Abstract
In the last few years, considerable attention has been given to in vitro models in an attempt to reduce the use of animals and to decrease the rate of preclinical failure associated with the development of new drugs. Simple two-dimensional cultures grown in a dish are now frequently replaced by organotypic cultures with three-dimensional (3-D) architecture, which enables interactions between cells, promoting their differentiation and increasing their in vivo likeness. Microengineering now enables the incorporation of small devices into 3-D culture models to reproduce the complex microenvironment of the modeled organ, often referred to as organs-on-a-chip (OoCs). This review describes various OoCs developed to mimic liver, brain, kidney, and lung tissues. Current challenges encountered in attempts to recreate the in vivo environment are described, as well as some examples of OoCs. Finally, attention is given to the ongoing evolution of OoCs with the aim of solving one of the major limitations in that they can only represent a single organ. Multi-organ-on-a-chip (MOC) systems mimic organ interactions observed in the human body and aim to provide the features of compound uptake, metabolism, and excretion, while simultaneously allowing for insights into biological effects. MOCs might therefore represent a new paradigm in drug development, providing a better understanding of dose responses and mechanisms of toxicity, enabling the detection of drug resistance and supporting the evaluation of pharmacokinetic–pharmacodynamics parameters.
Introduction
With only 10% of potential anticancer drugs progressing through the different clinical phases, there is a huge financial burden of failure for potential drugs, which is estimated to cost between 800 million and 1.4 billion US dollars. Hence, there is an urgent need to develop more accurate and more physiologically relevant in vitro models that will aid progression of drugs through clinical phases with a better success rate. 1,2
Current in vitro models for preclinical testing rely on two-dimensional (2-D) or simple three-dimensional (3-D) culture systems. Two-dimensional cell culture systems are widely used as fast, simple, and cost-effective models to perform toxicological studies with low maintenance demands and rather unproblematic application of treatments. Although useful in the early development phase of potential drugs (e.g. compound screening), their simplistic nature can be a major drawback because the results obtained from such a simple model can be misleading and do not reflect the mechanisms occurring under physiological conditions. 3,4
During the last decade, 3-D models have been developed to improve the resemblance to the in vivo tissue environment. In principle, 3-D culture systems can be described as the assembly of primary cells, cell lines, or stem cells into a 3-D structure using scaffolds or specific cell culture plates. The spatial organization within the 3-D culture systems allows the cells to exhibit cell–cell interactions and the buildup of an extracellular matrix, which consequently provides a physiological microenvironment where cells are subjected to polarization and zonation. 5 Several 3-D models are currently available and new ones are increasingly being developed to model almost all the organs of the human body. Each 3-D culture model has advantages and disadvantages; nonetheless, they can still be adapted to be useful tools to address particular scientific questions. For example, air–liquid interface (ALI) airway models can be used to test the impact of cigarette smoke exposure on cilia function and the secretion of mediators 6 but are not suitable for investigating the effect of breathing as the tissues are grown on static membranes. Ultimately, all 3-D cultures share more similarity with their in vivo counterparts than with the original 2-D cultures. 4
Despite improvements in 3-D culture engineering, the cultures are currently only maintained in a static condition, which is different from the physiological reality. Lung epithelial cells stretched by breathing, intestinal cells exposed to peristaltic movement, or liver cells exposed to blood flow are all examples of key physiological forces occurring in the human body, which are rarely, or not at all, observed in vitro. As cells have to grow, differentiate, and survive under particular mechanical constraints in vivo, recreating such environments in vitro would likely improve the physiology of the cells. More realistic models with engineered microenvironments have therefore been developed during the last few years to mimic better the key aspects of a tissue in vivo. 7,8 Results from such models have demonstrated that creating a more realistic cellular environment can induce a behavior closer to the physiological situation. The human intestinal Caco-2 cell line, which forms a 2-D cell layer when grown in a dish, has, for example, undergone spontaneous villus differentiation if the cells are subjected to peristaltic movement. 9 Blood flow on endothelial cells 10 and physical pressure on osteoblasts 11 are other examples of how specific microenvironmental features can modulate cellular function. The addition of such features to 2-D cell systems has enabled the transition to more physiologically relevant 3-D models, and artificial engineering of such microenvironments has helped create even more realistic models, giving rise to biochips or organs-on-a-chip (OoC). 8
OoC systems can be described as the combination of a culture, in general, a 3-D tissue to benefit from the in vivo resemblance, with an engineered cell culture platform or biochip. 8 An electronic unit integrated into the chip supports the recreation of the physiological microenvironment by, for example, directing a flow of medium or applying pressure on a membrane to cause a mechanical stress (Figure 1). The overall complexity of the chip can be tailored depending on the need. Some chips are developed by simply integrating a peristaltic pump, while others integrate various sensors that can simultaneously and continuously record the pH and oxygen consumption, monitor the morphology, and detect soluble proteins released into the culture medium. 12 Until recently, almost all chips that are commercially available are built using poly(dimethylsiloxane) (PDMS). PDMS is biocompatible (it is not toxic nor has any deleterious effects on tissues), is transparent, permeable to gas, exhibits low autofluorescence, and is fairly inexpensive. 13 As PDMS can be easily patterned using soft lithography, there are no limitations regarding the chip design. Thus, some devices have been designed to accommodate specific cell types in either mono- or co-culture. For example, there are chips for the maintenance of specific cells types on a stretchable membrane at the ALI, 14 while other chips are more modular and can house multiple organ cultures. µOrgano is an example of a micro-module chip where different cell types can be injected into micro-sized wells. 15 Conversely, Quasi Vivo® is a macro-module system that connects different chambers, each of which can harbor tissue from a different organ type, in various combinations. 16,17 One of the major drawbacks of PDMS is its ability to absorb small hydrophobic compounds. 18,19 Therefore, the material is not suitable for all toxicity studies. For example, if the drug to be tested is absorbed and retained by the PDMS, then the actual concentration of drug delivered is lower, and potential toxicity might be underestimated. Alternatively, other thermoplastic materials, such as polystyrene, polycarbonate, and poly(methyl methacrylate) (PMMA), can be used to develop biochips, because they are not only optically clear but also generally less absorbent than PDMS. 20,21 However, they are not as durable and are costly, which can be a disadvantage, particularly for a high-throughput setting. Three-dimensional printing technology offers an interesting alternative for the development of biochips, as demonstrated recently. 22 A chip can be easily designed using computer software and printed in various “ink materials,” such as silicone, polycaprolactone, or Pluronic® F127. Because the printing occurs layer-by-layer, sensors and mechanical parts can be incorporated into the chip at any point or position. The ever-increasing use of 3-D printers for laboratory applications has led to new printing technologies with higher precision and new ink materials with better biocompatibility. 23 At the same time, the materials have become less expensive and hence more able to be considered for routine use.
