Abstract
Introduction:
While the efficacy of endovascular thrombectomy (EVT) in large core infarcts has been established, the influence of blood pressure (BP) management on functional outcomes based on infarct volume remains unclear.
Patients and methods:
We conducted a secondary analysis of the Outcome in Patients Treated With Intra-Arterial Thrombectomy–Optimal Blood Pressure Control (OPTIMAL-BP) trial, which compared intensive (systolic BP < 140 mmHg) versus conventional (systolic BP 140–180 mmHg) BP management within the first 24 h following successful recanalization. Patients were grouped based on an infarct volume cut-off of 50 ml, assessed 24 h post-EVT. The primary efficacy outcome was functional independence (modified Rankin Scale of 0–2) at 3 months. Change of predicted probability for functional independence between BP managements, as infarct volume varied, was assessed.
Results:
Of the 300 patients, 222 (74.0%) were in the infarct volume ⩽50 ml group and 78 (26.0%) were in the infarct volume >50 ml group. The conventional management was significantly associated with a higher rate of functional independence in the infarct volume ⩽50 ml group (adjusted odds ratio [AOR], 2.06 [95% CI, 1.12–3.86]). In the infarct volume >50 ml group, the proportion of patients with functional independence was not significantly different between BP managements (AOR, 1.52 [95% CI, 0.46–5.04]). The interaction effect between the infarct volume groups and BP managements was not significant. As infarct volume increased, the difference in predicted probability of functional independence between BP managements decreased.
Discussion and conclusions:
Conventional BP management showed greater benefits for achieving functional independence at 3 months when infarct volumes were smaller. As infarct volume increased, the impact of BP management strategies on functional outcomes decreased.
Registration:
ClinicalTrials.gov (NCT04205305)
Introduction
Endovascular thrombectomy (EVT) is a highly effective treatment for acute stroke patients with large vessel occlusion, achieving approximately 80% successful recanalization. 1 With the increasing success rates of the EVT, interest has grown in understanding how post-procedural blood pressure (BP) management affects outcomes. Retrospective studies have shown an association between controlling BP below specific thresholds or lowering systolic BP (SBP) after successful EVT and improved functional outcomes.2–5 To date, this hypothesis has been tested in four multi-center and one single center randomized clinical trials (RCTs).6–10 Recent meta-analyses have shown that conventional BP management resulted in better functional outcomes compared to intensive BP management without increasing symptomatic intracerebral hemorrhage (ICH). 11
Approximately 20% of patients with large vessel occlusion stroke present with a large core infarct,12,13 which increases their susceptibility to developing ICH in the infarcted area after reperfusion.14–16 Despite these concerns, five RCTs for patients with large core infarct have demonstrated that EVT leads to more favorable outcomes than best medical treatment.14–18 However, three out of these trials reported a higher incidence of ICH following EVT.14–16 Given that current guidelines for spontaneous ICH recommend targeting a SBP of 140 mmHg,19–21 the question of optimal BP management in patients with large core infarct after successful reperfusion remains crucial. Effective BP control in such patients may help reduce the risk of ICH and reperfusion injury, but there is no established guideline for post-EVT BP management in this high-risk population.
Herein, using data from the Outcome in Patients Treated With Intra-Arterial Thrombectomy–Optimal Blood Pressure Control (OPTIMAL-BP) trial, in which conventional BP management showed favorable functional outcomes after successful EVT, we aim to explore how the effects of BP management on functional recovery differ based on 24-h infarct volume.
Methods
Study design
This study was a post-hoc secondary analysis of the OPTIMAL-BP trial, designed after the main trial’s completion and not specified in the original protocol. The OPTIMAL-BP trial was a multicenter, randomized, open-label trial with a blinded end point evaluation trial which was conducted across 19 stroke centers in South Korea from June 2020 to November 2022. 8 The OPTIMAL-BP trial was approved by the ethics committee at Severance hospital (approval number: 4-2019-1208) and all participating centers. Written informed consent was obtained from participants or their legally authorized representatives before the study began. The details of the protocol, statistical analysis plan, and the results of the trial were reported elsewhere.8,22 This trial is registered at ClinicalTrials.gov (NCT04205305).
