this study were to synthesize nβ-TCP and assess its efficacy in the rabbit tibial defect model.
Materials and methods
Synthesis of nβ-TCP and material characterization
nβ-TCP was synthesized using a wet chemical precipitation process with calcium nitrate tetrahydrate (Ca(NO3)2·4H2O) and diammonium hydrogen phosphate ((NH4)2HPO4) (Sigma-Aldrich, St Louis, Missouri, USA) as the calcium and phosphorus precursors, respectively. 21,22 Briefly, 0.4 mol (NH4)2HPO4 solution was vigorously stirred at room temperature, and 0.6 mol Ca(NO3)2 solution with pH 7.3 was added dropwise over 150–180 min to produce a white precipitate. Throughout the stirring process, the pH of the system was maintained at pH 10.8 by adding 25% ammonia solution (Sigma-Aldrich). The obtained white suspension was stirred for 8 h at room temperature. The synthesized precipitate was washed with distilled water and then with 100% ethanol (Sigma-Aldrich). The suspension was filtered and dried at 80 °C for 24 h. The dried powder was crushed using a mortar and pestle and calcined at 800°C for 2 h. The powder was autoclaved and stored under sterile conditions for use in further experiments.
The crystalline phase of the synthesized powder was evaluated using an AXS D8 Advance X-Ray Diffractometer (Bruker, Billerica, Masschusetts, USA). Each substance has a unique X-ray diffraction (XRD) pattern. A collection of the XRD patterns of many known substances has been archived in the Joint Committee on Powder Diffraction Standards (JCPDS) database by the International Centre for Diffraction Data. Thus, by comparing the XRD pattern of an unknown substance with the known substance in the JCPDS database, the identity of an unknown substance can be revealed.
The synthesized nβ-TCP was also subjected to Philips/FEI CM12 transmission electron microscopy (FEI Company, Eindhoven, Netherlands) to determine the size and morphology of the particles. The transmission electron microscopy (TEM) was equipped with an analysis Docu Version 3.2 image analysis system (Hillsboro, Oregon, USA). Finally, the particle size distribution of the synthesized nano β-TCP particles was measured using a Malvern Zetasizer Nano Series Nano-2 S (National Appliance Co., Portland, Oregon, USA).
Rabbit tibial defect model
Eight New Zealand white male rabbits (6 months old, weight 2.3–2.7 kg) were used in this study, which was approved by the Animal Ethics Committee of Universiti Sains Malaysia (AECUSM) (application number: USM/Animal Ethics Approval/2013/(86)(452)). The rabbits were housed in cages separately with water and food ad libitum. The room temperature was standardized, and the rabbits were subjected to a 12-h light/12-h dark cycle. Prior to the surgery, the rabbits were acclimated for 7 days to ensure their health and stability.
Surgical and implantation procedure
The rabbits were randomly divided into two groups (n = 4 for each group) and anesthetized intramuscularly by injecting a combination of xylazine (5 mg/kg body weight) and ketamine (35 mg/kg body weight). The surgical fields were shaved and disinfected. After gentle separation of subcutaneous tissue and periosteum, a defect site with dimensions of 10 mm length × 3 mm width × 2 mm depth was made on the dorsomedial part of the left tibia of each rabbit using an orthopedic hand drill machine with a drill bit size of 1.5 mm. 23 For the test item-treated group, the defect site was packed with 100 mg nβ-TCP. In the sham control group, the defect site was left empty. 23,24 The periosteum, subcutaneous tissue, and skin were then sutured with simple interrupted sutures, and the rabbits were placed back in their individual cages.
An antibiotic (enrofloxacin, 5 mg/kg body weight) was given intramuscularly once daily for 7 days. An anti-inflammatory drug (meloxicam, 0.2 mg/kg body weight) and a pain reliever (tramadol, 2 mg/kg body weight) were injected intramuscularly once daily for 3 days postoperatively. The surgical site was examined daily for complications, such as swelling, sepsis, pain, and abnormality of gait.
Digital radiography and computed tomography analysis
Lateral radiographs of the entire tibia in both groups were taken immediately after the surgery, and every 2 weeks thereafter for 16 weeks using a Philips DigitalDiagnost device (Hamburg, Germany). Computed tomography analysis was conducted at the same time points using a Siemens Somatom Defination AS+ (Muenchen, Germany). Samples were scanned with an X-ray tube (120 kV, 35 µA) with 360° rotation and a nominal single collimation width of 0.6 mm. The data sets were reconstructed and segmented for subsequent image processing using an OsiriX MD device (Bernex, Switzerland), and the estimated bone volume (cm3) of the whole surgical field for both groups was calculated.
Histological examination
Sixteen weeks after implantation, both groups of rabbits were euthanized with sodium pentobarbital (100 mg/kg), and the defect site was removed for histological examination. The defect site was washed with normal saline and fixed in 10% neutral formalin for 7 days. 24 10% Nitric acid was used to decalcify the bones until they were easily penetrable by pins, and then 4 µm sections were cut from the decalcified bones using Thermo Scientific Microm HM355 S Microtome (Kalamazoo, Michigan, USA). The sections were embedded into wax block using a Sakura Finetek Tissue-Tek® TEC™ Tissue Embedding Console System (Tokyo, Japan) and stained with hemotoxylin and eosin using a Leica Autostainer XL (Wetzlar, Germany).
Statistical analysis
Estimated bone volume results are given as the mean ± standard deviation. Results were compared using Mann–Whitney test to evaluate the difference in the estimated bone volume between the nβ-TCP-treated group and the sham control group at each specific time point. The value of p < 0.05 was considered to be statistically significant.
