Abstract
Objectives:
To investigate the impact of gastrointestinal involvement in systemic sclerosis affecting the quality of life and systemic sclerosis-related disability using patient-reported outcome measurements (PROMs).
Methods:
Data from 160 consecutive systemic sclerosis patients were collected, including clinical characteristics and self-assessment questionnaires. The severity of gastrointestinal involvement was determined by the University of California Los Angeles Scleroderma Clinical Trials Consortium gastrointestinal tract 2.0 (UCLA-GIT 2.0): patients were classified as having “none-to-mild” symptoms (total score = 0.00–0.49: Group A), “moderate” symptoms (Group B), and “severe-to-very severe” symptoms (Group C). All investigations were repeated following 1 year to check the reliability (n = 149).
Results:
Quality of life (EuroQol-5 Dimension anxiety/depression, Physical Component Summary, and Mental Component Summary of Short-Form Health Survey-36) and disability questionnaires (Health Assessment Questionnaire Disability Index, Scleroderma Health Assessment Questionnaire), Scleroderma Impact of Disease showed moderate significant correlation with UCLA-GIT 2.0 (rho: 0.400, −0.484, −0.468, 0.400, 0.436, 0.646, respectively). These correlations remained significant when tested in the two main subgroups (limited and diffuse cutaneous systemic sclerosis). Disease duration and UCLA-GIT 2.0 score did not show any correlation (rho: .055, p = 0.488). The average gastrointestinal involvement complaint of early systemic sclerosis (disease duration < 3 years, n = 24) subgroup measured by the UCLA-GIT 2.0 was not different from the rest of the cases (0.13 (0.03-0.46) vs 0.22 (0.08-0.45), p = 0.280). All investigated PROMs regarding quality of life and disability showed worse results in Group B when compared with patients in Group A. Similarly, patients in Group C have worse results compared to Group B. These correlations with the UCLA-GIT 2.0 score were confirmed during the 1-year follow-up.
Conclusion:
Moderate/severe gastrointestinal involvement symptoms based on the UCLA-GIT 2.0 are associated with worse quality of life and disability. This correlation holds for both systemic sclerosis subgroups and independent of disease duration: moderate/severe gastrointestinal involvement complaints develop early in systemic sclerosis parallel with early onset of decreased quality of life and disability.
Keywords
Introduction
Gastrointestinal involvement (GI) is a significant source of morbidity and mortality in systemic sclerosis (SSc).1,2 Both limited cutaneous SSc (lcSSc) and diffuse cutaneous SSc (dcSSc) show similar prevalence of GI manifestations.2,3 Assessing GI tract impairment is complex due to variability regarding involved organs and non-specific symptoms (decreased oral aperture, sicca symptoms, esophageal and intestinal dysmotility, decreased gastric emptying, reflux disease, vascular ectasias, bacterial overgrowth, and decreased sphincter functions). Few publications compare different patient-reported outcome measurements (PROMs) for GI involvement. 4 The University of California Los Angeles Scleroderma Clinical Trials Consortium gastrointestinal tract questionnaire (UCLA-GIT 2.0) is the only validated PROM for GI tract involvement in SSc, demonstrating good sensitivity to change.
Impaired quality of life (QoL) in SSc is multifactorial, including internal organ involvement, fatigue, exertional dyspnoea, decreased work ability, and social participation.5,6 GI symptoms, severity of Raynaud’s phenomenon, and fatigue are major contributors to decreased QoL in SSc.7–9
QoL can be assessed by general tools (e.g. Short-Form Health Survey-36 (SF-36), Health Assessment Questionnaire Disability Index (HAQ-DI)) and disease-specific (e.g. Scleroderma Health Assessment Questionnaire (SHAQ) and Scleroderma Impact of Disease (ScleroID)) or even organ-specific tools (e.g. UCLA-GIT 2.0). 10 Developing PROMs is essential for complicated diseases such as SSc, aiding physician decision-making and patient satisfaction. 11
General health in SSc (measured with SF-36) is worse when compared to healthy subjects, 12 and higher levels of disability have also been reported.5,7,13,14 EuroQol-5 Dimension (EQ-5D) is a widely used survey assessing mobility, self-care, usual activities, pain/discomfort and anxiety/depression, all indicating health-related QoL and showing responsiveness to effective therapy. 15
UCLA-GIT 2.0 assesses GI symptoms covering reflux, distention/bloating, fecal soilage, diarrhea, social functioning, emotional well-being, and constipation. Total UCLA-GIT 2.0 scores better reflect GI tract impairment severity than individual components (except the reflux score). 16 The single UCLA-GIT 2.0 scales had a high sensitivity (80% to 94%) but very low specificity (0%–20%) when compared to the objective measures. 17 The UCLA-GIT 2.0 also correlates with depression, 18 supporting findings in which GI involvement also has a substantial impact upon mental health.
Our study aims to investigate the impact of GI involvement on QoL and SSc-related disability based on PROMs among SSc patients.
