Abstract
Objective:
The Health Assessment Questionnaire–Disability Index is an important outcome measure reflecting functional disability, but knowledge on its course over time in patients with systemic sclerosis is scarce. Therefore, we investigated the long-term course of the Health Assessment Questionnaire–Disability Index and its association with baseline characteristics in systemic sclerosis patients.
Methods:
Systemic sclerosis patients, fulfilling the European League Against Rheumatism and the American College of Rheumatology 2013 criteria, were included from the Leiden Combined Care in Systemic Sclerosis cohort with annual assessments including the Scleroderma Health Assessment Questionnaire–Disability Index (range = 0–3). The course of the Health Assessment Questionnaire–Disability Index was evaluated over the total follow-up (baseline to last available Health Assessment Questionnaire–Disability Index) and between yearly visits. Based on a minimal clinical important difference of 0.22, courses were categorized into worsening, stable or improvement. The course of the Health Assessment Questionnaire–Disability Index over time was evaluated with linear mixed models. Baseline characteristics were compared between patients with a worsening or improvement of the Health Assessment Questionnaire–Disability Index over the total follow-up period with logistic regression analyses.
Results:
A total of 517 systemic sclerosis patients were included, with a median follow-up of 7 years (interquartile range = 4–9; 2649 visits) and a baseline Health Assessment Questionnaire–Disability Index of 0.625 (interquartile range = 0.125–1.25). On group level, the Health Assessment Questionnaire–Disability Index is stable with an annual increase of 0.019 (95% confidence interval = 0.011 to 0.027). Looking at subgroups, patients >65 years or who died/were physically unable to come during follow-up had a worse mean Health Assessment Questionnaire–Disability Index. In individual courses from baseline to the last follow-up, the proportions of patients with a clinically meaningful worsening, stable or improved Health Assessment Questionnaire–Disability Index were 35%, 42% and 23%, respectively. Patients with immunosuppressants (odds ratio = 0.5, 95% confidence interval = 0.3 to 0.9) or gastrointestinal involvement (odds ratio = 0.6, 95% confidence interval = 0.4 to 0.9) at baseline showed a reduced chance of worsening of the Health Assessment Questionnaire–Disability Index over the total follow-up period.
Conclusion:
Over time, the average course of the Health Assessment Questionnaire–Disability Index was stable in systemic sclerosis patients. However, individual courses vary, with worsening occurring in one-third. Worsening occurred less often in individuals using immunosuppressants or with gastrointestinal involvement at baseline.
Key messages
Over time, the Health Assessment Questionnaire–Disability Index in systemic sclerosis patients remained stable on group level.
Patients >65 years or who died/were physically unable to come during follow-up had a worse mean Health Assessment Questionnaire–Disability Index over time.
Individual trajectories varied considerably, with one-third of patients experiencing a clinically meaningful worsening on the Health Assessment Questionnaire–Disability Index, and one-quarter experiencing clinically meaningful improvement.
Worsening was less likely in individuals using immunosuppressive therapy of with gastrointestinal involvement at baseline.
Introduction
Systemic sclerosis (SSc) is a severe rheumatic autoimmune disease 1 affecting multiple organ systems that elicits a broad range of symptoms.2,3 Functional disability is a hallmark of SSc, with a severe impact on patients’ health-related quality of life (HRQoL).4,5 The Health Assessment Questionnaire–Disability Index (HAQ-DI) is one of the most commonly used measures of functional disability in rheumatic and musculoskeletal diseases, 6 including SSc.5,7,8 The HAQ-DI is a self-reported questionnaire on daily-life activities, that is scored according to the level of experienced difficulty. For SSc, five SSc-related Visual Analogue Scales (VASs) have been added to more accurately reflect disease activity in SSc-specific domains (Scleroderma Health Assessment Questionnaire SHAQ). 9
The HAQ-DI has been used as an outcome measure in clinical trials,8,10,11 and several studies have investigated cross-sectional associations of the HAQ-DI with disease characteristics in SSc.9,12–15 However, only two studies evaluating the course of the HAQ-DI in SSc are available: an American study from 1997 with 1250 SSc patients and a Dutch study from 2017 with 215 SSc patients. Both demonstrated little change in the HAQ-DI over 3 and 4 years, respectively.9,16 Obtaining information regarding functional disability over a longer period of time is important, as preventing functional disability is a primary aim of SSc treatment.
Therefore, this study aimed to describe the long-term course of functional disability in SSc patients from the Leiden Combined Care in Systemic Sclerosis (CCISS) cohort, 17 specifically to investigate which patients have a worsening functional ability, and which characteristics are risk factors for worsening of functional ability.
Methods
Study design and patients
This study included patients who participated in the ongoing and prospective CCISS cohort at Leiden University Medical Center 17 until 1 January 2022, fulfilled the European League Against Rheumatism and the American College of Rheumatology (ACR/EULAR) SSc 2013 criteria, 18 had ⩾1 follow-up visit and completed the baseline HAQ. In the CCISS cohort, all SSc patients undergo an annual assessment for organ involvement. All patients included in the cohort provided written informed consent. The cohort was approved by the LUMC Ethics Committee (CME no. B16.037, REU 043/SH/sh, P09.003/SH/sh).