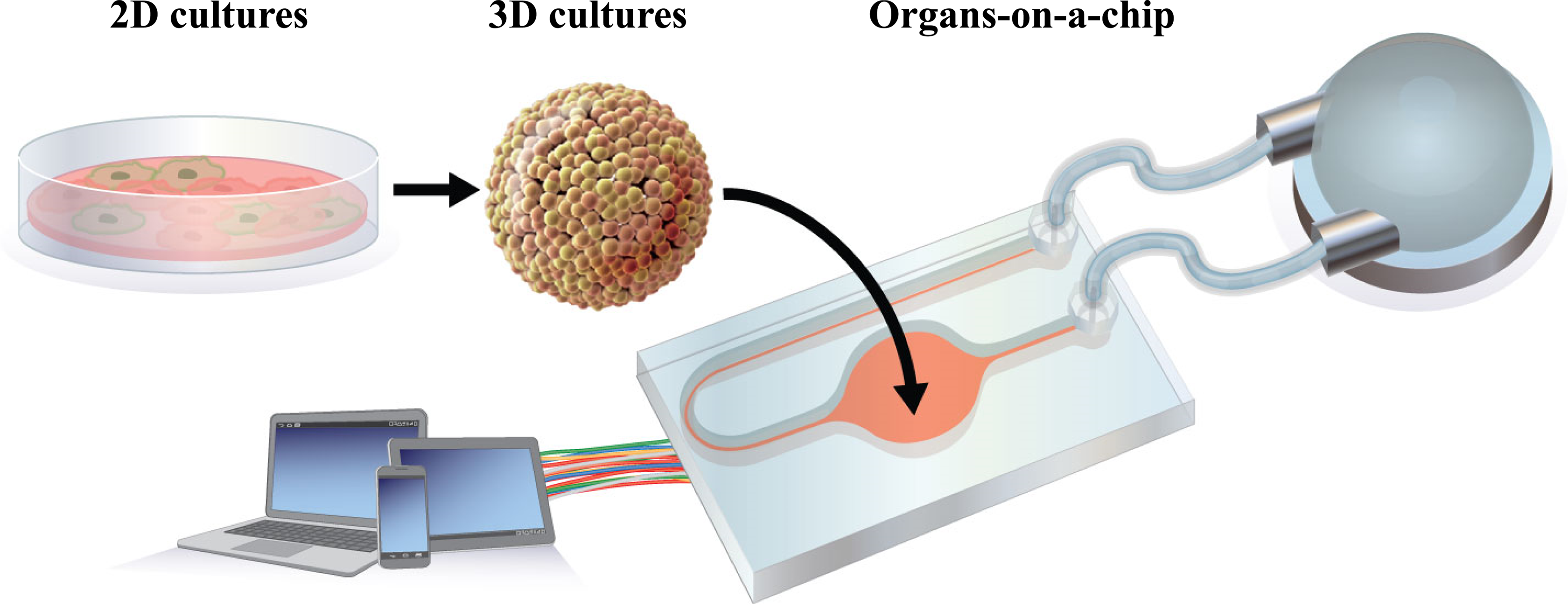
Evolution of in vitro models from simple monolayer cultures to OoCs. Monolayer cultures have previously been the choice for in vitro models for toxicological studies because of their legacy value and ease of maintenance. The development of new materials and matrices has enabled the growth of 3-D organotypic models with improved physiological characteristics. Microengineered devices, incorporating fluidics and sensor technologies, have then permitted the transfer of these 3-D tissues to a dynamic environment, creating organs-on-a-chip that more closely resemble human organs in vivo. OoCs: organs-on-a-chip.
Organs-on-a-chip: New in vitro tools
Interest in OoC technology for drug development, risk assessment, and exploratory, mechanistic research applications has grown rapidly in recent years. New chips are constantly being developed that cover almost all organs present in the human body. In the following section, we briefly describe models of primary OoCs for liver, brain, lung, and kidney tissues. Key challenges that were encountered during their development are discussed.
Liver-on-a-chip
The liver is the second largest organ in the human body, which not only regulates energy use; produces bile, hormones, and blood plasma proteins but also metabolizes xenobiotic compounds. The capacity of liver tissue to metabolize xenobiotics emphasizes the need for a liver-culture system that is physiologically relevant. Xenobiotic metabolism can occur in most tissues of the human body, but the liver is the most active. Once xenobiotic drugs are absorbed, they enter the blood circulation and reach the liver, where they undergo a two-step process to transform a generally lipophilic compound into a more hydrophilic molecule. 24,25 During phase I metabolism, xenobiotics are oxidized, reduced, or hydrolyzed. 26 The cytochrome P450 (CYP) family of enzymes, of which there are more than 50 members, are major regulators of phase I metabolism, which display substrate specificity. 27 Phase II xenobiotic metabolism involves glucuronidation, sulfation, and acetylation reactions, mainly catalyzed by transferases that further modify the previously transformed substrates. 24,25 The resulting product has higher water solubility and thus can be actively transported across the membrane, leading to excretion.
Studies have shown that several drugs, which are known to be inert, can be biotransformed in the liver and converted into reactive compounds (such as free radicals) that may induce local and/or systemic toxicity. 28 Phase I and II xenobiotic metabolism can occur in different regions of the lobule. Hepatocytes present in the pericentral zone, where nutrient and oxygen levels are low, express enzymes necessary for phase I metabolism, whereas hepatocytes located in the periportal zone, where nutrients are abundant, express phase II enzymes. 29 Ideally, a liver biochip should exhibit this nutrient zonation reproducing the physiological metabolism of drugs. Such a gradient of nutrients can be implemented in vitro by forming liver spheroids. 30 In the spheroid model, cells located near the core have limited access to nutrients compared with the cells located more peripherally. 31
With respect to liver OoCs, the need for an engineered microenvironment came from the observation that the liver is one of the most highly perfused organs in the human body, acting as a bioreactor to metabolize compounds delivered by the blood at a high rate. 32 Vascularization is therefore essential for hepatocyte function and ultimately also for metabolic waste removal. As blood flow in the liver exerts shear stress on hepatocytes, leading to changes in the expression of several genes, 33 researchers and engineers have developed a perfused liver-on-a-chip model to recreate such a mechanism. 34 Today, many liver-on-a-chip systems are available, which can be attributed to the following: (a) the liver is an important organ for drug metabolism, (b) recent improvements in the development of 3-D liver models, and (c) the relatively simple requirement for creating the desired microenvironment (i.e. a simple medium flow).