Methods
One hundred and sixty consecutive SSc patients fulfilling the 2013 American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) criteria 19 were recruited at the Department of Rheumatology and Immunology, Medical School, University of Pécs, Hungary. Patient enrollment took place between April 2017 and September 2018, and the 1-year follow-up was conducted between October 2018 and August 2020. Demographic, clinical, and laboratory items were recorded based on a standard protocol as previously described.20,21 The median age was 46 years (interquartile range (IQR) = 43;56). Median disease duration estimated from the first non-Raynaud symptom was 6.5 years (IQR = 4;12). Early scleroderma was diagnosed with disease duration of less than 3 years (n = 24). Fifty-five percent of patients (n = 88) had dcSSc.
To evaluate patients’ opinion regarding their disease, a large set of self-assessment questionnaires were completed including HAQ-DI, 22 SF-36, 23 SHAQ, 24 EQ-5D-3L, 25 UCLA-GIT 2.0, 26 and ScleroID. 27 Physician’s global assessment by a visual analogue scale (VAS), different pains, and fatigue scores were also recorded. Different activity indices were also calculated, that is, the revised European Scleroderma Trials and Research Group Activity Index (r-EUSTAR AI) 28 and the simplified activity index by our research group (Pecs-8.5).29,30
The severity of GI involvement was determined by UCLA-GIT 2.0: patients were classified as having “none-to-mild” symptoms (total score 0.00-0.49: Group A), “moderate” symptoms (total score 0.50-1.00: Group B), and “severe-to-very severe” symptoms (total score 1.01-3.00: Group C). 26
Autoantibody levels were measured using commercially available enzyme-linked immunosorbent assay (ELISA) blot (ORG 546 and 633 Orgentec Diagnostika, Mainz, Germany), according to the manufacturer’s instructions.
All the above tests were repeated on the entire patient population 1 year apart with the aim of confirming the consistency of our results (in time, this is short—in most patients—for a meaningful clinical change to occur). Clinical characteristics are detailed in Supplementary Table 1.
Ethics
All patients gave their informed written consent to the study, which was conducted in full accordance with the Declaration of Helsinki and was approved by the National Research Ethics Committee (30636-3/2017/EKU).
Statistical analysis
For continuous variables, comparisons between two SSc subgroups were performed using the Mann–Whitney U-test, while comparisons among three groups were conducted using the Kruskal–Wallis test. The chi-square test was used in the case of nominal variables. Correlation analysis was performed with Spearman’s correlation analysis. As most of the continuous variables showed non-Gaussian distribution, data are given in median (lower quartile; upper quartile). In consideration of these calculations, IBM SPSS Statistics version 27 (2020, Chicago, IL, USA) was used.
Results
The median of UCLA-GIT 2.0 score was 0.2033 (0.0833;0.4546) (for comparison with the results of previous studies, the mean value of UCLA-GIT 2.0 was 0.3395 (±0.3754)). Among our 160 patients afflicted with SSc, we identified 123 patients (76.9%) with none-to-mild GI symptoms by UCLA-GIT 2.0. Twenty-two patients belonged to the group with moderate GI signs (13.8%), and only 15 patients (9.4%) experienced severe GI complaints.
When patients were divided into three groups according to the UCLA-GIT 2.0 score (Group A vs Group B vs Group C), there was a significant difference between Group B and Group C in 6-minute walk test (6MWT) (452 m (317;538) vs 249 m (160;386), p = 0.014). When patients with moderate and severe GI complaints were pooled into one group (Group B + Group C), the length of 6MWT was shorter, the prevalence of hypertension, malabsorption and enteral feeding were higher, while renal functions were lower in this pooled group when compared to those with no or minimal GI complaints (Group A) (Table 1).
Clinical characteristics in patients with no/mild (Group A) and moderate + severe (Group B + C) gastrointestinal symptoms at the beginning of our study.
