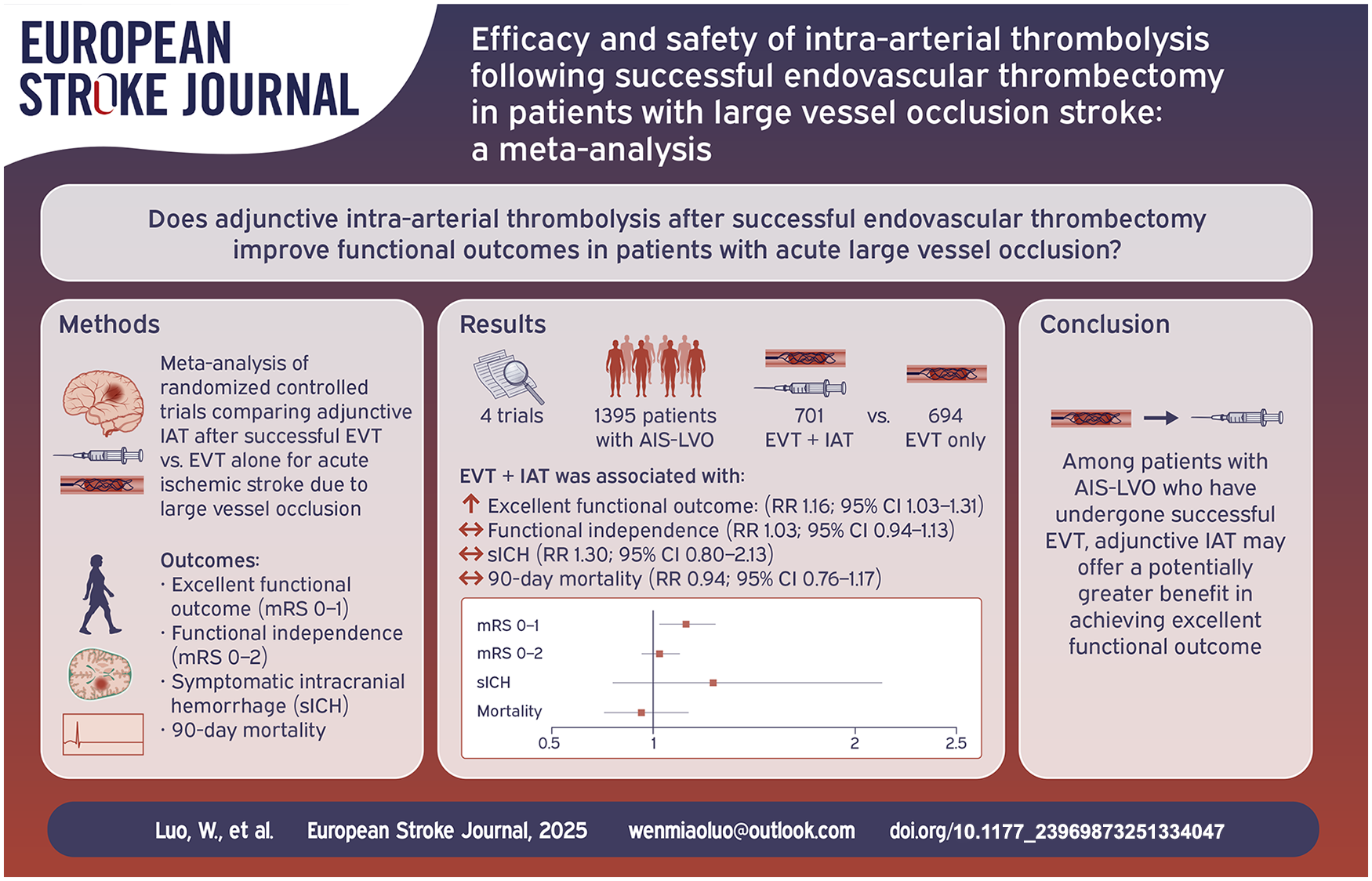
Abstract
Introduction:
The impact of intra-arterial thrombolysis (IAT) following successful endovascular thrombectomy (EVT) in patients with acute ischemic stroke (AIS) due to large vessel occlusion (LVO) remains uncertain. This study aims to assess the efficacy and safety of IAT as an adjunct to EVT in patients with AIS-LVO.
Patients and methods:
We searched PubMed, Embase, and Cochrane databases to identify randomized controlled trials (RCTs) that compared EVT + IAT with EVT-only for AIS-LVO. The primary outcome was an excellent functional outcome, defined as a modified Rankin Scale (mRS) score of 0–1 at 90 days. Secondary outcomes included functional independence (mRS 0–2), symptomatic intracranial hemorrhage (sICH), and mortality. The pooled data were analyzed using random-effects models.
Discussion and conclusion:
A total of four RCTs, involving 1395 patients, were included in the analysis. The results showed that patients who received EVT + IAT had a significantly higher likelihood of achieving an excellent functional outcome at 90 days compared to those who received EVT-only (risk ratio [RR], 1.16; 95% confidence interval [CI], 1.03–1.31). No statistically significant differences were observed between the EVT + IAT and EVT-only groups in terms of 90-day functional independence (RR, 1.03; 95% CI, 0.94–1.13), sICH (RR, 1.30; 95% CI, 0.80–2.13), or 90-day mortality (RR, 0.94; 95% CI, 0.76–1.17).