Results and discussion
Material characterization
nβ-TCP particles were synthesized using the wet precipitation method. Figure 1 shows the XRD spectra pattern of the sample. The diffraction pattern of the synthesized sample matched well with the standard XRD pattern of β-TCP (JCPDS reference 01-070-2065), indicating that the synthesized sample was β-TCP. To determine its morphology, the nβ-TCP sample was examined under TEM. The sample consisted of single and almost spherical nanoparticles. Figure 2 shows the average particle size distribution (Z-average) of the synthesized nβ-TCP sample. The sample contained two different particle size distributions; the Z-average of peak 1 was 1.414 nm (65.3%) and the value for peak 2 was 2199 nm (34.7%). The overall average particle size distribution (d 50) of the synthesized sample was 38.38 nm.
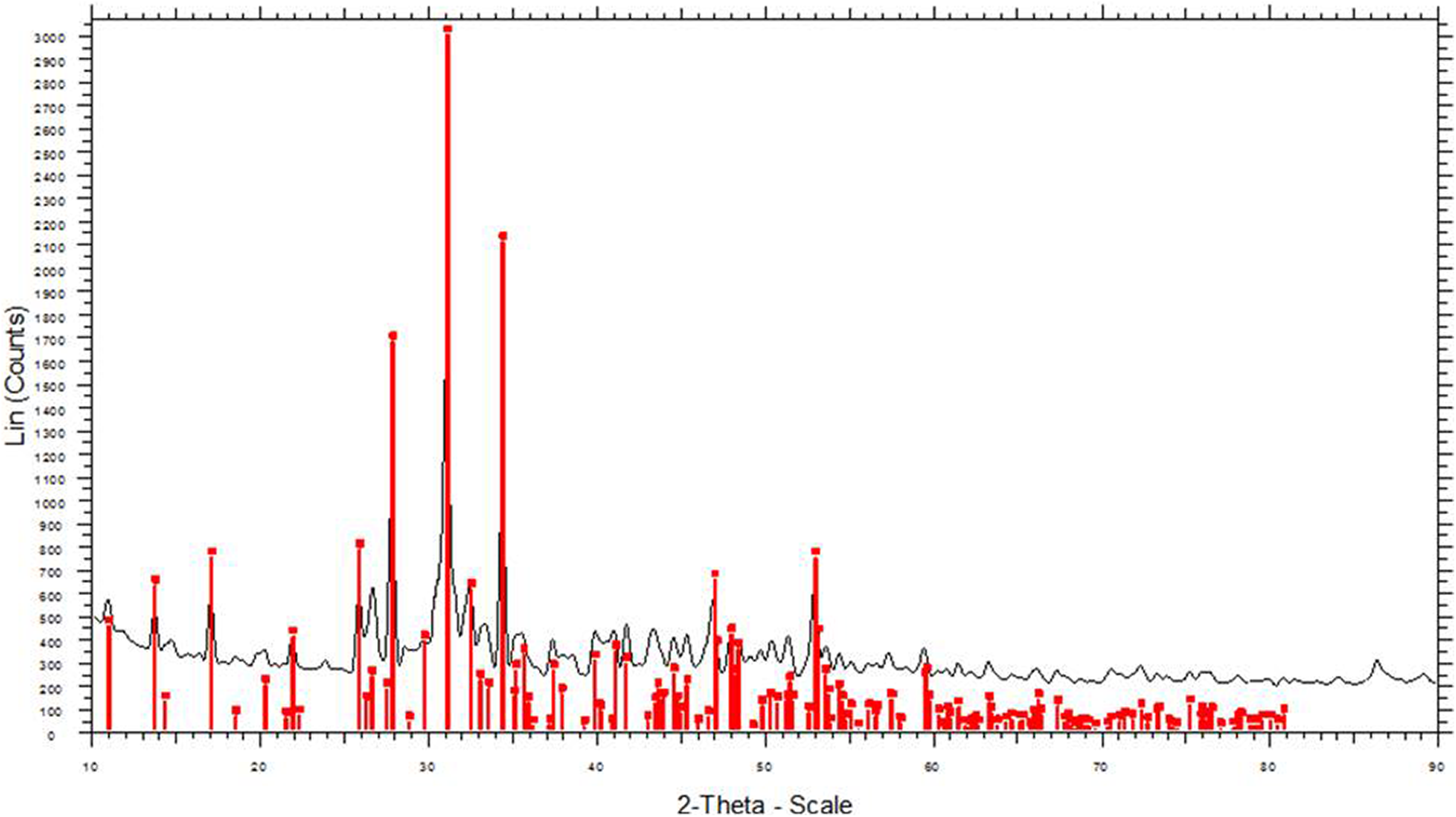
XRD spectrum pattern peaks of the synthesized nβ-TCP particles (black line) and the JCPDS reference (01-070-2065, red line). XRD: X-ray diffraction; nβ-TCP: nano-β-tricalcium phosphate.