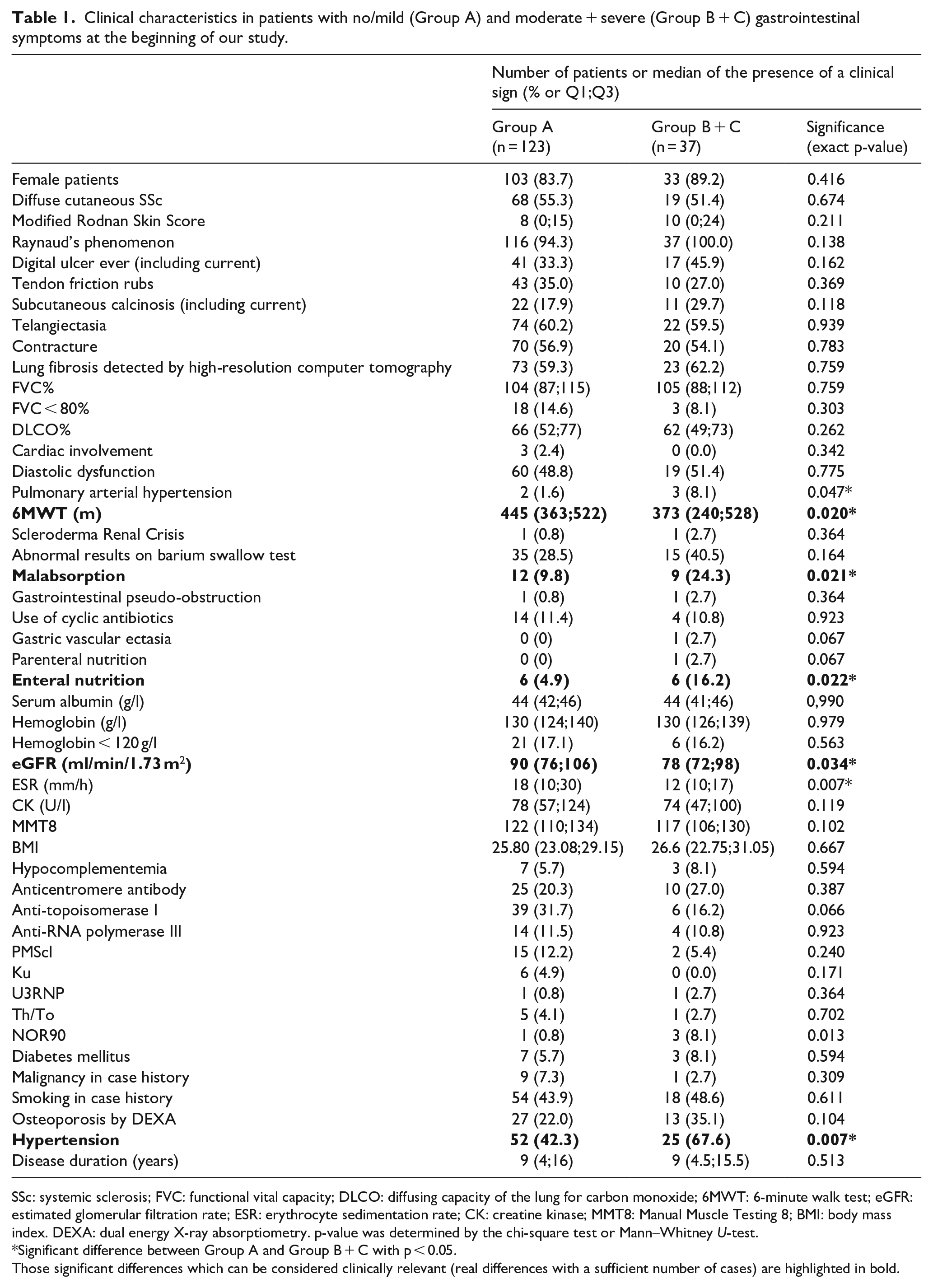
SSc: systemic sclerosis; FVC: functional vital capacity; DLCO: diffusing capacity of the lung for carbon monoxide; 6MWT: 6-minute walk test; eGFR: estimated glomerular filtration rate; ESR: erythrocyte sedimentation rate; CK: creatine kinase; MMT8: Manual Muscle Testing 8; BMI: body mass index. DEXA: dual energy X-ray absorptiometry. p-value was determined by the chi-square test or Mann–Whitney U-test.
Significant difference between Group A and Group B + C with p < 0.05.
Those significant differences which can be considered clinically relevant (real differences with a sufficient number of cases) are highlighted in bold.
All investigated PROMs regarding QoL and disability correlated well with the UCLA-GIT 2.0 score, both in whole SSc group and in the main subgroups (lcSSc and dcSSc) (Table 2).
Correlation analysis of different self-assessment questionnaires and UCLA-GIT 2.0.
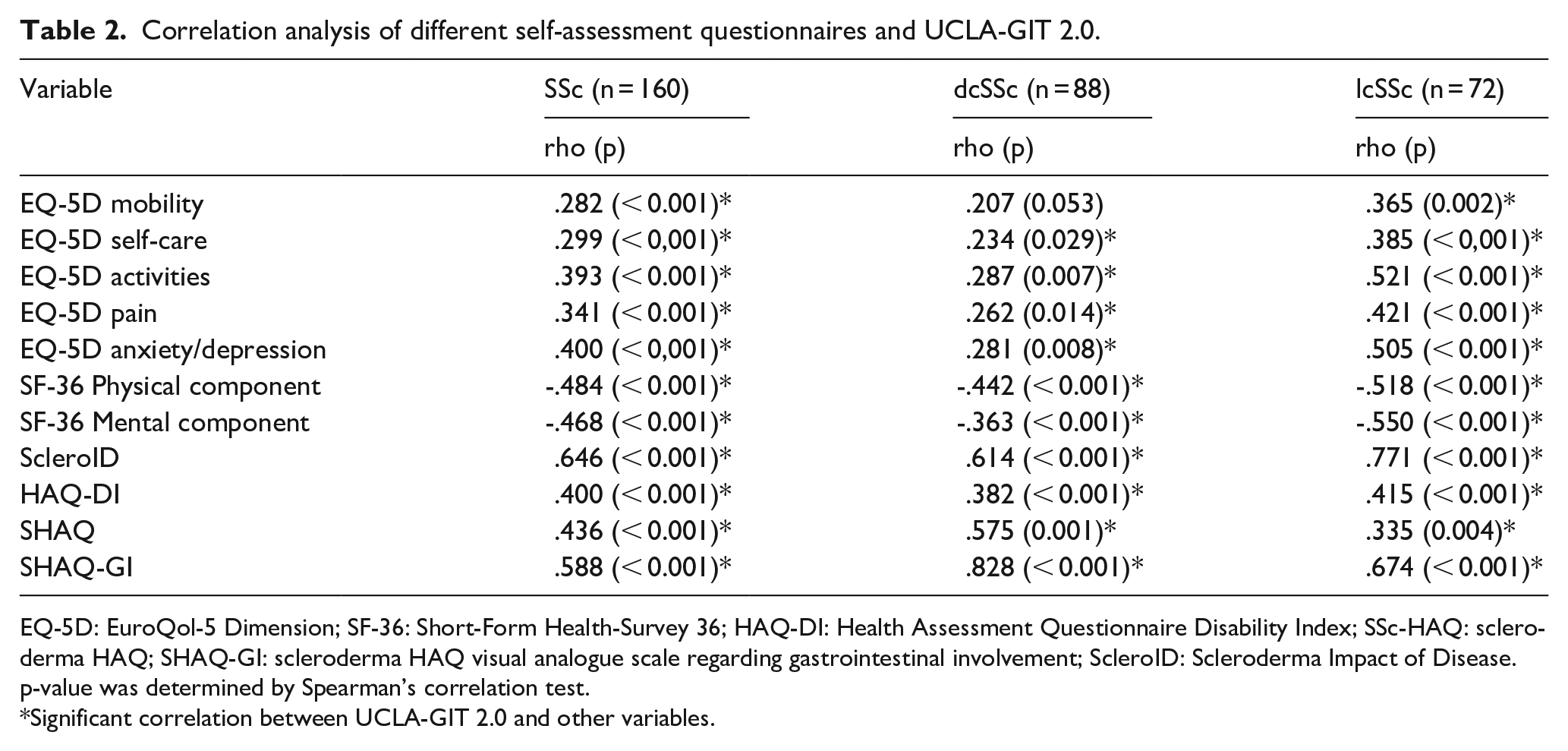