Bhise et al. have recently reported a liver-on-a-chip platform using bioprinted HepG2/C3A spheroids as a 3-D liver model. 35 The PDMS chip has three chambers: a central bioreactor, containing the spheroids and two external chambers connected to the inlet/outlet of a syringe pump. Albumin, transferrin, ceruloplasmin, and alpha-1-antitrypsin production by the spheroids was monitored and found to be stable over a 30-day period. The response of the spheroids to acute acetaminophen exposure was reportedly similar to that observed in animal studies, demonstrating that drug-induced liver injury can be mimicked by OoC. Bavli et al. used a similar system to create a liver-on-a-chip using PDMS micro-wells covered with a glass layer and connected to a pump. 36 To monitor the liver tissues in real-time, they incorporated sensors in the plate that were combined with oxygen-reactive micro-particles embedded in the tissues to measure the levels of glucose and lactate. 36 In addition, using an off-chip sensor unit, glucose consumption and lactate release were measured every 20 min. The results demonstrated a switch from the oxidative to the glycolytic pathway after the spheroids were exposed to rotenone or troglitazone. Troglitazone treatment resulted in adverse effects on the spheroids that occurred below the known threshold of troglitazone toxicity. This work showcases how an OoC can better predict the toxicity of compounds than a simpler in vitro model. Several other liver-on-a-chip systems have been developed and investigated for various applications using a similar design but different types of tissues. 37–42 The results from these systems have been encouraging and have stimulated the development of novel liver chips. For example, alcoholic liver disease has been modeled in a liver chip, using a 48-h exposure of liver spheroids to different concentrations of ethanol followed by a 3-day recovery period. The exposed spheroids exhibited impaired viability, morphology, and albumin secretion compared with control tissues. Incorporation of hepatic stellate cells in the liver spheroids exposed to ethanol resulted in the formation of fibrous structures similar to those observed in vivo during the progression of liver fibrosis. 43 Another application of liver-on-a-chip models is to identify metabolites generated from compound breakdown and to investigate potential secondary toxic effects. For example, coupling of liver microsomes with 2-D HepG2 cultures, exposed to medium flow in a microfluidic device with integrated micro-solid phase extraction columns, has been used to detect secondary metabolites and simultaneously to assess the toxicity elicited by the metabolites. 44 It should be noted that although this approach can be used to detect the main metabolites resulting from compound metabolism, other degradation products, potentially present in very low concentrations, could be missed because of the rather low cell numbers and relatively low concentrations of test compounds required for the format of the device.
Attention needs be paid to the choice of cell type used in a liver-on-a-chip model. Primary human hepatocytes, for example, are currently considered the most physiologically relevant cell type; however, their availability is limited. Primary human hepatocytes also have greater variability (donor dependent), for example, in regard to the expression of genes encoding xenobiotic enzymes, which becomes a disadvantage in larger scale experiments. Alternatively, HepG2 cells can be used; however, the expression levels of xenobiotic enzymes vary across passages. Moreover, HepG2 cells have low CYP inducibility in response to drugs, which is a limitation in toxicological assessments. The HepG2-derived C3A cell line, which is a more differentiated hepatic model, has been shown to be more metabolically active than the HepG2 cell line. 45,46 More recently, the HepaRG™ cell line has been developed, which has superior characteristics compared with either HepG2 or C3A cells. HepaRG™ cells highly express xenobiotic enzymes, secrete albumin abundantly, and have an extended life span compared with the previously mentioned cells. 47 The properties of HepaRG™ are comparable to primary human hepatocytes, thus offering a suitable alternative to primary liver cells. 48 A study showed that spheroids composed of primary hepatocytes performed similarly to HepaRG™ cells in regard to detecting compounds with cholestatic liability. 49 However, other studies have found HepaRG™ cells to be more sensitive to hepatotoxic agents than primary human hepatocytes. 50,51
Several studies have demonstrated that 3-D structures can improve key functions of the hepatic model compared with 2-D cultures. 52,53 Signs of cytotoxicity were detected with lower drug concentrations compared with 2-D cultures in standard plates, 54 or on a chip, 36 providing proof that 3-D cultures with their engineered microenvironments might help to identify early signs of hepatotoxicity and thus allow for the development of drugs with a better prediction of their margin of safety. Finally, spheroids composed of mixed cell populations can be used to improve the model physiology 55 and to understand diseases caused by the intricate interactions between hepatocytes and immune cells. 56
Brain-on-a-chip
Brain models are of particular interest for toxicological testing because the brain’s structure and function are different from those of other organs in the human body. A high level of complexity caused by the presence of the blood–brain barrier (BBB), which excludes up to 98% of small molecules 57 ; the interaction of multiple cell types 58 ; and the variable ratio of these cells in the different regions of the brain, 59 means that the development of a drug able to enter the brain and target only a specific region without causing side effects is challenging. Indeed, drugs targeting the central nervous system have a probability of success lower than 10%. 60
In vitro models of the BBB are useful for the development of drugs capable of crossing this barrier. Over time, the development of such models has benefited from several innovations, each increasing the in vivo likeness of the models. Initial models consisted of endothelial cells grown on a porous membrane to form a barrier between the apical side of the insert (the luminal compartment) and the basal side (the abluminal compartment). This physiological disposition of the cells allowed them to develop apical/basal polarization. 61 A major improvement involved the co-culture of endothelial cells grown on porous membranes with astrocytes. The astrocytes were found to increase the expression of transporters and tight junction proteins, increasing cell–cell interactions and consequently transendothelial resistance. 62 Because endothelial cells forming the BBB are subjected to blood flow, shear stress was hypothesized as also possibly being an important stimulus for these cells. Endothelial cells co-cultured with astrocytes on a porous hollow fiber and exposed to a physiological shear stress resulted in a barrier with a permeability similar to that seen in vivo. 63 Shear stress also promoted the expression of P450 enzymes, drug transporters, and tight and adherens junctions. 64 Based on these developments, several BBBs-on-a-chip have been designed, using the same approach but different chip formats, 65–69 and used, for example, to investigate drugs that may directly protect endothelial cells from deleterious events 70 or to improve the understanding of mechanisms underlying brain metastasis. 71 Future generations of engineered BBB models, such as those leveraging the potential of stem cells, 72 will undoubtedly help scientists to better predict the permeability of this barrier to newly developed compounds and help in creating BBB-permeant drugs.