Participants
The design and procedures of the trial, along with the detailed inclusion and exclusion criteria, have been previously reported.8,22 Briefly, the trial enrolled patients with acute ischemic stroke due to large vessel occlusion who attained successful reperfusion of the occluded artery and exhibited an elevated SBP (⩾140 mmHg) within 2 h after reperfusion. Patients who had a 3-month modified Rankin Scale (mRS) score available and had infarct volume assessed via brain magnetic resonance imaging (MRI) or computed tomography (CT) after EVT were included in this study. CONSORT guidelines were used to report the findings.
Randomization
Study participants were randomized in a 1:1 ratio, stratified by hospital and level of neurological impairment (National Institutes of Health Stroke Scale [NIHSS] score at admission, <15 points vs ⩾15 points). A centrally administered, password-protected web-based system was used for computerized random sequence generation, with permuted block randomization applied in block size of 4.
Study group
In the OPTIMAL-BP trial, the mean infarct volume was 51.9 ml (standard deviation [SD], 84.2 ml). Three cut-off points (30, 50, and 70 ml) were used to divide groups with infarct volumes either above or below each threshold. Based on the large core trial, which proposed 50 ml as the cut-off for large core infarct, 17 and the mean infarct volume, we used the 50 ml cut-off for the main analysis in this study. Additionally, cut-offs of 30 and 70 ml were used for sensitivity analysis. A number of patients did not undergo perfusion CT or diffusion-weighted imaging before EVT, preventing the assessment of core volume prior to the intervention. Thus, infarct volume was assessed using brain imaging (MRI or CT) at 24 ± 12 h post-EVT.
Measurement of infarct volume
Follow-up imaging was performed at 24 h (with a window of ±12 h) after randomization. The imaging data were submitted to an independent core laboratory for blinded adjudication. Infarct volume was measured using the open-source software 3D Slicer (available at https://www.slicer.org). An infarcted area was defined as a region with an apparent diffusion coefficient value of less than 620 × 10−6 mm2/s on MRI or a hypodense area on CT. The software’s drawing tool was used to trace the boundaries of the infarcted area on all relevant slices, with care taken to exclude areas of artifact or hemorrhage.
Outcome measures
The primary efficacy outcome was a binary analysis of the mRS score at 3 months, categorizing scores as either 0–2 for functional independence or 3–6 for functional dependence or death. The primary safety outcomes were symptomatic ICH within 36 h and mortality related to the index stroke within 3 months. Symptomatic ICH was defined according to the European Cooperative Acute Stroke Study III criteria as any presence of extravascular blood in the brain or cranium associated with clinical worsening, evidenced by an increase of 4 or more points in the NIHSS score or resulting in death. 23 Secondary outcomes included a shift analysis of the distribution of mRS scores at 3 months, health-related quality of life at 3 months using the 3-level EuroQoL 5-Dimension Self-Report Questionnaire (EQ-5D-3L), and any ICH within 36 h. Local certified medical staff masked to treatment allocation and imaging findings collected mRS scores at 3 months in person or via telephone.