EQ-5D: EuroQol-5 Dimension; SF-36: Short-Form Health-Survey 36; HAQ-DI: Health Assessment Questionnaire Disability Index; SSc-HAQ: scleroderma HAQ; SHAQ-GI: scleroderma HAQ visual analogue scale regarding gastrointestinal involvement; ScleroID: Scleroderma Impact of Disease. p-value was determined by Spearman’s correlation test.
Significant correlation between UCLA-GIT 2.0 and other variables.
No disease activity parameter was found to show at least a moderate correlation with the UCLA-GIT 2.0 score. Only the r-EUSTAR AI showed a low correlation with gastrointestinal complaints, which remained significant in both subgroups (lcSSc and dcSSc) (Table 3).
Correlation analysis of different disease activity and the UCLA-GIT 2.0.
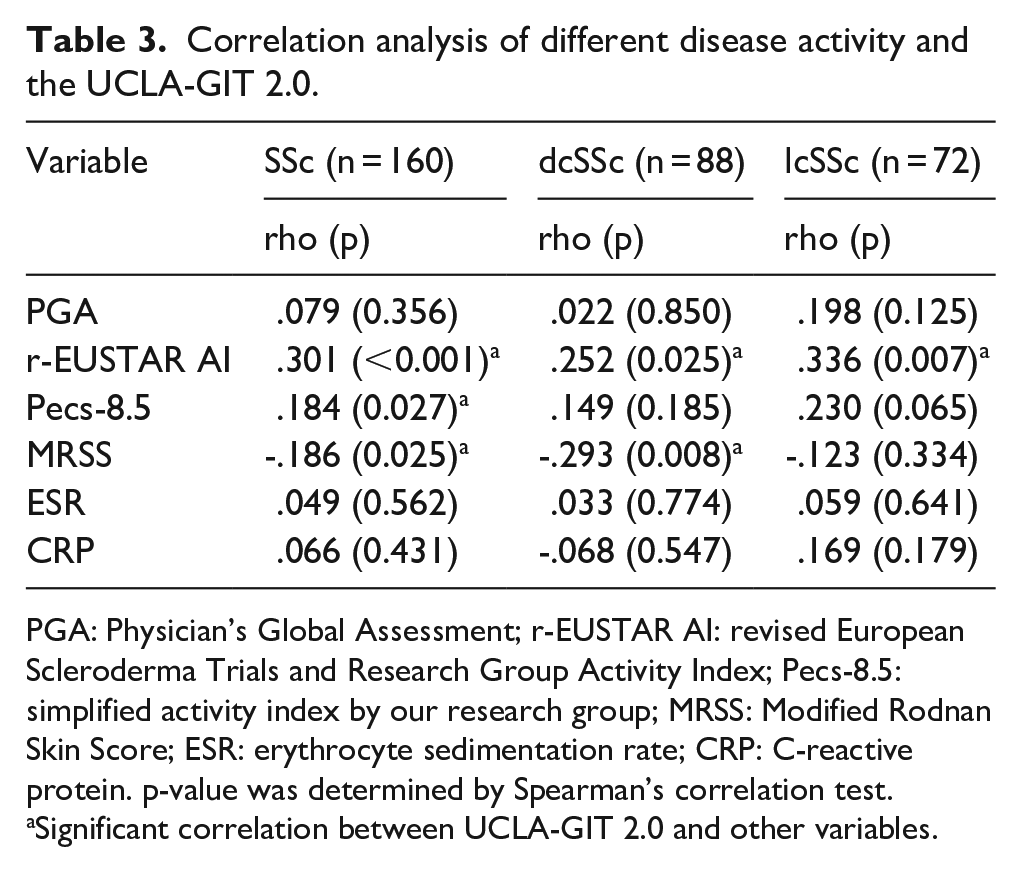
PGA: Physician’s Global Assessment; r-EUSTAR AI: revised European Scleroderma Trials and Research Group Activity Index; Pecs-8.5: simplified activity index by our research group; MRSS: Modified Rodnan Skin Score; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein. p-value was determined by Spearman’s correlation test.
