Abstract
Objectives:
To evaluate (1) the association between nailfold capillaroscopy pattern and 5-year risk for incident interstitial lung disease and (2) the association between transition in nailfold capillaroscopy pattern and risk of incident interstitial lung disease.
Methods:
Data of adult patients from the EUSTAR database fulfilling the ACR-EULAR criteria with a disease duration ⩽5 years, having a scleroderma pattern at nailfold capillaroscopy with high-resolution computed tomography confirmed absence of interstitial lung disease (i.e. baseline) was used. Interstitial lung disease-free survival was assessed for up to 5 years of follow-up with a Cox proportional hazards model stratified on nailfold capillaroscopy pattern at baseline. The association of annual transition in nailfold capillaroscopy pattern on the risk to develop interstitial lung disease was assessed with a mixed logistic regression analysis.
Results:
Out of 771 eligible patients, 283 (37%) had an early pattern, 377 (49%) had an active pattern, and 111 (14%) had a late pattern. The Cox proportional hazard model including the identified confounders did not show an association between severity of nailfold capillaroscopy pattern at baseline and increased risk for interstitial lung disease during 5-year follow-up (hazard ratio (95 confidence interval; p value): 1.09 (0.86–1.39; 0.47)). The mixed logistic regression analysis revealed an increased annual risk for incident interstitial lung disease with increasing severity of capillaroscopic pattern (odds ratio (95% confidence interval); p value 3.76 (1.99–7.11; <0.01)).
Conclusion:
Our study shows that worsening of nailfold capillaroscopy has a strong association with an increased annual risk to develop interstitial lung disease. Of note, a worse scleroderma pattern at nailfold capillaroscopy is not associated with the long-term risk to develop interstitial lung disease.
Introduction
Systemic sclerosis (SSc) is characterized by a triad of inflammation, vascular damage and fibrosis. 1 It is most detrimental for patients when this pathophysiological interplay takes place in the lungs, given that interstitial lung disease (ILD) accounts for the highest disease-related mortality. 2
Since ILD mainly occurs in the first 5 years after disease onset, all SSc patients are closely monitored in the first years of the disease.3–5 An important unmet clinical need is the feasibility to early identify patients at risk to develop ILD, in order to implement personalized monitoring strategies. Although no validated predictive algorithm is yet available in patient care, nailfold capillaroscopy is emerging as a promising candidate biomarker to predict the onset of ILD. 6 In patients with SSc, typical deviations can be observed during nailfold capillaroscopy, including enlarged capillary loops, loss of capillaries and haemorrhages. In addition to this quantitative assessment, the nailfold capillaroscopy findings can also be classified as a scleroderma pattern, which is categorized as early, active and late patterns.7,8 These patterns, early, active and late, indicate progression of vasculopathy, with the most severe vasculopathy observed in the late pattern. 7
Cross-sectional studies indicate an association between loss of capillaries and ILD.9–11 With respect to the presence of giant capillaries, abnormal capillary morphology or microhaemorrhages, no significant association with ILD has been found.10–12 In contrast, for the presence of avascular areas and neoangiogenesis, that is, late pattern morphologies, several studies have reported a significant association with ILD.10–13 Several longitudinal studies have been initiated to obtain insight into sequential changes of nailfold capillaroscopy patterns and pulmonary pathology. In a longitudinal study with 12 years of follow-up data of 34 patients with an early scleroderma pattern, it was found that evolution of an early pattern towards a more severe pattern is associated with an increased risk of ILD. 8 This study, however, does not provide in-depth insight on the risk to develop ILD based on the scleroderma pattern, since at baseline all patients had an early pattern. In another study based on EUSTAR data of 334 patients with SSc, a more severe nailfold capillaroscopy pattern was significantly associated with a twofold increased risk to develop severe and/or progressive organ involvement. 14 Remarkably, no association between nailfold capillaroscopy pattern and the risk to develop ILD or progressive ILD was found. In this study, almost half of the study population was already diagnosed with ILD at baseline. This might explain the low incidence of ILD (5.3%) during follow-up, potentially obscuring any longitudinal association between nailfold capillaroscopy and incident ILD. Therefore, in this study, we intended to investigate this association in a cohort at risk to develop ILD, that is, patients without ILD and a short disease duration. The aim of this study was twofold: (1) to evaluate the longitudinal association between nailfold capillaroscopy and incident ILD during 5 years of follow-up and (2) to evaluate the association between transitions in nailfold capillaroscopy pattern and incident ILD during 5 years of follow-up.
Methods
Patients and study design
We performed a post hoc analysis of prospectively collected data in the EUSTAR database (Eustar CP project 145). A detailed description of the structure of the database, the collected data set and definitions of clinical variables have been published previously.2,15 In compliance with local regulations, the pre-existing database was approved by the Ethical Committees at each centre and patients provided written informed consent. According to Dutch law and regulations, this observational, non-interventional study was exempt from the requirement of approval by a medical ethics committee. Data were extracted on 1 April 2023, and all data registered up to the timepoint of extraction were used in this study.