SHAQ
Functional disability was evaluated using the SHAQ, an SSc-specific extension of the HAQ. The HAQ was developed for adults with arthritis and consists of 20 questions split across 8 domains: rising, eating, walking, hygiene, dressing, reach, grip and usual activities. For each category, patients report the amount of difficulty they experience with that activity on a scale of 0 to 3, where 0 indicates ‘without difficulty’ and 3 indicates ‘unable to do’. The final composite score (disability index, known as HAQ-DI) ranges from 0 (no disability) to 3 (maximal disability). 6
The SHAQ adds five VAS scores (range = 0–100), which assess the level of impairment due to common non-musculoskeletal SSc complications: Raynaud’s phenomenon (RP), digital ulcers (DUs), gastrointestinal (GI) symptoms, respiratory symptoms and the overall disease severity from the patients’ perspective. 9 The SHAQ is a reliable and valid measure of functional disability in SSc.5,7,9,19
As proposed by the previous literature, the minimal clinical important difference (MCID) of the HAQ-DI in SSc is −0.037 for improvement and +0.140 for worsening. 20 However, with the aim of a higher specificity in classifying the patients who truly improve in this study, we decided to use a more stringent cut-off of 0.22, which is the MCID of the HAQ-DI determined in rheumatoid arthritis patients.21,22
Study variables
Sociodemographic characteristics included age at baseline (years), sex (female/male), socioeconomic status (low/high), current smoker (yes/no), cardiac or pulmonary comorbidities (yes/no) and body mass index (BMI) (kg/m2).
Disease characteristics included disease duration in months since RP and first non-RP symptom (=non-RP). Incident cases were defined as a non-RP duration <24 months. The presence of anti-centromere (ACA) or anti-topoisomerase antibodies (ATA) was tested. Skin involvement was measured using the modified Rodnan Skin Score (mRSS), 22 categorizing patients into sine scleroderma (no previous or current skin fibrosis), limited (current or previous skin fibrosis limited to the hands, face, feet and forearms) and diffuse cutaneous (current or previous skin fibrosis affecting the upper arms/legs or trunk) accordingly. 23 Cardiac involvement was defined as at least two of the following: arrhythmias (>2% ventricular or supraventricular arrhythmia, atrial fibrillation), conduction problems, decreased left ventricular ejection fraction <54%, diastolic or systolic dysfunction, pericarditis or pericardial effusion. Pulmonary arterial hypertension (PAH) was defined as a mean pulmonary arterial pressure ⩾25 mm Hg at rest as assessed by right heart catheterization, including the presence of pre-capillary pulmonary hypertension (PH), defined by a pulmonary capillary wedge pressure ⩽15 mm Hg and a pulmonary vascular resistance >3 Wood units on right heart catheterization. Interstitial lung disease (ILD) was determined based on the presence of interstitial fibrosis or ground glass opacities on high-resolution computed tomography of the thorax combined with a forced vital capacity <80%. GI involvement was defined as the presence of gastric antral vascular ectasia OR parenteral feeding OR anaemia and a BMI <20 and one of the following symptoms: reflux, bloating, distension, diarrhoea or faecal incontinence. Renal involvement was defined as a clinical diagnosis of a renal crisis. Musculoskeletal involvement was defined as the presence of synovitis, myositis or tendon friction rubs. Furthermore, the presence of joint contractures was evaluated and immunosuppressive medication use was collected. In addition, the 6-min walking distance in metres was recorded.
Statistical analysis
Descriptive statistics were used to present the demographic and clinical characteristics of the included patients. Continuous variables were presented as mean (standard deviation (SD)) or median (interquartile range (IQR)) and categorical variables as number (percentage).
The primary aim of the study was to describe the long-term course of the HAQ over time using three methods. First, the difference between HAQ at the last visit and HAQ at the baseline was calculated for each patient. In case the HAQ had not been recorded at the last visit, the penultimate value was considered. The course of the HAQ-DI was then categorized into three groups of clinically meaningful changes in HAQ-DI using the MCID: worsening (⩾+0.22), stable (between −0.21 and +0.21) or improvement (⩽−0.22).
Second, to take into account all visits, a linear mixed model was performed with HAQ-DI dependent on time to evaluate the course of the HAQ-DI over time for the total group, yielding a β representing the average change per year. Third, to describe if and how the HAQ-DI scores fluctuated per visit, the change in HAQ-DI compared to the previous visit was calculated for every follow-up visit. Based on the MCID, the change between two visits was then categorized as worsening (⩾+0.22), stable (between −0.21 and +0.21) or improvement (⩽−0.22). Heat maps were constructed to display this information.