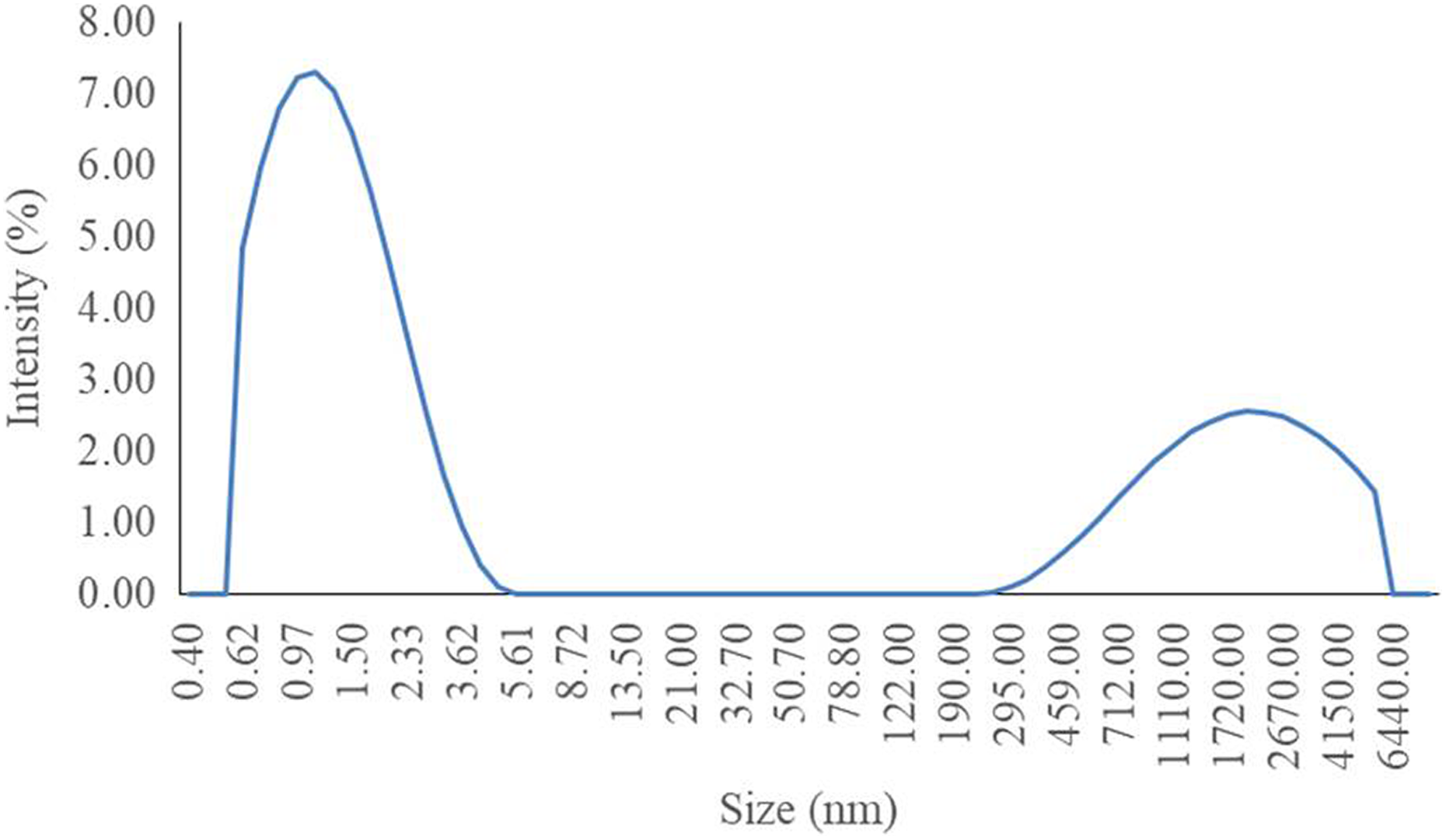
Particle size distribution of the synthesized nβ-TCP. nβ-TCP: nano-β-tricalcium phosphate.
Calcination temperature plays an important role in controlling the size and morphology of β-TCP particles. 21 An increase in calcination temperature produces highly reactive spherical particles, which leads to agglomerates of larger sized β-TCP particles. The individual spherical particles were shown to join together through “budding” or neck growth at high temperature. Thus, particle size increases with increased calcination temperature from the nano range at 800°C to the micron range at 1000°C calcination. 21 Because the primary objective in the current study was to synthesize nβ-TCP particles, lower calcination temperature (i.e. 800°C) was more favorable and therefore was used.
Radiography and computed tomography analysis
The defect site of both groups was radiolucent after surgery (week 0) (Figure 3). Radiographs taken at week 10 showed that the defect site of the sham control group was radiopaque, whereas that of the nβ-TCP-treated group appeared to be radiolucent throughout the experiment. Computed tomography scans also revealed that the defect site of the nβ-TCP-treated group was not fully recovered after 16 weeks as compared to the sham control group (Figure 4). Full bone recovery was achieved in the sham control group at week 10, and the radio-dense area was increased at week 16. In contrast, a clear gap between the trabecular bones was observed in the nβ-TCP-treated group at week 16, indicating that the bone regeneration process was hindered by the nβ-TCP particle implantation.