Patients entered the study at the first registered ILD-free visit with a documented scleroderma pattern at nailfold capillaroscopy (i.e. baseline) and were followed for a period of up to 5 years. Patients were classified as ILD-free during a visit if they had an high-resolution computed tomography (HRCT) scan at or after that visit without any signs of ILD, or if an HRCT scan-confirmed ILD diagnosis occurred a year or later after that visit. After cleaning and structuring the data (for details see Supplementary Table 1), the final cohort included patients meeting the following criteria: age ⩾18 years, fulfilling ACR-EULAR criteria, 16 disease duration from first non-Raynaud phenomenon ⩽5 years, having a scleroderma pattern (i.e. early, active, late) at nailfold capillaroscopy, 7 HRCT confirmed absence of ILD at baseline, available forced vital capacity (FVC) at baseline, documented start date of immunosuppressive treatment, no previous autologous hematopoietic stem cell transplantation and follow-up duration ⩾1 year.
Baseline demographic and disease characteristics data were derived from visits with a maximum difference of 1 year from baseline. We used data on demographic and disease characteristics including cutaneous subsets of SSc according to LeRoy criteria, 17 antibody status, fulfilment of ACR-EULAR criteria, 16 pulmonary function tests, New York Heart Association (NYHA) functional class, organ involvement, C-reactive protein (CRP) status (elevated vs normal) and treatment status. With respect to treatment status, two binary variables were created, namely immunosuppression usage (yes/no) and vasodilatory treatment (yes/no). Immunosuppressive/antifibrotic agents considered for this analysis were cyclophosphamide, mycophenolate mofetil, methotrexate, rituximab, tocilizumab, nintedanib, azathioprine, imatinib, leflunomide, pirfenidone, riociguat, TNF-alfa-antagonist, and abatacept, whereas vasodilatory agents considered were amlodipine, bosentan, dihydropyridine, diltiazem, felodipine, nicardipine, nifedipine, other calcium channel blockers, sildenafil, vardenafil, and iloprost.
As patients with an unfavourable prognostic phenotype are more likely to develop ILD, we defined a priori disease predictors through literature search and identified the following confounders based on authors’ consensus: that is, gender, diffuse cutaneous subtype, Caucasian ethnicity, presence of anti-topoisomerase I antibodies, presence of anti-RNA polymerase III antibodies, higher age, lower FVC % predicted and lower DLCO % predicted. 18
Scoring of nailfold capillaroscopy images
The nailfold capillaroscopy images were not reassessed centrally in this study. As we relied on the overall capillaroscopic pattern, which is easily identifiable, we used the capillaroscopic pattern entered in the database for the current analysis. Moreover, EUSTAR and EULAR regularly arrange courses to coach, update and standardize the assessment of SSc patients, including coaching sessions for capillaroscopy, to guarantee the quality and standardization of the clinical and capillaroscopic assessments.
Patient and public involvement
Patients were not involved with this analysis.
Statistical analysis
Baseline demographic and clinical characteristics were compared with one-way analysis of variance (ANOVA), Kruskal–Wallis test or Pearson’s chi-square test as appropriate between the three nailfold capillaroscopic patterns. For multivariate analyses, missing baseline values were imputed using chained imputations and predictive mean matching. Based on the maximum fraction of missing data (FMI) and coefficient of variation (CV (SE)), we calculated that 54 imputations were needed. All statistical tests were performed two-sided (α = 0.05). The analyses were carried out using Stata statistical software version 18. For the following analysis, scleroderma pattern at nailfold capillaroscopy was encoded as an ordinal variable (i.e. 1 = early, 2 = active, 3 = late) assuming an exponential relationship exists within nailfold capillaroscopy patterns from early to late.19,20
Primary outcome: ILD diagnosis was defined as an HRCT scan showing the presence of ILD, honeycombing, lung fibrosis, ground glass opacification or reticular changes. Patients with a visit-free interval of ⩾2 years were censored on their last visit date.
Association between nailfold capillaroscopic pattern at baseline and the time to ILD during 5-year follow-up
First, we assessed ILD-free survival stratified by the scleroderma pattern at baseline using unadjusted Kaplan–Meier analysis on complete cases. Then, we performed a Cox proportional hazard analysis to correct for confounding by indication on imputed data. The base Cox proportional hazard model was defined using the a priori selected set of confounders. The other baseline characteristics were assessed for potential confounding using a ‘change-in-estimate’ (CIE) approach using a cut-off point of 10%.21,22
Association between transition in nailfold capillaroscopic pattern during follow-up and risk to develop ILD
For this analysis, distinct observation periods with consecutive visits occurring with an interval of maximum 1 year ± 3 months were established for each patient. Specifically for this analysis, only data of patients with minimally one registered nailfold capillaroscopy during follow-up were used. In instances where multiple observation periods met the inclusion criteria per patient, the final observation period was systematically selected based on the highest number of transitions occurring in nailfold capillaroscopy pattern and follow-up period. Given the relatively low number of transitions in the scleroderma pattern, the number of dynamics in nailfold capillaroscopy pattern was prioritized over the follow-up period during the selection procedure. Using multilevel mixed-effects logistic regression, the association between incident ILD and transition of scleroderma pattern was assessed for each year up to 5 years of follow-up. The final multilevel mixed effects logistic regression consisted of the predefined confounders and confounders identified through the ‘CIE’ approach using a cut-off point of 10%.
