Abstract
Introduction:
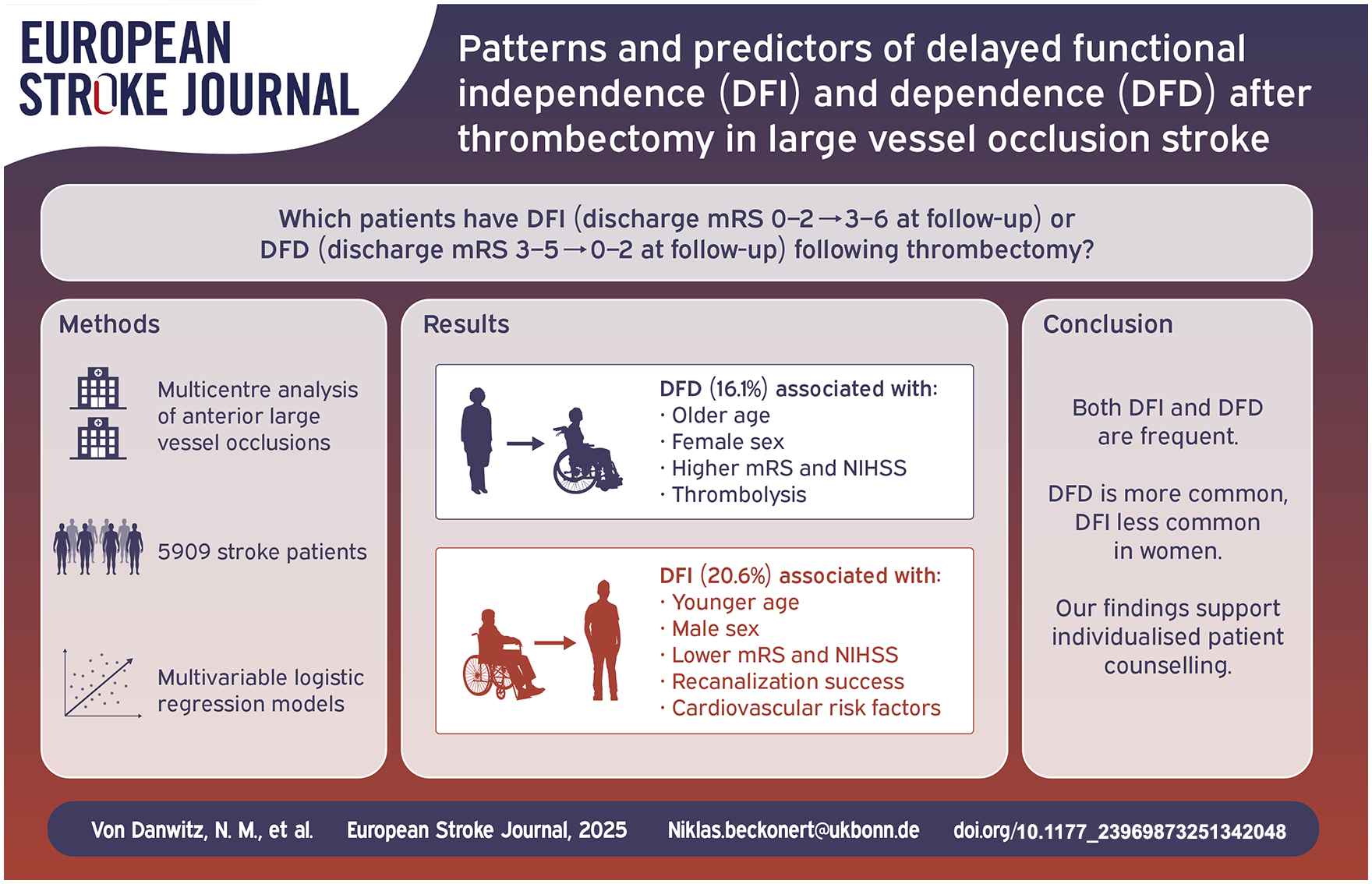
Predicting functional outcomes following endovascular treatment (EVT) for large-vessel occlusion stroke (LVOS) is challenging. Some patients achieve functional independence (modified Rankin Scale (mRS) 0 -2) at 90 days despite being dependent at discharge, termed delayed functional independence (DFI), while others lose independence after discharge, termed delayed functional dependence (DFD). This study explores patterns and predictors of DFI and DFD in LVOS patients undergoing EVT.
