Abstract
Interstitial lung disease is a common complication and cause of mortality in patients with systemic sclerosis. Pharmacotherapy for systemic sclerosis–associated interstitial lung disease was mostly limited to off-label use of immunosuppressive drugs until recently, when two drugs became licenced for this condition: nintedanib, an antifibrotic agent, and tocilizumab, a targeted anti-inflammatory/immunomodulatory therapy licenced in the United States. In chronic diseases, communication between physicians and patients is associated with treatment adherence, patient satisfaction, and clinical outcomes. This review of physician–patient communication during systemic sclerosis–associated interstitial lung disease treatment covers key issues identified by studies in Japan, the United States and Europe, as well as the clinical experience, opinion, and recommendations of the physician and patient advocate authors. As discussed, recent surveys in Japan found low usage of guideline-recommended immunosuppressive drugs for systemic sclerosis–associated interstitial lung disease and physician dissatisfaction with them. Physicians and patients in Japan also had differing perceptions about what had been said during consultations, suggesting the need to improve physician–patient communication. Other studies in Japan, the United States and Europe made several key findings. Notably, most patients feel uneasy at the diagnosis of systemic sclerosis–associated interstitial lung disease, and both physicians and patients avoid discussing prognosis and mortality. Furthermore, a white-coat barrier hinders patients raising topics important to them. For physicians, listening and empathy may be key for building rapport with patients. Importantly, physicians and patients have different cognitive models of systemic sclerosis–associated interstitial lung disease, creating communication challenges. There are also similarities and differences in clinical practice and physician–patient communication between countries that are important to consider. From the patient’s perspective, key factors include the quality of the first consultation, physician empathy and active listening, and space to ask questions. Efforts to improve physician–patient communication include peer mentoring, patient self-education (such as the ‘Self-Manage Scleroderma’ website from the University of Michigan), and shared decision-making – although not all activities will necessarily be appropriate everywhere.
Keywords
Introduction
Interstitial lung disease (ILD) is a common complication of systemic sclerosis (SSc), normally manifesting within a few years of SSc onset.1,2 ILD prevalence in SSc patients is estimated at 30%–90%, depending on diagnostic technique. 3 Characterised by chronic inflammation and fibrosis in the lung parenchyma,3,4 SSc-associated interstitial lung disease (SSc-ILD) is a chronic pulmonary condition with a heterogeneous clinical course ranging from stable disease in some patients to progressive respiratory failure and death in others.4,5 Overall, however, ILD is a leading cause of premature mortality in SSc patients,6,7 accounting for over 30% of deaths,3,8 and those with this complication have nearly 3-fold higher risk for premature mortality than SSc patients without ILD.7,9
SSc-ILD patients requiring treatment have limited options. Haematopoietic stem cell transplantation is reserved for those with early-stage disease at high risk of rapid progression due to a high likelihood of developing respiratory failure, whereas lung transplantation is generally restricted to advanced refractory cases at high risk of mortality.10,11 Pharmacotherapy is therefore the most common treatment modality for SSc-ILD. However, few drugs are actually licenced for this condition, and off-label non-specific immunosuppression with cyclophosphamide or mycophenolate mofetil has historically been the most common therapy.10,11
In 2019, nintedanib, an orally administered anti-fibrotic agent, became the first pharmacotherapy to be licenced for treatment of SSc-ILD when it was approved in Japan and the United States; 12 it was subsequently approved in the European Union (EU) in 2020. Nintedanib is a small-molecule inhibitor of multiple receptor tyrosine kinases involved in pro-fibrotic signalling pathways, including platelet-derived growth factor receptors α and β, fibroblast growth factor receptors 1, 2 and 3, and vascular endothelial growth factor receptors 1, 2 and 3.13,14 Nintedanib has therapeutic effects in animal models of SSc-ILD, 12 and reduced the annual rate of decline in forced vital capacity by 44% in a multinational, randomised, phase III clinical trial in SSc-ILD patients (SENSCIS). 15 Subsequently, tocilizumab, a biologic interleukin-6 receptor antagonist, was approved for the same indication in the United States based on the multinational, randomised, phase III FocuSSced trial. 16 However, tocilizumab was not approved for SSc-ILD in the EU following review by the European Medicines Agency.
Updated recommendations and guidelines for physicians on managing SSc-ILD have been issued by societies and experts in Japan, the United States and Europe,4,17 –22 aspects of which need to be communicated by physicians to patients and carers. Furthermore, these guidelines generally do not focus on communication between physicians and patients, although evidence from other areas of medicine suggests that it can influence treatment adherence, patient satisfaction and clinical outcomes, and is particularly important in the management of chronic and rare diseases.23 –25 In this narrative review, we identify and discuss key issues in physician–patient communication during treatment of SSc-ILD, based on relevant studies from Japan, the United States and Europe. As physicians (n = 3 from Japan, the United Kingdom and the United States) and patient/patient advocates (n = 2 from the United Kingdom and Italy), we also provide our clinical experience, expert opinion and recommendations.
Incidence and prevalence of SSc and SSc-ILD
The estimated incidence and prevalence of SSc varies somewhat between studies. In Japan, there were 27,647 patients with SSc-specific medical expenses coverage cards in the fiscal year 2020. 26 In a recent survey using the Japanese Medical Data Centre (JMDC) claims database, the incidence of SSc was 6.6 per 100,000 person-years and the overall prevalence was 37.0 per 100,000 people. 27 Studies of insurance claims databases in the United States estimated an SSc incidence of 8.8 28 and 16.4 29 per 100,000 person-years, and a prevalence of 41.5 28 and 24.4 29 per 100,000 people. In other studies in North America, the reported incidence of SSc was 1.4–5.6 per 100,000 person-years and prevalence was 13.5–44.3 per 100,000 people. 2 In Europe, the estimated incidence was 0.6–2.3 per 100,000 person-years and the prevalence was 7.2–33.9 per 100,000 people, based on a systematic review of five and 11 studies, respectively. 2
Similarly, the estimated incidence and prevalence of SSc-ILD vary slightly between studies. In the JMDC study, the incidence and prevalence of SSc-ILD in Japan was 1.9 per 100,000 person-years and 13.9 per 100,000 persons, respectively. 27 The US studies of claims databases estimated the SSc-ILD incidence to be 1.2 29 and 1.6 28 per 100,000 person-years, and the prevalence to be 6.9 29 and 13.3 28 per 100,000 people. Taking these data together, the proportion of SSc patients with ILD was 29%–30% in Japan in the JDMC study, 27 and 32%–47% and approximately 52% in Europe and North America, respectively. 2
Overall, allowing for methodological differences between these studies, the incidence and prevalence of SSc and SSc-ILD seem to be similar in Japan, the United States and Europe. According to these data, SSc is a rare disease and consequently SSc-ILD is slightly rarer, although frequent among the SSc patient population.