Significant correlation between UCLA-GIT 2.0 and other variables.
Individual domains of the UCLA-GIT 2.0 score showed a significant correlation with most PROMs related to QoL and disability; however, all individual UCLA-GIT 2.0 domains showed lower correlation coefficients with these parameters than the total UCLA-GIT 2.0 score (Supplementary Table 2).
Disease duration and UCLA-GIT 2.0 score did not show any correlation (rho: .055, p = 0.488). The average GI complaint of the early SSc (< 3 years, n = 24) subgroup measured by UCLA-GIT 2.0 was not different from the late group (0.13 (0.03-0.46) vs 0.22 (0.08-0.45), p = 0.280) (Supplementary Table 3). The mean disease duration was comparable in Group B + C and Group A (Group B + C: 9.0 (4.5–15.5) years vs Group A: 9.0 (4.0–16.0) years, p = 0.513). The proportion of Group B + C was similar in both the early and late disease stages (21% vs 24%, p = 0.833). None of the PROMs (for QoL: EQ-5D anxiety/depression, SF-36, ScleroID; for disability: HAQ-DI, SHAQ, SHAQ-GI) showed any significant difference between patients in the early and late phases of the disease (Supplementary Table 3). However, if we considered the onset of Raynaud phenomenon as the beginning of the SSc, the UCLA-GIT 2.0 score was found to be significantly higher in late SSc compared to early SSc (0.23 (0.10–0.49) vs 0.04 (0.00–0.14), p = 0.002). In contrast, no significant differences were observed in the PROMs (Supplementary Table 4). Different UCLA-GIT 2.0 score items were examined in patients with limited and diffuse SSc but found no significant differences in any of the UCLA-GIT 2.0 items (data not shown). Subsequently, a subgroup analysis was performed to investigate the correlations between UCLA-GIT 2.0 items and the examined PROMs within the limited and diffuse SSc groups (see Supplementary Tables 5 and 6). Subgroup analyses by SSc subtype revealed that correlations between specific gastrointestinal symptoms and QoL/disability were generally stronger in the lcSSc cohort. Reflux and bloating were the most frequently reported symptoms in the limited SSc group. However, as we did not assess long-term outcomes, further investigation is needed to draw more definitive conclusions.
Regarding the impact of severe GI complaints on the QoL, all investigated self-assessment questionnaires referencing QoL and disability showed worse results among patients with moderate gastrointestinal symptoms (Group B) when compared with patients with none or mild symptoms (Group A). Similarly, patients with severe gastrointestinal complaints by UCLA-GIT 2.0 (Group C) have worse results compared to patients with moderate symptoms (Group B) (Table 4 and Figure 1).
Results of the investigated PROMs related to QoL and disability in SSc patients divided based on the UCLA-GIT 2.0 scores at baseline.