Statistical analysis
The main analysis compared the baseline characteristics of intensive and conventional BP managements based on a cut-off infarct volume of 50 ml. Additionally, baseline characteristics were compared for each BP management using cut-off infarct volumes of 30 and 70 ml. Continuous variables were presented as a mean ± SD or median (interquartile range [IQR]), and categorical variables as numbers (%). The
The primary outcomes were tested using binomial logistic regression to estimate the odds ratios (ORs) and 95% CIs. Multivariable logistic regression analyses were performed to adjust for age, sex, time from stroke onset to randomization, and NIHSS score greater than 15 before EVT, as these variables were adjusted in the OPTIMAL-BP trial. Ordinal, binary logistic, and linear regression analyses were conducted for the secondary outcomes, with adjustments using the same variables as for the primary outcomes. Interaction analyses were conducted. To illustrate the relationship between infarct volume and functional outcome at 3 months according to BP managements, adjusted predicted probabilities (range, 0–1) of binary outcome (mRS score of 0–2 or 3–6) were calculated. Adjustments included age, sex, time from stroke onset to randomization, baseline NIHSS > 15 before EVT, and the interaction between infarct volume and intensive versus conventional BP management. We further examined whether the intersection of the regression lines for each BP management could be a meaningful cut-off point. A generalized linear model with a three-way interaction test was used to assess the significance of differences in predicted probabilities for binary outcomes as infarct volume varied between BP managements. A two-sided
Results
Of the 306 patients who were enrolled in the OPTIMAL-BP trial, one patient withdrew consent immediately after randomization, three patients were lost to follow-up within 24 h, and two patients were excluded because follow-up infarct volume could not be measured. Among the 300 included patients (median age, 75 [IQR, 66–82] years; 179 [59.7%] men), the infarct volume ⩽50 ml group comprised 222 (74.0%) patients (108 intensive management and 114 conventional management) and the infarct volume >50 ml group comprised 78 (26.0%) patients (47 intensive management and 31 conventional management; Figure S1). Among the included patients, infarct volume was measured by MRI in 295 (98.3%) and by CT in 5 (1.7%) patients.
Baseline characteristics