An important aspect of the brain is the presence of several regions with specific functions and anatomy. The ratio of neurons to glial cells, 59 metabolic processes, 73 and electrical activity 74 varies between these structures, and this suggests that each region of the brain might exhibit cellular or proteomic specificities. Once a compound has crossed the BBB, it might then exert different effects depending on the regions it reaches. To study this phenomenon, the Wyss Institute at Harvard University created a multiregional brain-on-a-chip. 75 Initially, three different (traditional) neuronal cultures from the hippocampus, prefrontal cortex, and amygdala were characterized, demonstrating that even in vitro, each region had different cell compositions, protein expression, metabolism, and electrical activity. Then these cultures were transferred to a glass coverslip with a PDMS mask to create three separate chambers. Once the PDMS mask was removed, cells from each region grew axons and connected with the two other regions influencing their electrical activity. The injection of a drug known to cause schizophrenia-like symptoms was found to induce different electrical activities depending on the neuronal cell population tested. A potential drug could therefore be tested in this system to observe any potential region-specific effects. 75
Another aspect specific to the brain is that it reaches maturation only after 20 years. Prior to this, brain cells reorganize and make new connections essential for learning. 76 Any compound crossing the BBB and affecting brain cell maturation could induce permanent injuries, which poses a risk to the normal brain development of children and adolescents. 77 The brain-on-a-chip model might therefore prove valuable in studying potential toxic effects on the brain when testing drugs that may be given to children or adolescents. For example, methylphenidate, a drug for the treatment of hyperactive disorder in children, which currently suffers from a lack of evidence regarding its effects on brain maturation, 78 could be assessed using such a platform. A first prototype combining brain tissue and a BBB, developed by Kilic et al., was composed of a brain compartment, containing neuronal and glial cells differentiated from human embryonal carcinoma cells, and a vascular compartment separated from the brain compartment by a PDMS membrane. The membrane was covered by human endothelial cells forming a simplified BBB. After 4 weeks, cells within the brain compartment formed neuronal clusters interconnected with sparse astrocytes, having a similar structure to the human brain parenchyma. Neuronal progenitor cells in the brain compartment were sensitive to chemokines and shown to be attracted by CXCL12 only when the neuronal microenvironment was mimicked. This chip therefore provided a suitable tool for studying the development of different neuronal cell types from a single progenitor, in the presence, or absence of, drugs. In addition, the chemotaxis displayed by the progenitor cells might also prove useful for studying central nervous system regeneration. 79 A similar but miniaturized system composed of four distinct channels each filled with a different neuronal cell population has been developed by another group and could be used for the same type of studies as described above. 80
Different from liver-on-a-chip models, which all have a fairly similar design, brain-on-a-chip models have different architectures depending on the mechanism being studied. As a given brain-on-a-chip can currently only cover one specific region of the brain, the type of chip to be used will mainly depend on the type of mechanism or the region to be studied. The ability of a new drug to cross the BBB would need to be studied using a BBB-on-a-chip, while the global impact on the brain would require the use of another chip containing different brain regions. The currently available chips may have to be further miniaturized and adapted to be suitable for high-throughput screening in the context of preclinical testing. The choice of cells and their origin is important because of the species difference between human and animal neuronal cells, especially regarding astrocytes. 81 Induced pluripotent stem cells (iPSCs) could be an alternative source of human neuronal cells because they can be induced to form neurons or astrocytes. 82 With the chips described above and human neurons derived from patients with a specific disease, it may be possible to specifically interrogate the underlying mechanisms of the disease and find new treatments. Narcolepsy, Parkinson’s and Huntington’s diseases are examples that could possibly benefit from such an approach. 83
Kidney-on-a-chip
The levels of all compounds, ions, and water present in the blood have to be finely regulated. This essential function is achieved by the kidney using simple mechanisms of filtration, reabsorption, and secretion. While on the pathway to excretion, drugs can cause adverse effects, such as cytotoxicity and inflammation. Consequently, having an accurate renal model is important to understand the mechanisms of nephrotoxicity and to decrease the high instances of drug-induced nephrotoxicity observed in older adults. 84,85
Each kidney is composed of millions of nephrons, which are the basic functional unit. Each of these units is composed of a renal corpuscle (the initial filtering unit) and a renal tubule where remaining molecules are reabsorbed back into the blood stream. Each segment of the tubule expresses specific genes and therefore possesses a highly specific function. 86 Unlike in the liver where zonation is responsible for hepatocyte specialization, fluid shear stress is responsible for renal cell gene expression along the tubule, linked to apical/basal polarization or cytoskeletal reorganization. 87,88 In vitro renal cell polarization was first recreated using porous membranes as substrates for cells to grow on. The attachment of cells to the membrane was sufficient to induce a mild apical/basal polarization. 89 Since then, almost all studies have used a porous membrane as support. These kidney models have been substantially improved based on in vivo observations that renal cells are constantly exposed to blood flow. 90 Therefore, all kidneys-on-a-chip designed so far have, in addition to a porous membrane, an apical compartment for medium circulation. Using such a design, Jang and Suh showed that inner medullary collecting duct cells subjected to shear stress displayed a strong apical/basal polarization, based on the expression of aquaporin 2 and the Na+-K+ pump. 91 The flow of medium was shown to affect the cytoskeleton and increase localization of adherens junction proteins at the cell edges. In addition, cells maintained in the chip were less affected by hydrogen peroxide exposure than cells in a static condition. 91 Similar improvements in epithelial parameters were seen in co-cultures of Madin-Darby canine kidney (MDCK) epithelial cells. The co-culture was sufficient to increase cell height, cilia formation, and Na+-K+ pump membrane expression. Medium flow applied to the co-culture allowed for fluid exchange and was shown to impact microtubule distribution. 92
A critical aspect of renal cells is their ability to strictly control the reabsorption and secretion of molecules within the tubules. 93 However, recreating such a mechanism in vitro has proved difficult. Sciancalepore et al. used tubular adult renal stem cells in a fluidic system to demonstrate that these polarized cells formed a monolayer, decreasing the permeability of urea and creatinine, compared with membrane-grown cells only. 94 More evidence that such a kidney-on-a-chip can be used to study transport across cells came from Ingber’s group 95 who observed an active reabsorption of glucose and albumin from the tubular compartment. The transport was twofold higher than under static conditions. In addition, the utility of the system for drug toxicity testing was also demonstrated with two different scenarios. In the first, the renal cell response to cisplatin was demonstrated to be similar to in vivo observations with an improved recovery compared with static cultures. Second, the activity of the ATP-binding cassette membrane transporter, a protein responsible for multidrug resistance, was increased in the dynamic conditions of the OoC. These results support the usefulness of kidney-on-a-chip platforms for the future development of specific inhibitors of multidrug transporters to improve the efficacy of several existing drugs.