Results
Patient characteristics
We identified 771 patients fulfilling the inclusion criteria (Figure 1). Demographic and clinical characteristics of all eligible patients are presented in Table 1 (see online Supplementary Table 2 for details about missing data rate). The median (interquartile range (IQR)) age of the overall study population was 52.3 (41.6–62.1) years, with a male:female ratio of 1:4. The median (IQR) follow-up duration was 3.4 (2.1–4.4) years and not different between patients with an early, active or late scleroderma pattern at nailfold capillaroscopy. In total, 25.1% of the patients were diagnosed with a diffuse cutaneous subtype, median (IQR) disease duration was 1.8 (0.9–3.2) years and median (IQR) modified Rodnan skin score (mRSS) at baseline was 5.0 (2.0–11.0) units in the whole population. Regarding the capillaroscopy, 283 (37%) of the patients had an early pattern, 377 (49%) had an active pattern and 111 (14%) had a late pattern at baseline. Patients with a more severe scleroderma pattern (e.g. active or late) had more frequently prognostic unfavourable characteristics, including male gender, diffuse cutaneous subtype, lower FVC% predicted, pulmonary hypertension, higher mRSS, digital ulcers, renal crisis and immunosuppressive treatment. There were no differences between the groups for the prevalence of gangrene or upper gastro-intestinal symptoms.
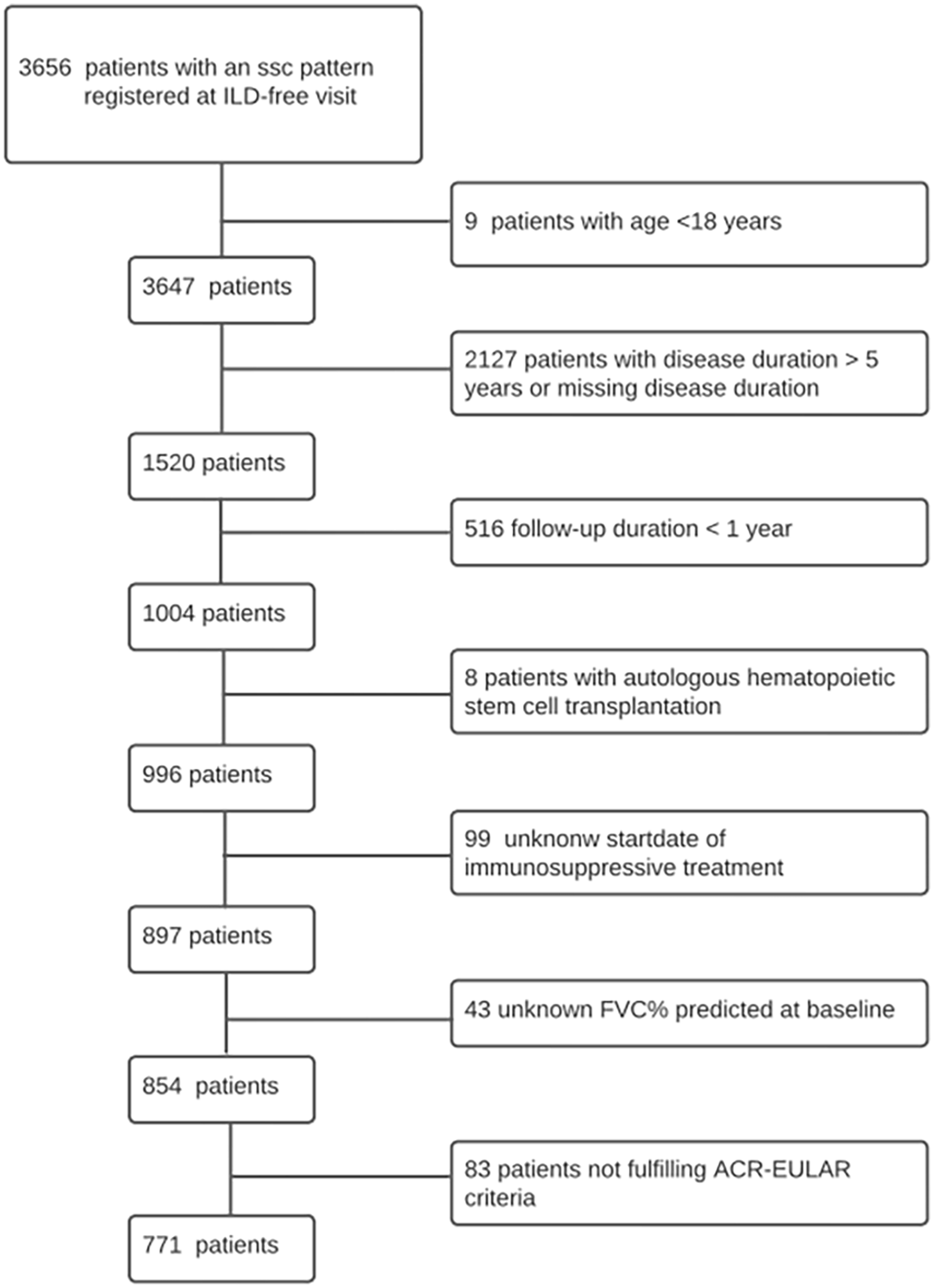
Flowchart of patient selection in EUSTAR database.