Patients in the infarct volume ⩽50 ml group had a median age of 75 (IQR, 66–82) years; 131 (59.0%) were men. Patients in the infarct volume >50 ml group had a median age of 75.5 (IQR, 64.5–82) years; 48 (61.5%) were men. The infarct volume ⩽50 ml group had a lower proportion of patients with NIHSS scores >15, Alberta Stroke Program Early CT Score (ASPECTS) <6, and poor collateral status. There were no significant differences between infarct volume groups in demographics, medical conditions, or time intervals (Table S1). In the ⩽50 ml infarct volume group, baseline characteristics were similar between BP management strategies, except for collateral status, where the intensive BP management had a higher proportion of good collateral status. In the infarct volume >50 ml group, there were no significant differences in baseline characteristics between BP managements (Table 1).
Baseline characteristics according to cut-off infarct volume of 50 ml.

ASPECTS: Alberta Stroke Program Early CT Score; BP: blood pressure; EVT: endovascular thrombectomy; IQR: interquartile range; IV-tPA: intravenous tissue-type plasminogen activator; mTICI: modified treatment in cerebral infarction; NIHSS: National Institutes of Health Stroke Scale.
BP measurements and infarct volumes
At the time of enrollment, the mean SBP did not significantly differ between the conventional and intensive BP managements, regardless of whether the infarct volume was ⩽50 ml or >50 ml (Table S2). The mean between-BP management difference in SBP over 24 h was 6.29 mmHg (95% CI, 1.67–10.91;
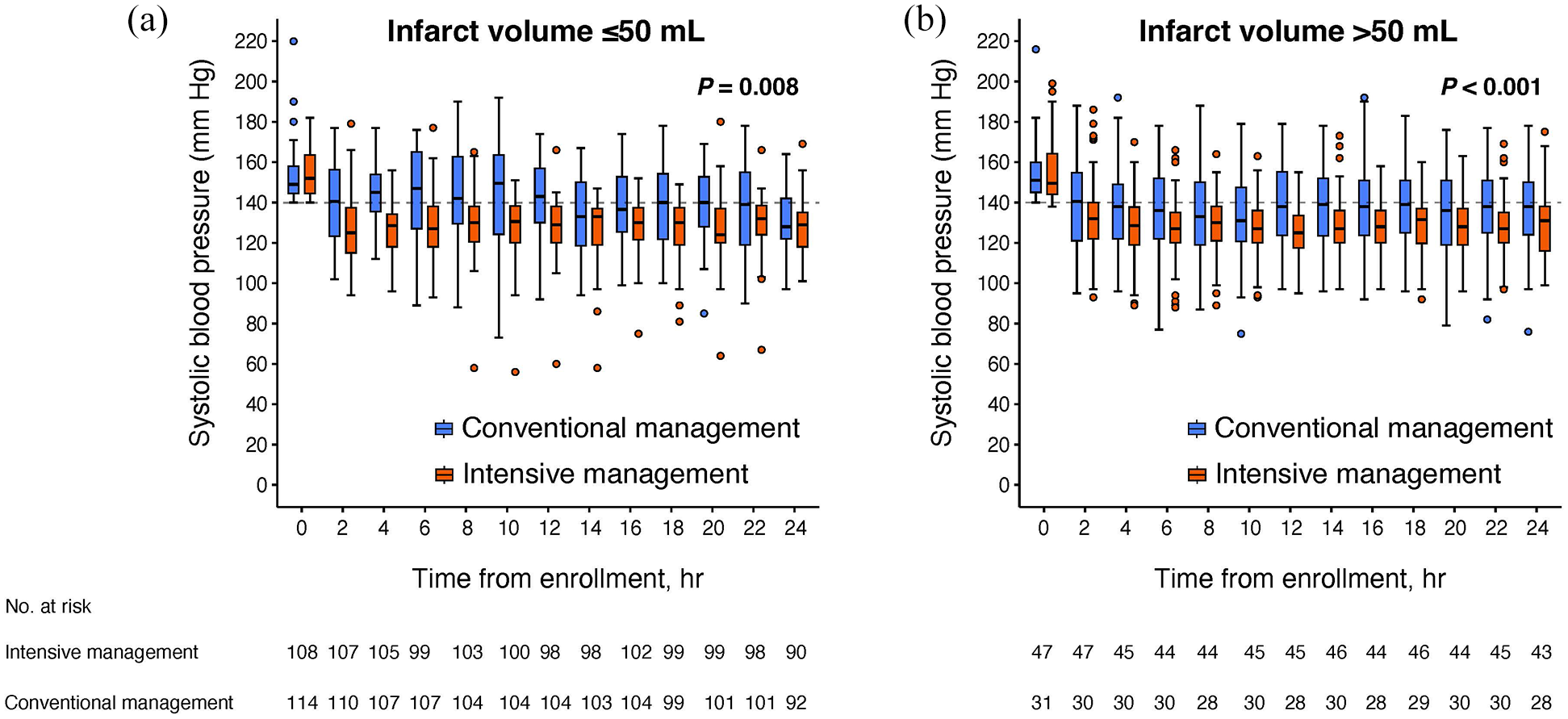
Changes in systolic blood pressure from enrollment to 24 h according to cut-off infarct volume of 50 ml. The mean between-management difference in systolic blood pressure over 24 h was: Infarct volume ⩽50 ml: 6.29 mmHg (95% CI, 1.67–10.91;
Cut-off infarct volume of 50 ml and presence of hemorrhagic transformation.

HI: hemorrhagic infarction; ICH: intracerebral hemorrhage; IQR: interquartile range; PH: parenchymatous hematoma.
Hemorrhagic transformation is classified by Heidelberg Bleeding Classification.
Primary efficacy outcome
In the infarct volume ⩽50 ml group, conventional BP management was significantly associated with a higher rate of functional independence at 3 months (conventional management: 62.3% vs intensive management: 48.1%; adjusted OR [AOR], 2.06 [95% CI, 1.12–3.86];
Outcomes according to cut-off infarct volume of 50 ml.

Abbreviations: EQ-5D-3L, 3-level EuroQoL 5-Dimension Self-Report Questionnaire; EVT, endovascular thrombectomy; ICH, intracerebral hemorrhage; IQR, interquartile range; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale.
Data are presented as number (percentage) of patients for categorical variable and median (IQR) for ordinal variable.
Treatment effects are analyzed with binary logistic regression adjusted for age, sex, onset to randomization time, and NIHSS score before EVT.
Treatment effect is analyzed with ordinal shift regression adjusted for age, sex, onset to randomization time, and NIHSS score before EVT.
Treatment effects are analyzed with linear regression adjusted for age, sex, onset to randomization time, and NIHSS score before EVT.