Physician–patient communication in Japan, the United States and Europe
SSc-ILD treatment and physician–patient communication in Japan
The survey in Japan covered September 2015–August 2019 27 (before nintedanib’s approval). During that time, the survey found a low rate of guideline-recommended treatments such as cyclophosphamide and mycophenolate mofetil, with less than 15% of patients receiving these medications. 27
To investigate if issues in physician–patient communication contributed to this suboptimal use of guideline-directed medical therapy, a survey of physicians and SSc-ILD patients was conducted in Japan in 2021. 30 The objective was to explore consistencies or inconsistencies in communication between physicians and their patients with SSc-ILD. Responses were received from 121 rheumatologists treating SSc-ILD and 58 SSc-ILD patients who had been introduced by participating physicians. Information obtained from the patients was blinded to the physicians. Of these 58 patients, 81% were women, 65% were over the age of 60 years, 57% visited the hospital every 2 months and 29% visited every month. In terms of treatment, 70% of patients were on medication (oral glucocorticoids: 41%; tacrolimus: 17%; nintedanib: 15%; cyclophosphamide: 14%).
The survey revealed discrepancies between what information on SSc-ILD and its treatment the physicians said they had explained at diagnosis and what the patients thought had been explained (Figure 1(a)). However, there was general agreement between physicians and patients regarding which information is important (Figure 1(b)). These findings were similar at treatment initiation, with many physicians responding that ‘I explain this to all of my patients’ or ‘I explain this to most of my patients’, whereas few patients responded that ‘they received explanations’.
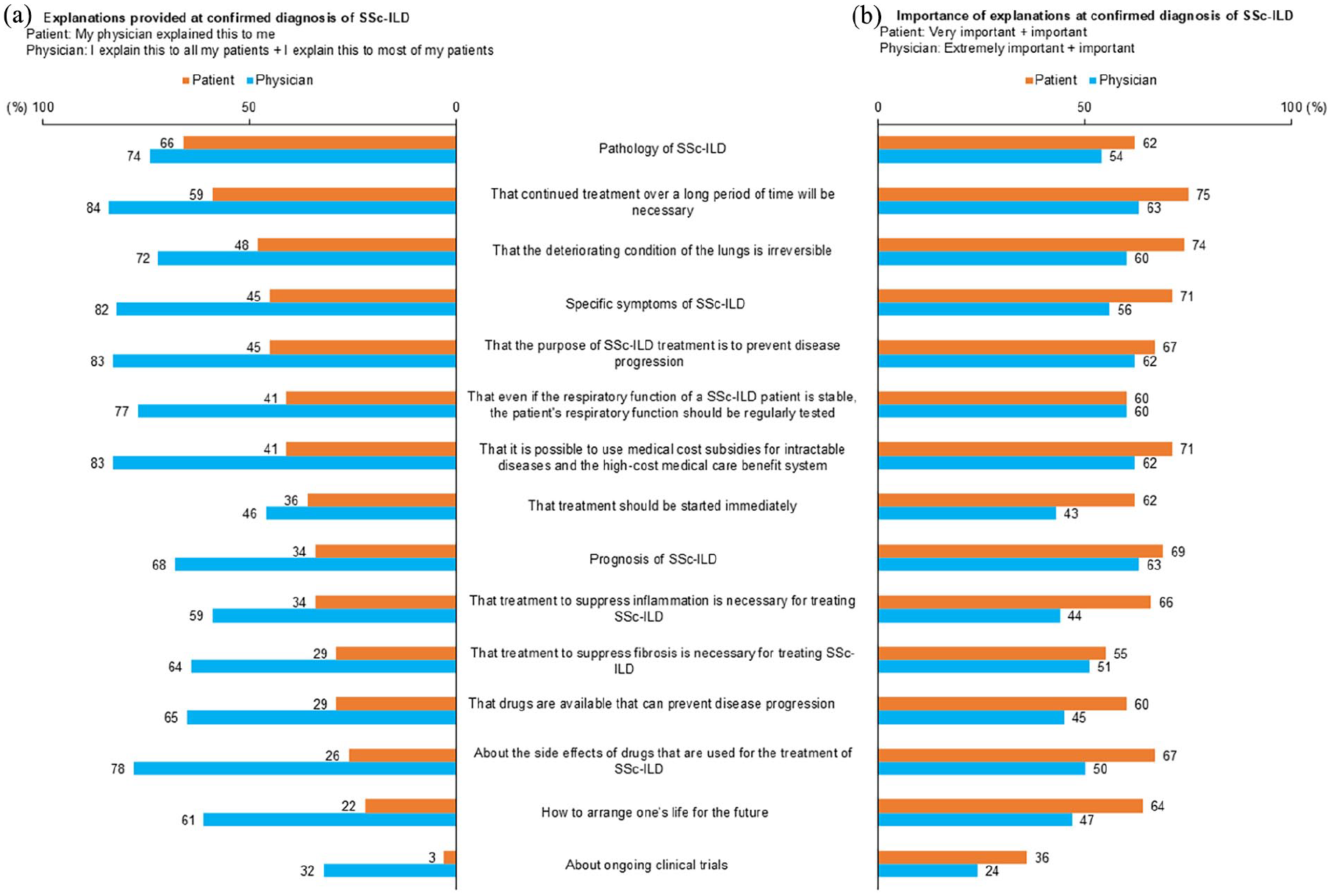
(a) Explanations provided by Japanese rheumatologists to their SSc-ILD patients at the time of confirmed diagnosis of SSc-ILD and (b) their importance. An average of 69% of physicians responded that ‘I explain this to all my patients’ or ‘I explain this to most of my patients’ when the diagnosis of SSc-ILD was made, but only an average of 37% of patients responded that ‘My physician explained this to me’. From a survey conducted in Japan in 2021 involving 121 rheumatologists treating SSc-ILD and 58 SSc-ILD patients; 30 republished with permission.
The survey also found that most patients (78%) felt ‘uneasy’ when SSc-ILD was diagnosed and approximately one quarter felt ‘fear’, ‘nervous’, ‘surprised’ or ‘sad’ (Figure 2(a)). However, by the time treatment was initiated, fewer patients felt ‘uneasy’ (55%) and more felt ‘satisfied’ (31%), ‘positive’ (16%), or had ‘hope’ (10%) (Figure 2(b)). This improvement in patient mind-set between diagnosis and treatment initiation may reflect good physician communication.
