Abstract
Objective:
Systemic sclerosis, a rare disease characterized by chronic multisystem fibrosis, requires lifelong management, necessitating enough insurance coverage for the patient. Official drug approval is the first step to ensuring that the drug is covered by insurance. In this study, we investigated the approval status of essential therapeutic drugs for systemic sclerosis across eight countries and compared it with that of drugs for rheumatoid arthritis.
Methods:
The essential therapeutic drug lists for systemic sclerosis and rheumatoid arthritis were taken from the guidelines of the American College of Rheumatology and the European Alliance of Associations for Rheumatology. Official drug approval status for the selected drugs was confirmed by searching representative Internet databases from eight countries: the United States, the United Kingdom, Germany, France, Italy, Switzerland, Japan, and the Republic of Korea.
Results:
A total of 21 and 16 drugs were selected for systemic sclerosis and rheumatoid arthritis, respectively. The drug approval rates of the 21 drugs for systemic sclerosis varied among countries. Most drugs used to treat pulmonary arterial hypertension, which were developed recently and are expensive, are approved by most countries; however, most older drugs—which are still essential for management of Raynaud’s phenomenon, digital ulcers, interstitial lung disease, and skin fibrosis—are not approved by most countries. By contrast, almost all of the 16 drugs used to treat rheumatoid arthritis, whether old or new, are approved by most countries.
Conclusion:
Approval rates for drugs used to treat systemic sclerosis, a rare disease, are much lower than those for drugs used to treat rheumatoid arthritis. Thus, approval rates of essential therapeutic drugs for systemic sclerosis need to improve, which will benefit patients by increasing the number of drugs covered by insurance.
Key messages
Systemic sclerosis uses significant healthcare resources and is an economic burden. Official approval rates of drugs essential for systemic sclerosis are very low across many countries when compared with drugs for rheumatoid arthritis.
Approval of more drugs for systemic sclerosis will benefit patients by ensuring more comprehensive insurance coverage.
Introduction
Systemic sclerosis (SSc) is a chronic inflammatory disease characterized by progressive fibrosis of the skin and internal organs. It is one of the rarest rheumatic diseases, with a prevalence of 13.5–44.3 per 100,000 individuals in North America and 7.2–33.9 in Europe.1,2
Due to the progressive, irreversible, and multisystem nature of the disease, SSc is one of the costliest rheumatic disease to manage. The average annual total cost per patient ranges from US$26,000 to US$30,000 across several countries, with medication costs comprising 22%–26% of the total. 3 Medical costs continue to increase as new drugs become available, and the annual cost increases further if patients have major organ involvement or complications. 4 Such patients are often unemployed, making them more susceptible to economic hardship; thus, most patients with SSc require insurance coverage. Unfortunately, insurance coverage for such a rare disease is poor due to lack of clinical trial data regarding treatments and drugs. In most countries, the first step to insurance coverage is drug approval by the government’s official drug agency. Sound scientific evidence for efficacy, coupled with pharmacoeconomic evaluation, is factors to be considered when evaluating a drug for insurance coverage. Pharmacoeconomic evaluation varies according to a country’s economic status and the importance of a particular condition to pertinent populations; however, the drug approval process is broadly similar in most countries.
Official treatment guidelines issued by a reliable academic society usually encompass most scientific evidence showing that the drug is suitable for the indicated use. The American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) release treatment guidelines for rheumatologic diseases. The drugs listed in the ACR or EULAR guidelines are usually considered essential for the treatment of a disease. Drug approval status can be verified through countries’ official drug agencies. Here, we investigated the approval status of essential drugs for SSc recommended by academic societies and compared it with those for a more prevalent disease, rheumatoid arthritis (RA).
Methods
The approval status of therapeutic drugs essential for SSc in eight countries was compared with that for RA. First, essential therapeutic drug lists were chosen from representative treatment recommendations or guidelines. In the case of SSc, the updated (2016) EULAR recommendations for the treatment of SSc were used. 5 New therapeutic agents developed since 2016, particularly those for SSc-related interstitial lung disease (ILD), were obtained from the 2020 evidence-based European consensus statements for SSc-ILD and also included. 6 In accordance with the 2016 EULAR recommendations, the SSc medication list was classified as follows: drugs for SSc-RP (Raynaud’s phenomenon), drugs for SSc-DU (digital ulcer), drugs for SSc-PAH (pulmonary arterial hypertension), and drugs for skin and lung disease. Drugs used to treat scleroderma renal crisis and gastrointestinal symptoms were excluded if they are not disease-specific (such drugs include angiotensin-converting enzyme inhibitors, proton pump inhibitors, prokinetics, and antibiotics). The therapeutic drug list for RA was based on the updated 2019 EULAR recommendations for management of RA 7 and on the 2021 ACR guidelines for the treatment of RA. 8 Therapeutic drugs for RA were classified as follows: conventional synthetic disease-modifying antirheumatic drugs (DMARDs), biologic DMARDs, and targeted synthetic DMARDs.