Overall baseline demographic and clinical characteristics of all SSc patients and stratified by nailfold capillaroscopy pattern at baseline.
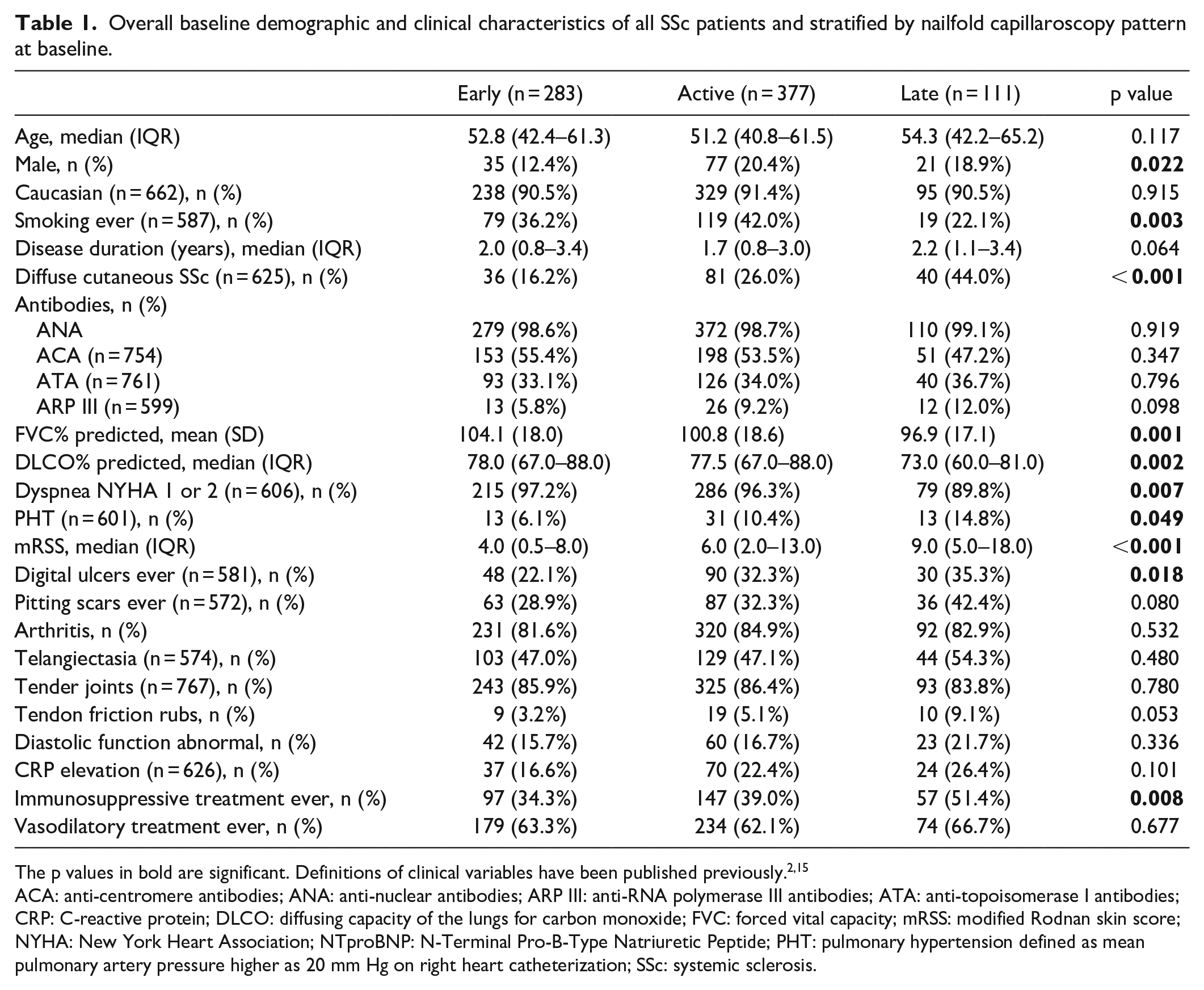
The p values in bold are significant. Definitions of clinical variables have been published previously.2,15
ACA: anti-centromere antibodies; ANA: anti-nuclear antibodies; ARP III: anti-RNA polymerase III antibodies; ATA: anti-topoisomerase I antibodies; CRP: C-reactive protein; DLCO: diffusing capacity of the lungs for carbon monoxide; FVC: forced vital capacity; mRSS: modified Rodnan skin score; NYHA: New York Heart Association; NTproBNP: N-Terminal Pro-B-Type Natriuretic Peptide; PHT: pulmonary hypertension defined as mean pulmonary artery pressure higher as 20 mm Hg on right heart catheterization; SSc: systemic sclerosis.