Patients and methods:
We analyzed anterior circulation LVOS patients from the prospective multicenter German Stroke Registry. Multivariable logistic regression models identified independent predictors of DFI and DFD.
Results:
Of 5909 patients, 2346 were independent at discharge, with 16.1% experiencing DFD at 90 days. DFD was associated with older age (median 78 vs 69 years,
Discussion and conclusion:
Both DFD and DFI are frequent in clinical practice, with higher DFD and lower DFI rates in women, which warrants further investigation. Understanding these predictors can enhance individualized patient counseling and management strategies.
Keywords
Introduction
Endovascular treatment (EVT) is a cornerstone of acute ischemic stroke with large vessel occlusion with well-established efficacy across diverse patient populations, including those treated in extended time windows and with lower Alberta Stroke Program Early CT Scores (ASPECTS).1 –3 The primary goal of EVT, both in clinical trials and in real-world practice, is the achievement of functional independence, defined by a modified Rankin Scale (mRS) score 0–2. 4 This outcome is not only a measure of treatment success but also meaningful for patients and their caregivers.
While patients achieve functional independence already at discharge, referred to as early functional independence (EFI), others may attain this outcome later during follow-up, termed delayed functional independence (DFI). 5 Conversely, patients who are functionally independent at discharge may deteriorate and experience delayed functional dependence (DFD). Despite the clinical importance of these trajectories, DFI and DFD remain understudied, with limited data on their incidence, predictors, and impact on health-related quality of life.
Understanding the factors influencing DFI and DFD may inform the optimization of treatment strategies, patient counseling, and long-term care planning. However, existing studies investigating DFI after EVT are limited by small sample sizes, monocentric designs, or a focus on specific thrombectomy devices.5,6 Moreover, DFD has not been systematically explored, leaving relevant gaps in our knowledge of outcome trajectories post-EVT.
To address these limitations, we analyzed data from the German Stroke Registry (GSR-ET), a large multicenter registry of LVOS patients treated with EVT. This study aims to (1) characterize the patterns and predictors of DFI and DFD, and (2) assess their impact on living conditions and health-related quality of life.
Methods
Study design and setting
We analyzed data from the German Stroke Registry - Endovascular Treatment (GSR-ET; NCT03356392). The GSR-ET is an ongoing, academic, open-label, multicenter registry that prospectively collects data on LVOS patients undergoing EVT for acute ischemic stroke across 25 certified stroke centers in Germany. The study period spanned from June 2015 to December 2021, and data were collected and maintained according to a standardized protocol. The study was conducted in accordance with the Declaration of Helsinki and was approved centrally by the Institutional Review Board of the Ludwig-Maximilians-University Munich (689-15) and by local institutional review boards. Detailed methods of the GSR-ET have been published previously.7,8
Patient population
Patients included in this analysis were adults (⩾18 years) treated with EVT for anterior circulation LVOS and a premorbid mRS of 0–2. Inclusion criteria required documentation of functional outcomes at discharge and 90 days, measured by the mRS. Patients were excluded in case of death before discharge or if follow-up data were incomplete or if EVT was not performed.
Data collection
Data were extracted from the GSR-ET database and included demographic information, clinical characteristics, imaging findings, procedural details, and functional outcomes. Stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS), while successful reperfusion was defined as a modified Thrombolysis in Infarction (mTICI) score of 2b-3. 9 Baseline infarct size was assessed using the Alberta Stroke Program Early CT Score (ASPECTS). Functional outcomes and premorbid disability were assessed using the mRS. 4
The mRS at 90-day follow-up was obtained during outpatient visits to a vascular neurologist or, if unavailable, by structured telephone interview from patients or, if unavailable, from their caregivers. 8 The vascular neurologists and study nurses involved have been trained through the Bluecloud MRS certification course and hold an MRS certificate. The follow-up visit/structured telephone interview included an assessment of living status and health-related quality of life. Therefore, responses were obtained to the established and previously used three-level European QoL-five dimensions (EQ-5D-3L) in stroke cohorts. 10 The EQ-5D utility index for each health state is based on a country-specific value set for each health state, with higher values indicating better health-related quality of life. Death was assigned an index score of 0 per instrument validation, with 1 representing optimal health. A representative, validated German-specific score is not available, but the UK population score was used with reasonable agreement, as described previously. 10
Adverse events were assessed during EVT, at 24 h, at discharge, and at 90-day follow-up. Symptomatic intracranial hemorrhage (sICH) was defined as intracranial hemorrhage detected on a 24-h follow-up CT scan and at least a 4-point increase in NIHSS score. 11 Stroke etiology was rated by vascular neurologists according to the TOAST classification. 12
Outcomes
EFI was defined as mRS 0–2 at discharge. 5 The primary outcomes were:
(1) DFI: mRS score of 0–2 at 90 days among EFI patients discharged with an mRS of 3 or higher
(2) DFD: mRS score of 3 or higher at 90 days, among non-EFI patients discharged with an mRS of 0–2.