Applications of kidney-on-a-chip systems are not only limited to toxicological assessments—new treatments for various nephropathies could also be developed using OoC technology. Wei et al. used human proximal tubular cells combined with a PDMS microfluidic device to mimic calcium phosphate stone formation in kidneys. 96 Variation in the tubular flow speed could initiate some nephropathies. 90,97 This mechanism was studied in vitro by Rydholm et al. using a chip made out of either PMMA or silicon glass and with several crossing channels to produce a controlled laminar flow across MDCK cells. Calcium variations within the cells were monitored to show that variations in flow speed can change the kinetics of cellular calcium responses. 98 Other kidney diseases could be studied on chip platforms in the future. However, this may require the use of renal cells or iPSCs from patients with renal disease, for example, polycystic kidney disease or Alport syndrome. 99 Finally, in the context of renal insufficiency, the mimicry of renal cell physiology derived from the controlled microenvironment in the chip might allow for the development of an artificial and simplified kidney working as a portable dialyzer. 100
Together, these data indicate that with the use of cells with a high level of in vivo similarity, in 3-D organization, 101 and medium flow with similar characteristics as in the renal tubules, a kidney-on-a-chip might be useful in identifying drugs that may cause nephrotoxicity, in understanding their mechanism of action and in finding biomarkers to accurately predict renal toxicity. 102 One of the major current limitations of kidney-on-a-chip models is that they reproduce only a specific region of the nephron with a specific function, that is, filtration or reabsorption. Consequently, potential drug effects, including toxicity, are only assessed in the context of this specific region rather than the whole filtration unit. A nephron-on-a-chip would therefore be a promising tool for the future, perhaps with the help of 3-D bioprinting to enable the precise distribution of cells in a spatially controlled manner. 103–105
Lung-on-a-chip
Understanding how compounds can enter the body to eventually reach the bloodstream is an essential aspect of drug development because it determines bioavailability and toxicity. Different exposure routes are known: compounds can be taken orally and digested, absorbed by the skin, directly injected into the bloodstream, or inhaled. The way inhaled aerosols can enter the bloodstream is especially relevant because everybody is exposed daily to numerous volatile compounds present in the air. As a consequence of our breathing, gases, vapors, dusts, droplets, and volatile organic compounds can reach the respiratory tract where they will be absorbed. 106 The first experiments performed on submerged 2-D cultures were only poorly representative of the human lung environment. The development of porous membranes allowed for the development of ALI cultures: Primary human lung cells are cultured on a porous membrane and, once confluent, exposed to air on their apical side. The presence of factors inducing differentiation in the medium, such as retinoic acid and exposure to air, allows for the formation of a pseudostratified epithelium. 107 Depending on the origin of the cells, 3-D lung models can mimic the upper (large) or the lower (small) airways. 108–110 These models, also referred to as organotypic lung models, have been widely used in studies, including investigating the impact of pathogens on the lung, 111,112 the damage caused by aerosols and air pollutants on the lung epithelium, 6,113,114 and intrinsic mechanisms. 115,116 Initially, lungs-on-a-chip were designed, either to recreate a specific function of the lung or to mimic the blood flow observed in vivo. For example, blood oxygenation provided by the lung was modeled by interconnected artificial alveolar structures. Each structure was composed of an ultrathin PDMS membrane to allow for gas exchange between the medium and the air. 117 Shear stress caused by blood flow was recreated in vitro with a fluidic chip and cells maintained on porous membranes to provide an ALI with the basal side exposed to medium flow. An integrated automated fraction collector allowed for the analysis of cellular response kinetics after stimulation with pollen extract over a 24-h period. 118
The OoCs described above do not recreate breathing, which is a major constraint to which lung epithelia are constantly exposed. Breathing movements observed in the lung are difficult to capture in vitro. Such interactions are important, however, to reflect better the physiology of the lung tissue because they play a role in the transport of fluids, nutrients, immune cells, and other regulators in the tissue and influence cell differentiation and expansion. 119
The lung-on-a-chip model of the Wyss Institute aims to mimic the physiology of a breathing lung. 14 The model is designed to imitate the structural, functional, and mechanical properties of the human alveolar–capillary interface. The system consists of a microfluidic system containing two microchannels that are separated by a thin, porous, and flexible PDMS membrane lined with fibronectin or collagen. Human alveolar epithelial cells are seeded on the top side of the membrane, this upper compartment is the air chamber, while human pulmonary microvascular endothelial cells are cultured on the bottom side. Two vacuum microchambers are located on the lateral side of the cell-containing microchannels. These two vacuum chambers control the stretching of the PDMS membrane (and thus the adherent tissue layers). A computerized system is used to regulate the vacuum, by which the stretch of the PDMS membrane is controlled. For example, the normal inspiration, subatmospheric, pressure-driven stretching of the membrane (alveoli) can be followed. As a result, the dynamic mechanical distortion of the alveolar–capillary interface, which occurs normally during breathing, can be simulated in the system.
The breathing lung-on-a-chip model expresses junctional complexes, such as the epithelial and endothelial junctional proteins, occludin, and vascular endothelial cadherin. 14 Following introduction of air to the upper chamber (i.e. the epithelial-containing microchannel), both the alveolar epithelial and pulmonary endothelial cells were reported to be viable for more than 2 weeks. Simultaneously, increased surfactant production by the alveolar epithelium was reported. The presence of surfactant increased the stability of the thin liquid layer, as evidenced by elevated electrical resistance across the tissue layers. This lung-on-a-chip also exhibited functional pulmonary inflammatory responses, that is, the early increase in cytokine levels, upregulation of leukocyte adhesion molecules, and subsequent leukocyte infiltration into the alveolar space from the microcirculation. Moreover, the innate cellular response to bacterial infection (investigated by stimulating the alveolar microchannel with Escherichia coli) could induce neutrophil transmigration through the underlying endothelium into the alveolar microchannel, followed by engulfment of bacteria by the neutrophils. 14 Because of its capability to “breathe” (stretch), the model can reflect the physiology of mechanical breathing and how this impacts biological effects following exposure to a noxious insult. First, the breathing motion can further elevate ICAM-1 levels in endothelial cells following exposure of the alveolar epithelium to nanoparticles (ultrafine airborne particles). Second, the breathing motion can further increase the levels of reactive oxygen species following nanoparticle exposure, although this effect seems to be specific for certain types of nanoparticles (e.g. silica nanoparticles and carboxylated quantum dots). When tested alone, neither nanoparticle nor mechanical strain impacted reactive oxygen species production. Finally, the mechanical breathing influenced the translocation of small molecules (e.g. albumin and nanoparticles) from the alveolar epithelium to the lower chamber. This observation emphasizes the importance of the inherent mechanical activity in transporting airborne particles from the alveolar compartment into the bloodstream. In a more clinically relevant application of this model, physiological mechanical forces exacerbated interleukin (IL)-2-induced edema, 120 providing further insight into the mechanism that limits the therapeutic potential of IL-2 for anticancer therapy. 121