ILD-free survival based on nailfold capillaroscopic pattern
In our cohort, 152 (19.7%) of the patients developed ILD after a median (IQR) follow-up period of 1.7 (1.1–2.9) years. The unadjusted Kaplan–Meier analysis showed that the ILD-free survival was distinctive in patients with a late scleroderma pattern compared to patients with an early scleroderma pattern (Figure 2). The mean (95% confidence interval (CI)) incidence of ILD in the early, active and late group was 17.0% (12.3%–21.9), 19.9% (16.0–24.3) and 26.1% (18.2–35.3), respectively.
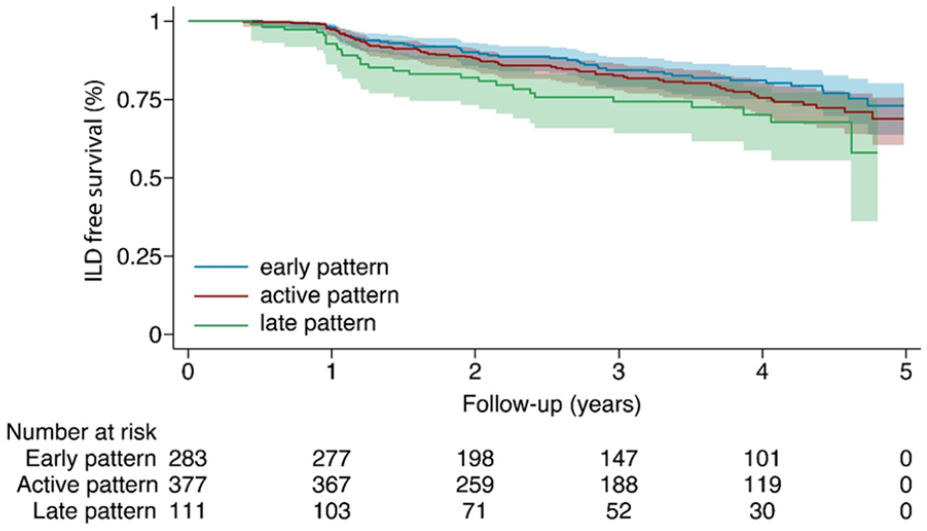
Kaplan–Meier ILD-free survival curve stratified by nailfold capillaroscopy pattern at baseline.
The univariate Cox proportional hazards analysis demonstrated an increasing risk to develop ILD with worsening severity of the nailfold capillaroscopic pattern (hazard ratio (HR) (95 CI; p value): 1.32 (1.04–1.66; 0.02)). For the final Cox proportional hazard model, no additional confounders were identified through CIE approach (see Supplementary Table 3). In the final Cox proportional hazard model, corrected for gender, diffuse cutaneous subtype, Caucasian ethnicity, presence of anti-topoisomerase I antibodies, presence of anti-RNA polymerase III antibodies, higher age, FVC % predicted and DLCO % predicted, no significant association between the severity of the scleroderma pattern and ILD-free survival was observed (HR (95 CI; p value): 1.09 (0.86–1.39; 0.47)) (Figure 3).
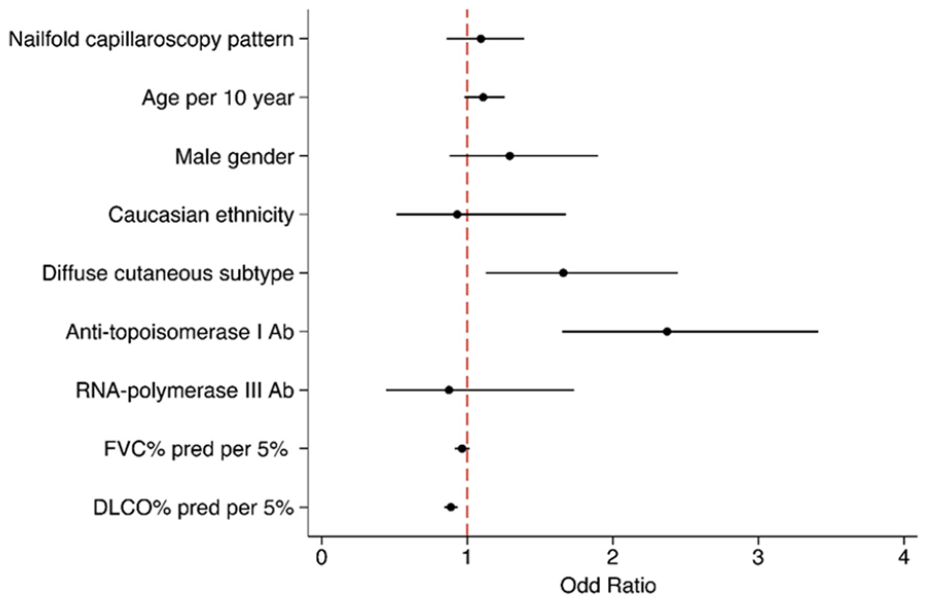
Final Cox proportional hazard model for the association between nailfold capillarosopy pattern at baseline and ILD risk during 5 years of follow-up. Nailfold capillaroscopy pattern at baseline is an ordinal variable, coded as followed: 1: early pattern, 2: active pattern, 3: late pattern.