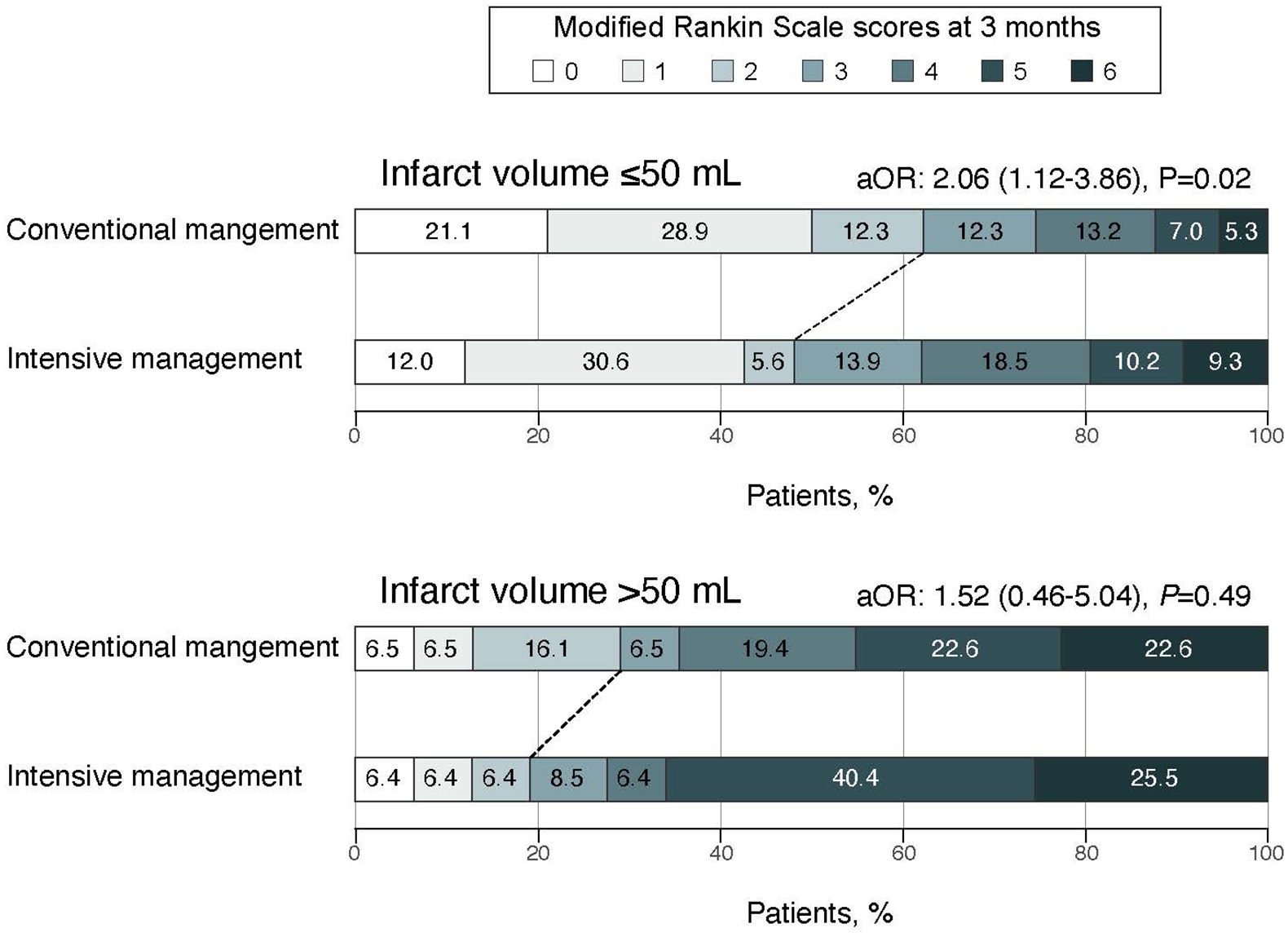
Distributions of modified Rankin Scale scores at 3 months according to the BP managements stratified by cut-off infarct volume of 50 ml.