A patient inclusion flow chart illustrating DFI and DFD is in Figure 1, and a schematic representation of DFI and DFD is presented in Figure 2(a).

Flow chart for patient inclusion.
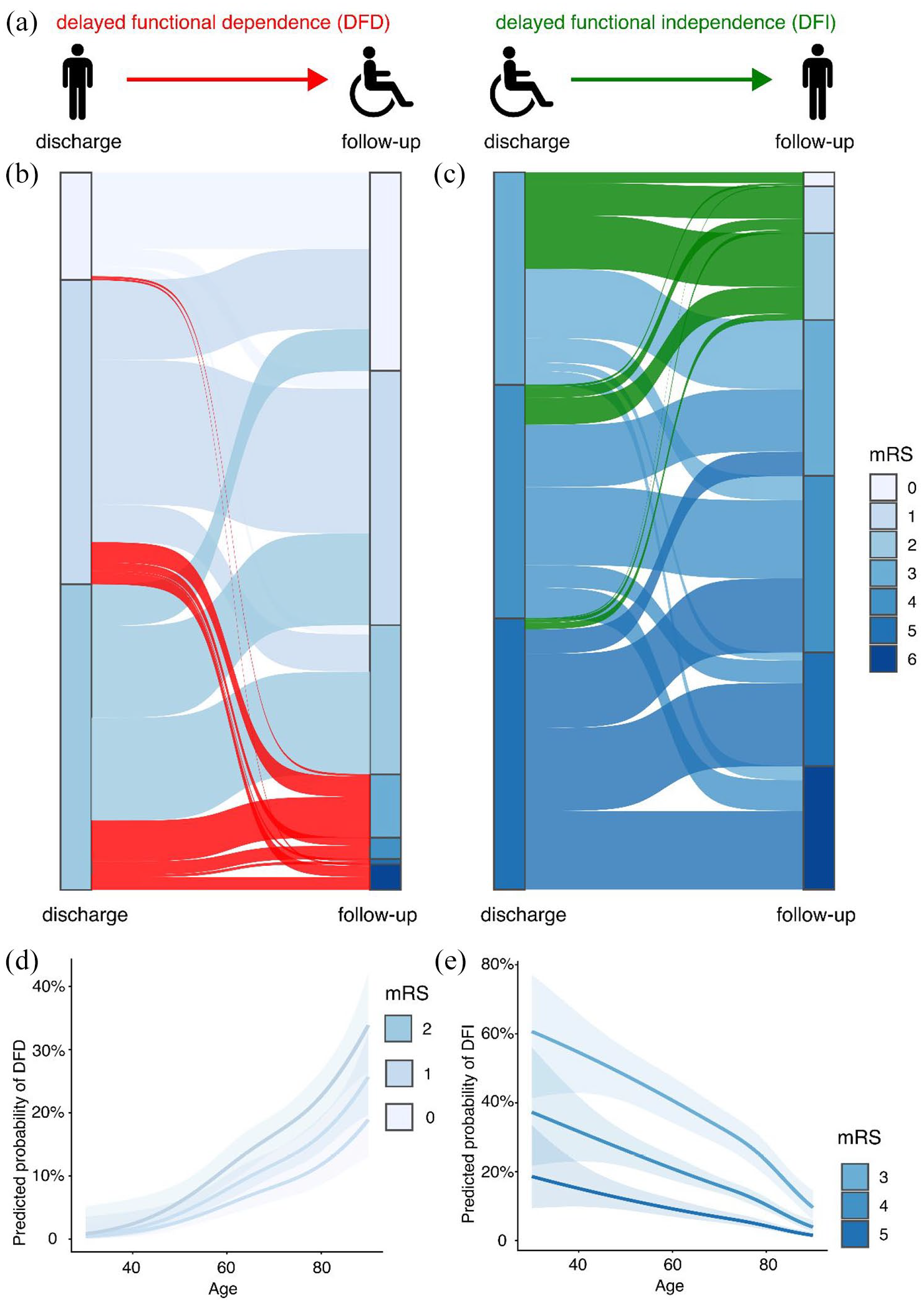
(a) Patients independent at discharge (mRS ⩽ 2) but dependent at 90-day follow-up (mRS > 2) show delayed functional dependence (DFD), while those dependent at discharge but achieving functional independence at follow-up show delayed functional independence (DFI). The Sankey plot illustrates the corresponding mRS shift for DFD in (b) and for DFI in (c). (d) Predicted probability of DFD (d) and DFI (e) as a function of discharge mRS and age, modeled using a natural cubic spline in a multivariable logistic regression model. Shaded areas represent the 95% confidence interval.
Statistical analysis
Standard descriptive statistics were provided, and group differences were assessed using unpaired Student’s t-test, Mann-Whitney U test, Fisher’s exact test, and chi-squared test, as appropriate. Multivariable logistic regression models were constructed including significant differences between patients achieving versus not achieving the respective outcome change category, followed by stepwise backward selection according to Akaike’s Information Criterion. Missing data were addressed using a complete-case analysis without imputation. Regression coefficients are presented as odds ratios (OR) with 95% confidence intervals. To explore the non-linear relationship between age and DFD or DFI, updated models including a natural cubic spline regression term with 3 degrees of freedom for the predictor age were trained. The spline fit and marginal effects of age