Association between ILD risk and transition of nailfold capillaroscopic pattern
Next, we analysed the association between transition in nailfold capillaroscopic pattern and incident ILD. In total, 275 patients were included for this analysis with a median (IQR) follow-up duration of 2.0 (1.1–3.1) years and a median (IQR) of 2(1-3) nailfold capillaroscopies during follow-up. Due to a changed observation window for this analysis, the baseline for this analysis is different from the previous objective. As a result, overall, there was a higher proportion of patients with a diffuse cutaneous subtype. In addition, patients with an active pattern had more frequently pitting scars and arthritis, while patients in the early group received more frequently vasodilatory treatment. No other relevant differences were observed (see also Supplementary Tables 4–6).
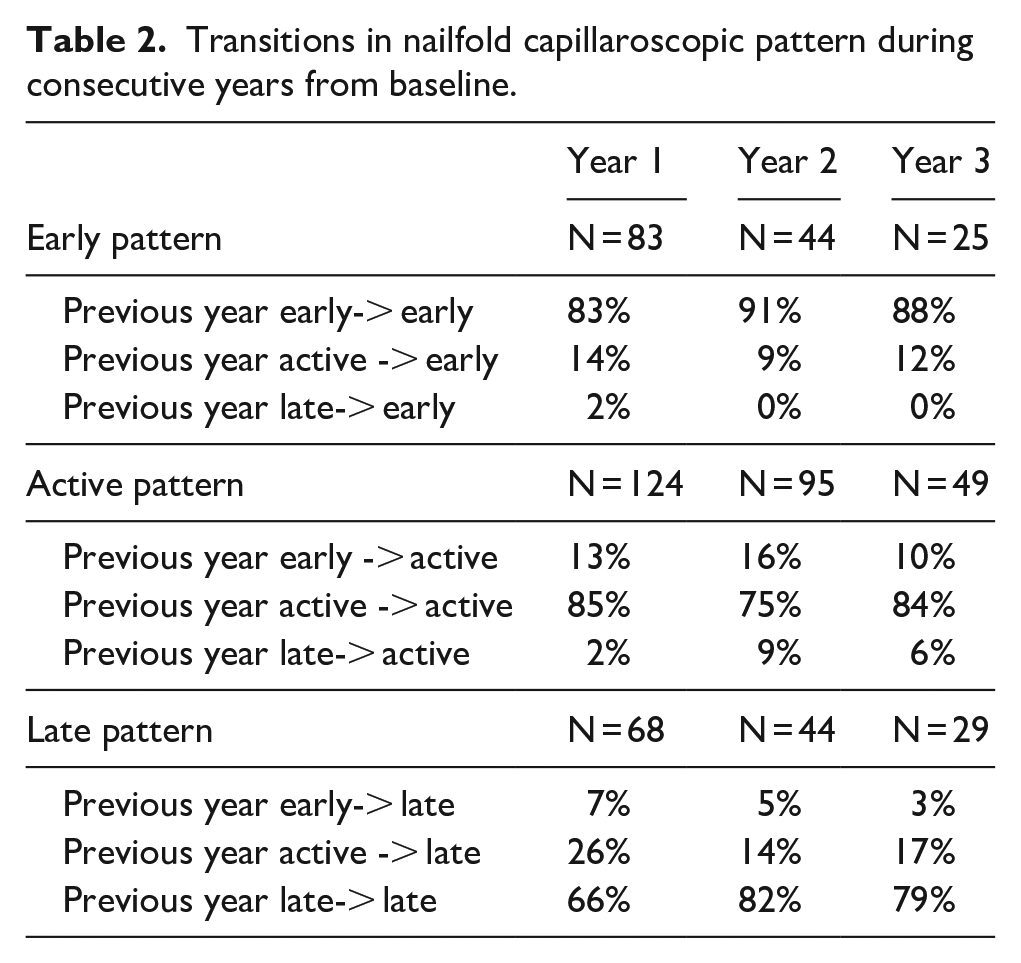
In Table 2, the transition of the nailfold capillaroscopic pattern in the first three consecutive years after baseline is presented. This evaluation showed that the nailfold capillaroscopic pattern remains stable in the majority of the patients (~81%). Remarkably, one patient transitioned from a late to an early pattern.The final mixed logistic regression model, incorporating the predefined confounders and mRSS as additional confounder identified through ‘CIE’ approach (see also Supplementary Table 7), revealed a rise in the annual risk for incident ILD with worsening severity of nailfold capillaroscopic pattern (odds ratio (OR) (95% CI); p value 3.76 (1.99–7.11; <0.01)) (Figure 4). Thus, these results indicate an increased risk for incident ILD after transitions towards a deteriorating nailfold capillaroscopic pattern, along with a reduced risk for incident ILD when transitioning to an improved nailfold capillaroscopic pattern.