Conclusions:
Among patients with AIS-LVO who have undergone successful EVT, the use of adjunct IAT, compared to no additional treatment, was associated with a greater likelihood of achieving an excellent functional outcome at 90 days.
Systematic review registration:
CRD42024602099
Background
Acute ischemic stroke (AIS) caused by large vessel occlusion (LVO) is a significant contributor to disability and mortality in adults. 1 Previous randomized controlled trials (RCTs) have shown that endovascular thrombectomy (EVT) significantly reduces disability and mortality at 90 days compared to medical therapy alone for patients with AIS-LVO.2,3 Despite successful reperfusion achieved in 71% of patients through EVT, only 27% are free from disability at 90 days. 4 Key determinants of functional outcomes after EVT include the no-reflow phenomenon, initial tissue damage, cerebral edema, reperfusion injury, procedural factors, and variations in postoperative management. 5 Among these, the no-reflow phenomenon is modifiable and may benefit from intra-arterial thrombolysis (IAT) to reduce clot burden in the microcirculation. However, data on the efficacy and safety of adjunctive IAT following successful EVT in AIS-LVO patients has been limited and conflicting.6–9
The CHOICE trial 6 demonstrated that, in patients with AIS-LVO who underwent successful EVT, IAT significantly increased the proportion of patients achieving excellent functional outcome at 90 days, assessed by a modified Rankin Scale (mRS) score of 0–1, compared to those receiving placebo. However, the trial was prematurely terminated after enrolling only 60% of the planned participants, potentially affecting its applicability to clinical practice. Recently, three published trials (ATTENTION-IA, 7 POST-UK, 9 and POST-TNK 8 ) showed no statistically significant reduction in disability at 90 days with IAT after successful EVT. Notably, the POST-TNK trial 8 observed a significantly higher incidence of intracranial hemorrhage in the IAT group compared to the control group. To gain further insights, a pooled analysis was conducted, combining the available evidence from these RCTs.
Methods
The study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 10 and was registered in the International Prospective Register of Systematic Reviews with the identifier CRD42024602099.
Eligibility criteria
Comparative studies published in English in peer-reviewed journals, adhering to the Population, Intervention, Comparison, Outcome, and Study design framework were considered eligible for inclusion.
Population: Adult patients with AIS-LVO who underwent successful EVT, defined as achieving Thrombolysis in Cerebral Infarction grades 2b–3. Interventions: IAT as an adjunctive procedure following successful EVT. Comparison: Procedures that were terminated without adjunctive IAT. Outcomes: The primary outcome was an excellent functional outcome, defined as an mRS score of 0–1 at 90 days. Secondary outcomes included functional independence (mRS 0–2), symptomatic intracranial hemorrhage (sICH), and mortality. Study design: RCTs. Meeting abstracts were excluded due to the possibility of insufficient information.
Search strategy
We conducted a search in PubMed, Embase, and the Cochrane Central Register of Controlled Trials for articles published between January 1, 2015, and January 20, 2025. The search terms included “Acute Ischemic Stroke,” “Large Vessel Occlusion,” “Mechanical Thrombectomy” or “Endovascular Therapy,” and “Intra-Arterial Thrombolysis.” The complete search strategy was detailed in eTable 1. Additionally, we manually reviewed the reference lists of previous reviews and the included studies.
Data extraction
All search results were managed using EndNote software. Two independent reviewers screened studies based on predefined eligibility criteria, categorizing them as eligible, ineligible, or uncertain. Exclusion criteria were established in accordance with the Population, Intervention, Comparison, Outcome, and Study design framework, beginning with a review of the title and abstract. 11 Studies that could not be excluded based on title and abstract screening were deemed potentially relevant and underwent full-text review. Final inclusion required consensus between both reviewers regarding eligibility. When persistent disagreements occurred after discussion, a third reviewer arbitrated the decision.
The extracted information comprised essential elements such as study characteristics, baseline patient data, and outcomes of interest. Study characteristics encompassed the trial name, year of publication, country of origin, number of patients, and patient-specific information. Baseline patient data included age, gender, prior medication history, medical history, National Institutes of Health Stroke Scale (NIHSS) score, Alberta Stroke Program Early CT Score (ASPECTS), and initial systolic blood pressure. Outcomes of interest were sICH assessed both neurologically and radiologically, as well as the mRS score at 90 days. If disagreements could not be resolved through discussion, a third reviewer mediated to ensure consensus.
Risk of bias assessment
The Cochrane Risk of Bias Tool were used to assess the risk of bias in the studies, taking into account factors such as sequence generation, allocation concealment, blinding, and other potential sources of bias. 12 The overall risk of bias rating for each study was determined by the lowest rating across all the evaluated criteria. For example, if any domain was rated as high risk of bias, the study was classified as having high overall bias. Any inter-reviewer disagreements during these steps were resolved through iterative discussions until consensus.