The secondary aim of this study was to investigate factors associated with the overall course of the HAQ-DI. Baseline characteristics between the worsening and improvement groups as described above were compared with logistic regressions adjusted for age, sex and disease duration. In addition, the association with all timepoints of the HAQ-DI was evaluated for the following variables: age above 65 years (above vs lower than 65 years), sex (female vs male), incident case (=duration of symptom other than RP <24 months, yes vs no), autoantibody status (ACA and ATA negative vs ACA and ATA positive), disease subset (non-cutaneous vs limited cutaneous and diffuse cutaneous) and death/physically unable to come (yes vs no).
As a sensitivity analysis, we performed the analyses in a subgroup of patients classified as incident cases, that is, a disease duration of non-RP of less than 24 months.
To evaluate the five SSc-specific VAS scores of the SHAQ over time, linear mixed models were performed for the total group and the different strata (age above 65 years, sex, autoantibody status, incident case, disease subset and death/physically unable). A p-value of less than 0.05 was considered statistically significant. Statistical analyses were performed with STATA SE16 (StataCorp, College Station, TX, USA).
Results
Patients
In total, 517 of the 708 (73%) SSc patients from the Leiden CCISS cohort were included in this study (Supplemental Figure S1). Compared to the 191 excluded patients, the included patients were more often females (74% vs 81%, p = 0.048) and were on average younger (57 (SD = 15) vs 54 (SD = 14) years, p = 0.028; Supplemental Table S1).
Table 1 shows the baseline characteristics of the included patients. The median follow-up duration was 7 years (IQR = 4–9). The mean baseline HAQ was 0.77 (SD = 0.65).
Baseline characteristics of 517 SSc patients from the Leiden CCISS cohort.
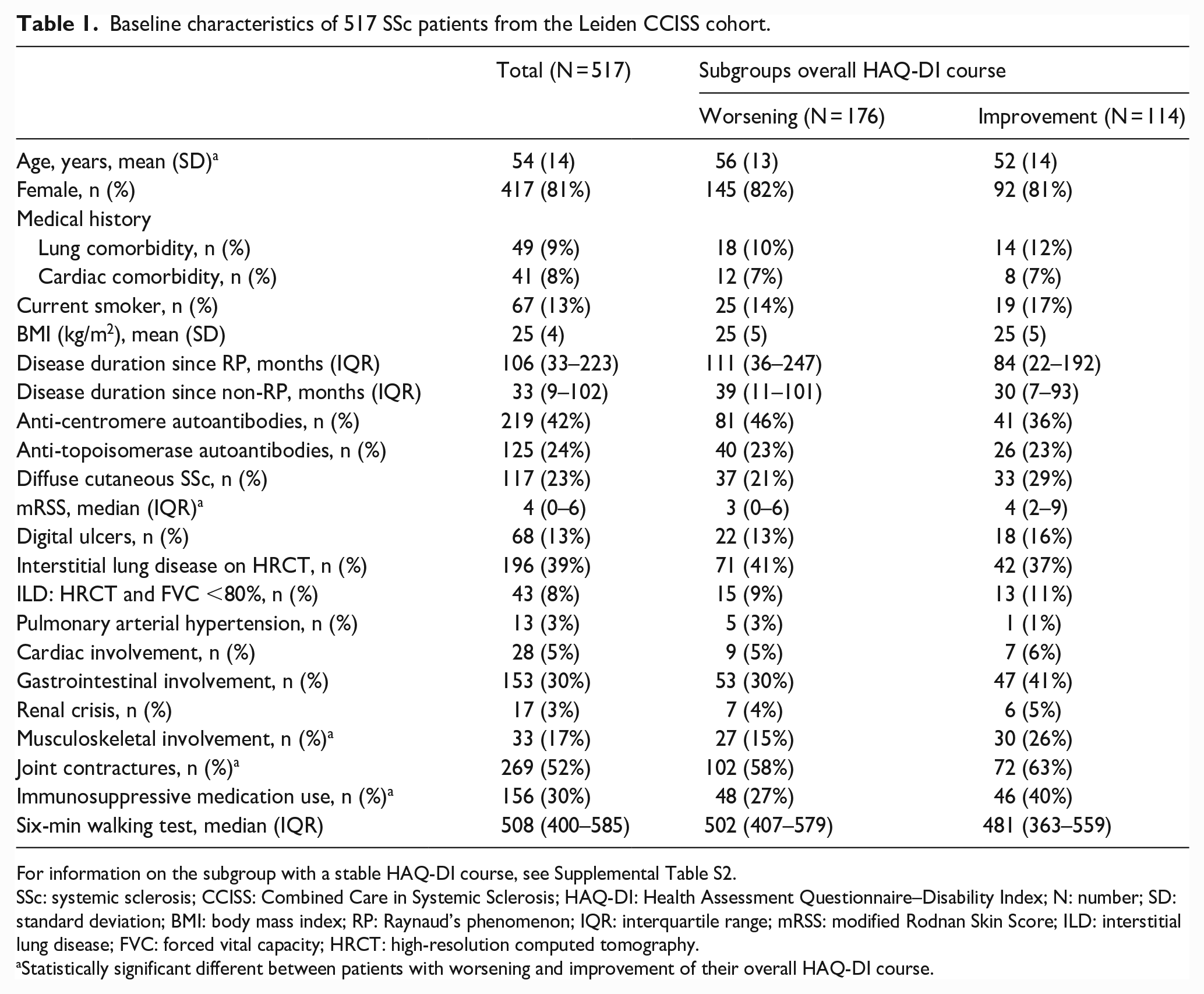
For information on the subgroup with a stable HAQ-DI course, see Supplemental Table S2.