Transitions in nailfold capillaroscopic pattern during consecutive years from baseline.
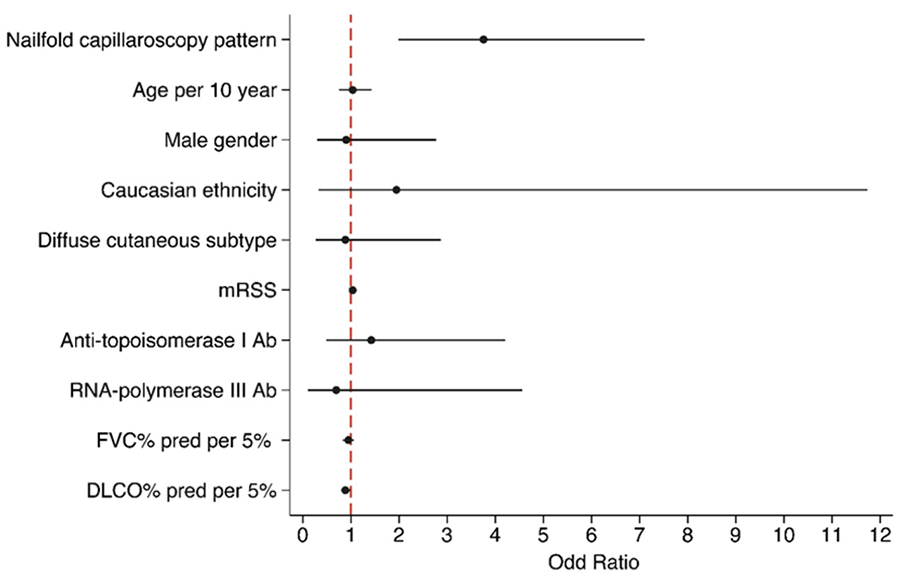
Final mixed logistic regression model for the association between nailfold capillaroscopy pattern and annual ILD risk.
Discussion
This observational study of prospectively collected data analysing the association between nailfold capillaroscopy pattern and incident ILD showed that a more severe nailfold capillaroscopy pattern is associated with a higher annual risk to develop ILD. In addition, patients who transition to a more severe capillaroscopic pattern face a higher risk of developing ILD within the following year, while those showing an improvement in their pattern experience a reduced risk of ILD during the same period. Interestingly, nailfold capillaroscopy pattern at baseline is not significantly associated with 5 years of risk to develop ILD.
These findings are contrary to that of Vanhaecke et al 14 who found no significant association between a severe nailfold capillaroscopy pattern and risk to develop ILD using data of patients from the EUSTAR database. In their study, the baseline was defined as the first date of registration in the EUSTAR database and presence of nailfold capillaroscopy. Furthermore, they used data from selected EUSTAR centres, resulting in a cohort with a relatively long disease duration and a high prevalence of ILD at baseline. Although our study has an arbitrary defined baseline, that is, first registered nailfold capillaroscopy pattern at an ILD-free visit, the additional inclusion criteria resulted in a relatively homogeneous cohort. This issue calls for attention in future research: establishing a relatively homogeneous patient population is crucial to avoid potential associations or effects being overshadowed by the high variation within this heterogeneous disease.
This study underscores the significance of nailfold capillaroscopy as a supplementary diagnostic tool in identifying patients at risk to develop ILD at the early disease stage. This aligns with the results of Sulli et al 23 who showed that SSc patients progressing to a more severe nailfold capillaroscopy pattern have a higher prevalence of ILD. Other studies have also hinted at an association between nailfold capillaroscopy and ILD. Given that these studies used a composite endpoint, that is, ILD and pulmonary hypertension or incident ILD and progressive ILD, these results are difficult to interpret.20,24,25 We found the most robust association between nailfold capillaroscopy patterns and the annual risk of ILD. We observed an association between nailfold capillaroscopy on the short term (i.e. mixed model) and not long term (i.e. Cox proportional hazards model), suggesting that primarily the transitions in nailfold capillaroscopy are informative. Of note, these transitions have not been taken into account into the Cox proportional hazards model as this is based on the nailfold capillaroscopy pattern at baseline. Large-scale prospective observational studies focusing on early disease stages are imperative to thoroughly assess the predictive capacity of nailfold capillaroscopy in addition to other established risk factors and ascertain the specific role of nailfold capillaroscopy within predictive algorithms.