Although airway inflammatory diseases have been modeled using other in vitro systems (e.g. co-cultures of airway epithelium and tissue-resident immune cells, such as macrophages or dendritic cells), in vivo lung inflammation is fundamentally mediated by organ-level responses, including not only the complex tissue–tissue interactions between the lung epithelium and the underlying microvascular endothelium but also the active fluid flow. In another adaptation of the lung-on-a-chip model, human small airway epithelial cells were used, incorporating an ALI culture in the upper compartment of the chamber. 122 The top small airway epithelium-containing microchannel was designed to have a dimension similar to the radius of a human bronchiole (height and weight of 1 mm). The small airway epithelial cells were seeded on a porous polyester membrane, coated with type I collagen, and initially submerged in the culture medium. After 5 days, air was introduced to trigger the differentiation of the epithelium into a pseudostratified epithelium with a constant flow of growth medium in the bottom channel. With the capability of simulating “blood” flow in the bottom channel, this lung-on-a-chip model permits the study of human lung inflammation in a more physiologically relevant system compared with static cell cultures. Primary human lung microvascular endothelial cells were incorporated onto the opposite side of the membrane. This co-culture can be sustained for several weeks. The small airway epithelium of this lung-on-a-chip model was reported to express the tight junction protein ZO-1, whereas the endothelium expresses the adherens junction protein PECAM-1. 122 The frequency of ciliary beating and mucociliary transport closely mimicked those observed in the human airways. Moreover, following IL-13 stimulation, goblet hyperplasia and mucus hypersecretion could be induced. Increased secretion of G-CSF and GM-CSF and reduced ciliary beating frequency were also observed after the IL-13 stimulation. These findings correlate well with features of the airway mucosa in asthmatic patients. As such, this lung-on-a-chip lends itself to the assessment of new experimental therapeutics. For example, stimulation with tofacitinib (a potent inhibitor of JAK1, JAK2, and JAK3) was shown to reverse the IL-13-induced phenotypes. Moreover, a new anti-inflammatory drug (a bromodomain-containing protein-4 inhibitor of nuclear factor kappa B signaling) was able to inhibit neutrophil adhesion under dynamic flow conditions, whereas no impact was observed in static Transwell® cultures, further highlighting the importance of simulating the dynamic flow in in vitro culture systems. Subsequent work has demonstrated functionally intrinsic cross talk between the epithelium and endothelium. Treatment of the small airway epithelium with polyinosinic:polycytidylic acid (poly (I:C)), an analogue of the double-stranded RNA produced in infected cells during respiratory viral replication, resulted in increased levels of secreted RANTES, IL-6, and interferon-gamma-inducible protein 10 (IP-10) in the underlying basal media. Without the endothelium co-culture, lower levels of RANTES, IL-6, and IP-10 were detected. Furthermore, elevated levels of E-selectin and VCAM-1 were observed in the underlying endothelium and linked to augmented recruitment of circulating human neutrophils (added into the lower channel). Of note, when using epithelial cells isolated from an individual with chronic obstructive pulmonary disease (COPD) for the assembly of the lung-on-a-chip, the pathological features of COPD could be simulated. 122 Treatment with poly (I:C) or with lipopolysaccharide endotoxin induced an upregulation of IL-8 and M-CSF. This upregulation was not seen in chips developed using the small airway cells of healthy individuals. 123
To better reproduce the movements of physiological breathing, further adaptation of the small airway on-a-chip model has been reported, whereby the microfluidic system was connected to a smoking instrument. This instrument allowed the simulation of a breathing parameter (inhaling and exhaling) of the upper small airway epithelium-containing microchannel. 124 Because of the similar dimensions of the epithelium-lined channel and a human bronchiole, the channel geometry, air volume, and shear stress were able to be scaled to reflect the expected values found in the human airway. A software replicating human smoking behavior was integrated to specifically regulate the smoking parameters. Moreover, a peristatic pump controlling the medium perfusion to the bottom endothelial cell-containing channel allows the system to be maintained in dynamic flow conditions. Initial work demonstrated that acute exposure to cigarette smoke (75 min) resulted in oxidative stress in the epithelial cells, as evidenced by the increased expression of heme oxygenase 1 mRNA and phosphorylation of the transcription factor nuclear factor (erythroid-derived 2)-like 2 (NRF2) 24 h post-exposure, as compared with unexposed controls. Gene microarray analysis demonstrated that the oxidation–reduction pathway alterations detected in the in vitro small airway-on-a-chip were found to be similar to those seen in human smokers. Moreover, mucociliary clearance and ciliary beating frequency were reported to be similar to that of nasal brushings or tissue explants cultured ex vivo. Clinical exacerbations in smokers with COPD were investigated using a microfluidic chip that was generated using epithelial cells obtained from COPD patients. When the chip was exposed to cigarette smoke, large increases in IL-8 secreted in the basal media were observed (which were not seen in the normal chip developed using cells obtained from non-COPD individuals).
A first step toward a miniaturized and easy to handle lung-on-a-chip was taken by constructing a chip composed of three circuits, each of which has two compartments separated by a thin, porous PDMS membrane. 125 A small diaphragm present in the basal compartment can be stretched using a pneumatic setup. Because the basal chamber is closed, pressure applied on the diaphragm is transferred to the PDMS membrane mimicking breathing behavior. Lung epithelial cells were seeded on the apical side of the porous membrane and endothelial cells on the basal side. Stretching of the membrane was found to increase the permeability of the epithelial tissue to small molecules such as IL-8. Given its miniaturized format, such OoC could be adapted to multi-well formats and therefore amenable to high-throughput assays.
All OoCs described so far recreate mechanisms observed in vivo. Cells used in these systems have benefited from important gains in physiological authenticity with improved life span, in vivo like morphologies, improved metabolic activity, and have even gained functions not observed previously in vitro. Despite their impressive potential, the use of OoCs for drug testing remains rare and to date has mainly served to confirm previously obtained results. Although this field can still be considered in its infancy, several groups have already explored the next step in the evolution of this platform: the MOC.
Multi-organ-on-a-chip
The single OoCs presented so far have permitted in vitro studies using models displaying a level of physiological resemblance not reached previously. Extension of the cultures’ life span is one of the major advantages brought about by these chips, making this technology potentially amenable to chronic toxicological studies. However, despite the level of technology incorporated on these chips, they still do not replicate the human body in its completeness. The human body is a complex machinery composed of multiple organs, each responsible for a specific essential function. Testing a drug on a single OoC, without taking into account its metabolism or transformation by other organs, might lead to unwanted effects when tested in humans. With this in mind, the “next generation” of organ chips was developed, connecting multiple organs (Figure 2).
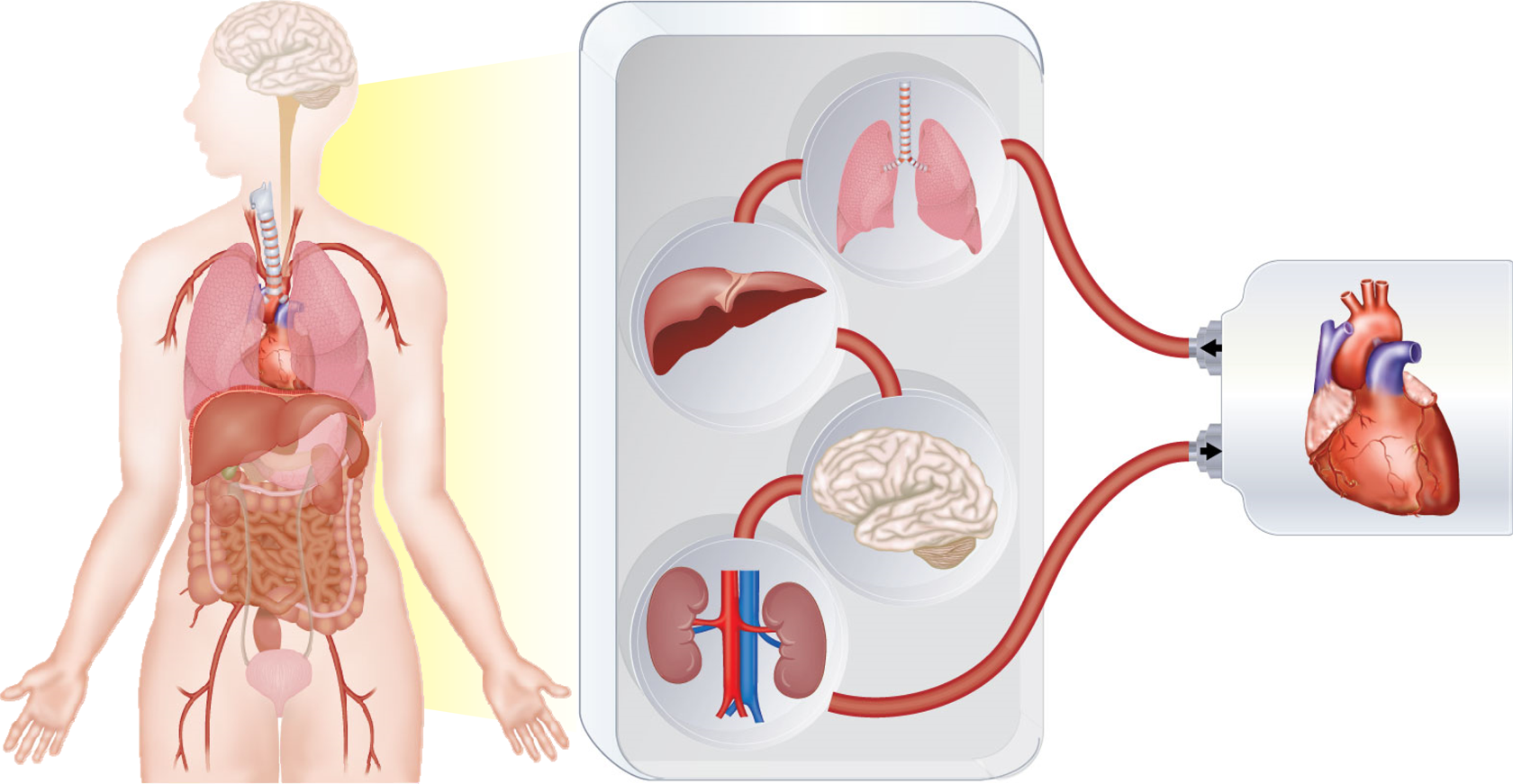
The future of OoCs. MOCs are devices developed to recreate organ connections observed in the human body. Each compartment of the device houses a specific organ model and is connected to the other chambers via artificial vessels. A peristaltic pump ensures that the medium flows through all the compartments, as the blood pushed by the heart flows in vivo. MOC: multi-organ-on-a-chip; OoCs: organs-on-a-chip.