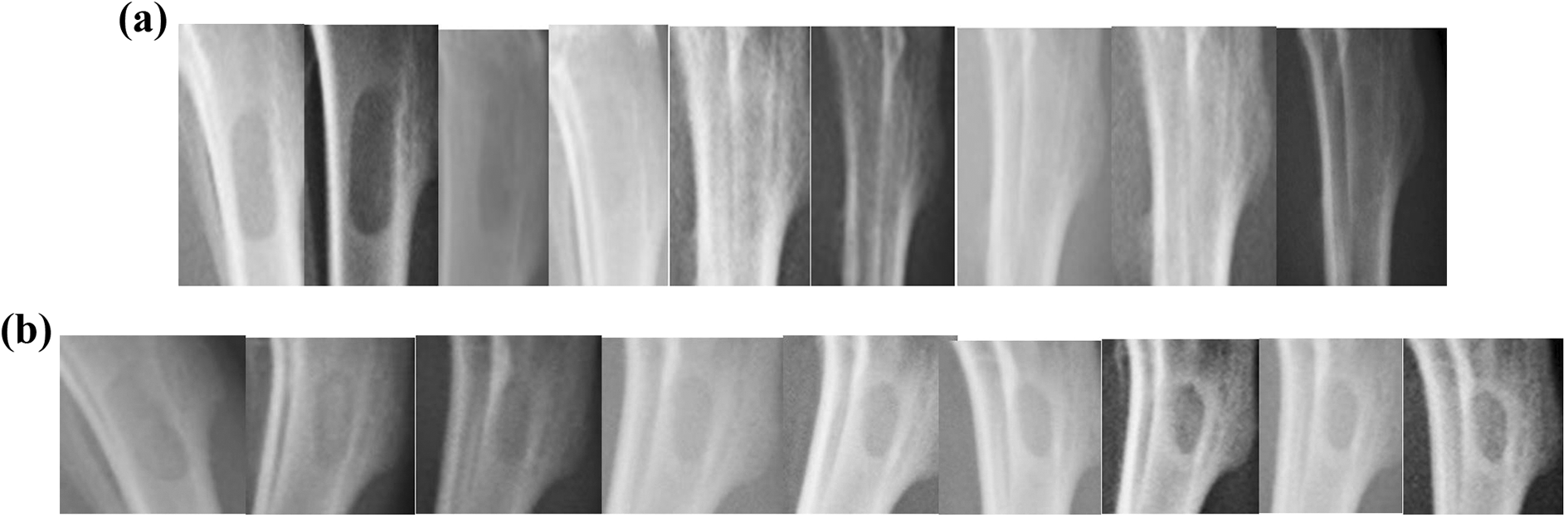
Tibia radiograph scans of the (a) sham control group and (b) nβ-TCP-treated group from week 0 (postsurgery) to week 16. nβ-TCP: nano-β-tricalcium phosphate.
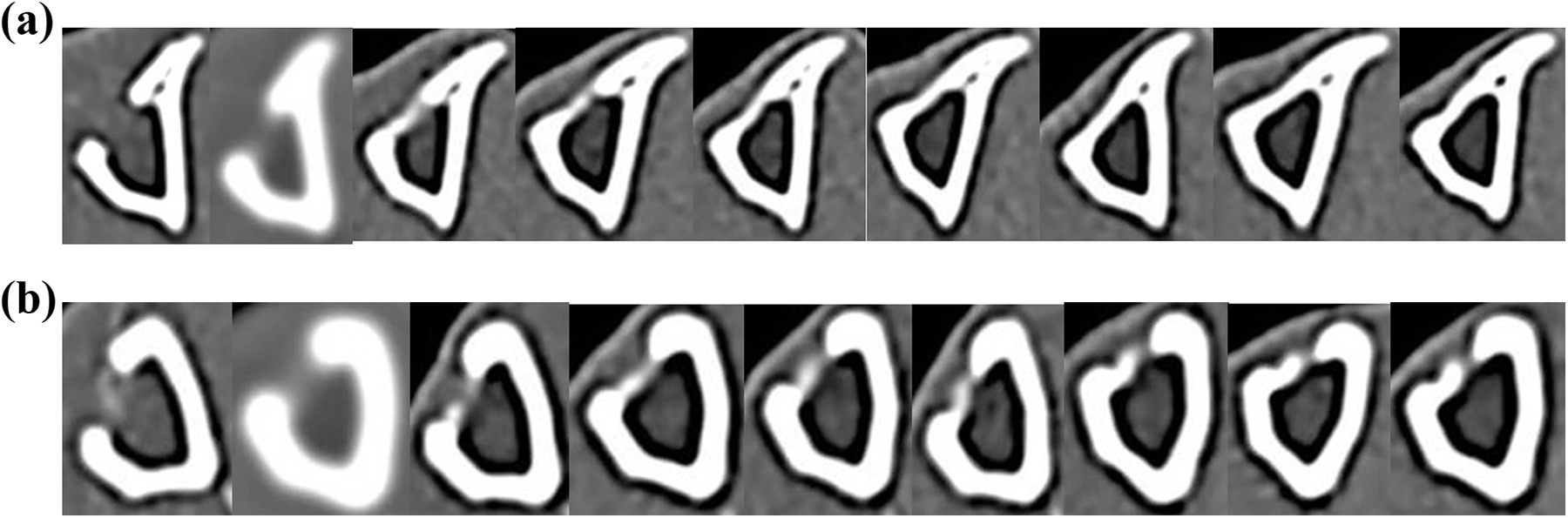
Tibial computed tomography scans of the (a) sham control group and (b) nβ-TCP-treated group from week 0 (postsurgery) to week 16. nβ-TCP: nano-β-tricalcium phosphate.
In the sham control group, the estimated bone volume of the defect site increased from 0.272 cm3 at week 0 (postsurgery) to 0.458 cm3 at week 8, and the final bone volume recorded on week 16 was 0.465 cm3 (Figure 5). On the other hand, the recovery progress of the nβ-TCP-treated group clearly lagged behind, and the estimated bone volume only slightly increased from 0.302 cm3 at week 0 to 0.342 cm3 at week 16. The defect site of the nβ-TCP-treated group still exhibited incomplete restoration at week 16, whereas full bone recovery was observed in the sham control group.