In this study, the potential role of nailfold capillaroscopy may have been underestimated. Our study revealed that transition of nailfold capillaroscopy pattern is associated with an increased risk to develop ILD. A prospective longitudinal study has reported that a subset of SSc patients show rapid progression of microangiopathy.9,23 This subset of patients might have a different risk to develop ILD. However, as we did not take the time to transition into account, we could not replicate this analysis. Furthermore, our analysis solely relied on the qualitative assessment of nailfold capillaroscopy, neglecting quantitative assessments such as capillary density. The inclusion of such assessments could offer additional valuable insights, given that several studies have reported an association between reduced capillary density and ILD.9–11 Also, it is conceivable that variations in clinical practice concerning the indication and timing of HRCT scans, in particular, during active disease, have impacted our results. Finally, we have only used data of patients with a scleroderma pattern in this analysis and did not include patients with a normal nailfold capillaroscopy pattern or non-specific changes in their nailfold capillaroscopic pattern. This might have resulted in an underestimation of the association between nailfold capillaroscopy pattern and ILD, given that we lack a subset of patients with no detectable vasculopathy at nailfold capillaroscopy. So, in conclusion, a significant association between scleroderma pattern at nailfold capillaroscopy and the risk to develop ILD has been found, although certain other aspects of nailfold capillaroscopy might increase this association.
Major strengths of our study are the large sample size of patients at risk to develop ILD. Furthermore, we investigated the added value of nailfold capillaroscopy in a population with a high unmet clinical need to identify patients at risk for ILD. A limitation of our study, however, is that the nailfold capillaroscopy images were not centrally assessed, resulting in an intra-observer variability, possibly yielding less precise results. Nonetheless, our patients are followed in EUSTAR excellence centres and are representative for the common clinical practice. Furthermore, we focused on easy-to-assess scleroderma pattern instead of quantitative measurements.
In conclusion, our study shows that nailfold capillaroscopy has a strong association with the annual risk to develop ILD and that transitions in nailfold capillaroscopy patterns are informative. Of note, a worse scleroderma pattern at nailfold capillaroscopy is not associated with the long-term risk to develop ILD. These findings indicate that regular nailfold capillaroscopy assessments may guide clinicians in risk stratification, facilitating early interventions and personalized treatment strategies.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983241307692 – Supplemental material for Longitudinal association between nailfold capillaroscopy and incident interstitial lung disease: A EUSTAR database analysis
Supplemental material, sj-pdf-1-jso-10.1177_23971983241307692 for Longitudinal association between nailfold capillaroscopy and incident interstitial lung disease: A EUSTAR database analysis by Arthiha Velauthapillai, Jeska K de Vries-Bouwstra, Joerg Henes, Laszlo Czirjak, Lorenzo Dagna, Gabriela Riemekastan, Yannick Allanore, Rosario Foti, JJ Alegre-Sancho, Britta Maurer, Giovanna Cuomo, Julia Spierings, Thierry Martin, ME Truchetet, Cornelia HM van den Ende and Madelon C Vonk in Journal of Scleroderma and Related Disorders
Footnotes
Authors’ Note
The Editor/Editorial Board Member of JSRD is an author of this paper, therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Board member had no involvement in the decision-making process.
Author Contributions
Criterion 1:
(a) Substantial contributions to study conception and design: AV, CHME, MV.
(b) Substantial contributions to acquisition of data: All authors.
(c) Substantial contributions to analysis and interpretation of data: All authors.
Criterion 2: Drafting the article or revising it critically for important intellectual content: All authors.
Criterion 3: Final approval of the version of the article to be published: All authors.
Data availability
data are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.V.: none declared. J.K.B.: received consulting fees from Abbvie, Janssen and Boehringer Ingelheim, and received research grants from Roche, Galapagos Janssen, NVLE and ReumaNederland; all payments were made to her institution. J.H.: speakers bureau and consulting fees from ABBVIE,Astra-Zeneca, Boehringer Ingelheim, BMS, GSK, Janssen Pharmaceutical, Novartis, Roche, Pfizer and UCB. L.C.: none for this manuscript. L.D.: none for this manuscript. G.R.: none declared. Y.A.: received consulting fees from Boehringer Ingelheim, Janssen Pharmaceutical Companies, Astra-Zeneca, Topadur, Abbvie, Galderma, Medsenic, Prometheus regarding the management of systemic sclerosis. R.F.: none declared. J.J.A-S.: none for this manuscript. B.M.: grants from Novartis, consulting fees from Novartis, Boehringer Ingelheim, Jannsen-Cilag, GSK; speaker fees from Boehringer Ingelheim, GSK, Novartis, Otsuka, MSD; congress support from Medtalk, Pfizer, Roche, Actelion, Mepha, and MSD; and patent mir-29 for the treatment of systemic sclerosis (US8247389, EP2331143). G.C.: none for this manuscript. J.S.: received research grants from Boehringer Ingelheim and ReumaNederland; all payments were made to her institution. T.M.: none for this manuscript. M.E.T.: received consultancy and speaker fees from Boehringer Ingelheim, Pfizer, Galapagos, MSD and Lilly. C.H.M.E.: none declared. M.C.V.: received research grants from Boehringer Ingelheim, Ferrer and Galapagos; received consulting fees from Boehringer Ingelheim and Janssen Pharmaceutical Companies of Johnson & Johnson; received speaker fees from Boehringer Ingelheim, GSK, Janssen Pharmaceutical Companies of Johnson & Johnson, MSD, and Novartis; and is treasurer of EUSTAR and steering committee member of the ERN ReCONNET.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki. All participating EUSTAR MEDS database centres have ethics committee approval.
Informed consent
All participants provided written informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