Several examples of two OoCs have been described to date. Given the crucial role of the liver in metabolizing, and thereby determining the half-life, of a drug, these chips all connect a liver model with another tissue. TissUse have developed a dynamic chip connecting liver spheroids, composed of HepaRG and hepatic stellate cells, with tissue from a human skin biopsy. The tissues were placed in two separate compartments of a PDMS chip, with a microfluidic channel connecting the compartments. 126 Both cultures remained stable for 28 days as demonstrated by glucose consumption and lactate release. The skin compartment was observed to consume the albumin produced by the liver providing evidence of tissue crosstalk. 126 Such a system might be beneficial when studying, for example, the administration of drugs via the skin and their potential effect on the liver. The same type of chip platform was then used to connect NTera-2/cl.D1 cell neurospheres with liver spheroids. 127 Co-cultivation of the neural tissues with liver spheroids did not affect the expression of tissue-specific markers, including albumin, CYP2B6, and 3A4 (liver spheroids) or nestin, Oct4, and β-tubulin 3 (neurospheres). Glucose consumption, lactate, and lactate dehydrogenase (LDH) production were monitored to demonstrate successful co-culture for 14 days. The connection of the two cultures was found to increase the toxicity associated with 2,5-hexanedione exposure. Necrotic factors released by one tissue that affected the other tissue were suggested as being responsible for the heightened sensitivity to exposure. 127
Recently, a two-channel PDMS chip was used to co-culture intestinal CaCo-2 cells in a monolayer with HepG2 cells. 128 To form an epithelial barrier, the intestinal cells were seeded on a membrane separating the two channels: The first channel running on the apical part of the intestinal cells, representing the lumen, with the second channel running below the intestinal cells, containing the hepatic cells, and representing the interstices. Similar to observations made using single OoC, both tissue types displayed more physiological characteristics once exposed to a flow of medium as shown, for example, by decreased intestinal permeability and increased hepatic and intestinal CYP3A4 expression. Apigenin was used as a test compound to demonstrate epithelial barrier transport and metabolism by the two different cultures. Apigenin was found to be mainly metabolized by the intestinal cells but was also metabolized by hepatic cells once it crossed the epithelial barrier. The hepatic culture produced additional apigenin metabolites, not observed with the intestinal cells alone, but similar to those observed in rats. 128 This study demonstrates how the combination of two different organ cultures in vitro can provide more predictive and accurate toxicological readouts. Another interesting study focused on the impact of liver metabolism on cancer treatment and was used to generate a pharmacokinetic–pharmacodynamics (PK–PD) model. 129 A PDMS chip was constructed, composed of two chambers the size of a well of a 96-well plate connected via a fluidic channel. Medium flow in the chambers was controlled by a plate rocker. Monolayer cultures of HeLa cells were connected to HepG2 2-D cultures to mimic a tumor and a liver tissue, respectively. All metabolites resulting from HepG2-dependent luteolin metabolism were first measured to demonstrate the liver’s metabolic activity. In a second step, luteolin and its metabolites were applied to the HeLa cells. While luteolin itself was shown to have a potent cytotoxic effect on the HeLa cells, stronger cytotoxic effects were observed once luteolin metabolites were incorporated in the medium. This observation further highlights the beneficial role of having a liver bioreactor when developing or testing drugs. Moreover, all parameters used for the experiments on the chip (e.g. medium volume, flow rate) and the data from the luteolin metabolism (e.g. metabolization rate, cytotoxicity) were used to create a physiological PK–PD model, indicating the utility of this type of chip platform for predicting drug effects.
While the three examples mentioned above use submerged organ cultures, such MOC platforms are also amenable to cultures (e.g. lung cultures) that have an ALI. The lung is known to express several xenobiotic-metabolizing P450 enzymes and could therefore bioactivate inhaled compounds, transforming an inert molecule into a compound that is potentially toxic for another organ. 130 To allow for the co-culture of a submerged tissue with an air-exposed tissue, Coppeta et al. developed an open MOC composed of six modules. 131 They used four chambers to maintain lung cultures and one as a liver compartment. The challenge to providing a common media was addressed by considering the specific needs of each tissue. For example, concentrations of epinephrine, bovine pituitary extract, and bovine serum had to be decreased, compared with the original airway epithelial culture medium, as they would have affected the liver cells. Medium flow between the chambers was programmable, and at first, only the lung chambers were connected. After a few days, the lung tissues were connected to the liver compartment. Together, both cultures remained viable for 14 days, as demonstrated by the presence of ciliated cells and a high transepithelial electrical resistance in the airway compartment, and high CYP3A4 expression in the liver compartment. 131
While useful for the toxicological testing of single compounds, the MOCs discussed above do not allow for high-throughput experiments. µOrgano is a small plug and play MOC system composed of single chambers. 15 Each chamber possesses an inlet and outlet to allow for a syringe pump to create a flow of medium. The chambers can have different designs to accommodate different types of cultures. The idea behind the system is that each chamber can be either isolated, to allow, for example, the culture to settle and mature, or can be connected to other chambers using a simple connector that creates a bridge between the chambers. With the connector and the flow created by the syringe pump, the two chambers can exchange medium and metabolites. In an example, Mathur and co-workers reproduced a previously described 132 heart-on-a-chip in series. Cardiomyocytes derived from iPSCs were first seeded in separate chambers to form tissues with homogeneous beating behavior. After 3 days, the two chambers were connected using a linear connector with a syringe pump to ensure medium circulation. After 24 h, the two heart tissues were found to beat at physiological rates demonstrating the possibility of maintaining two hearts-on-a-chip simultaneously. 15 Because the chambers housing the cell cultures can have various designs, different types of cultures can first be grown independently and connected once mature. The very low amount of medium circulating on the chip is also an advantage because lower amounts of test compounds (which can be expensive) can be used.