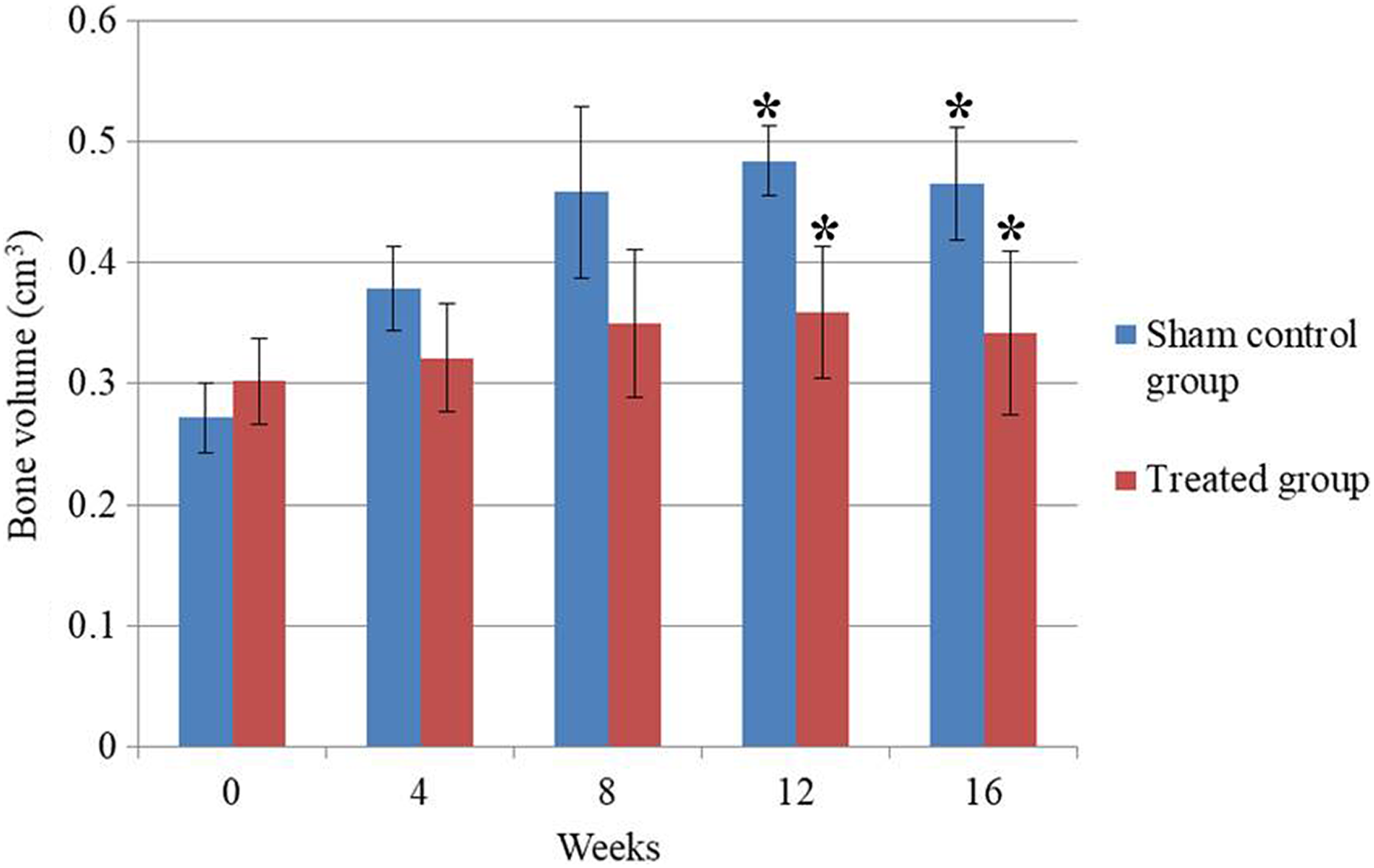
Estimated bone volume (cm3) of the sham control group and nβ-TCP-treated group from week 0 (post-surgery) to week 16 based on data from the computed tomography scan. The values represent the mean of three independent measurements (mean ± standard deviation). Asterisks indicate significant difference at p < 0.05 (Mann–Whitney test). nβ-TCP: nano-β-tricalcium phosphate.
Gross examination
All rabbits were euthanized at week 16, and the bone with the surgical site was extracted. Gross examination revealed that the sham control group showed full recovery (Figure 6(a)). The healed bone was not penetrable by pins, indicating that the defect gap was filled with hard tissue. In contrast, the surgical site of the nβ-TCP-treated group was only filled with soft tissue (Figure 6(b) and (c)). The surgical site was easily penetrated with pins, and the defect gap was not fully recovered. However, the dimension of the defect site was slightly reduced in the nβ-TCP-treated group.