Next, the approval status of the listed drugs for use as treatments for SSc and RA was investigated in eight countries: the United States, the United Kingdom, Germany, France, Italy, Switzerland, Japan, and Korea. In the Republic of Korea, a drug approval process begins with the application from the drug company to the Ministry of Food and Drug Safety, and it finally approves the drug after review of their safety and effectiveness from a risk–benefit perspective. The drug approval status in each country was obtained from online databases. Table 1 shows the providers and the online databases used to investigate drug approval status in each country. If a disease or manifestation was mentioned in the indications for a specific drug, the drug was considered to be approved for that disease. If a related product was indicated, that drug class was also considered to be approved. In some cases where drug approval status is not clear, press release from European Medicines Agency or specific drug companies was also referred to. When a specific drug was not listed in a database, it was denoted as “not applicable (NA).” The research was performed by two independent investigators (S.H.H. and K.W.M.) from 1 April 2023 to 23 June 2023. Where discrepancies existed, a discussion was held until consensus was reached, and a final decision was made by a reviewer (E.B.L.).
Online databases used to investigate drug approval status.
Results
Drugs listed as approved for treatment of SSc and RA are summarized in Table 2. And the specific drug database and access time to the database are summarized in Supplementary Table 1. In total, 21 drugs were selected for SSc and 16 for RA. If the same drug was also recommended for different organ involvement, it was described separately. Table 3 shows the approval status of therapeutic drugs for SSc in eight countries. The number of approved drugs varied among countries: the United States, 13 (61.9%); the United Kingdom, 11 (52.4%); Germany, 11 (52.4%); France, 12 (57.1%); Italy, 13 (61.9%); Switzerland, 13 (61.9%); Japan, 13 (61.9%); and Korea, 9 (42.9%). With respect to organ involvement, therapeutic drugs for SSc-PAH, most of which were developed recently, are approved by most countries; however, their use was permitted only in limited situations. Most drugs for RP are not approved in most countries. For example, Nifedipine, which is the most common drug used to treat RP, is approved only in France and Italy. Bosentan (for DUs) is approved in the United Kingdom, Germany, France, Italy, and Switzerland, but not in the other countries. Interestingly, intravenous Iloprost, which showed efficacy against DU in a randomized trial, 9 is not approved for this purpose by any country. Approval of drugs used to treat skin and lung involvement varied among countries, but most drugs are not approved except recently introduced Nintedanib. Table 4 shows the approval status for drugs used to treat RA. In contrast to drugs for SSc, almost all of the therapeutic drugs for RA are approved by most countries. Hydroxychloroquine has not been approved for treatment of RA in Japan, 10 and certolizumab pegol and sarilumab are not available in Korea.
List of drugs recommended by guidelines as a treatment for SSc and RA.