Quality of evidence
Two reviewers independently evaluated the quality of evidence using the Grading of Recommendations, Assessment, Development, and Evaluation system (GRADE), 13 taking into account factors such as risk of bias, inconsistency, indirectness, imprecision, and publication bias. The evidence was subsequently categorized into four levels: very low, low, moderate, or high. Disagreements were adjudicated by a third reviewer through structured deliberation.
Statistical analysis
Statistical analyses were conducted using Review Manager 5.4 software. Summary measures for categorical outcomes were reported as risk ratios (RRs) with 95% confidence intervals (CIs). Given clinical and methodological heterogeneity, pooled analysis was performed using a random-effects model. 14 Statistical heterogeneity across trials was assessed using the Cochran Q test (p < 0.1 indicating significance) and quantified by the I² statistic. An I² value >50% was considered indicative of significant heterogeneity. 15 Subgroup analyses were conducted for the primary outcome based on gender, age, stroke etiology, intravenous thrombolysis (IVT) status before EVT, artery occlusion site, and thrombolytic agent type. Sensitivity analyses using the leave-one-out method were performed to investigate the influence of each study on the overall effect size estimate. Additional sensitivity analyses were conducted by incorporating additional gray trials and excluding the two trials that prohibited IVT. Due to the limited number of studies included in each analysis (<10), an Egger regression test to assess publication bias was not feasible. The threshold for statistical significance was set at a p-value <0.05.
Results
Trial selection and characteristics
A total of 472 potentially eligible records were identified through electronic screening, with an additional 5 records discovered through citation searches. Ultimately, four RCTs meeting the inclusion criteria were selected: CHOICE, 6 POST-TNK, 8 POST-UK, 9 and ATTENTION-IA. 7 Figure 1 illustrates the study selection process. POST-TNK 8 and POST-UK 9 trials involved participants with acute anterior circulation stroke. CHOICE trial 6 included participants with acute anterior and posterior circulation strokes, with outcomes reporting only two cases of posterior circulation stroke receiving adjunctive IAT. ATTENTION-IA trial 7 focused on acute posterior circulation stroke. The thrombolytic agents used were alteplase (0.225 mg/kg) in the CHOICE trial, 6 tenecteplase (0.0625 mg/kg) in the POST-TNK 8 and ATTENTION-IA 7 trials, and urokinase (100,000 IU) in the POST-UK trial. 9 IVT was permitted for eligible participants in the CHOICE 6 and ATTENTION-IA 7 trials, but not in the POST-TNK 8 and POST-UK 9 trials. The number of participants in each trial ranged from 52 to 271, with an average age of 65–73 years, and the proportion of female patients varied from 19% to 46%. The included participants had an average NIHSS score ranging from 15 to 23 and an average ASPECTS score ranging from 8 to 9. Table 1 summarizes the characteristics of the included RCTs.
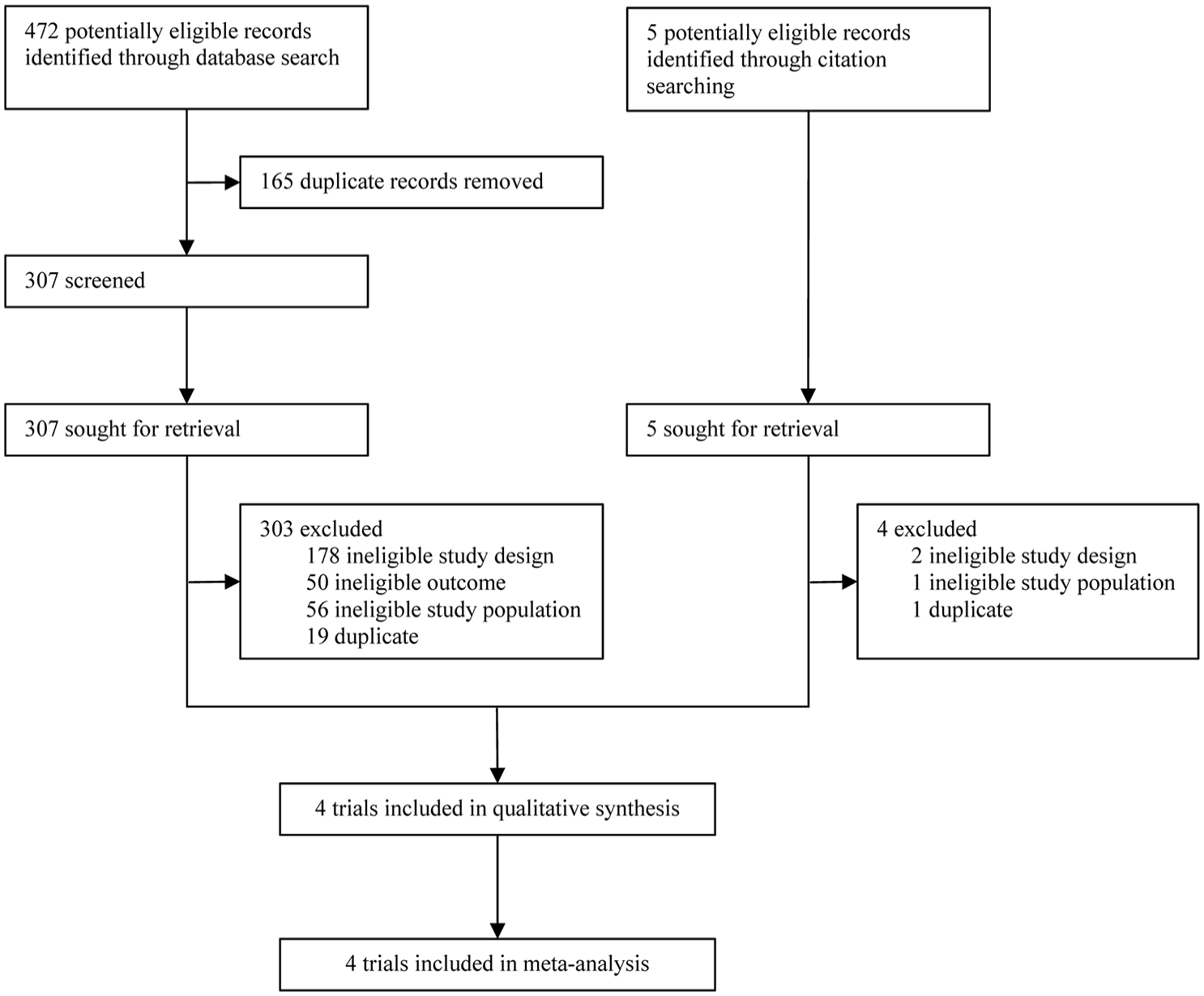
Study flow diagram.