SSc: systemic sclerosis; CCISS: Combined Care in Systemic Sclerosis; HAQ-DI: Health Assessment Questionnaire–Disability Index; N: number; SD: standard deviation; BMI: body mass index; RP: Raynaud’s phenomenon; IQR: interquartile range; mRSS: modified Rodnan Skin Score; ILD: interstitial lung disease; FVC: forced vital capacity; HRCT: high-resolution computed tomography.
Statistically significant different between patients with worsening and improvement of their overall HAQ-DI course.
The course of the HAQ-DI
The mean change in HAQ-DI from baseline to last measurement was 0.07 (SD = 0.50). The overall course for the total and covariates is shown in Figure 1.
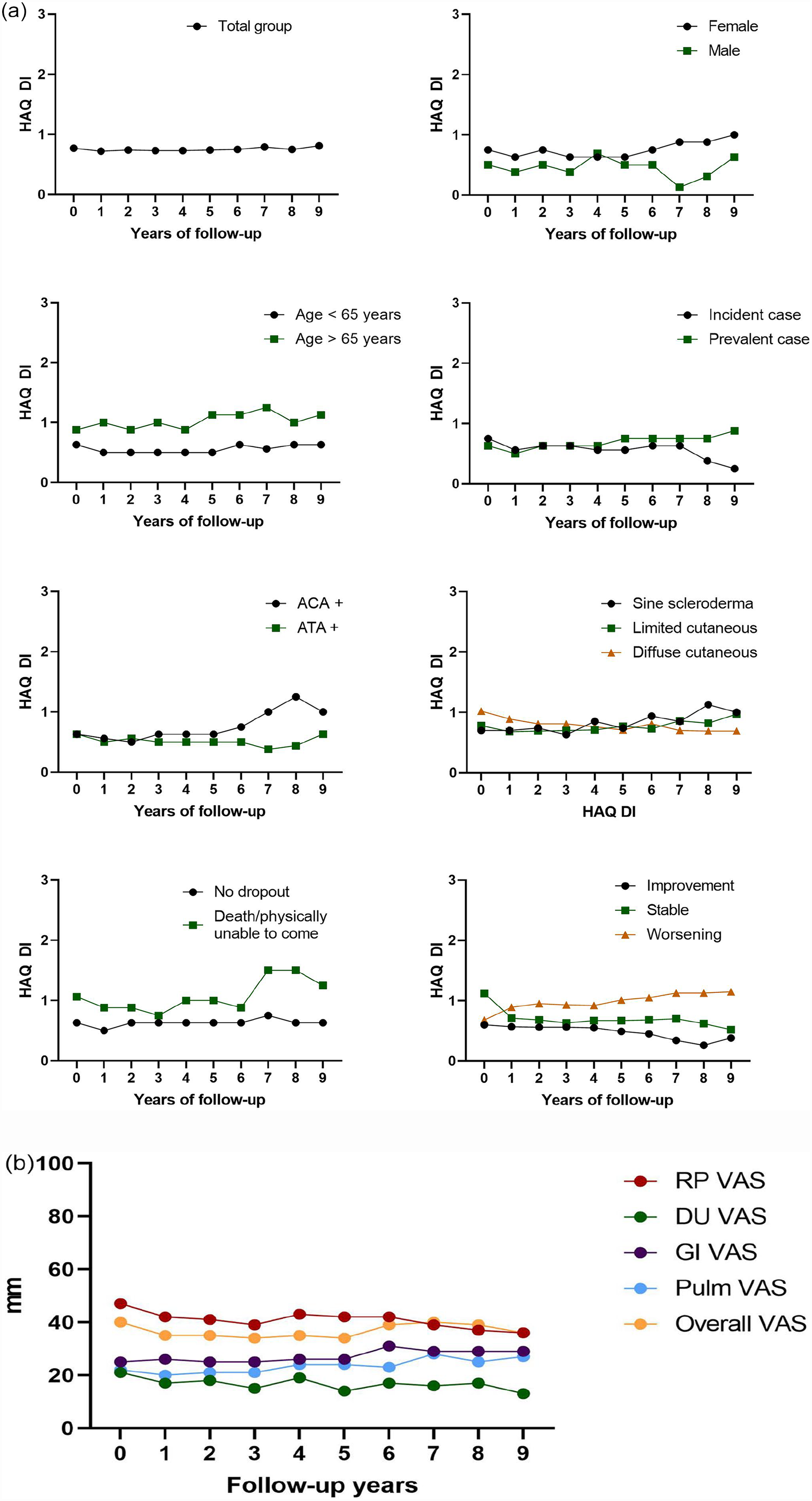
Health Assessment Questionnaire over time in SSc patients. (a) Median HAQ-DI over time. (b) Scleroderma-related VAS scores (mean) of the HAQ over time in SSc patients.