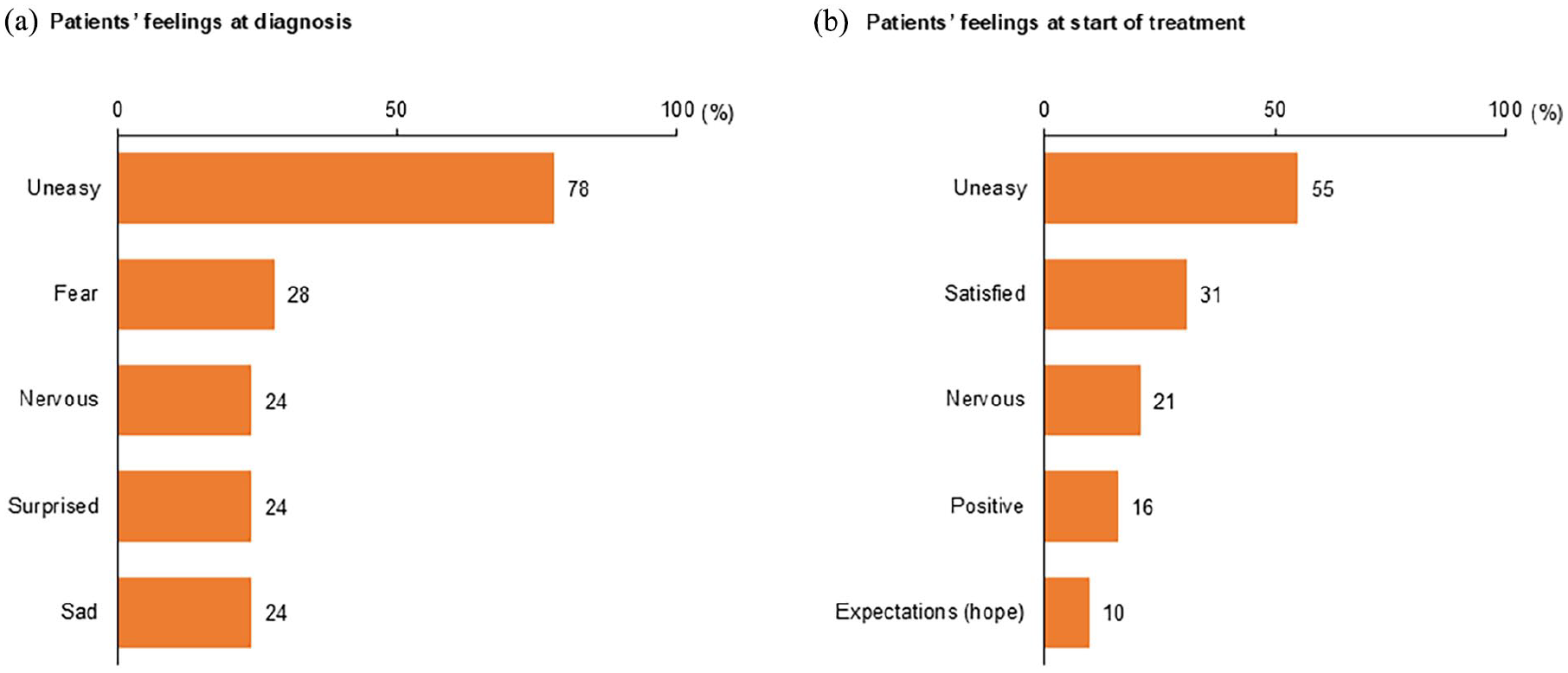
Most common feelings of Japanese SSc-ILD patients at (a) the time of diagnosis and (b) start of treatment. From a survey conducted in Japan in 2021 involving 121 rheumatologists treating SSc-ILD and 58 SSc-ILD patients; 30 republished with permission.
What is noteworthy was satisfaction with SSc-ILD treatment. Although it is difficult to compare the physician and patient responses because the questions were different, 73% of patients answered ‘somewhat satisfied/satisfied/very satisfied’ with SSc-ILD treatment, whereas less than 35% of physicians were satisfied with the various aspects of treatment (Figure 3). Furthermore, many physicians thought there were issues with physician–patient communication, although few patients were aware of them. In particular, most physicians felt that ‘it is difficult to predict the course of disease and future prognosis’ and ‘there is no explanatory material’.
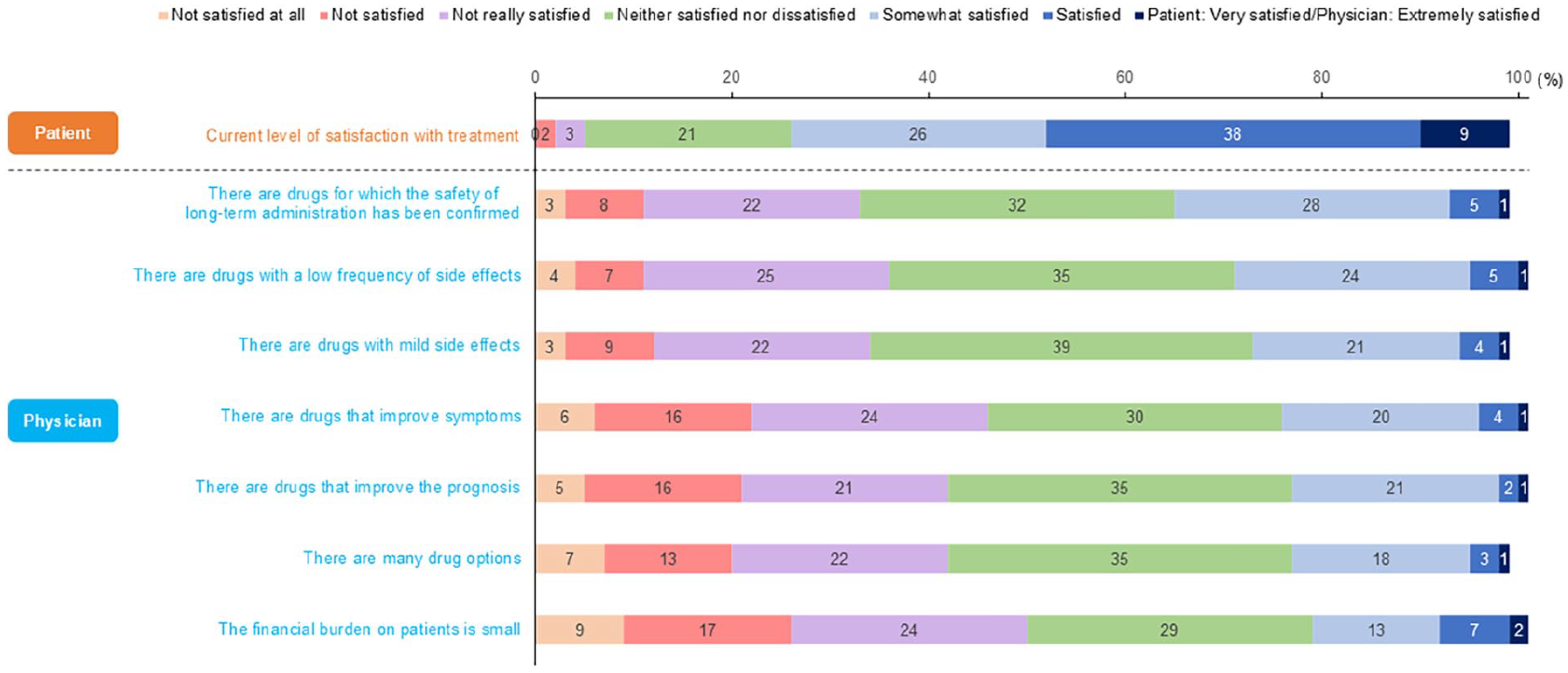
Japanese physician and patient satisfaction with SSc-ILD treatment. Patients were only asked about their overall level of satisfaction, but 73% answered that they were ‘somewhat satisfied’, ‘satisfied’, or ‘very satisfied’. Less than 35% of physicians were ‘somewhat satisfied’, ‘satisfied’, or ‘extremely satisfied’ for all items regarding the current SSc-ILD treatment environment. From a survey conducted in Japan in 2021 involving 121 rheumatologists treating SSc-ILD and 58 SSc-ILD patients; 30 republished with permission.