were visualized with the ggeffects library using effect plots for age and discharge mRS for the different discharge mRS categories, while all other covariates were held at their mean values (continuous variables) or reference category (categorical variables).
13
Statistical significance was accepted at
The study was reported in accordance with the recommendations of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). The data supporting the results of this study are available from the corresponding author upon reasonable request and pending authorization of the study sites and ethics committees.
Results
Of the 13,082 patients in the GSR-ET registry, 5909 were included in the present analysis. Among these, 734 achieved DFI, while 378 experienced DFD. Baseline, periprocedural, and outcome characteristics of the included patients are presented in Supplemental Table 1. The patient inclusion process and classification are detailed in Figure 1.
Delayed functional dependence
Among 2346 patients with functional independence at discharge (mRS 0–2), 378 (16.1%) transitioned to dependency at 90 days and were classified as DFD (Figures 1, 2(a) and (b)). Compared to patients with preserved independence (hereafter termed non-DFD), DFD patients were more frequently female (61.4% vs 46.1%, both
Multivariable regression analysis identified older age, female sex, higher premorbid mRS, and higher discharge mRS and NIHSS scores as independent predictors of DFD, while patients receiving IVT were less likely to experience DFD (Table 1).
Multivariable logistic regression models for predicting delayed functional dependence in patients with functional independence at discharge.