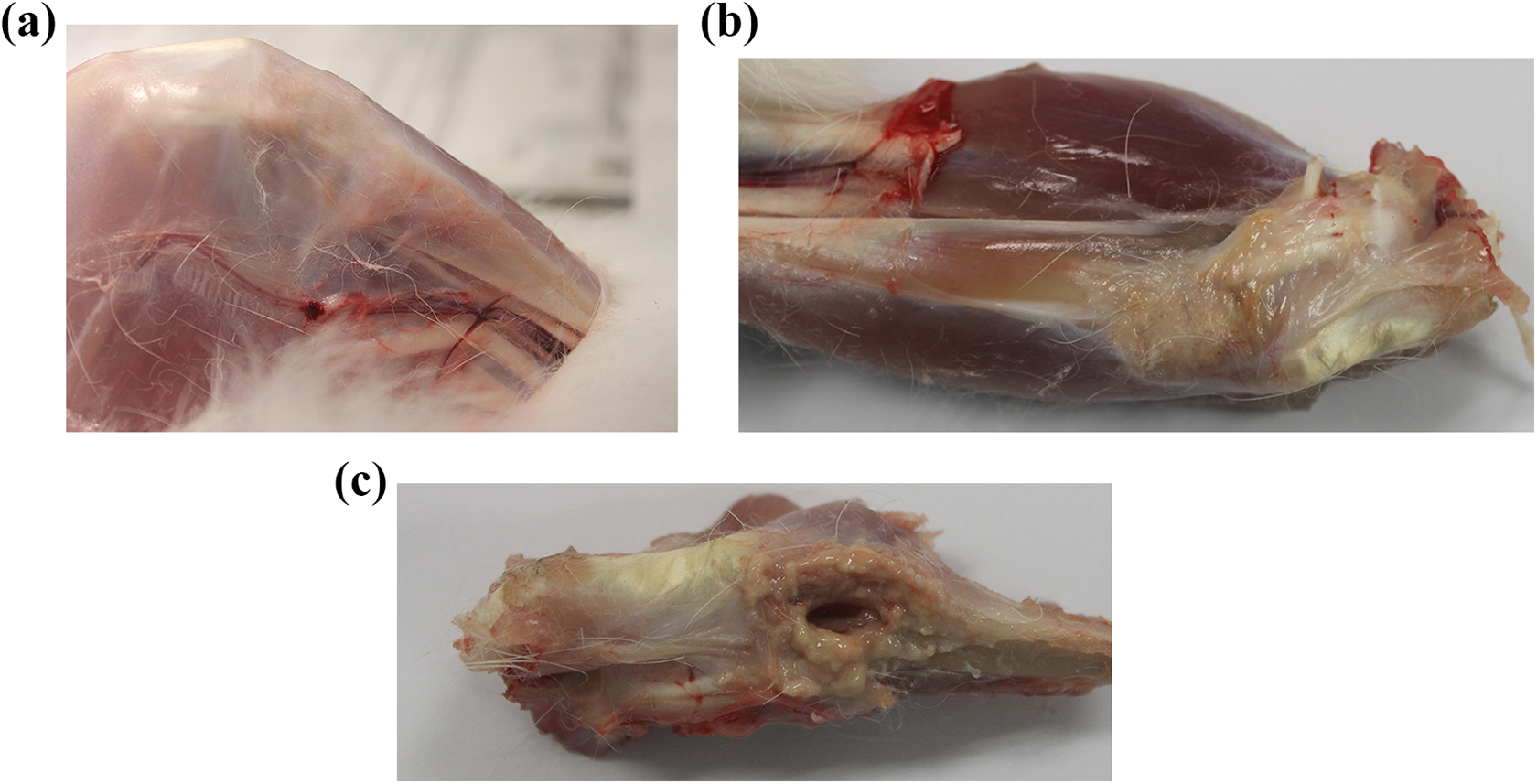
Gross examination of the surgical site in the (a) sham control group and (b and c) nβ-TCP-treated group. No soft tissue was present in the tibia of the sham control group (a), whereas soft tissue was present at the defect site of the nβ-TCP-treated group (b), and the defect site was easily penetrated by a pin (c). nβ-TCP: nano-β-tricalcium phosphate.
Histological evaluations
Sixteen weeks after implantation, the sham control group showed complete healing, with the formation of lamella bone without residual inflammatory changes (Figure 7). However, the nβ-TCP-treated group showed marked granulomatous inflammation that was composed of sheets of histiocytes and scattered multinucleated giant foreign body type cells in the surgical site (Figure 8). Clefts with structures similar to cholesterol clefts were also observed. The granulomatous inflammation spread into the Harversian system and beyond the gap in the surface of the bone and into the surrounding soft tissue. However, there was no destruction of the surrounding existing bone and soft tissue, and a focus of new bone formation was found in the center of the defect site.