Over time, linear mixed models showed that the HAQ-DI of the total group of patients worsened with 0.019 per year (95% confidence interval (CI) = 0.011 to 0.027) (Table 2). Patients ⩾65 years (β = 0.27, 95% CI = 0.1 to 0.4) and patients who died/were physically unable to come during follow-up (β = 0.34, 95% CI = 0.2 to 0.5) had a significantly higher HAQ-DI over time than, respectively, patients younger than 65 years and patients who did not die (Table 2). The plots in Figure 1 show that ACA-positive patients experience a worsening in HAQ-DI over time, especially after 5 years of follow-up.
Linear mixed models to evaluate the change in HAQ-DI over time in SSc patients.

Incident case: disease duration since first non-Raynaud’s phenomenon symptom <24 months. Range of HAQ-DI: 0 (no disability) to 3 (maximal disability). A β of 0.27 means that patients >65 years have a 0.27 higher HAQ-DI than patients <65 years per follow-up year.
HAQ-DI: Health Assessment Questionnaire–Disability Index; SSc: systemic sclerosis; CI: confidence interval; ACA: anti-centromere autoantibody; ATA: anti-topoisomerase antibody.
Over the entire period of follow-up, 176 (35%) patients experienced worsening, 202 (42%) remained stable and 114 (23%) experienced improving HAQ-DI. The changes in the HAQ-DI per annual follow-up visit are displayed in a heat map (Figure 2). A total of 1777/2649 (67%) visits were evaluated, 451 of which (25%) indicated worsening, 954 (54%) a stable situation and 372 (21%) an improvement. This pattern is in line with the percentages of patients in the three categories for the total follow-up period. However, the heat map shows that there is considerable variation in the course per visit, also within the subgroups of courses of patients classified as worsening, stable and improvement based on difference in HAQ between baseline and last follow-up. When analysing the categories of changes per yearly visit per patient, the HAQ-DI was always stable for 123 patients (27%) and always worsened for 37 (8%). In 51 cases (11%), the HAQ-DI was either stable or improved during follow-up, in 67 (14%), it was stable or worsened, while 32 patients (7%) experienced both worsening and improvement. Finally, in 153 patients (33%), follow-up was characterized by a fluctuating trend of HAQ-DI between stability, improvement and worsening.
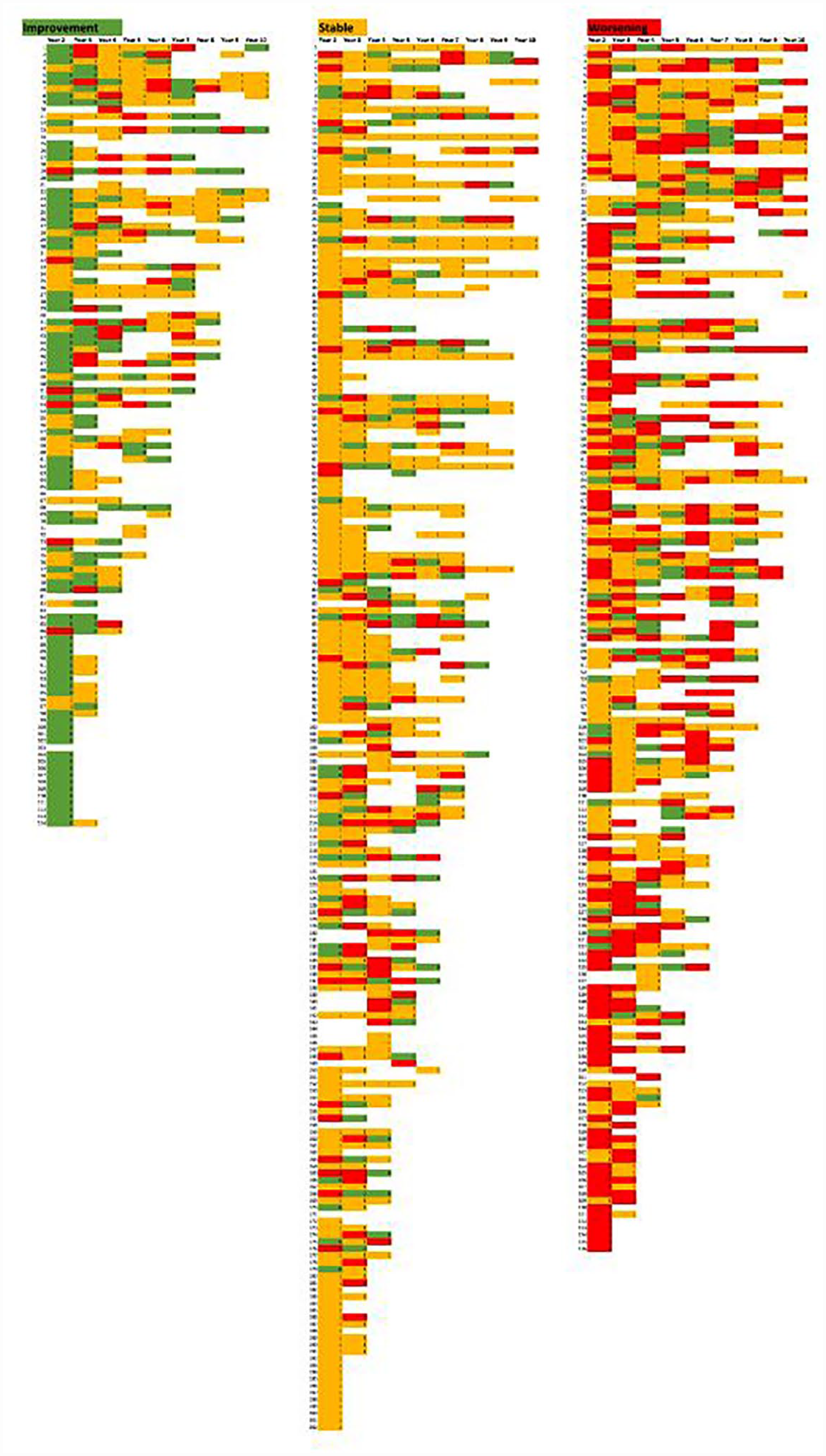
Heat map of the change per visit in HAQ-DI.