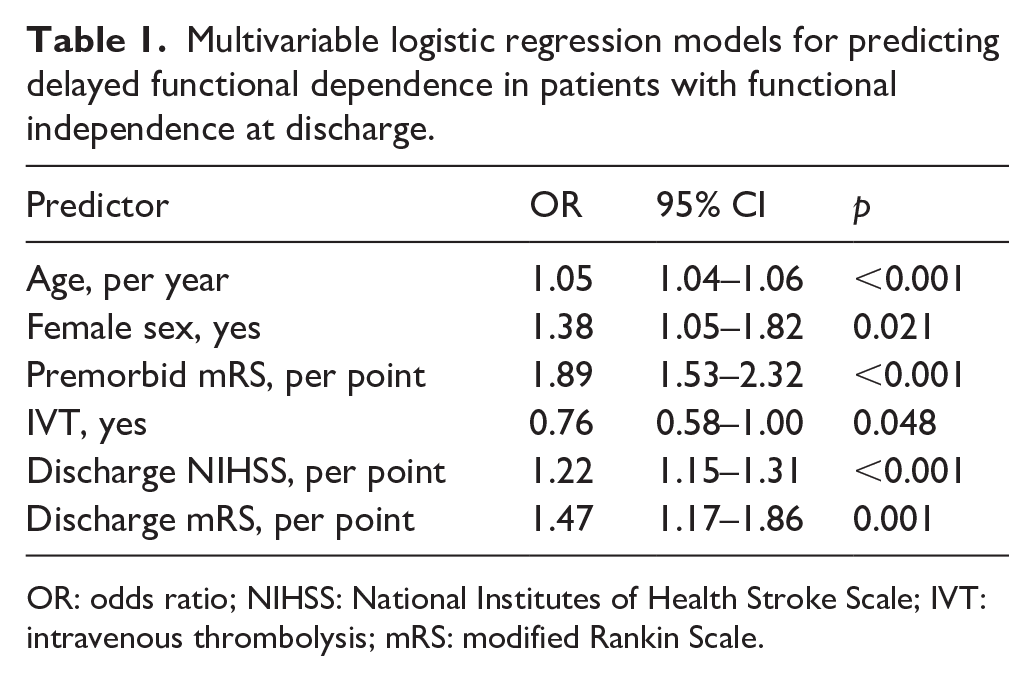
OR: odds ratio; NIHSS: National Institutes of Health Stroke Scale; IVT: intravenous thrombolysis; mRS: modified Rankin Scale.
Figure 2(d) illustrates the combined impact of age and discharge mRS on DFD in the multivariable logistic regression model, with restricted cubic spline regression allowing for a non-linear impact of age on DFD. The predicted probability of DFD rose steeply with age. For patients aged <50 years, the probability of DFD was 5% or less, but this increased to 20% or more in elderly patients with a discharge mRS of 1 or 2.
Delayed functional independence
Among the 3563 patients dependent at discharge (mRS > 2), 734 (20.6%) achieved functional independence at 90 days and were classified as DFI (Figure 2(c)). Compared to patients with persistent dependency or death at 90 days (hereafter termed non-DFI), DFI patients were younger (median age 71 vs 77 years,
DFI patients had milder strokes at admission (median NIHSS score 13 vs 16,
Multivariable regression analysis confirmed younger age, lower premorbid mRS, lower admission NIHSS, a lower number of thrombectomy maneuvers, successful recanalization, as well as lower discharge mRS and NIHSS as independent predictors of DFI (all