Based on these findings from Japan, there are differences in perception between physicians and patients about explanations at diagnosis and treatment initiation. Furthermore, most physicians are dissatisfied with current SSc-ILD treatment, and many found it difficult to explain the condition and its prognosis to patients. Therefore, efforts to resolve physician–patient communication gaps are needed.
Physician–patient communication in Europe and the United States
A qualitative study in Germany, Italy, Spain, the United Kingdom and the United States explored communication between SSc-ILD patients and healthcare professionals to identify information and communication needs of patients and whether they are being met. 31 The study involved individual interviews and observed conversations between pairs of physicians, patients and nurses/carers, as well as interviews following simulated conversations between physicians and patients discussing experiences of SSc-ILD. The participants comprised 42 rheumatologists or pulmonologists treating SSc-ILD patients, 21 SSc-ILD patients, 16 specialist nurses, and five carers. The study found that the main unspoken topics that patients wanted to discuss were prognosis and mortality, symptom management and well-being, family issues, daily life and personal issues (Table 1). All these topics are difficult for physicians to predict and answer, but are important for patients to plan their own lives.
Key topics that SSc-ILD patients want to discuss with healthcare professionals that are often omitted from consultations. a
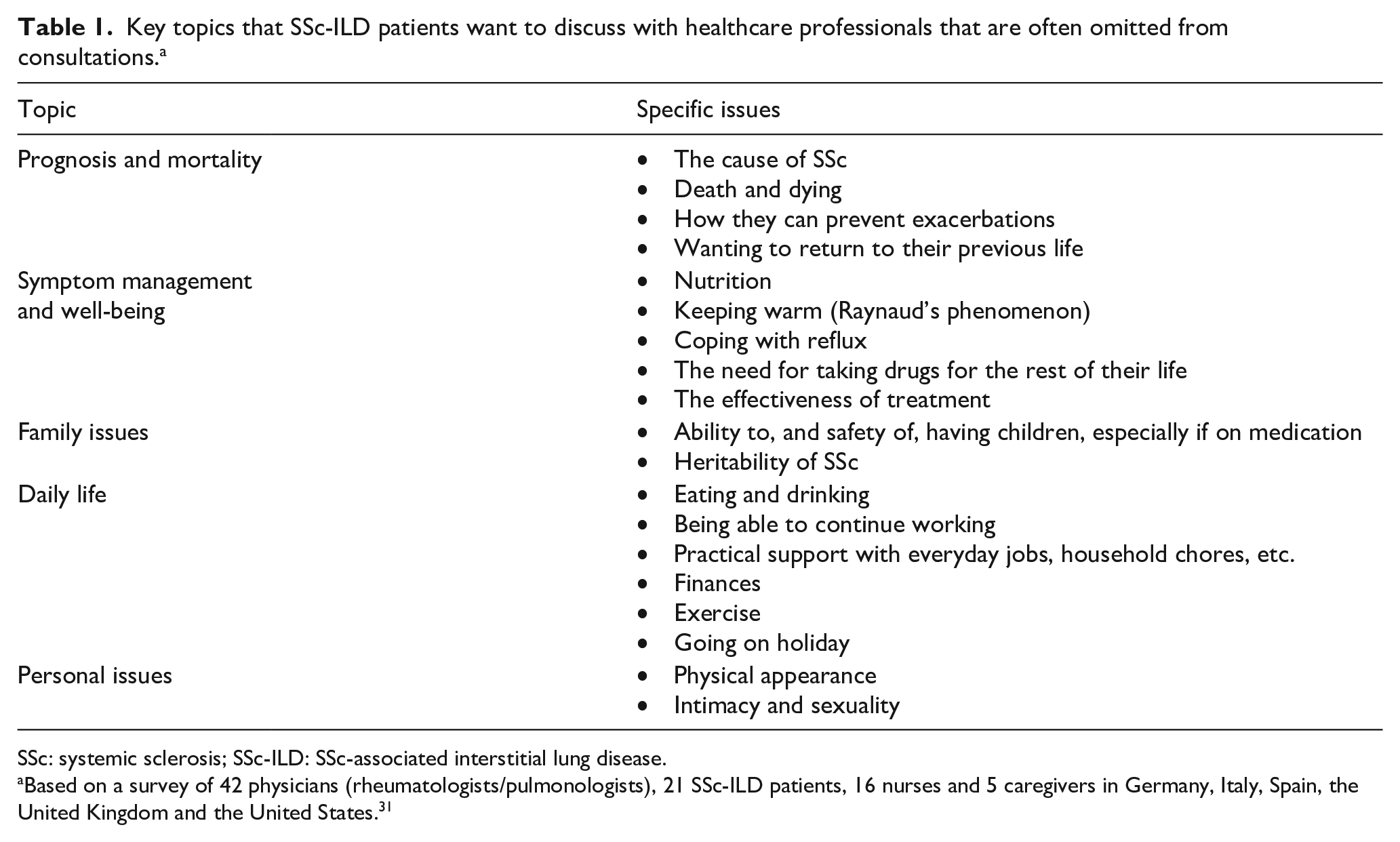
SSc: systemic sclerosis; SSc-ILD: SSc-associated interstitial lung disease.