Baseline characteristics of included trials.
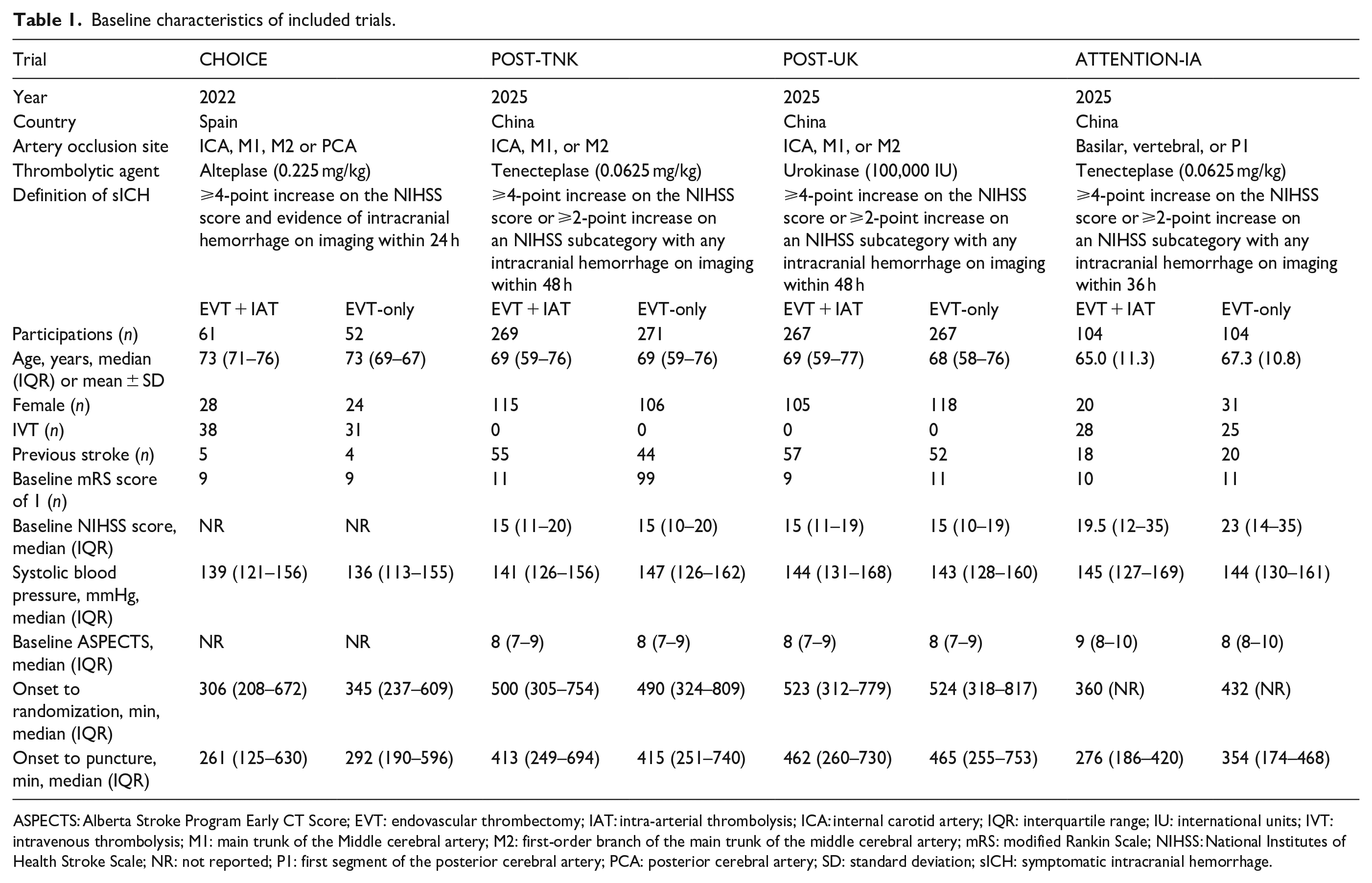
ASPECTS: Alberta Stroke Program Early CT Score; EVT: endovascular thrombectomy; IAT: intra-arterial thrombolysis; ICA: internal carotid artery; IQR: interquartile range; IU: international units; IVT: intravenous thrombolysis; M1: main trunk of the Middle cerebral artery; M2: first-order branch of the main trunk of the middle cerebral artery; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; NR: not reported; P1: first segment of the posterior cerebral artery; PCA: posterior cerebral artery; SD: standard deviation; sICH: symptomatic intracranial hemorrhage.