Primary safety outcomes
In both infarct volume ⩽50 and >50 ml groups, there was neither a significant difference in the symptomatic ICH within 36 h (infarct volume ⩽50 ml group, AOR, 0.19 [95% CI, 0.01–1.21];
Secondary outcomes
In the ⩽50 ml infarct volume group, a shift toward better 3-month mRS outcomes was observed with conventional management compared to intensive management (AOR, 1.67 [95% CI, 1.04–2.70];
Sensitivity analysis
Aside from a higher proportion of good collateral status in the intensive management for patients with infarct volumes below the cut-off, baseline characteristics and infarct volume were similar between BP managements. (Tables S3–S7 and Figure S2). In the infarct volume ⩽30 and ⩽70 ml groups, the proportion of patients achieving the functional independence at 3 months was significantly higher with conventional BP management compared to intensive BP management. However, in the infarct volume >30 and >70 ml groups, no significant differences were observed between the BP managements (Table S8 and Figure S3). These findings consistent with the results of the main analysis using the infarct volume of 50 ml cut-off. In the infarct volume >50 ml subgroup, the rate of modified Treatment In Cerebral Infarction (mTICI) 3 achievement immediately after EVT differed based on BP management. Therefore, we conducted an analysis using a model that included mTICI 3 achievement as an additional adjustment variable. The results remained consistent with the main analysis (Table S9).
Changes of BP management effect according to infarct volumes
Adjusted predicted probability showed that a significant association between BP managements and functional independence at 3 months depending on infarct volume at 24 h. As infarct volume increased, the difference in predicted probability of functional independence between conventional and intensive BP managements decreased (Figure 3). The intersection of the regression lines at 120 ml was used as a critical cut-off point. Below this threshold, the difference in predicted probability for achieving functional independence between the two managements was statistically significant, favoring conventional management (3-way interaction test, slope difference;
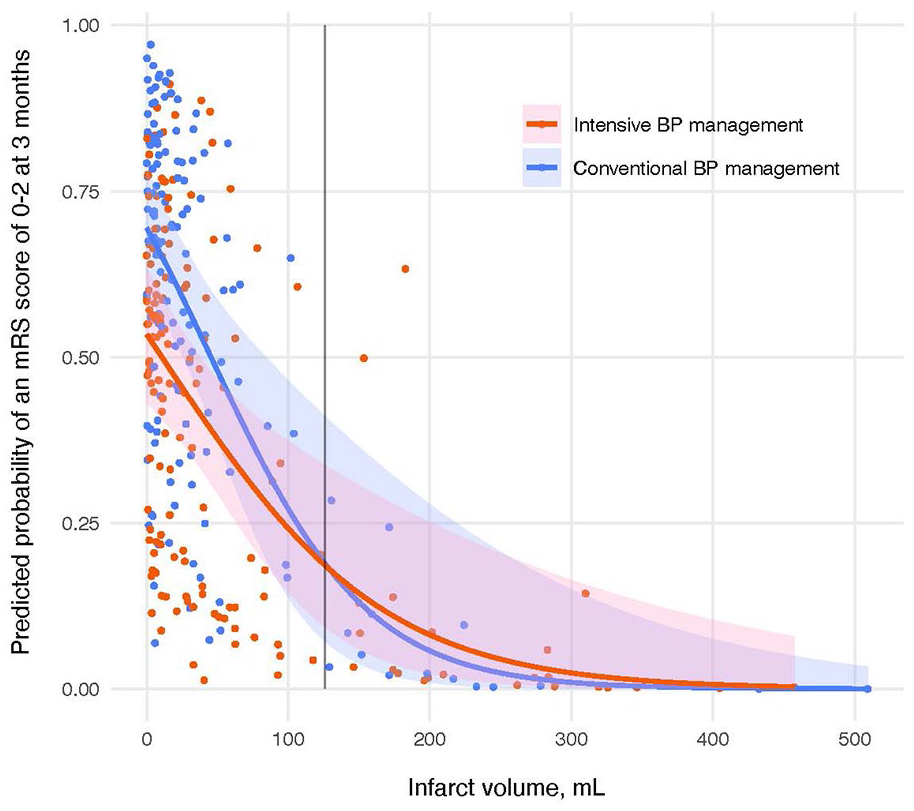
Functional outcome association of the blood pressure management with infarct volumes. The regression curve and confidence region from a logistic regression analysis estimate the probability of achieving functional independence at 3 months (mRS score 0–2) based on infarct volume and BP management with interactions. Predicted probabilities are shown from a regression model adjusted for age, sex, onset-to-randomization time, infarct volume, NIHSS score > 15 before EVT, and BP management, including interaction between infarct volume and BP management. Cut-off values were determined by the crossing point of the regression lines.