Factors associated with the HAQ-DI course
Compared to patients experiencing an improvement in HAQ-DI, patients classified as having a worsening HAQ-DI were older (52 (14) vs 56 (13) years, p = 0.020), had a lower skin score (4 (2–9) vs 3 (0–6), p = 0.043), less often had musculoskeletal involvement (26% vs 15%, p = 0.022), less often had joint contractures (63% vs 58%, p < 0.001) and less often used immunosuppressive medications (40% vs 27%, p = 0.020) (Table 1).
Logistic regressions, adjusted for age, sex and disease duration, revealed that patients with immunosuppressive medication at baseline (odds ratio (OR) = 0.5, 95% CI = 0.3 to 0.9) or GI involvement at baseline (OR = 0.6, 95% CI = 0.4 to 0.9) were associated with a lower chance of worsening HAQ-DI (Table 3). A trend was seen for the presence of PAH (OR = 2.3, 95% CI = 0.3 to 20.8) and worsening of the HAQ-DI course (Table 3).
Logistic regressions to evaluate associations between the subgroups of the overall course of HAQ-DI and baseline disease characteristics in SSc patients.
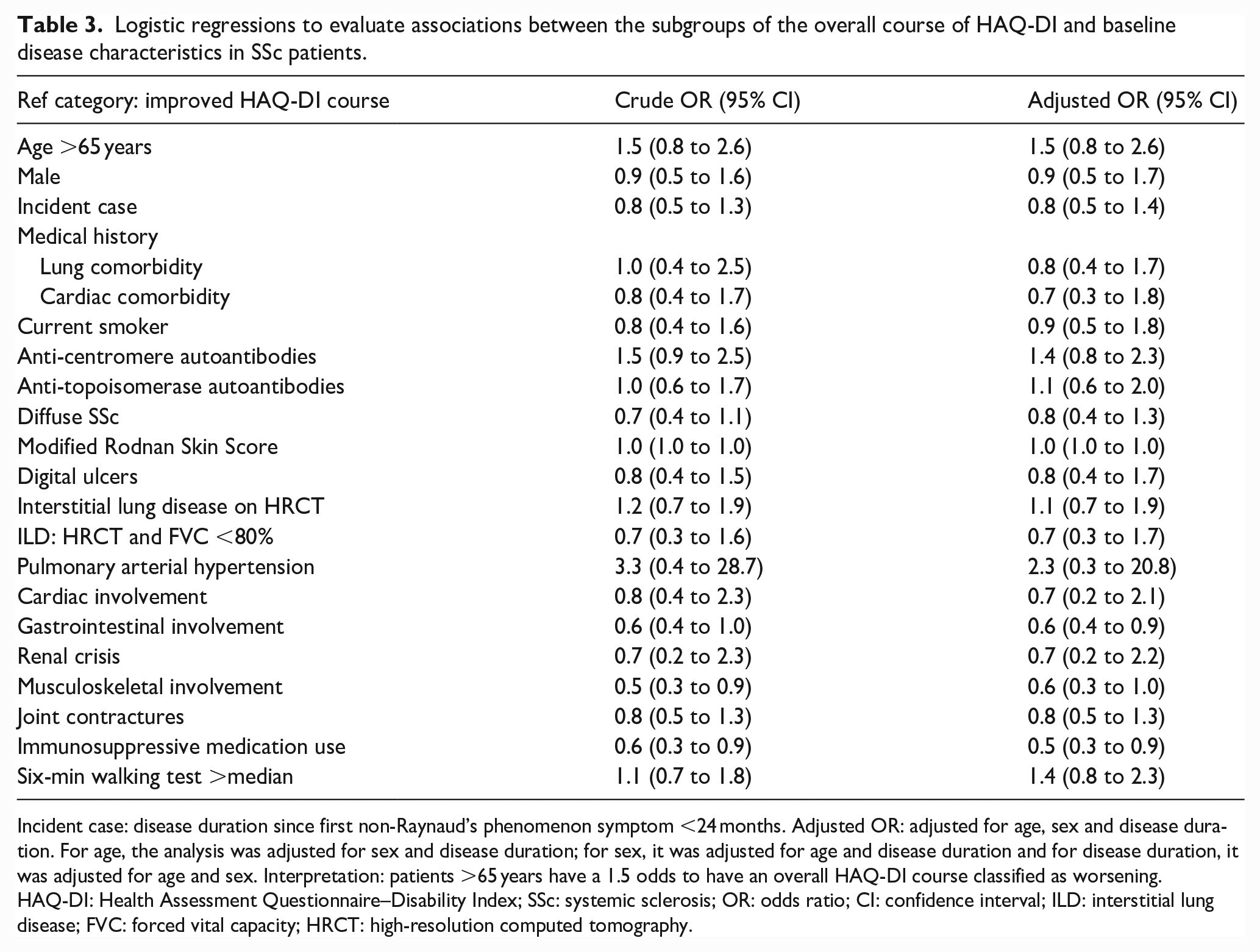
Incident case: disease duration since first non-Raynaud’s phenomenon symptom <24 months. Adjusted OR: adjusted for age, sex and disease duration. For age, the analysis was adjusted for sex and disease duration; for sex, it was adjusted for age and disease duration and for disease duration, it was adjusted for age and sex. Interpretation: patients >65 years have a 1.5 odds to have an overall HAQ-DI course classified as worsening.
HAQ-DI: Health Assessment Questionnaire–Disability Index; SSc: systemic sclerosis; OR: odds ratio; CI: confidence interval; ILD: interstitial lung disease; FVC: forced vital capacity; HRCT: high-resolution computed tomography.
The sensitivity analyses only including patients with a non-RP duration of less than 24 months are in line with these results (Supplemental Tables S3–S5).
SSc-related VAS scores
At baseline, the mean (SD) VAS was 47 (32) for RP, 21 (31) for DU, 25 (31) for GI problems, 22 (27) for pulmonary problems and 40 (32) for general problems related with SSc. The overall course of the VAS scores is shown in Figure 1(b). All were stable (Table 4).
Linear mixed models to evaluate change in SSc-related VAS scores over time in SSc patients.

Incident case: disease duration since first non-Raynaud’s phenomenon symptom <24 months. VAS: scale of 0 (no burden) to 100 (high burden). A β of 4 means that patients >65 years have a 4 higher Raynaud’s phenomenon VAS than patients <65 years over time.