Risk of bias
The analysis for risk of bias indicated a low risk of bias for the POST-TNK, 8 POST-UK, 9 and ATTENTION-IA 7 trials. However, the CHOICE trial 6 had a small sample size and was terminated prematurely, which posed a moderate risk of bias. Details of the risk of bias are presented in eFigure 1.
Primary outcome
Across all four studies included,6–9 rates of excellent functional outcome (mRS 0–1) were compared between the EVT + IAT and EVT only groups among 1392 patients. The EVT + IAT group had a higher rate of 90-day mRS scores of 0–1 (46.3%; 324/700) compared with the EVT only group (39.6%; 274/692), resulting in a statistically significant RR of 1.16 (95% CI, 1.03–1.31; p = 0.01; Figure 2). No notable heterogeneity was detected (I² = 0%, p for heterogeneity = 0.56).
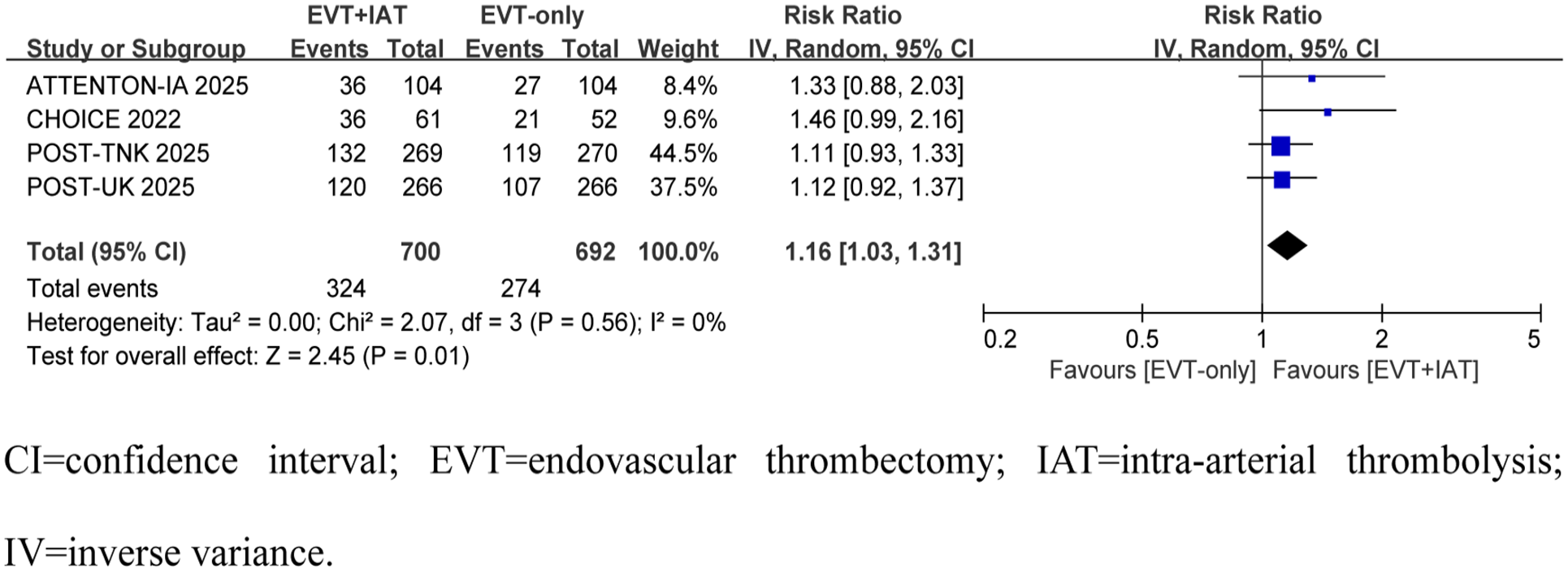
Forest plots for the primary outcome of modified Rankin Scale Score of 0–1 at 90 days.
Subgroup analyses of primary outcome
Figure 3 displays the results of prespecified subgroup analyses for the primary outcome. The ATTENTION-IA, 7 POST-TNK, 8 and POST-UK 9 trials provided adjusted RR values for confounding factors, whereas the CHOICE trial 6 did not. The CHOICE trial reported only two cases of posterior circulation patients who received adjunctive IAT. Due to data limitations, we classified them into the anterior circulation group. Notably, sex-related differences in IAT efficacy were observed. Specifically, EVT + IAT, as a first-line treatment, significantly benefited female patients in achieving an mRS score of 0–1 (RR, 1.41; 95% CI, 1.15–1.71), but not male patients (RR, 1.02; 95% CI, 0.88–1.19; p for heterogeneity = 0.01). No statistically significant interactions were found in additional exploratory analyses of the tested subgroups.