Based on a survey of 42 physicians (rheumatologists/pulmonologists), 21 SSc-ILD patients, 16 nurses and 5 caregivers in Germany, Italy, Spain, the United Kingdom and the United States. 31
Notably, both patients and physicians tended to avoid discussing prognosis and mortality, the most important topic. Patients were afraid to question their physician about this and unsure how to ask, whereas physicians avoided this topic because it is difficult to give a prognosis, as it depends on the underlying SSc-ILD pathology, and because they were reluctant to cause their patients distress and make them lose hope. Potential cross-country differences were not explored in this study.
As patient advocates, the authors (S.F, I.G) feel that physician–patient communication quality at the first visit is crucial, as it sets the tone for the future relationship (‘first impressions last’). Although there are few direct studies of this issue in SSc-ILD, evidence from elsewhere suggests that physician–patient communication at the first visit is indeed critical for influencing attitudes and behaviours that affect the subsequent relationship, including the establishment of trust and rapport, and may reduce the patient’s psychological distress and enhance their self-care behaviours.32 –34 This phenomenon may partially reflect ‘thin slicing’, i.e. the process of quickly inferring details about an individual or situation based on minimal information.35,36 It would be of interest for future studies to investigate physician–patient communication at the first visit for SSc-ILD, particularly given the importance of patient self-care for this condition. Ideally, at the first visit, the physician can help the patient understand that they are on their side. Furthermore, patients should be encouraged to ask questions, both to address the important topics mentioned above that would otherwise go unspoken and to pre-empt the patient stumbling across misinformation elsewhere. In other words, the physician can guide the conversation but leave space for patient questions. However, the patient needs to understand that the physician cannot always provide definitive answers for a disease as complex as SSc.
As physicians, the authors (M.K., C.P.D., D.K.) find the biggest difficulty in communicating with patients is that SSc-ILD is a disease where it is extremely difficult to give an individual prognosis with high certainty but patients want clear answers from their physicians. It is particularly difficult to advise patients early after diagnosis, and – when asked – we often suggest that we first monitor their condition for a year, unless there are indications to initiate treatment immediately, such as severe or progressing ILD, diffuse cutaneous SSc, or elevated inflammatory markers. We agree that the first patient encounter is indeed very important, and if there is a problem such as the physician unavoidably being late, they can reassure the patient that this will not happen in future.
This study suggests that both physicians and patients need to recognise that progression and severity of SSc-ILD is heterogeneous and thus difficult to predict with certainty, and physicians should help patients understand this. However, physicians can also reassure patients that they will monitor their condition closely and suggest appropriate treatment if it shows signs of worsening.
This study also revealed a ‘white-coat barrier’ between SSc-ILD patients and physicians. Patients feel uncomfortable and embarrassed about raising topics important to them and their caregivers, such as the daily management of symptoms, relationships, family, work, etc., but which seem not directly related to the physician’s consultation (Table 1). For example, most SSc-ILD patients are women, who do not usually talk about topics related to their sex life. Changes in appearance (mainly facial) are a major problem for patients but are rarely discussed. Under these circumstances, specialist nurses – when available – should be the ones to break through the ‘white-coat barrier’ for patients; however, specialist nurses themselves think that communication with patients is difficult because they do not have sufficient knowledge to support patients or answer all their questions. This study concluded that communication by multi-disciplinary teams is necessary to meet patient needs. 31 This finding was echoed in an in-depth Italian study of the patient journey where a major finding was the need for a multi-disciplinary approach for SSc-ILD.37,38 The same study also recommended offering psychological support to patients and caregivers, which we fully support.
Cross-cultural study of physician–patient communication in Japan, Europe and the United States
A study in Germany, Italy, Japan, Spain, the United Kingdom and the United States evaluated challenges in physician–patient communication by observing consultations and analysing communication patterns, needs and levels of understanding using linguistic techniques. 39 Table 2 shows actual statements from physicians and patients during and immediately after a consultation.
Quotes from physicians and SSc-ILD patients during and immediately after consultations. a

SSc-ILD: systemic sclerosis-associated interstitial lung disease.
Based on a study in Germany, Italy, Japan, Spain, the United Kingdom and the United States that observed 23 medical consultations spanning 20 min involving 20 physicians (10 rheumatologists, 8 respiratory specialists, 1 dermatologist, 1 general internist), 19 SSc-ILD patients ex-Japan and 4 actors in Japan. 39
Overall, the study found that consultation styles could be classified into three types. The first type was ‘empathy and rapport building’, a flowing dialogue format where the physician listens attentively to the patient’s story and shows empathy. This type of consultation style builds a rapport (trusting relationship between the physician and patient). The second type was a ‘question-and-answer session’, where physicians followed a screening questionnaire and asked questions sequentially: ‘When did the Raynaud phenomenon begin?’ ‘When did the skin hardening start?’, and so on. As such, the focus was on topics related to clinical symptoms. It was reported that patients were sometimes confused and felt that the physicians were in a hurry because they occasionally asked multiple questions at once. The third type of consultation style was the ‘consultation pattern’, a format where the physician talked unilaterally. Here, the physician started explaining without listening to the patient and used the consultation as an opportunity to educate the patient. In our opinion, no style is necessarily right or wrong: rather, different styles may suit different stages of the patient journey and different individual patients. That said, physician empathy and building rapport with the patient do generally seem to be crucial aspects of physician–patient communication. All three consultation styles were observed across all countries except Japan where the style was exclusively a question-and-answer session.
This study showed that the information patients share and how they feel supported by their physician will vary depending on how the consultation is performed. Patients often felt they were given few opportunities to explain their concerns or ask questions during the consultation. One overarching message physicians should take is that patients are there to tell a story, and physicians should try to sit back and listen carefully to them.