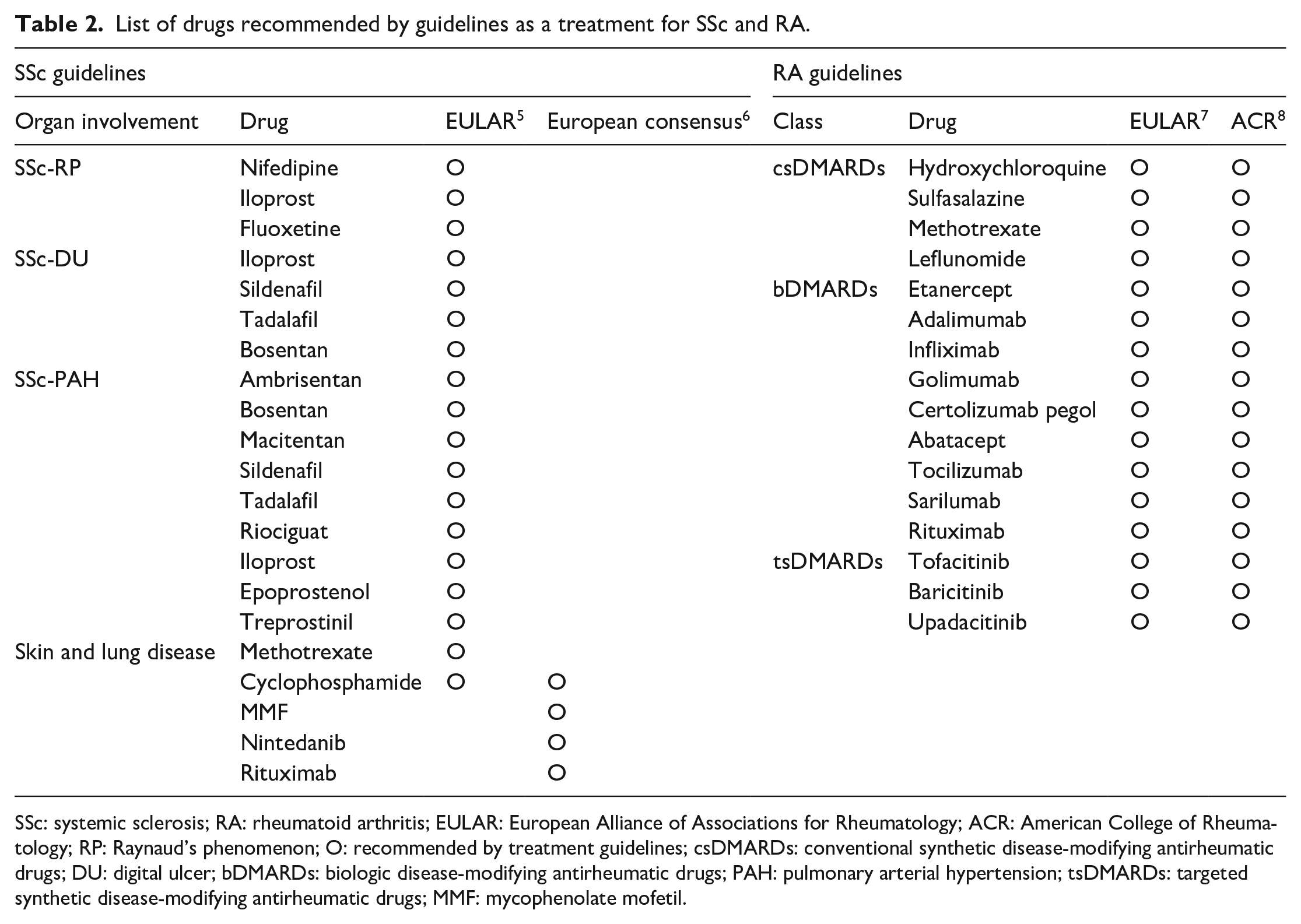
SSc: systemic sclerosis; RA: rheumatoid arthritis; EULAR: European Alliance of Associations for Rheumatology; ACR: American College of Rheumatology; RP: Raynaud’s phenomenon; O: recommended by treatment guidelines; csDMARDs: conventional synthetic disease-modifying antirheumatic drugs; DU: digital ulcer; bDMARDs: biologic disease-modifying antirheumatic drugs; PAH: pulmonary arterial hypertension; tsDMARDs: targeted synthetic disease-modifying antirheumatic drugs; MMF: mycophenolate mofetil.
Approval status of therapeutic drugs for systemic sclerosis across eight countries.