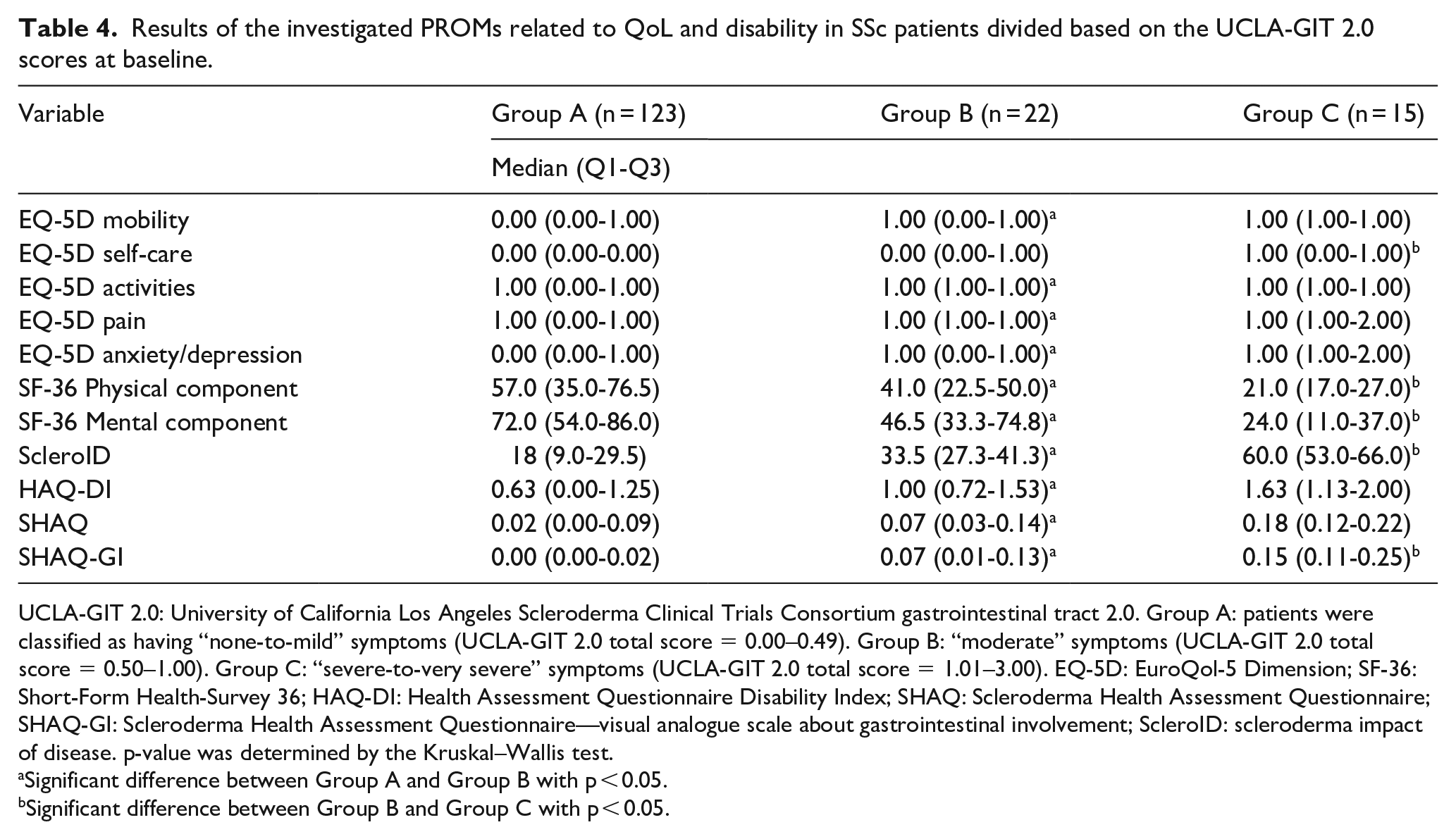
UCLA-GIT 2.0: University of California Los Angeles Scleroderma Clinical Trials Consortium gastrointestinal tract 2.0. Group A: patients were classified as having “none-to-mild” symptoms (UCLA-GIT 2.0 total score = 0.00–0.49). Group B: “moderate” symptoms (UCLA-GIT 2.0 total score = 0.50–1.00). Group C: “severe-to-very severe” symptoms (UCLA-GIT 2.0 total score = 1.01–3.00). EQ-5D: EuroQol-5 Dimension; SF-36: Short-Form Health-Survey 36; HAQ-DI: Health Assessment Questionnaire Disability Index; SHAQ: Scleroderma Health Assessment Questionnaire; SHAQ-GI: Scleroderma Health Assessment Questionnaire—visual analogue scale about gastrointestinal involvement; ScleroID: scleroderma impact of disease. p-value was determined by the Kruskal–Wallis test.
Significant difference between Group A and Group B with p < 0.05.
Significant difference between Group B and Group C with p < 0.05.
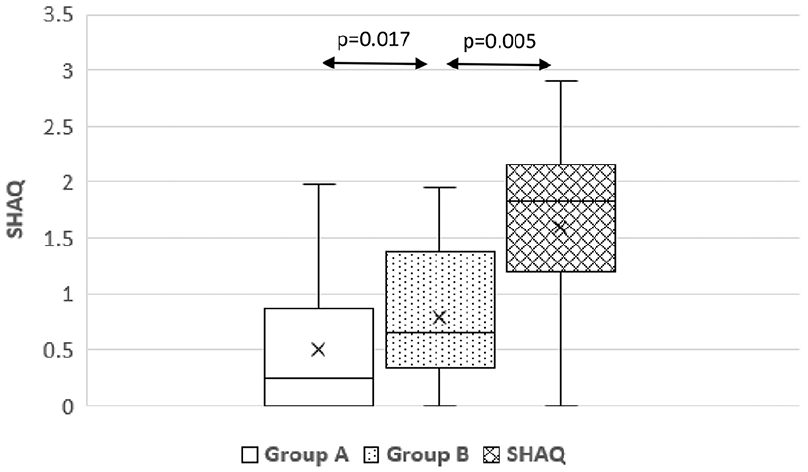
Relationship between the severity of GI symptoms based on the UCLA-GIT 2.0 and QoL based on the SHAQ in patients afflicted with systemic sclerosis.
Among the investigated activity parameters, there was no significant difference between patients with moderate and severe gastrointestinal symptoms (Supplementary Table 7).