This study also found that patients often felt that they are required to know enough to understand what is being said when physicians use a lot of technical terms (Table 2). Even when physicians initially used plain language in explanations, they often reverted to technical terms when explaining complex issues. However, metaphors helped the patients understand the issues more clearly. For example, one physician described the lungs of an SSc-ILD patient as ‘darker alleys, with constriction and small spaces, and instead of a balloon that you can push on, there are just more rigid walls, with some very narrow areas’. It was also reported that unless the physician showed empathy, patients tended to use functional, factual language. As described above, this may be because patients feel uneasy or fearful. 30 However, in the United States, patients do tend to express their feelings more emotionally. It is important to note that health literacy of patients may be a barrier. For example, a recent study in Italy found that 63% of SSc patients had inadequate or problematic overall health literacy skills. 40
Another informative finding from this study is the differences in understanding of SSc-ILD between physicians and patients (Figure 4). Whereas the physicians had cognitive models of SSc-ILD that reflect specialised medical knowledge and experience, patients’ cognitive models of its causes and symptoms were based on personal knowledge, beliefs and experience – with correct parts and other parts based on misconceptions. Since patients process information according to their own cognitive models, this will potentially affect their disease understanding and communication with physicians. However, if physicians realise that patients have different cognitive models, they may be able to correct misunderstandings and more easily comprehend patient anxieties, which in turn may improve the physician–patient communication. Issues from the physicians’ perspective included the fact that the cause of SSc has not been fully elucidated – as shown in their cognitive model (Figure 4) – and that it is difficult to give a prognosis. In addition, the physicians were frustrated by the limited treatment options for SSc-ILD and that they did not think there is a treatment they can feel confident about. These treatment-related issues may also hinder communication with patients.
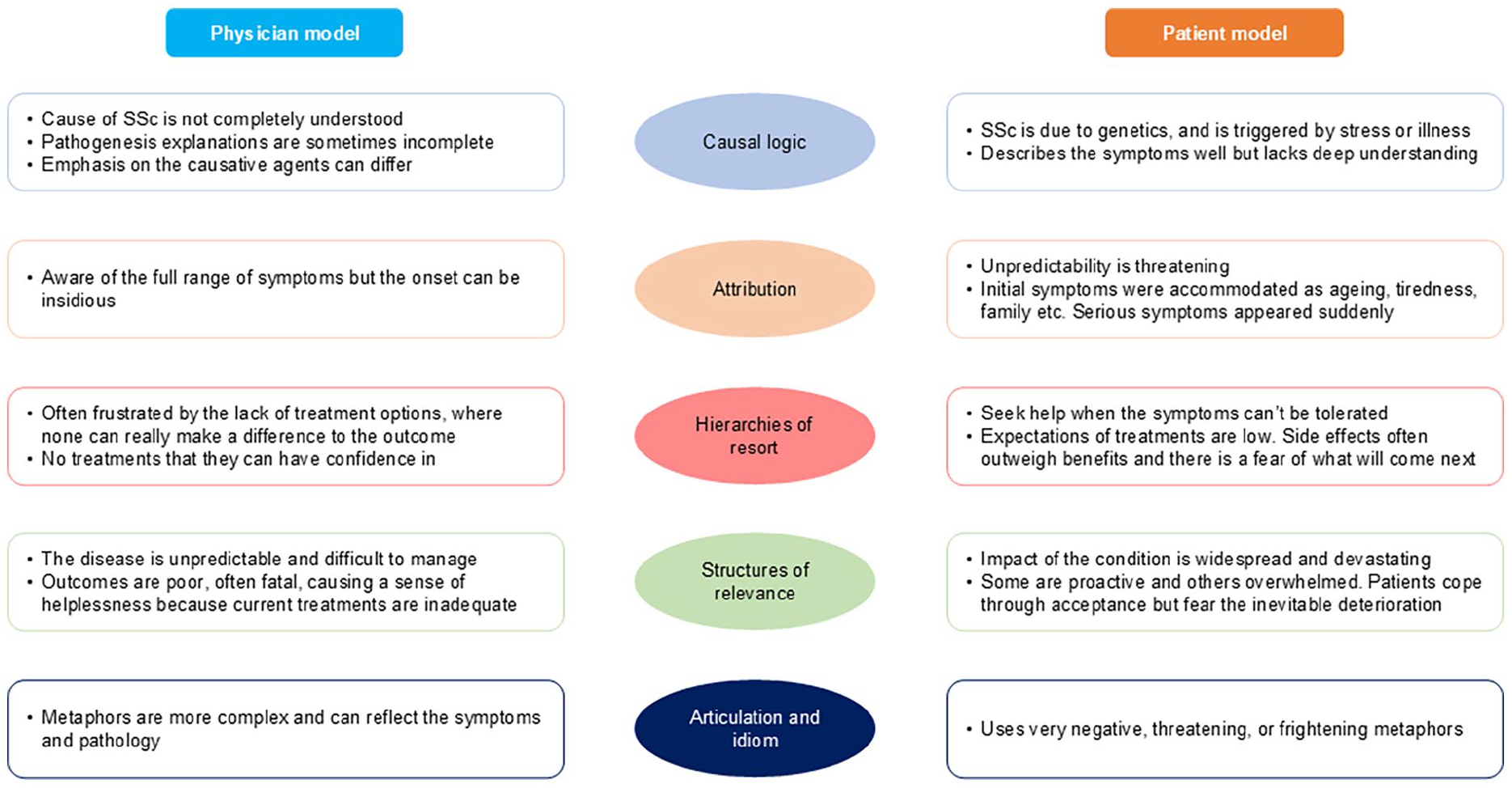
Physician and patient explanatory models in SSc-ILD. Models are derived from a study of physician–patient communication in Germany, Italy, Japan, Spain, the United Kingdom and the United States. 39 The study involved 23 medical consultations spanning 20 min involving 20 physicians (10 rheumatologists, 8 respiratory specialists, 1 dermatologist, 1 general internist) and 23 SSc-ILD patients (19 patients ex-Japan, 4 actors in Japan). Republished with permission.
Key messages and recommendations
Some key messages and recommendations from the above studies of physician–patient communication in SSc-ILD can be summarised as follows:
Most patients feel uneasy at the diagnosis of SSc-ILD.
The quality of physician–patient communication at the first visit is crucial.
Both physicians and patients avoid mentioning prognosis and mortality, so patients should be encouraged to ask questions, in order to address these and other important topics that would otherwise go unspoken.
The white-coat barrier interferes with communication.
Although different physician communication styles may suit different stages of the patient journey, listening and empathy are key for physicians to build rapport with patients.
Physicians should avoid using a lot of technical terms.
Patients have different perceptions (cognitive models) of SSc-ILD than physicians.