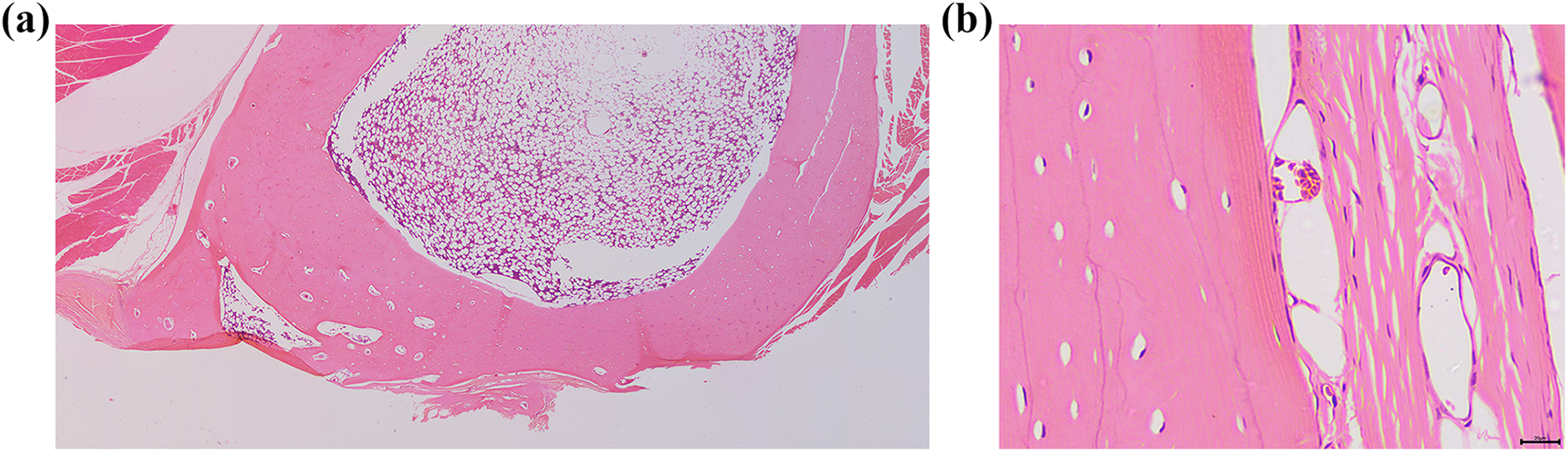
Hematoxylin and eosin-stained section from a non-treated rabbit 16 weeks after surgery. The created defect site was completely healed (a), and a zoom in at the highlighted area (b) showed the formation of lamella bone. (Magnification power: ×20, scale bar: 20 µm).
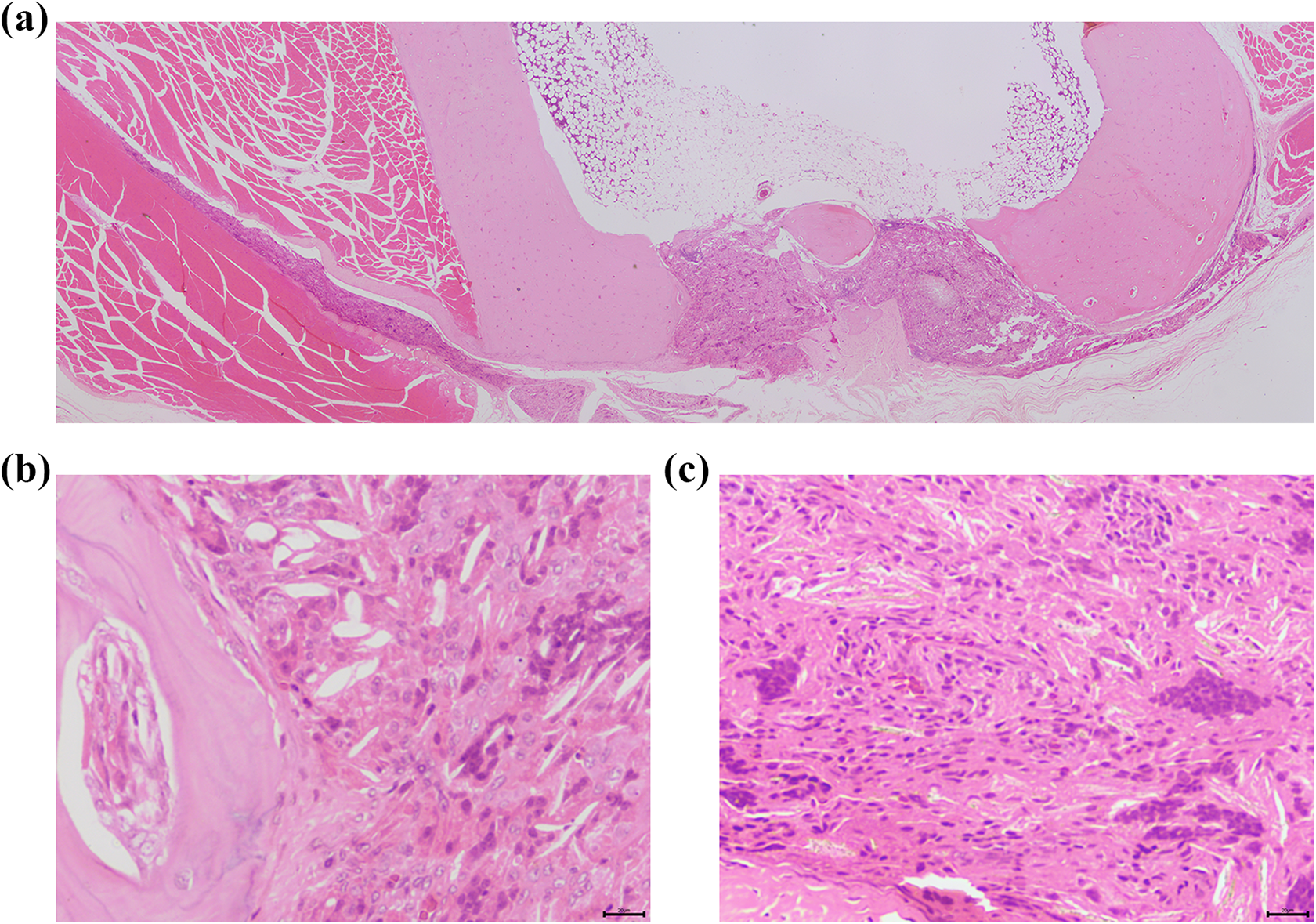
Hematoxylin and eosin-stained section of a nβ-TCP-treated rabbit 16 weeks after implantation. The created defect site was not healed (a), and a zoom in at the highlighted area (b) showed the presence of foreign body-type multinucleated giant cells at the gap and also in the surrounding soft tissue (c). (Magnification power: ×20, scale bar: 20 µm.).