Multivariable logistic regression models for predicting delayed functional independence in patients with large vessel occlusion stroke receiving endovascular treatment.
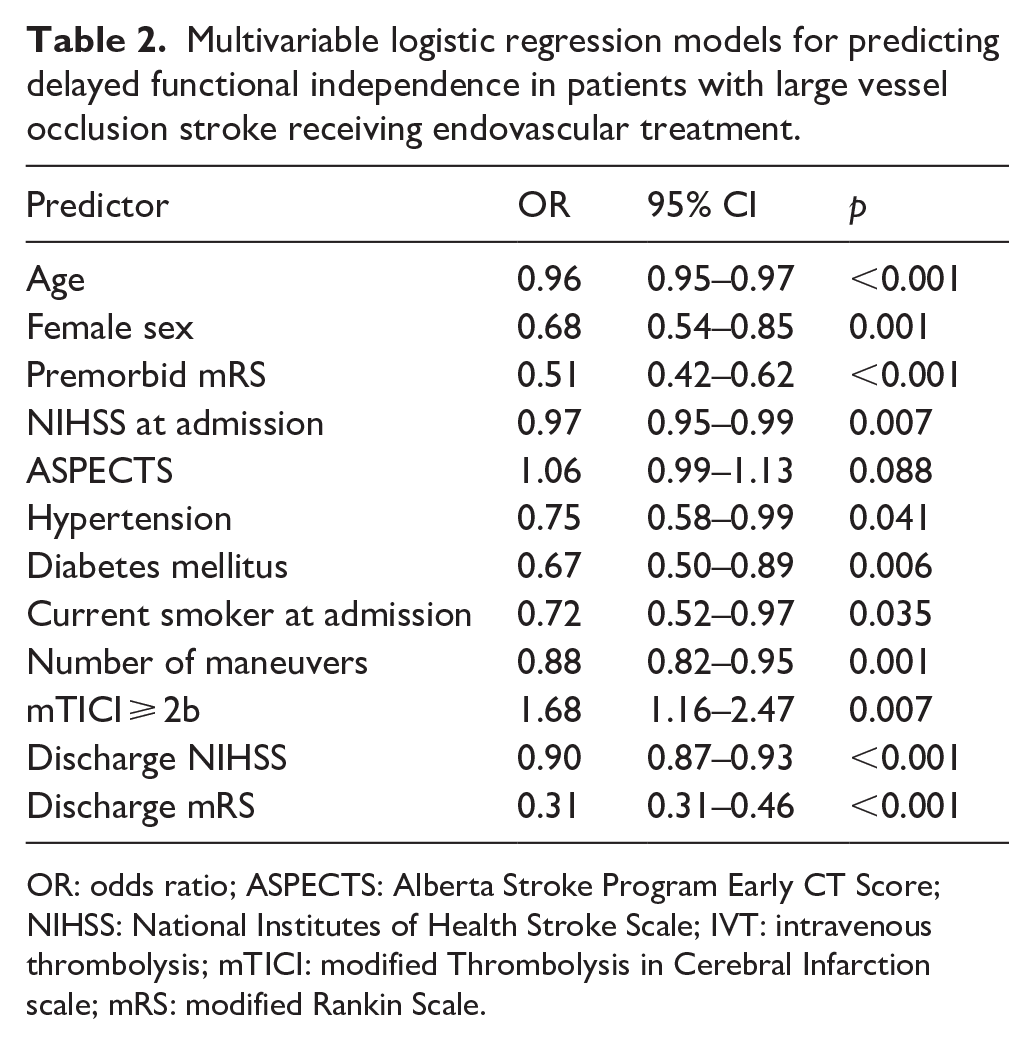
OR: odds ratio; ASPECTS: Alberta Stroke Program Early CT Score; NIHSS: National Institutes of Health Stroke Scale; IVT: intravenous thrombolysis; mTICI: modified Thrombolysis in Cerebral Infarction scale; mRS: modified Rankin Scale.
Figure 2(e) illustrates the impact of age and discharge mRS on DFI. The probability of DFI was < 10% in patients aged ⩾60 years with a discharge mRS of 5 but exceeded 25% for those with a discharge mRS of 3, up to 80 years of age. For patients <50 years with a discharge mRS of 3, the likelihood of DFI exceeded 50%.
Impact of DFD and DFI on living form and health-related quality of life
To assess the impact of DFD and DFI on patient-reported outcomes, we further assessed the form of living as well as health-related quality of life.
At 90 days, patients achieving DFI were four times more likely to live at home (85.7% vs 19.5%) and six times less likely to reside in nursing homes (6.1% vs 35.9%) than non-DFI patients (
Living status and health-related quality of life assessed at 90-day follow-up.