Discussion
This secondary analysis of the OPTIMAL-BP trial found that smaller infarct volumes were more favorable for achieving 3-month functional independence with conventional BP management compared to intensive management. As infarct volume increased, the effectiveness of BP management strategies on functional outcomes decreased.
In this study, we also revealed that as infarct volume progressively increased, the difference in the effects of BP management strategies on functional outcomes diminished. Two RCTs on BP management after successful EVT demonstrated that conventional BP management had a higher likelihood of achieving functional independence at 3 months compared to intensive BP management.7,8 Among these trials, the ENCHANTED2/MT (Intensive Blood Pressure Control After Endovascular Thrombectomy for Acute Ischemic Stroke) measured pre-EVT core volume using CT perfusion, with a median core volume of 8 (IQR, 0–27) ml in the intensive BP management and 7 (IQR, 0–28) ml in the conventional BP management. Similarly, in the OPTIMAL-BP trial, which also showed positive results, the proportion of ASPECTS 9–10 was 56.6% in the intensive management and 62.7% in the conventional management, suggesting a likely small core volume in both groups. In contrast, the BP-TARGET (Blood Pressure Target in Acute Stroke to Reduce Hemorrhage After Endovascular Therapy) trial, which yielded neutral results, did not provide data on core volume or ASPECTS. However, it reported a higher baseline NIHSS score compared to aforementioned two trials, suggesting the presence of a larger core infarct. The BEST-II (Blood Pressure After Endovascular Stroke Therapy-II) trial, with a median ASPECTS of 7–8 and a core infarct volume ranging from a median of 5–20.5 ml across groups, focused on patients with larger infarcts compared to previous two studies. In this context, RCTs with smaller infarction volumes demonstrated that conventional BP management led to better outcomes compared to intensive BP management. In contrast, as core infarct volume increases, trials tend to yield neutral results. These results underscore the importance of considering infarct volume when tailoring BP management to improve functional outcome after successful reperfusion by EVT.
This study suggests that the benefits of conventional BP management over intensive BP management may be better in patients with an infarct volume of ⩽50 ml, rather than those with >50 ml. The favorable outcomes associated with conventional BP management in cases of smaller infarct volumes are consistent with the findings of the OPTIMAL-BP study. 8 Higher BP within a certain threshold in oligemic area may enhance perfusion, supporting brain tissue in areas at risk of ischemia. 24 In contrast, with larger infarct volume, much of the hypoperfused area has already progressed to ischemic core, explaining why intensive BP reduction was associated with negative impact on the oligemic state. Furthermore, in cases of larger infarct volume, the potential benefits of improved perfusion from conventional BP management may be offset by an increased risk of ICH, compared to intensive BP management. 25 This suggests that tailored BP management based on infarct volume threshold is crucial, as our results indicate that permissive BP control, aiming to keep SBP below 180 mmHg, was beneficial for patients with small infarct volume.
Post-EVT infarct volume can reflect the rate of infarct growth, whether rapid or slow, which is a key determinant of functional outcome.26,27 In our study, the observation that more patients with an infarct volume >50 ml received intensive BP management than conventional BP management (47 in intensive management vs 31 in conventional management) suggests that intensive management may have contributed to faster infarct growth. Consequently, accelerated infarct growth due to intensive BP management in patients with smaller infarct volumes could be a possible mechanism underlying our study’s findings.
Little is known about BP management after EVT in patients with large core. In one large core trial, approximately 58% of patients experienced any ICH, with 8% developing symptomatic ICH following EVT. 14 In this study, 52.3% of patients experienced some form of ICH, including 8% with symptomatic ICH, yet intensive BP management did not yield better outcomes compared to conventional BP management. In cases of spontaneous ICH, it is recommended to maintain acute phase SBP below 140 mmHg.20,28 However, there are no established BP guidelines for managing hemorrhagic transformation after successful reperfusion therapy. Thus, further research with additional hypotheses is necessary to determine the optimal BP management strategy following successful EVT in patients with a large core infarct.