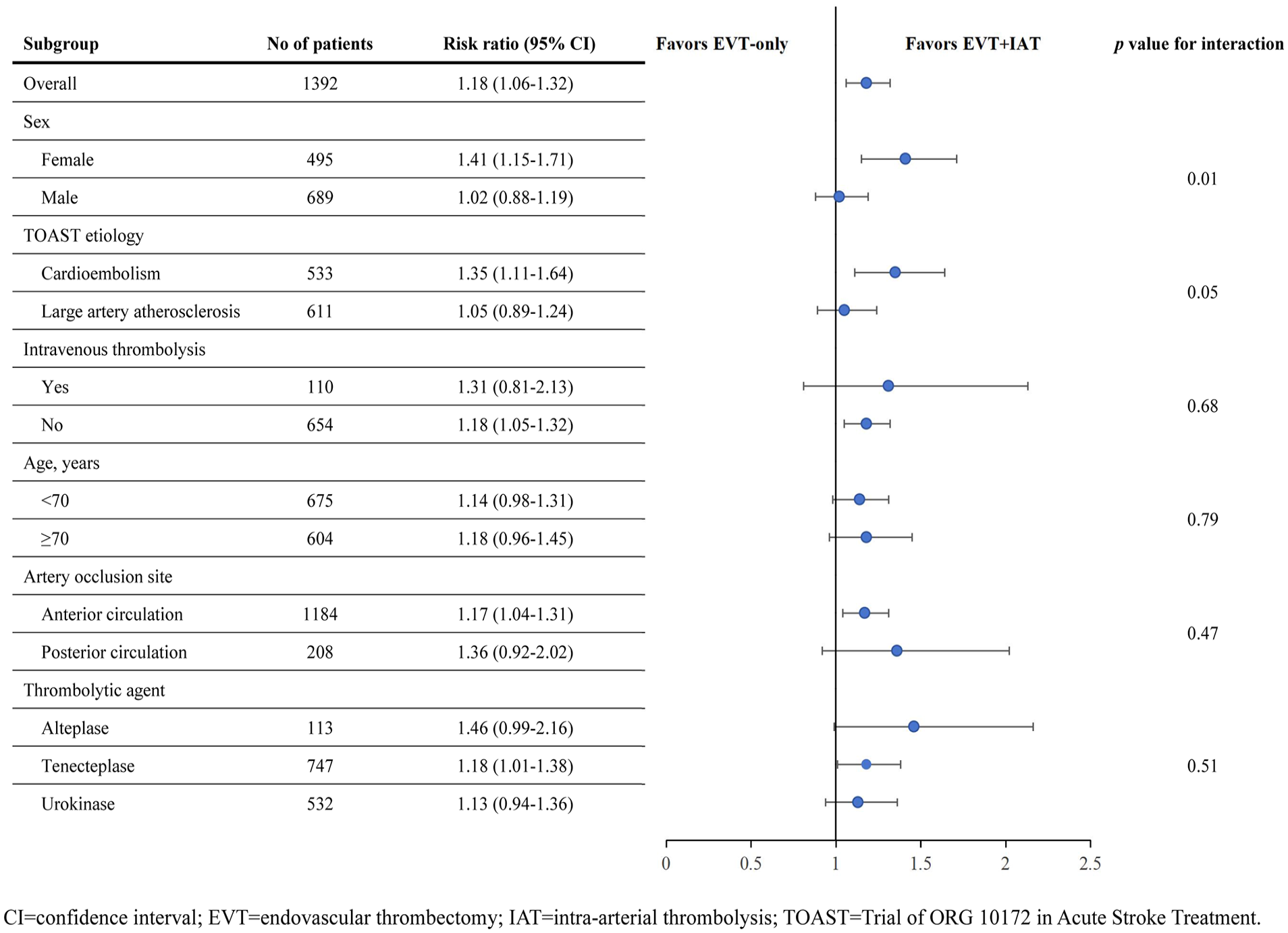
Subgroup analysis for the primary outcome of modified Rankin Scale Score of 0–1 at 90 days.
Secondary outcomes
Data on secondary outcomes were provided by all four studies.6–9 In the EVT + IAT group, 389 of 700 patients (55.6%) achieved functional independence (mRS 0–2), compared to 373 of 692 patients (53.9%) in the EVT only group. The calculated RR of 1.03 (95% CI, 0.94–1.13; p = 0.51) showed no statistical significance (Table 2 and eFigure 2A). No heterogeneity was found among the studies (I² = 0%, p = 0.97).
Summary of all secondary outcomes.