Following 1 year, only 7% of participants of the original study had not been retested, 11 patients did not complete our reinvestigations due to loss during follow-up (n = 6) or mortality (n = 5) (i.e. the total number of retested patients: n = 149). The results of the baseline study were overwhelmingly supported by the 1-year follow-up data, with only minor differences (Supplementary Tables 8 to 10 and Supplementary Figure 1): All QoL and disability-related correlations with the UCLA-GIT 2.0 score were confirmed during the 1-year follow-up (rho: 0.263, −0.493, −0.477, 0.405, 0.499, 0.541, respectively). The differences of SHAQ among patients with no/mild, moderate or severe GI symptoms also remained consistent (SHAQ: 0.12 vs 0.68 vs 1.28, p < 0.05).
Discussion
The UCLA-GIT 2.0 instrument is the only widely used questionnaire to establish the presence and severity of GI tract involvement of SSc, which is fully validated.4,31 The average of the total UCLA-GIT 2.0 score of our patients (mean = 0.33) was nearly the same as in an earlier Italian cohort (0.34) 32 and lower than in a French manuscript (0.53). 33 Twenty-three percent of our patients showed moderate-to-severe GI tract involvement, which is similar to the previously reported results: in a recently published longitudinal investigation regarding risk factors for progression of GI involvement, the authors described this ratio as 28%. 34 Severe GI involvement alone has a relatively low incidence in our cohort (9%), which is very similar to the earlier published results of another cohort (Norwegian: 8%, Dutch: 6%). 35
Other clinically significant differences found between those with at least moderate GI involvement (Group B + C) and those with less GI complaint (Group A) were those patients with more severe GI complaints experiencing more hypertension, significantly lower renal function and lower distance during the 6MWT. The association between hypertension and a decline in renal function in scleroderma has also been previously reported; 36 however, to our knowledge, the association between hypertension and GI complaints has not yet been described.37–39 Furthermore, the fact no difference in BMI, in muscle strength, or in cardiopulmonary involvement was detected among the patients despite their differences in 6MWT raises the possibility the differences may be due to vascular abnormalities associated with advanced GI complaints. However, there is only limited evidence to suggest that patients with higher UCLA-GIT 2.0 severity scores experience an impact on the 6MWT due to vascular abnormalities associated with GI, or a potential link between gastrointestinal complaints and macroangiopathy. One possible explanation is that gastrointestinal vascular involvement in SSc, such as ischemia-reperfusion injury, impaired microcirculation, or vascular remodeling 40 may lead to malabsorption and chronic anemia which can contribute to reduced physical capacity. Since anemia is defined by a decreased hemoglobin level, 41 we compared this parameter between patients with no/mild and moderate/severe gastrointestinal symptoms, both as a continuous and as a categorical variable (see Table 1), but found no significant difference. However, we did observe a significant difference in the prevalence of malabsorption, which supports our hypothesis. And also, if the disease duration considered from the beginning of the Raynaud phenomenon, gastrointestinal complains became more prominent, probably due to vascular damage. Although we did not directly assess vascular abnormalities in the gastrointestinal tract, this remains an important area for future research.
No SSc-specific autoantibodies were found which can distinguish these three groups from one another, and only uncertain differences were detected due to the low number of patients (details in Table 1, Supplementary Table 11, and Supplementary Figure 2).
Only few papers evaluate the UCLA-GIT 2.0 and SHAQ-GI in parallel, for the assessment of GI symptoms.4,42,43 In our study, we found a high correlation between SHAQ-GI and UCLA-GIT 2.0 (rho: 0.588, p < 0.001) in our total patient population, as well as our groups of patients with none-to-mild (Group A), moderate (Group B), and severe GI complaints (Group C) based on the UCLA-GIT 2.0 scores showed significant differences in their SHAQ-GI scores (Table 4). Similarly, in a recent publication, both total UCLA-GIT 2.0 and average SHAQ values significantly reflected the particular SSc clusters based upon their organ involvements. 44
Data on the correlation between the UCLA-GIT 2.0 and the two most accepted PROMs regarding QoL (SF-36, EQ-5D) in scleroderma patients32,33,45 are conflicting. In our study, both instruments achieved moderate correlation (rho: SF-36 PCS = −0.484, SF-36 MCS = −0.468, EQ-5D anxiety/depression = 0.400, Table 2) with UCLA-GIT 2.0. These data were similar in most of the previously published papers: rho values of correlation between UCLA-GIT 2.0 and SF-36 PCS were −0.136 −0.342, −0.37, −0.43, −0.482 in different cohorts.33,43,45–47 The correlation between UCLA-GIT 2.0 and SF-36 MCS was also moderate in these papers (−0.34, −0.37, −0.39, −0.45, −0.54). Our results referencing this correlation between UCLA-GIT 2.0 and SF-36 (and EQ-5D) were consequent not only in the entire patient cohort but also separately in the lcSSc and dcSSc subsets (Table 2). On the contrary, in the Italian cohort, SF-36 did not show any significant association with total UCLA-GIT 2.0, only SF-36 MCS showed a moderate correlation with the “emotional well-being” component of UCLA-GIT 2.0 score. The authors suggested, the GI involvement has greater impact upon mental when compared to physical health in SSc, 32 since our results, the SF-36 PCS showed a stronger correlation with the UCLA-GIT 2.0 score than the correlation we could demonstrate for the SF-36 MCS. Furthermore, in consideration of EQ-5D, its anxiety/depression component showed the strongest association with gastrointestinal complaints (Table 2). A novel disease-specific questionnaire was recently introduced for SSc (ScleroID) 48 , which showed an even stronger correlation (rho: 0.646, p < 0.001) between patients’ QoL and severity of GI involvement compared to previous, non–disease-specific questionnaires, in which we have already published in a previous publication. 