This study has several limitations. First, the analysis in this study was conducted using infarct volume measured at 24 ± 12 h from randomization after EVT, rather than the core infarct volume measured prior to EVT. Additionally, since post-EVT BP management could have influenced the final infarct volume, we were unable to fully account for its impact. However, our study observed that as final infarct volume decreased, the difference in outcomes between the BP management strategies became more pronounced. Considering that the mean infarct growth after successful EVT is approximately 15 ml, 29 it is likely that differences in outcomes between BP management strategies would be more evident in patients with a smaller initial infarct core. Future research is needed to investigate the impact of BP management based on initial core volume prior to EVT. Second, the relationship between BP variability and study outcomes was not evaluated. Since BP variability during the first 24 h after EVT is associated with 90-day mortality and disability, 30 caution is needed when interpreting the results, as BP variability could act as a significant confounder. Third, reduced sample size as partitioned between the two groups may have limited ability to detect a difference. The results of these secondary group analyses are considered exploratory, given the potential for type I errors due to multiple comparisons. Finally, given that all participants in the OPTIMAL-BP trial were from South Korea, the results need to be interpreted while considering potential ethnic differences.
Conclusions
This secondary analysis of the OPTIMAL-BP trial revealed that smaller infarct volumes were linked to greater benefits from conventional BP management in achieving 3-month functional independence compared to intensive BP management. In contrast, as infarct volume increased, the influence of BP management strategies on functional outcomes decreased. Further studies are needed to optimize post-EVT BP management for patients with large core infarcts following successful reperfusion.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251335204 – Supplemental material for Blood pressure management based on infarct volume after successful endovascular thrombectomy
Supplemental material, sj-docx-1-eso-10.1177_23969873251335204 for Blood pressure management based on infarct volume after successful endovascular thrombectomy by Jae Wook Jung, Kwang Hyun Kim, Jaeseob Yun, Haram Joo, Young Dae Kim, JoonNyung Heo, Hyungwoo Lee, Byung Moon Kim, Dong Joon Kim, Na Young Shin, Bang-Hoon Cho, Seong Hwan Ahn, Hyungjong Park, Sung-Il Sohn, Jeong-Ho Hong, Tae-Jin Song, Yoonkyung Chang, Gyu Sik Kim, Kwon-Duk Seo, Kijeong Lee, Jun Young Chang, Jung Hwa Seo, Sukyoon Lee, Jang-Hyun Baek, Han-Jin Cho, Dong Hoon Shin, Jinkwon Kim, Joonsang Yoo, Minyoul Baik, Yo Han Jung, Yang-Ha Hwang, Chi Kyung Kim, Jae Guk Kim, Il Hyung Lee, Jin Kyo Choi, Chan Joo Lee, Sungha Park, Soyoung Jeon, Hye Sun Lee, Sun U Kwon, Oh Young Bang, Ji Hoe Heo and Hyo Suk Nam in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of Patient-Centered Clinical Research Coordinating Center (PACEN) funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2024-00398053).
Ethical approval
This study was approved by the ethics committee at Severance hospital (approval number: 4-2019-1208) and all participating centers.
Informed consent
Written informed consent was obtained from participants or their legally authorized representatives before the study began.
Guarantor
Hyo Suk Nam, MD, PhD
Contributorship
JWJ, JY, HSL, and HSN performed data analysis. JWJ, KHK, JY, HJ, YDK, JH, HL, BMK, DJK, NYS, BC, SHA, HP, SS, JH, TS, YC, GSK, KS, KL, JYC, JHS, SL, JB, HC, DHS, JK, JY, MB, YHJ, YH, CKK, JGK, IHL, JKC, CJL, SP, SUK, OYB, JHH, and HSN obtained local data. JWJ and HSN wrote the first draft of the manuscript or prepared the figures. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
The anonymized OPTIMAL-BP dataset will be available on reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