SSc: systemic sclerosis; VAS: visual analogue scale; CI: confidence interval; ACA: anti-centromere autoantibody; ATA: anti-topoisomerase antibody; HAQ-DI: Health Assessment Questionnaire–Disability Index.
Over time, patients who died/were physically unable to visit during follow-up had a 19 point higher pulmonary VAS (95% CI = 13 to 25) and a 13 higher general burden of SSc VAS (95% CI = 7 to 20) compared to patients who survived. In addition, ACA-positive SSc patients had a higher RP VAS and GI VAS compared to ACA- and ATA-negative SSc patients (RP VAS: β = 9; 95% CI = 4 to 14 and GI VAS: β = 10; 95% CI = 5 to 15), but a lower pulmonary VAS (β = −5; 95% CI = −9 to −0.5). For the DU VAS, incident SSc patients had a lower DU VAS compared to prevalent SSc cases (β = −10; 95% CI = −14 to −6). Diffuse cutaneous patients had a higher general burden of SSc VAS over time (β = 11; 95% CI = 6 to 16). Over time, male SSc patients had a lower GI VAS than female SSc patients (β = −9; 95% CI = −14 to −3).
Discussion
Summary
This study found that the overall long-term course of the HAQ-DI was relatively stable. However, individual courses in HAQ-DI varied strongly, with the HAQ-DI worsening in 35% of the patients, being stable in 42% and improvement in 23%. Patients with immunosuppressants or GI involvement at baseline less often showed worsening of the HAQ-DI.
Adaptation
We found a stable HAQ-DI over time on group level, which is in line with two previous cohort studies.9,16 Still, we observed that patients ⩾65 years and patients who died or were physically unable to come during follow-up had a less favourable HAQ-DI course. In addition, looking at individual courses, one-third of the patients showed worsening and one-quarter showed improvement of the HAQ-DI over time. It is noteworthy that the changes of the HAQ-DI compared to the previous visit were classified as stable in the majority of visits, but the yearly changes reflected the long-term development in both the worsening and improvement groups. This finding indicates that, with the interpretation of annual assessments, clinicians should consider not only the previous visit but also a longer period. It is also important to verify with the patients how they perceive the course of their functional ability both in the past year and from the diagnosis.