27 Moreover, in our study, all domains of the UCLA-GIT 2.0 score were correlated with QoL indicators; however, factually, the total UCLA-GIT 2.0 score was more strongly correlated with these than any of the domains alone (Table 2 and Supplementary Table 2), confirming the earlier premise in which the use of a total score by itself is a better reflection of the overall status of the GI system than individual domains alone. 17
In addition, the most used measurements of disease activity (r-EScSG-AI, Pecs-8.5) did show only weak correlation and only in different SSc subsets (Table 3). On the contrary, one of our key findings highlighted the severity of GI involvement measured by UCLA-GIT 2.0 was associated with worse QoL among patients afflicted with SSc (Table 2). Both investigated PROMs (EQ-5D and SF-36) indicated significantly more impaired QoL in Group B (patients with moderate GI involvement) when compared with Group A (no or mild GI manifestation). Similarly, patients with severe GI complaints (Group C) stated much lower level of QoL matched even with Group B (Table 4 and Figure 1). To the best of our knowledge, we are reporting this finding for the first time in published literature. The strong correlation between GI complaints and QoL indices, and the deterioration in QoL between those with no/mild complaints and those with more severe complaints, remained at the follow-up 1 year later; however, the significance between those with moderate and those with severe GI complaints was no longer significant. Comprehensively, the UCLA-GIT 2.0 score is considered to reflect QoL well and can distinguish patients with good QoL without complaints (Group A) from patients with marked GI complaints and poor QoL (Group B + C).
Regarding the relationship of overall disability and the impact of GI complaints assessed by the UCLA-GIT 2.0 questionnaire, all investigated indices (HAQ, SHAQ-GI) showed significant moderate correlation with UCLA-GIT 2.0 in the entire SSc cohort and the lcSSc and dcSSc subgroups as well (Table 2). The strength of these associations (HAQ rho: .400, SHAQ rho: .436) was very similar in previous papers (SHAQ rho: 0.347, 0.448, 0.559).42,43,46 Nevertheless, the cited publications did not investigate these correlations in the single SSc subgroups (lcSSc and dcSSc) separately, in this respect, our results can be considered as the first publication in this field. The SHAQ was able to discriminate between patients divided into three groups by the UCLA-GIT 2.0 score in both the first and second studies, that is, GI complaints were a major contributor to patient disability (Table 4, Figure 1, Supplementary Table 10, and Supplementary Figure 1).
GI complaints by UCLA-GIT 2.0 scores were not influenced by disease duration and disease duration did not correlate with QoL measured by PROMs. These findings suggest GI abnormalities develop early in the course of the disease. This is supported by the fact we did not observe any significant change in UCLA-GIT 2.0 results during our 1-year follow-up (see Supplementary Table 1).
In consideration of these results, we can conclude the results by UCLA-GIT 2.0 instrument are in accordance with the opinion of patients regarding their disability and impaired QoL in much higher proportion than with overall disease activity determined by physicians. These GI complaints develop early in the SSc and are parallel with early onset of anxiety, decreased QoL and disability. Our results also raise the possibility of a link between GI complaints and hypertension and related macroangiopathy.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983251345284 – Supplemental material for Patient-reported gastrointestinal involvement is associated with reduced quality of life and disability in systemic sclerosis
Supplemental material, sj-pdf-1-jso-10.1177_23971983251345284 for Patient-reported gastrointestinal involvement is associated with reduced quality of life and disability in systemic sclerosis by Patrik Kéringer, Katalin T Kovács, Gabriella Nagy, Ágnes Ágoston-Szabó, Kristóf Filipánits, Franciska Ilona Kiss, Attila Szabó and Gábor Kumánovics in Journal of Scleroderma and Related Disorders
Footnotes
Acknowledgements
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the “PEPSYS GINOP-2.3.2–15-2016–00050—Stratégiai K + F műhelyek kiválósága: A peptiderg szignalizáció komplexitása és szerepe szisztémás betegségekben” grant, EUSTAR Database Improvement Grant—2024 (![]() ), and TKP-2021-EGA-10 has been implemented with the support provided from the National Research, Development and Innovation Fund of Hungary, financed under the TKP-2021-EGA funding scheme.
), and TKP-2021-EGA-10 has been implemented with the support provided from the National Research, Development and Innovation Fund of Hungary, financed under the TKP-2021-EGA funding scheme.
Statement of ethics and consent
The study was conducted according to the Declaration of Helsinki and approved by the Ethics Committee of National Public Health Centre Institutional Committee of Science and Research Ethics (39672-6/2020/EÜIG, 2022.10.03, Budapest, Hungary).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