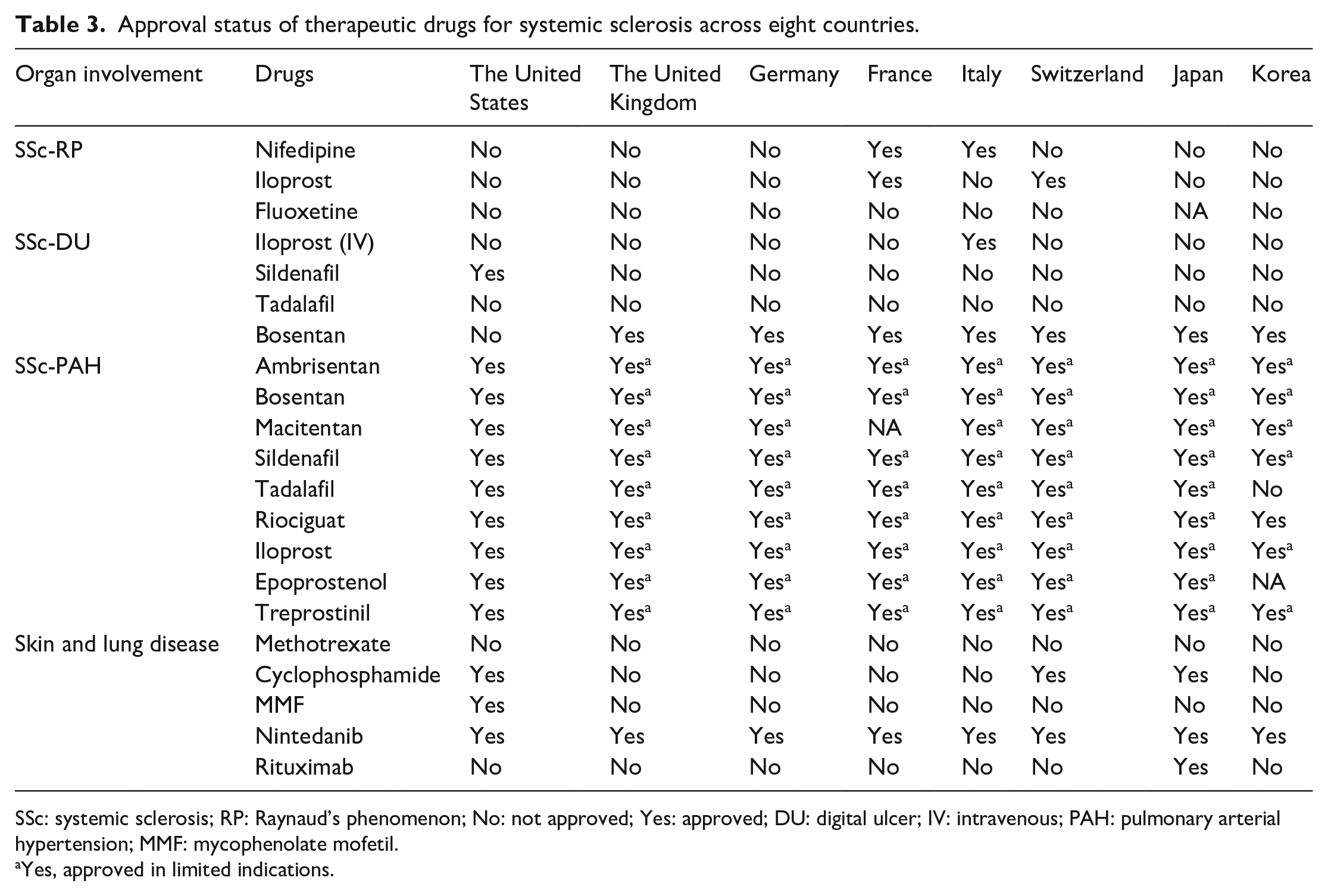
SSc: systemic sclerosis; RP: Raynaud’s phenomenon; No: not approved; Yes: approved; DU: digital ulcer; IV: intravenous; PAH: pulmonary arterial hypertension; MMF: mycophenolate mofetil.
Yes, approved in limited indications.
Approval status of therapeutic drugs for rheumatoid arthritis across eight countries.