Similarities and differences in healthcare systems and physician–patient communication in Japan, the United States and Europe: physician and patient perspectives
Similarities and differences between Japan, the United States and European countries including the United Kingdom in SSc-ILD treatment and physician–patient communication are important to consider. In this section, we illustrate these potential differences by discussing our experiences at three clinical centres in these countries (Table 3), albeit these do not necessarily reflect the situation at all centres in each country, and – equally importantly – our perspectives as patients/patient advocates.
Comparison of healthcare systems and physician–patient communication in SSc-ILD at three clinical centres in the United States, Japan, and the United Kingdom. a

ILD: interstitial lung disease; SSc: systemic sclerosis.
Based on the experience of the authors (M.K., C.P.D., D.K.).
Physician perspectives
First, the healthcare systems in these three countries are very different; therefore, there are differences in clinical practice. For example, the first author based in Japan (M.K.) examines 40–50 patients daily, with consultations lasting approximately 5 min each. Ideally, there would be a 1-h consultation the first time a patient is seen; however, it normally lasts only approximately 15 min. Furthermore, patients with other rheumatic diseases are also seen. In contrast, the author from the United States (D.K.) sees 20–25 patients daily with consultations lasting approximately 1 h for the first visit and 30 min for follow-up, which typically occurs every 3 months. Furthermore, only SSc patients are seen. Finally, the UK author (C.P.D.) typically sees 6–8 SSc patients daily in the outpatient clinic and 10–12 on the day ward, with a consultation of approximately 45 min for a new patient in the clinic and 20 min at follow-up. Although consultations are shorter in Japan, the patient visits the physician more often, generally once a month or every 2 weeks. This frequent follow-up facilitates communication – for example, the physician can explain the main points in the first visit, the patient can then go away and prepare questions, which the physician can answer at follow-up. This pattern can be repeated at subsequent visits.
There are also differences between Japan, the United States, and the United Kingdom in how patients respond. In the United States, patients may actively ask questions and express their feelings openly; most say they are so shocked at the diagnosis of SSc that their mind goes blank. From the physician’s perspective of that situation, it seems that nothing else said to the patient at that point really registers with them. Also, patients often cry in front of the physician and ask for a hug. In contrast, although patients in Japan are also usually shocked, they try to be calm and almost never cry in front of the physician. In the United Kingdom, most patients are anxious and frightened at the first consultation but have a more questioning approach at follow-up.
SSc-ILD–associated expenses, including treatment costs, may also be something that patients wish to discuss with their physicians in countries without universal health coverage.
As described above, the study of physician–patient communication in Europe and the United States 31 and the Italian study of patient perspectives37,38 both concluded that multi-disciplinary team support may be needed to meet patient communication needs. However, there are no SSc or ILD specialist nurses in Japan. In the United States, although there are no specialist nurses at the author’s facility (D.K.), there are coordinators who provide emotional support to patients, answer questions, give guidance and are generally involved in everything that cannot be done at the physician’s appointment alone. In the United Kingdom author’s facility (C.P.D.), specialist nurses are available who see all new patients for 10–20 min to address questions and provide disease information; the nurses also provide email/telephone contact details for communication between clinic visits. Ideally, nurses specialising in SSc and ILD will become involved in all patient-support systems in the future. Although there would obviously be associated costs, the oncology field can provide a model as it is common for nurses and pharmacists, for example, to be involved in multi-disciplinary team care of cancer patients.
Patient perspectives
In the experience of the patient/patient advocate authors (S.F., I.G.), there are several important issues in physician–patient communication. A consultation with an SSc patient may require more physician time than for patients with other rheumatic or pulmonary diseases, 41 and the patient needs to be confident in their interaction with the physician, which is reliant on the latter’s interpersonal skills. 42 We feel strongly that the first visit is a pivotal point for creating the trust and rapport necessary for a productive physician–patient relationship, but this opportunity is often missed. Ideally, the physician would effectively communicate that they are on the patient’s side. Furthermore, in agreement with the findings above, we have also found that topics that patients want to discuss are often not broached, such as changes in facial appearance and issues relating to sex life. Often patients are not encouraged to ask questions – for example, ‘do you understand what I said’ – but it would be better to do so than have the patient subsequently consult ‘Dr Google’ instead. 43 With regard to communication style, we have also found that some physicians may simply talk at patients rather than guide an interactive conversation, and we reinforce the importance of physician empathy during physician–patient communication. Conversely, patients often do not fully appreciate that physicians are not omniscient, particularly for a rare and complex disease like SSc-ILD where fundamental aspects of pathophysiology have yet to be elucidated, and thus may be unable to satisfactorily answer all the patient’s questions. A patient-advocate perspective in the United States identified education as key to empowering patients, 44 which would be expected to impact physician–patient communication. Overall, we believe there is much opportunity to better engage patients during consultations, which a wealth of evidence shows is associated with improved patient outcomes.
Efforts to bridge the communication gap between physicians and patients
Clearly, work is needed to close the physician–patient communication gap in SSc-ILD. Notable recent initiatives include peer-mentoring (patients talking to other patients) and patient self-education programmes, as well as increased emphasis on shared decision-making and social media.
Peer mentoring
The University of Michigan has a peer-mentoring system called the ‘Scleroderma Peer Mentor Program’. Peer mentors are SSc patients who serve as mentors (counsellors) for newly diagnosed patients. As described earlier, many patients who have just been diagnosed with SSc are in a state of great shock and confusion. Although the physician can answer some questions from patients, they do not have the lived experience of SSc to share with them. However, peer mentors were similarly shocked and confused at diagnosis, and therefore understand what it is like for new patients and can support them. So, if a new patient wants to talk to someone, they can be referred to a peer mentor. Generally, optimistic and positive people are invited to be peer mentors, and those who accept the invitation receive training. These individuals are volunteers who do not receive any financial support.
Patient self-learning and patient organisations
Patient self-learning can be facilitated by websites, pamphlets and other patient-education materials. The University of Michigan – in collaboration with the University of New Mexico, the National Scleroderma Foundation (NSF), the Scleroderma Research Foundation and the Patient-Centered Outcomes Research Institute – has developed a web-based programme called ‘Self-Manage Scleroderma’, which patients are encouraged to use. The programme is structured into chapters by topic (Table 4) and enables learning about SSc by watching videos.