A possible explanation for the relatively stable HAQ-DI over time might be that SSc is a chronic disease and that patients adapt to their symptoms. This has been defined as a ‘response shift’, which occurs due to changes in patients’ internal standards, values and/or the concept of the patient-reported outcome measure. 23 Moreover, given the stable HAQ-DI course, it can be argued that the HAQ-DI is not an appropriate outcome measure to evaluate functional disability over time in SSc patients. Originally, the HAQ was developed in 1978 and its basic contents have never been updated. The relevance of some activities can be questioned, such as the use of taps, which now have handles in most Western countries. On the contrary, other activities, such as the physical use of smartphones or computers, are not part of the HAQ, although they are currently highly relevant to patients. Recently, there have been increased efforts to develop outcome measures to be used in trials in SSc. These include the American College of Rheumatology Composite Response Index in SSc (ACR-CRISS)24,25 and the ongoing CRISTAL project (Combined Response Index for Scleroderma Trial Assessing Limited cutaneous SSc).26,27 It is reasonable that an updated version of the HAQ could better capture relevant disabilities in SSc patients.
Associations with disease characteristics
Regarding the comparison of factors associated with the course of the HAQ-DI, the previously mentioned American study found that the HAQ-DI score over time was associated with skin, cardiac and renal involvement, tendon friction rubs, hand contractures, proximal muscle strength and survival. 9 In addition, the Dutch study identified low and high disability subgroups based on baseline values rather than on the course of the HAQ-DI, which was overall stable in both groups. 16 Patients in the high disability group were more likely to be older and female. 16 Furthermore, one European Scleroderma Trials & Research Group (EUSTAR) study, including 690 diffuse cutaneous SSc patients, found that the 1-year worsening of the HAQ-DI was associated with baseline skin score and oesophageal symptoms. 28 In this study, we confirmed that patients with a higher age had a worse mean HAQ-DI over time. This study also suggests a lower risk of worsening of the HAQ-DI in individuals using immunosuppressive medication. Although one might expect that specifically during the first years of disease course the HAQ-DI might change, we could not identify an impact of disease duration on the course of the HAQ-DI. Intriguingly, in this study, specifically ACA-positive patients who in the majority of cases have limited cutaneous SSc show a worsening of the HAQ-DI during follow-up. In the majority of cases, ACA-positive SSc patients are older at disease onset, have a longer disease duration since RP and more often have sine scleroderma or limited cutaneous SSc. From clinical experience, these patients often show a more gradual decline in functional ability, without life-threatening disease complications. Unfortunately, for these SSc patients, less pharmacological treatment options are available and this group is frequently excluded from clinical trials evaluating new drugs. Worsening of the HAQ-DI over time could suggest insufficient treatment in these patients. However, it is good to keep in mind that this cohort was not designed to evaluate the effect of treatment on HAQ-DI. Future research should investigate (non-)pharmacologic interventions and their effect on functional disability over time in this patient group.
Strengths and limitations
The major study strength is the use of a well-defined and prospective SSc cohort with a high rate of data completeness, which contributes to the great generalizability of the results. However, the cohort design might also be considered a limitation of this study. The observational nature does not allow for any causal inferences, such as for the observed association between the use of immunosuppressive therapy and the course of functional ability. In addition, we did not have completed HAQ questionnaires for all time points of the included patients, and number of patients decrease per follow-up year. However, for example, at visit 8 still over a hundred patients are included. Moreover, the included patients were younger and more often female than the excluded patients. We cannot rule out that selection bias might have occurred and the results must be interpreted with caution. Finally, it must be noted that comparisons of associations among the present and other relevant studies are considerably hampered by the differences in study populations, the variables that were taken into account, the duration of follow-up and the type of statistical analyses that were employed.
Conclusion
To conclude, this study found that on group level, the HAQ-DI is relatively stable in SSc patients over time, but individual courses vary considerably and worsening occurred in one-third of patients. For clinical practice, it is important to realize that changes in HAQ occur slowly and may vary over time in individual patients. Therefore, when evaluating a patient, the course of functional ability over a longer period should be taken into account. Importantly, older patients have a higher chance of worsening in HAQ-DI while use of immunosuppressants and GI involvement at baseline decrease the risk of worsening in HAQ. Further prospective studies are warranted to elucidate the association between immunosuppressive treatment and course of HAQ-DI.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983231181719 – Supplemental material for The long-term course of the Health Assessment Questionnaire in patients with systemic sclerosis
Supplemental material, sj-pdf-1-jso-10.1177_23971983231181719 for The long-term course of the Health Assessment Questionnaire in patients with systemic sclerosis by Sophie IE Liem, Sytske Anne Bergstra, Jacopo Ciaffi, Coen van der Meulen, David A Ueckert, Marisca R Schriemer, Tom WJ Huizinga, Theodora PM Vliet Vlieland and Jeska K de Vries-Bouwstra in Journal of Scleroderma and Related Disorders
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by NVLE (Nationale Vereniging voor Lupus, APS, Sclerodermie en MCTD), a Dutch patient organization for rare systemic autoimmune diseases.
Data sharing statement
Data are available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