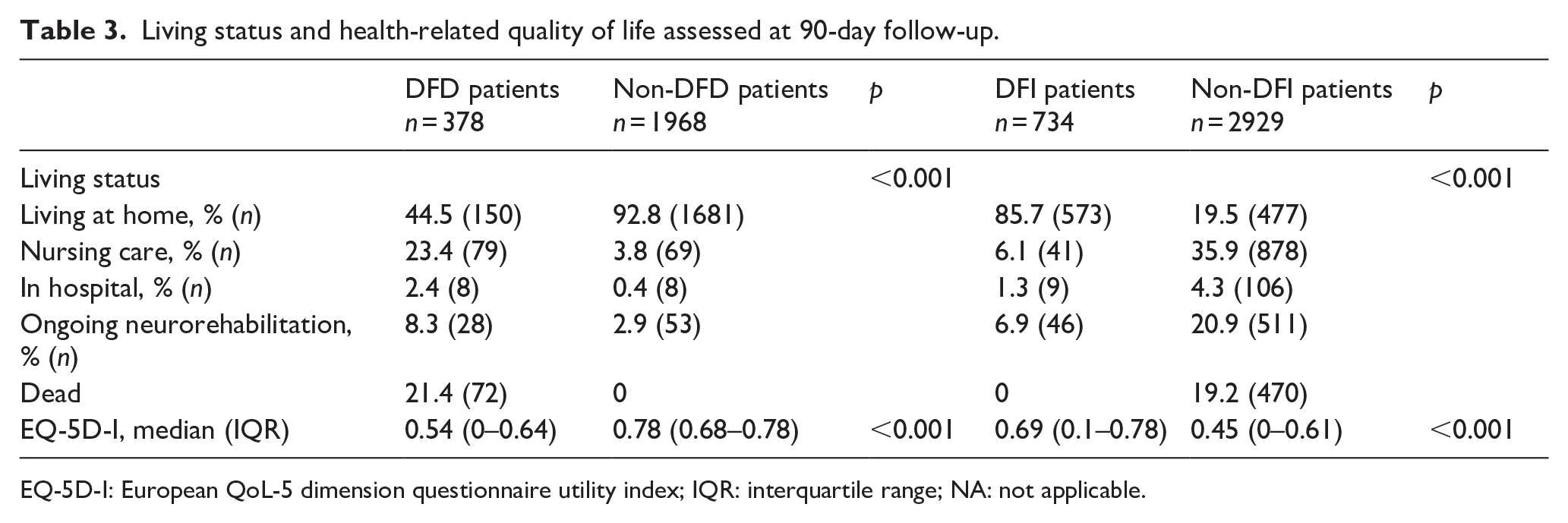
EQ-5D-I: European QoL-5 dimension questionnaire utility index; IQR: interquartile range; NA: not applicable.
In contrast, DFD patients were less likely to live at home (44.4% vs 92.8%) and more likely to require hospitalization (2.4% vs 0.8%) or nursing care (23.4% vs 3.8%) compared to non-DFD patients (
Discussion
Analyzing a large multicenter cohort of LVOS patients treated with EVT, we provide novel real-world insights into the predictors and outcomes of both DFI and DFD. Importantly, we find that younger age, better premorbid status including non-smoking, the absence of hypertension and diabetes mellitus, lower stroke severity, successful recanalization, and male sex increase the odds of achieving functional independence at 90 days despite being dependent at discharge. Conversely, patients with functional independence at discharge are more likely to experience subsequent dependency in the absence of IVT and with older age, female sex, worse premorbid mRS, and higher discharge NIHSS score/mRS. Highlighting the trajectories of functional recovery and dependency after EVT, our findings underscore the importance of tailoring clinical discussions and interventions to expected patient outcomes.
A recent meta-analysis analyzed a total of 3266 EVT patients with delayed neurological improvement, albeit hampered by heterogeneous definitions of improvement, also identified younger age and male sex, but also smoking as positive predictors for DFI.
14
Despite higher rates of smoking among DFI patients, smoking was associated with significantly
Sex differences of DFI and DFD
Our analysis revealed significant sex differences, with women being more likely to experience DFD and less likely to achieve DFI, consistent with previous studies.14,17,18 While women have higher rates of cardioembolic etiology and an often greater stroke severity,18 –20 our findings suggest that sex-specific factors, beyond baseline differences, may influence recovery trajectories. Further research is warranted to explore potential mechanisms, including biological, psychosocial, and healthcare-related factors, contributing to these disparities.
Age and outcome trajectories
Our analysis confirms that younger stroke patients have higher rates of DFI, likely due to greater rehabilitative potential, lower frailty, and overall better functional reserve. In contrast, older patients are more susceptible to DFD potentially due to diminished neuroplasticity, reduced physiological resilience, and a higher burden of comorbidities such as atrial fibrillation and cardiovascular risk factors. 21 A recent real-world analysis showed that in the 80+ age group, more than 50% of thrombectomy patients died within 1 year. 22 The heterogeneity of age-specific mRS trajectories until 90-day follow-up needs to be taken into account when designing or interpreting EVT trials. Furthermore, our findings challenge the suitability of using 3-month mRS outcomes in stroke trials involving elderly populations, as these outcomes may be disproportionately influenced by age-related factors. Alternative, earlier outcome measures may provide more robust and meaningful insights into treatment efficacy.23,24
Functional outcomes and health-related quality of life
The impact of DFI and DFD extends beyond functional status, influencing living status and health-related quality of life at 90 days. DFI patients were more likely to live at home and had markedly better health-related quality of life than their non-DFI counterparts. Conversely, DFD patients were more likely to require nursing care or hospitalization and reported lower health-related quality of life. These findings highlight the broader implications of DFI and DFD, emphasizing the need for targeted post-discharge rehabilitation and support strategies to improve long-term outcomes.