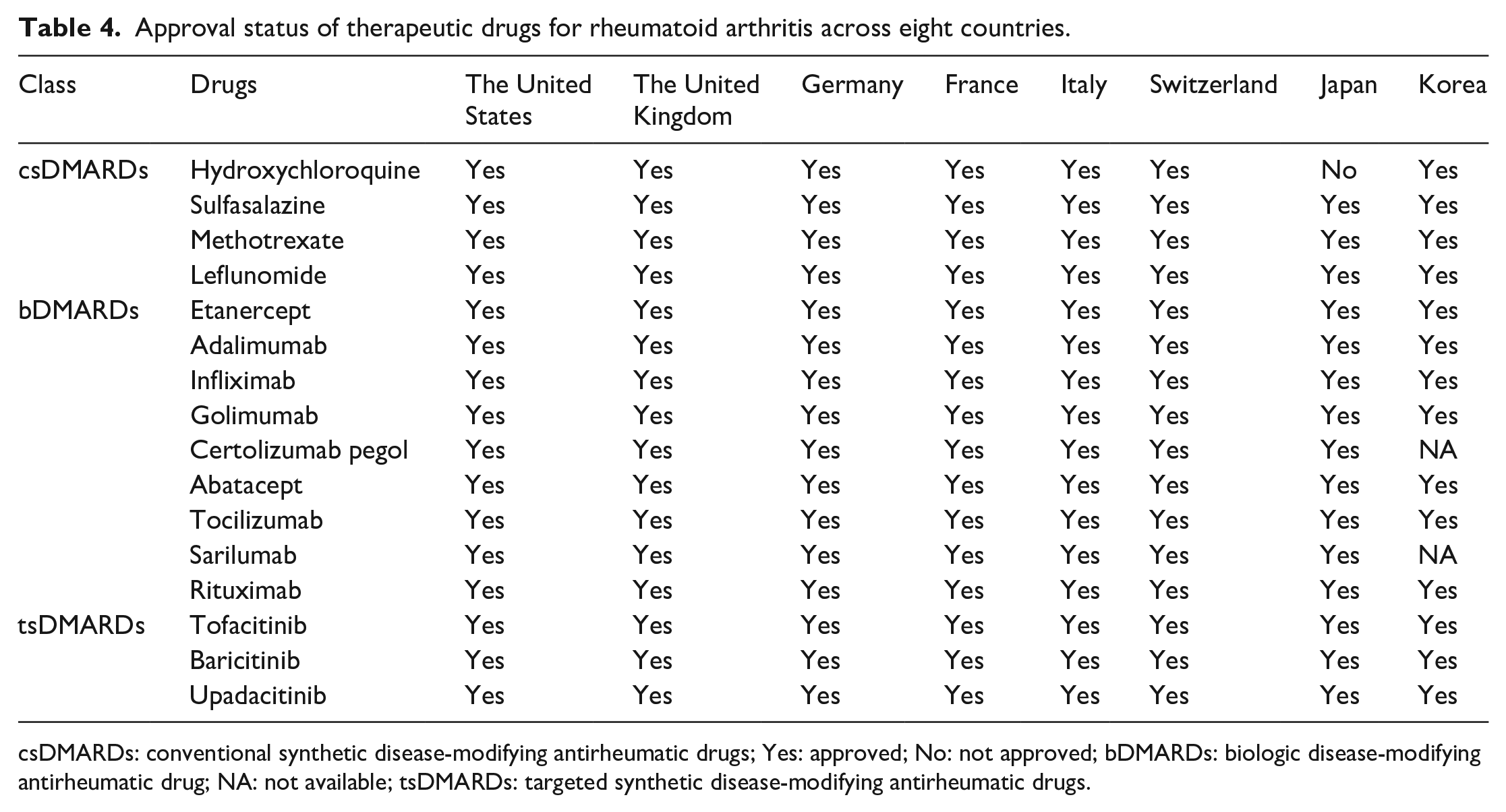
csDMARDs: conventional synthetic disease-modifying antirheumatic drugs; Yes: approved; No: not approved; bDMARDs: biologic disease-modifying antirheumatic drug; NA: not available; tsDMARDs: targeted synthetic disease-modifying antirheumatic drugs.
Discussion
Here, we compared the approval status for drugs used to treat SSc in eight countries and compared it with that of RA. Drug approval status varied among countries; however, a consistent finding was that fewer drugs were approved for SSc than for RA. SSc is a rare disease with high medical costs. Also, because the disease is rare, new drugs are few in number and expensive. In addition, management of the disease is very costly. For example, the annual medical cost per patient ranges from US$27,441–US$29,020 in the United States to US$4607–30,797 in Europe. 3 Li et al. 11 investigated the economic burden of 9714 Chinese patients from Sichuan province, all of whom had a rare disease. They reported that of these, the number of SSc patients was the highest (26.09%), and the cost of curative care was US$2.45 million. This ranked third in terms of medical cost for rare diseases. When SSc patients have major organ involvement, the cost increases further. A US study showed that the average total cost per month is higher for patients with connective tissue disease (CTD) with PAH, which is a common feature of SSc, than for those with CTD without PAH (US$16,854 vs US$4476, p-value < 0.001). 12 Due to the high cost of management, insurance coverage for essential therapeutic drugs is of great importance to patients.