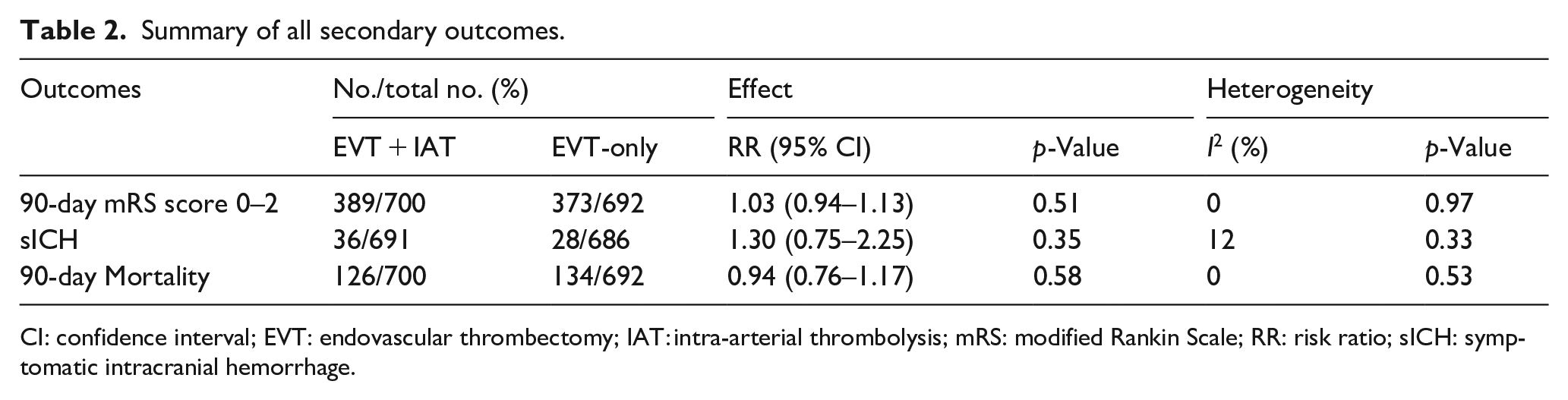
CI: confidence interval; EVT: endovascular thrombectomy; IAT: intra-arterial thrombolysis; mRS: modified Rankin Scale; RR: risk ratio; sICH: symptomatic intracranial hemorrhage.
The definition of sICH varied across the four studies. In the CHOICE trial, 6 sICH was defined as any hemorrhage with a ⩾ 4-point increase in NIHSS at 24 h. The POST-TNK 8 and POST-UK 9 trials specified sICH as an NIHSS increase of ⩾ 4 points or a subcategory increase of ⩾2 points with any intracranial hemorrhage on imaging at 48 h, based on the modified Heidelberg Bleeding Classification. Similarly, 16 the ATTENTION-IA trial 7 used the same criterion but at 36 h. The rate of sICH was higher in the EVT + IAT group (36 of 691 [5.2%]) than in the EVT-only group (28 of 686 [4.1%]), but the RR of 1.30 (95% CI, 0.75–2.25; p = 0.35) indicated no significant difference (Table 2 and eFigure 2B). Minimal heterogeneity was observed among the studies (I² = 12%, p = 0.33).
Death occurred in 126 of 700 patients (18.0%) in the EVT + IAT group and 134 of 692 patients (19.4%) in the EVT-only group (RR, 0.94; 95% CI, 0.76–1.17; p = 0.58). No significant differences were observed in safety outcomes (Table 2 and eFigure 2C), and no heterogeneity was found among the studies (I² = 0%, p = 0.53).
Sensitivity analyzes
The results of the sensitivity analyses aligned with the primary outcome. A comprehensive breakdown of each assessed outcome is provided in eTable 2 and eFigures 3 and 4.
Quality of evidence
The quality of evidence was rated as moderate for all outcomes. Detailed assessment information is outlined in eTable 3.
Discussion
Principal findings
In patients with AIS-LVO who underwent successful EVT, adjunctive IAT was more likely to lead to excellent functional outcome at 90 days compared to placebo. Importantly, we observed a gender-specific difference in the efficacy of IAT: EVT + IAT showed significant benefits in achieving excellent functional outcome among female patients, but not among males. There were no significant differences between the adjunctive IAT group and the control group in terms of 90-day functional independence, sICH, or 90-day mortality.
Relation to previous research
This pooled analysis aligns with previous research, emphasizing the potential role of IAT in patients with AIS-LVO who have undergone successful EVT. 6 Preliminary evidence from the CHOICE trial 6 suggested that adjunctive IAT after successful EVT increased the likelihood of excellent functional outcome at 90 days, hypothesizing that supplementary fibrinolysis could effectively clear microvascular residual thrombi and enhance tissue reperfusion. This hypothesis was supported by the mechanism of IAT, which targets thrombus dissolution through fibrinolytic system activation, potentially compensating for EVT’s limitations in addressing microvascular thrombi that may persist despite angiographically successful recanalization.17,18 However, the generalizability of the CHOICE trial 6 was limited by its small sample size and premature termination, necessitating further validation. 6 In contrast, the ATTENTION-IA, 7 POST-TNK, 8 and POST-UK 9 trials did not find statistically significant improvements in primary outcomes with adjunctive IAT, similar to studies on bridging IVT that showed a trend toward outcome improvement but did not reach statistical significance.19,20 Synthesizing these trials with our research results, we draw two key conclusions: Firstly, we found that adjunctive IAT following successful EVT provided moderate yet statistically significant benefits in improving excellent functional outcome, 13 further highlighting the importance of addressing reperfusion inefficacy mechanisms such as microvascular obstruction. Secondly, the observed functional improvement was limited, benefiting only approximately 10% of patients, underlining the critical importance of precise patient selection. Additionally, subgroup analyses revealed significant gender disparities in the effect of IAT, with female patients demonstrating greater benefits from adjunctive IAT after successful EVT. This finding may be attributed to gender differences in fibrinolytic activity, hormonal influences, or vascular biology. Estrogen and progesterone confer cardiovascular protection in females by regulating lipid metabolism and sustaining vascular homeostasis. Key mechanisms include enhancing endothelial function, suppressing vascular smooth muscle cell proliferation, and stabilizing coagulation-fibrinolysis equilibrium. These synergistic actions preserve vascular integrity and significantly mitigate the risk of cardiovascular disease. 21 While these mechanisms require further investigation, the results emphasize the potential of gender-specific factors in personalized treatment.