Clinical implications
Our findings have important clinical implications for patient management and counseling. Identifying patients at risk of DFD or with the potential for DFI can guide discussions about prognosis and inform personalized treatment and rehabilitation plans. For instance, younger patients with discharge mRS of 3 and successful recanalization may be reassured about the likelihood of DFI, while it is speculative if closer follow-up and additional support might prevent DFD in older female patients with high NIHSS scores.
Limitations and strengths
Several limitations of our study warrant consideration. The observational design of the registry may introduce selection bias, and unmeasured confounders could influence the observed associations. The generalizability is limited to comparable Western health care systems. Interrater variability of the mRS must be taken into account when interpreting these results and might explain some cases barely missing or reaching functional independence. Unexpected events between discharge and 90-day follow-up, such as early recurrent stroke, infection or trauma, may affect the dynamic recovery process, but these events are not systematically captured by the GSR-ET, representing an important limitation. As in the landmark EVT trials, follow-up information in the registry is provided at day 90; therefore, no conclusions can be drawn about the temporal distribution of dependence/independence up to and beyond this cut-off date. Furthermore, a substantial number of patients from our real-world cohort were still in neurorehabilitation at 90 days, emphasizing that functional recovery often takes more time. 25 The strengths of this study include the multicenter design and the use of real-word data from an established nationwide registry, providing the largest analysis of DFI and DFD to date.
Conclusion
DFI and DFD represent distinct frequent and clinically significant trajectories in patients treated with EVT for LVOS. Understanding their predictors and implications can inform individualized care strategies and optimize outcomes. Our findings also underscore the need for further research into sex disparities to refine prognostication and therapeutic approaches.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251342048 – Supplemental material for Patterns and predictors of delayed functional independence and dependence after thrombectomy in large vessel occlusion stroke: A multicenter analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873251342048 for Patterns and predictors of delayed functional independence and dependence after thrombectomy in large vessel occlusion stroke: A multicenter analysis by Niklas M von Danwitz, Felix J Bode, Omid Shirvani Samani, Hannah Asperger, Taraneh Ebrahimi, Johannes Kaesmacher, Julia Layer, Nils Lehnen, Louisa Nitsch, Julius N Meissner, Svenja Odensass, Sebastian Stösser, Christian Thielscher, Mousa Zidan, Franziska Dorn, Gabor C Petzold and Johannes M Weller in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NVD received travel grants from Sanofi and Viatris. FD is a consultant/proctor for Cerenovus/Johnson&Johnson, Balt, Cerus Endovascular and Phenox. She received speaker’s honoraria from Acandis, Stryker, Cerenovus/Johnson&Johnson, Asahi and research support from Cerenovus/Johnson&Johnson. JK reports Microvention consultancy within the framework of a corelab, financial support from Medtronic for the BEYOND SWIFT registry and SWIFT DIRECT trial; medication supply support and an unrestricted grant paid to the institution from Boheringer-Ingelheim for the TECNO trial, a research agreement with Siemens Healthineers regarding flat panel perfusion imaging and research grants from the Swiss National Science Foundation supporting the TECNO trial, Swiss Academy of Medical Sciences supporting MRI research and Swiss Heart Foundation supporting cardiac MRI in the etiological work-up of stroke patients. All other authors report no conflicting interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Participants or their legal representatives gave informed consent as described in the study protocol. 5
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki and was centrally approved by the institutional review board of the Ludwig-Maximilians University Munich (689-15) and institutional review boards according to local regulations.
Guarantor
JMW
Contributorship
NVD, FJB and JMW designed the study, NVD and JMW researched literature. NVD, FJB, OSS, HA, TE, JL, NL, LN, JNM, SO, SS, CT, MZ, FD, GSP acquired data. JMW analyzed the data. NVD and JMW drafted the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Trial registration
Clinicaltrials.gov NCT03356392
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