Additionally, in an attempt to miniaturize MOC models, InSphero has fabricated a multi-hanging drop plate where spheroids of different tissue origins can be suspended in a drop of medium. Because the drops are connected by a small channel and pressure equilibration ensures that all connected drops have the same size, any addition of medium within a drop will be immediately transferred to the next one, and so on. Medium can therefore circulate through the different drops without the need for a complex pump. As a proof of concept, the bioactivation of cyclophosphamide by liver spheroids and the subsequent impact of the metabolites on cancer spheroids was successfully demonstrated. 133
In parallel to the MOC miniaturization, other laboratories have aimed at increasing the number of organs connected to further increase the overall complexity of the system. In the future, PK/PD studies might especially benefit from these technologies, enabling the study of a given drug from absorption all the way through to excretion. Pioneered by TissUse, MOCs are available that connect four tissues mimicking the liver, skin, intestine, and the kidney. To make this MOC possible, chip platforms with three different circuits were created to accommodate three different media: the main blood circuit connecting the four organs, the intestinal lumen circuit, and the excretory circuit. LDH activity and glucose consumption remained impressively stable for 28 days of co-culture. The morphology and specific genes expressed by each organ were conserved, demonstrating the feasibility of connecting several organs in a closed system. 134 In another study using an MOC, Oleaga et al. developed a platform with multiple compartments to co-culture liver, cardiac, skeletal, and neuronal cells. 135 The low-cost chip used a rocker platform to ensure circulation of the common medium. The four different cultures remained viable for 14 days and were able to mimic the response of human organs to five different drugs, including atorvastatin, doxorubicin, and acetaminophen. 135 However, the lack of control experiments, where one or more tissues are absent from the chip, makes any conclusions on the role of each tissue in drug toxicity difficult to interpret.
These MOCs all connect two or more tissues using microchannels. As the cell culture medium in these MOC systems has to accommodate all organ cultures simultaneously, finding an appropriate medium may prove difficult. Even if only one organ culture does not draw sufficient nutrients from the chosen medium, the entire MOC may “underperform.” Ideally, future developments of MOC platforms would include blood or a blood surrogate that supports the growth and health of all the organs. Pending the development of such a sophisticated blood surrogate, Vernetti et al. used an alternative approach. The different tissues comprising their MOC were not directly connected. Instead, medium used in a given compartment to perform an assay was later mixed with the medium of another compartment. Several compounds were tested to confirm the usefulness of this approach by monitoring their absorption, distribution, metabolism, and excretion (ADME) profiles. For example, terfenadine was applied on the apical side of an intestinal mucosa model. Transport of terfenadine across the barrier and partial biotransformation into fexofenadine by the intestinal cells was successfully demonstrated 24 h later. The basolateral medium was then transferred to a liver compartment composed of a 2-D co-culture of hepatocytes, endothelial, Kuppfer, and stellate cells. In this compartment, 65% of the remaining terfenadine was converted to fexofenadine. The liver-conditioned medium was then used in a neuronal compartment mimicking the BBB, with endothelial cells seeded on one side of a porous membrane, and neurons and astrocytes grown on the other side. Fexofenadine was unable to cross the BBB and remained in the vascular compartment. Finally, liver-conditioned medium was perfused through a vascularized kidney model. Approximately 1% of fexofenadine was found to remain in the luminal compartment, while the rest was excreted. Similar results were obtained for trimethylamine and vitamin D3, which showed the robustness of this approach.
Challenges, limitations, and future prospects for fluidic devices
The OoCs and MOCs described in this review may facilitate the discovery of new mechanisms underlying diseases and the development of safer and more efficient drugs. As with previously developed in vitro models, microfluidic models will require improvements before they can be used as preclinical in vitro models. Regardless of the organ mimicked, tissue properties depend mainly on the source of the cells used to create the model and particular attention should be paid to the selection of cell type in relation to the desired endpoint. Using the liver as an example, primary hepatocytes in 2-D or 3-D culture would represent a first choice when seeking to identify metabolites resulting from compound breakdown, because xenobiotic enzyme expression in these cells is high and similar to in vivo conditions. 45 However, their shorter life span in 2-D culture makes their use for repeated drug exposure unfeasible. If repeated dosing is a requirement, cellular reorganization in 3-D, or use of more metabolically active cell lines, such as HepaRG™ should be considered. 51,54
The in vitro models currently used might benefit, first from the recent advances in the iPSC field to create chips with cells having either a human origin or a specific disease background 136 and second from the addition of different cell types within a single tissue. In their native environment, cells are surrounded by different cell types releasing mediators that can influence the physiology of the cells of interest. Examples have been presented in this review, including the co-culture of renal cells and adipose-derived stem cells, 92 and hepatocytes and Kuppfer cells. 43 Unfortunately, such heterocellular tissues might be difficult to maintain because of their complexity, and their use may be associated with increased costs.
The rapid rise in the number of publications regarding OoCs emphasizes the strong interest of the scientific community in this technology. Multiple challenges have already been faced to give rise to biochip platforms, but their real potential is probably still under evaluated as many reports describe the chip system, but lack of data concerning tissue stability over long periods of time or the application of the OoC models, for example, for toxicological testing. This could be related to technical issues, because connecting different organs (and maintaining them in combination) requires a blood surrogate suitable for all tissues, without inducing their dedifferentiation. Appropriate proportions of each organ and a correct ratio of surface-to-volume vascularization are other critical aspects that are currently not being taken into account. 137
In conclusion, having a human-on-a-chip with all organs constituting the human body connected on a device and amenable for toxicological and disease assessment is unlikely to be realized in the near future. However, MOCs will open a window for toxicological studies, in particular, for elucidating a given mechanism of toxicity or a given tissue–tissue interaction that may drive toxicity. MOCs could be further leveraged for ADME studies, which will improve the prediction of compound behaviors in users (consumers or patients). Such MOCs could combine intestinal, liver, and kidney tissues with an artificial blood flow to replicate the various compartments critical to ADME. Moreover, MOCs incorporating human models will avoid issues caused by differences in species, such as those seen in CYP enzyme induction. Some studies have indicated that in vitro CYP induction correlates well with the in vivo data (e.g. lack of induction in primary hepatocytes is consistent with the lack of induction seen in clinical studies). Clinical drug–drug interaction studies could be waived if a candidate compound has tested negative in a human in vitro CYP induction study. 138
MOCs might also represent a new paradigm for drug development, providing a better understanding of dose responses, enabling the detection of drug resistance, highlighting potential side effects, and supporting the evaluation of PK/PD parameters. 139,140 Future chips will benefit from the development of new tissue models with even better physiological resemblance to their in vivo counterparts. In association with computational modeling to transform in vitro data into in vivo predictions, MOCs will certainly help in decreasing the rate of clinical failure, through the development of even safer and more effective drug candidates, which is the essential and initial reason for the development of new, more physiologically relevant in vitro models.
Importantly, to further OoC and MOC applications, there is also a need to standardize models for each of the organs to be mimicked. Assays adapted to the more complex structures of the tissues currently used need to be optimized and validated to ensure accuracy, specificity, and reproducibility. Associated with a future potential decrease in the cost of fabrication because of the development of new manufacturing processes, OoCs and MoCs will most likely become highly efficient and standardized high-throughput platforms for toxicological evaluations.
Footnotes
Authors’ note
DB, AI, KL, JH, and MCP are full-time employees of Philip Morris International (PMI).
Acknowledgements
The authors are thankful to Antonin Sandoz and Samantha Elmurst for their contribution to creating the figures. The authors are also appreciative of the services of Edanz Editing for their assistance with language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