Immunological responses of hosts following implantation are a common problems in bone healing. 25 Upon implantation, the inflammatory reaction is the first stage of the bone healing whereby inflammatory cells (macrophages, monocytes, lymphocytes, and polymorphonuclear cells) are recruited and colonize the β-TCP surface. 6,25,26 Tai et al. demonstrated that subcutaneous implantation of β-TCP in normal B6 mice evoked extensive migration of immune cells into the area surrounding the implantation site. The β-TCP was initially colonized by neutrophils, followed by lymphocytes, histiocytes (macrophages), and fibroblasts, and finally by multinucleated giant cells. 6,26 Studies have reported that human peripheral blood mononuclear cells and J774A.1 cells (monocyte/macrophage cells derived from mice) incubated with β-TCP exhibited upregulation of the protein level of tumor necrosis factor alpha. 26,27 β-TCP also induced the protein level of macrophage inflammatory protein 1 alpha in J774A.1 cells. 26 Both proteins are known to stimulate recruitment of various immune cells to the site of inflammation, thus upregulation of these proteins in the presence of β-TCP triggers and enhances the migration of a huge number of immune cells towards the area surrounding β-TCP in vivo. 6,26
The recruitment of these immune cell types to the vicinity of β-TCP lead to degradation of β-TCP via phagocytosis. Eggli et al. found a cluster of macrophages packed with granular β-TCP on the implantation site in a rabbit model, which suggests that macrophages may play an active role in intracellular degradation of small detached ceramic particles. 28 Human peripheral blood mononuclear cells and J774A.1 cells were also found to incorporate β-TCP by phagocytosis, 26,27 and recent studies demonstrated the involvement of foreign body multinucleated giant cells (FBGCs) in degrading β-TCP. 29,30 FBGCs form by fusion of monocytes/macrophages only under pathological conditions or at the surface of a foreign material such as β-TCP. Ahmed et al. demonstrated that FBGCs were able to degrade β-TCP without expression of tartrate-resistant acid phosphatase and cathepsin K, which are the classical osteoclast markers involved in bone resorption; this result suggests that FBGCs have functional similarities with osteoclasts. 29 ten Harkel et al. observed similar functions of FBGCs in their study. 30 Thus, the presence of β-TCP, as a foreign material, triggered and enhanced the recruitment of immune cell types towards itself, which in turn led to the degradation of β-TCP and subsequent bone regeneration.
However, an imbalance of inflammation and bone formation may impede the bone regeneration process. 29,31,32 Chen et al. incorporated cobalt with β-TCP powders, and the material significantly upregulated the expression of pro-inflammatory cytokines (such as interleukin (IL)-1β and IL-6) and other bone destructive factors (such as macrophage colony-stimulating factor and hypoxia-inducible factor 2α) in a rat femur condyle defect model. 32 The cobalt ions stabilized the hypoxia-inducible factors, which are known to participate actively in the maturation of macrophages and their functional activities, and amplified the inflammation. Although the material also enhanced osteogenic differentiation of bone marrow stem cells, which is a positive effect for the bone regeneration process, the effect was canceled out by overexpression of the pro-inflammatory cytokines and bone catabolic factors released from the macrophages. High release of pro-inflammatory cytokines impeded osteogenic differentiation, and the inflammation went hand-in-hand with the destruction of bone tissue. 32
The extent of the inflammatory response caused by β-TCP depends greatly on the physicochemical properties of the β-TCP particles, which in turn affect the degradability of the implant (i.e. release of calcium and phosphate ions, precipitation of calcium phosphate crystals, and accumulation of microparticulate degradation products). 31,33 Particle size, pore size, porosity, and pore interconnectivity of the β-TCP scaffold are among the many physicochemical properties that not only influence the degradation of β-TCP but also subsequently affect the bone regeneration process. 34 For instance, Ghanaati et al. demonstrated that smaller-sized β-TCP initiated the generation of multinucleated giant cells faster than larger-sized β-TCP. 35 Because the nβ-TCP particles synthesized in this study have more surface area per unit mass and increased surface reactivity compared to bulk-sized particles, degradation of the synthesized nβ-TCP particles likely was significantly increased, leading to the spontaneous release of calcium and phosphorous ions compared to physiological conditions. Okuda et al. showed that excessive biodegradability of the β-TCP scaffold that caused a loss of bone tissue in the rabbit femur. 33 At week 12 after implantation, biodegradation of the β-TCP scaffold was accompanied by the new bone formation, but a significant loss of bone tissue was observed at 24 weeks postimplantation. 33 Results of an in vitro study showed that an imbalance between degradation of the β-TCP scaffold and bone formation was the reason for implant failure. 36 A study in sheep revealed that fast degradability of the β-TCP scaffold caused a loss of bone content and higher soft tissue content.
Conclusion
The in vivo results of this study indicated that the synthesized nβ-TCP particles were not an ideal bone void filler. The in vivo study revealed that the nβ-TCP particles caused an excessive and prolonged inflammatory response by the host that impeded the bone regeneration process in the rabbit tibial model.
Footnotes
Acknowledgements
The authors are indebted to the Advanced Medical and Dental Institute (AMDI) at Universiti Sains Malaysia for providing an AMDI Student Research Grant to finance this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support from Advanced Medical and Dental Institute (AMDI) for the publication of this article.