‘Self-Manage Scleroderma’ patient-education programme from the University of Michigan. a

Although approximately 1000 patients attend the NSF patient-association meeting in the United States every year, many hesitate to attend, often because it is very difficult for newly diagnosed patients who are doing well to confront their long-term prognosis, as well as travel barriers such as distance. However, while hope is necessary for patients, education is also essential. The Self-Manage Scleroderma programme is therefore designed to provide information to help patients self-manage SSc and overcome negative emotions.
Patient education is also important to combat misinformation about SSc, given the existence of incorrect information on the Internet. Organisations such as the American Society of Rheumatology and the NSF play an important role here as their websites are frequently visited in the United States, probably because they are recognised as providing accurate disease information.
Indeed, patient organisations already have a wealth of educational and practical aids that physicians can direct patients to, without having to ‘reinvent the wheel’ themselves. These aids include practical tools such as consultation planners to help formulate key questions before consultations to maximise the quality of the physician–patient conversation.
As mentioned, inadequate health literacy of patients with SSc may be a barrier to effective physician–patient communction; 40 however, this may also be an area where patient organisations can support physicians.
Shared decision-making
Patient education is also essential for shared decision-making. As lung fibrosis in SSc is irreversible, the expert consensus in Japan, Europe and the United States is that newly diagnosed SSc patients should be screened for ILD using chest high-resolution computed tomography (HRCT).4,17,18,45 However, shared decision-making here requires patients to understand there is a net benefit to screening with chest HRCT for early detection of SSc-ILD, despite the risk of radiation exposure. Shared decision-making is a core principle for other rheumatic diseases, including rheumatoid arthritis,46,47 but this is not yet the case for SSc.
Physicians can play a role in facilitating shared decision-making – for example, the physician could describe some drug options for SSc-ILD to the patient, but tell them to take their time to consider them before indicating their preference.
Disease-awareness campaigns with patient-oriented websites and print materials are also important for educating SSc-ILD patients and enhancing shared decision-making. Although pharmaceutical companies are already playing a role here, they could expand their efforts. Nevertheless, disease-awareness campaigns provide general information not individualised guidance, and patients should be encouraged to ask their physician questions through face-to-face or electronic communication.
Information technology and social media
The use of information technology also could enhance physician–patient communication. For example, visits can be conducted virtually when a physical meeting would be challenging, and important information can be disseminated to patients using email functionality in electronic medical record systems, where available. However, it is vital that platforms used for sharing sensitive data are secure, and that virtual consultations can be reimbursed.
Social media already play a role in patient education and thus influence shared decision-making, and this trend is likely to increase. In the United States, physicians have been encouraged to use Twitter (now called ‘X’) to promote their research, and many patients also use this platform. For example, one of the authors (D.K.) has a patient who underwent haematopoietic stem cell transplantation and has used Twitter to communicate its advantages and disadvantages to other patients globally who are considering this treatment option. Thus, patients sharing their experience can be beneficial for other patients, provided it is done in the right context alongside wider information. However, social media can be a double-edged sword, with misinformation, disinformation and fake news rife, so its potential disadvantages also should be considered.
Conclusion
In medicine generally, the potential influence of physician–patient communication on treatment adherence, patient satisfaction and clinical outcomes is well established, particularly for chronic diseases.23 –25 More specifically, a systematic review of studies in the United States, Japan and Europe evaluating physician–patient communication in rheumatology found evidence that it affects patient quality of life, medication adherence and disease activity. 48 Overall, positive patient participation in the consultation and high level of trust in the physician were associated with treatment satisfaction, fewer side effects, lower disease activity and less organ damage.
As we have discussed, recent studies in Japan, Europe and the United States30,31,39 suggest there is much room to improve physician–patient communication in SSc-ILD management. In Japan, at least, the gaps in physician–patient communication may partly explain the low usage of guideline-recommended treatments by patients with SSc-ILD. 27 Further studies exploring potential cross-country differences may be informative.
Patient education may be one of the best ways to enhance physician–patient communication, using various methods such as disease-awareness campaigns (including websites and printed materials) and peer-mentoring systems. Although many initiatives already exist, there is scope for healthcare organisations, patient organisations and pharmaceutical companies to increase such efforts. Notably, differences in culture and healthcare systems between countries lead to some contrasts in how physicians and patients communicate. However, it may be that SSc-ILD patients in most countries experience similar feelings but simply express them differently, due to different cultural environments. Therefore, although not all patient-education activities used in one country will necessarily be appropriate in others, patient-centred activities such as peer-mentoring systems may be generally successful.
From the patient’s perspective, the quality of the first consultation is a key influence on physician–patient communication (and thus patient outcomes), particularly the physician’s empathy, active listening, and allowance of space for the patient’s questions and answers.
Finally, efforts to enhance physician–patient communication in SSc-ILD may be informed by similar initiatives in other fields of medicine, particularly oncology.
Footnotes
Acknowledgements
Medical writing assistance was provided by Giles Brooke, PhD, CMPP, of Envision Pharma Group under the authors’ conceptual direction and based on feedback from the authors, and was contracted and compensated by Nippon Boehringer Ingelheim Co. Ltd. Nippon Boehringer Ingelheim was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations.
Author contributions
The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE) and were fully responsible for all content and editorial decisions, and were involved at all stages of manuscript development.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: The authors received no direct compensation related to the development of the manuscript. M.K. has received consulting fees, speaking fees and research grants from argenx, Asahi Kasei, AstraZeneca, Boehringer Ingelheim, Chugai, GlaxoSmithKline, Janssen, Kissei, MBL, Mitsubishi Tanabe, Mochida, Nippon Shinyaku, Ono Pharmaceuticals and Taisho. A.S. is an employee of Nippon Boehringer Ingelheim Co. Ltd. S.F. and I.G. have nothing to disclose. C.P.D. reports consulting fees from Roche, Boehringer Ingelheim, GlaxoSmithKline, Horizon Therapeutics, CSL Behring; honoraria from Boehringer Ingelheim and Janssen; and grants from Horizon Therapeutics, GlaxoSmithKline and Servier. D.K. reports consultancies with Amgen, argenx, Astra Zeneca BMS, Boehringer Ingelheim, Genentech/Roche, GSK, Mitsubishi Tanabi, Merck, Novartis and Zura Bio.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: this review is based on roundtable discussions between the authors in Tokyo, Japan (3 December 2022) and at the 8th Systemic Sclerosis World Congress in Prague, Czech Republic (14–16 March 2024), sponsored by Nippon Boehringer Ingelheim Co. Ltd and Boehringer Ingelheim, respectively.
The statement
The Editor/ Editorial Board Member of JSRD is an author of this paper, therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Board member had no involvement in the decision-making process.