Most drugs for SSc-PAH are approved by most countries; however, their use is restricted to certain cases. For example, many countries authorize the use of endothelin receptor antagonists or phosphodiesterase type 5 inhibitors for PAH cases only when the severity of dyspnea exceeds World Health Organization (WHO) functional class III or IV, despite the fact that clinical guidelines emphasize early detection and treatment of CTD-PAH, with combination therapy when necessary. 13 This shows that even authorized drugs have limitations with respect to SSc.
ILD is a major complication of SSc that affects more than half of patients and is a major cause of death. 1 A US study reported that SSc patients with ILD use more healthcare resources, attend more outpatient clinics, have more inpatient hospitalization days, attend more emergency room visits, and have higher annual direct medical costs than SSc patients without ILD. 14 However, most therapeutic agents for SSc-ILD—including cyclophosphamide, mycophenolate mofetil, and rituximab—are not approved for use in many countries despite the fact that the efficacy of cyclophosphamide and mycophenolate mofetil has been proven in well-designed randomized controlled trials.15,16 By contrast, Nintedanib, a newly developed and expensive drug, is approved by most countries. This strongly suggests that the drug approval process appears to be driven by profitability rather than scientific evidence or patient need.
RP is the most common initial symptom of SSc, and DUs cause a painful symptom. RP develops in over 95% of patients. 17 An Australian study involving 1085 SSc patients reported that 48.6% experienced DU over about 5 years, resulting in reduced quality of life and significant economic burden. 18 The prevalence and impact of these two conditions on quality of life suggest that there must be approved therapeutic drugs for these conditions; however, most therapeutic agents are not approved in most countries. Even Nifedipine, the most common drug for RP, is not approved by any country. Intravenous Iloprost, which was shown to be effective against SSc-DU in a randomized controlled trial, 9 is not approved either. Like drugs for ILD, it seems that older drugs, even those that show a clear benefit–risk ratio, cannot be approved without the effort by pharmaceutical companies.
Our data suggest that only more profitable drugs (with respect to price and market size) are approved by most countries. Most drugs for RA, which is much more prevalent than SSc, are approved by most countries. Even methotrexate, which is a cheapest anchor drug for RA, is approved by most countries. As stated above, older, cheaper drugs for SSc are not approved, whereas newer, more expensive drugs, including several drugs for PAH and Nintedanib for ILD, are approved. Medications for SSc, a rare disease, appear to be a low priority for pharmaceutical companies, government, and even society as a whole. Therefore, to ensure that patients with rare diseases such as SSc have access to medicines that improve quality of life, it is necessary to establish a drug approval process that is different from the status quo.
This study has some limitations. First, we used open online databases to search for drug approval status. The databases maintained by the formal drug agencies are not open or accessible to outside users. Therefore, the latest approval status of some drugs could not be evaluated. Second, we did not investigate insurance coverage or reimbursement status. In the Republic of Korea, certain drugs are covered by insurance despite the fact that they are not officially approved for use. However, insurance coverage is usually based on drug approval status. In addition, different insurance companies provide different coverage plans. Third, our search was restricted to only eight countries for which we could access databases. The countries chosen are all developed countries. Therefore, drug approval status in developing countries may be different. However, we do not expect that the approval of drugs for SSc will be more comprehensive in developing countries. Fourth, there are therapeutic drugs used in specific countries, so our drug lists cannot cover all of the drugs used in each country. Accordingly, real percentage of approved drugs may be different. However, for comparability among different countries, we only selected medications expected to be commonly used.
Conclusion
There is significant discrepancy between drugs recommended for treatment of SSc and those that are actually approved by government agencies, and the discrepancy between SSc and RA is stark. The drug approval state of SSc should be improved to come up with the current treatment guidelines, which would benefit SSc patients by expanding insurance coverage.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983231222368 – Supplemental material for Approval status of essential therapeutic drugs for systemic sclerosis versus that of drugs for rheumatoid arthritis
Supplemental material, sj-pdf-1-jso-10.1177_23971983231222368 for Approval status of essential therapeutic drugs for systemic sclerosis versus that of drugs for rheumatoid arthritis by Ki Won Moon, Soo-Hee Hwang, Jieun Yun and Eun Bong Lee in Journal of Scleroderma and Related Disorders
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a National Research Foundation of Korea grant, funded by the Korean government (Ministry of Science and ICT) (2021R1A2C2004874).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