Implications for clinicals and guidelines
These findings carry significant implications for clinical practice and guideline development. Although the CHOICE trial 6 initially hinted at the potential efficacy of adjunctive IAT following successful EVT, its limitations – including a small sample size and premature termination – have hindered its inclusion in clinical guidelines. Furthermore, the neutral results of the ATTENTION-IA, 7 POST-TNK, 8 and POST-UK 9 trials have fueled skepticism regarding the practicality of adjunctive IAT post-EVT and raised concerns about potential risks and limited benefits. Despite these uncertainties, this pooled analysis provides moderate-quality evidence supporting the safety and efficacy of adjunctive IAT after successful EVT, indicating a significant improvement in favorable functional outcomes without a notable increase in adverse events. Adjunctive IAT may serve as an effective tool to reduce disability among post-stroke patients, particularly those with successful EVT.22,23 The key limitation was the small number of studies included and the significant a priori heterogeneity in research designs. Additional sensitivity analyses were conducted by incorporating additional gray trials and excluding the trials that prohibited IVT to validate the stability of the outcomes. Ongoing studies, such as the Safety and Efficacy of Intra-Arterial Tenecteplase for Non-Complete Reperfusion of Intra-Cranial Occlusions (TECNO; NCT05499832) and Treatment of Persistent Distal Occlusion After Successful Proximal Recanalization in Thrombectomy (2BE3; NCT06034847), aim to evaluate the utility of adjunctive IAT in broader patient populations and diverse clinical settings.4,24 The results of these studies will be crucial for updating meta-analyses and refining clinical recommendations.
Strengths and weaknesses of the study
The strength of this pooled analysis lies in its pioneering comprehensive evaluation of the efficacy and safety of adjunctive IAT following successful EVT in patients with AIS-LVO.25,26 Secondly, the robustness of the findings was confirmed through predefined subgroup and sensitivity analyses, enhancing the reliability of the results. Furthermore, the application of the Grading of Recommendations Assessment, Development and Evaluation system to assess the certainty of the evidence further bolstered the clinical relevance of the findings.
Indeed, the study has several limitations. First, the key limitation was the inclusion of only four RCTs, with considerable variability in patient characteristics across the trials, such as differences in anterior versus posterior circulation stroke and the use of IVT before EVT. Despite conducting subgroup analyses and sensitivity analyses to mitigate the impact of these confounding factors, the limited number of studies hindered the statistical power needed to draw definitive conclusions. Secondly, Due to data limitations, we classified patients from the CHOICE trial into the anterior circulation stroke group for subgroup analysis of occlusion sites. We acknowledge the limitations of this subgroup analysis. Thirdly, while significant sex differences in the efficacy of adjunctive IAT were observed, the underlying mechanisms remain unknown, highlighting the complexity of biological responses to interventions and emphasizing the necessity for further mechanistic research. Finally, due to the lack of detailed individual-level data, we were unable to make finer adjustments for baseline characteristics of patients (such as NIHSS score, age differences, etc.), which may affect the interpretation of the results. Therefore, future research needs to collect more comprehensive individual-level data to allow for more detailed multivariate analysis, thereby enabling a more accurate assessment of the efficacy, cost-effectiveness, and potential mechanisms of adjunctive IAT.
Conclusion
This study evaluated the efficacy and safety of adjunctive IAT after successful EVT through through a pooled analysis. The results demonstrated that IAT significantly increased the incidence of excellent functional outcome, with potentially greater efficacy in female patients. There were no significant differences observed in improving functional independence or safety.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251334047 – Supplemental material for Efficacy and safety of intra-arterial thrombolysis following successful endovascular thrombectomy in patients with large vessel occlusion stroke: A meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873251334047 for Efficacy and safety of intra-arterial thrombolysis following successful endovascular thrombectomy in patients with large vessel occlusion stroke: A meta-analysis by Zeni Yang, Wentai Zhang, Yonggang Xu, Yuwei Ding, Chao Liu, Zhiyuan Shen, Jiwei Wu, Yu Guo and Wenmiao Luo in European Stroke Journal
Footnotes
Acknowledgements
Not applicable.
Abbreviations
ASPECTS Alberta Stroke Program Early CT Score
AIS Acute ischemic stroke
CI Confidence interval
EVT Endovascular thrombectomy
IAT Intra-arterial thrombolysis
ICA Internal carotid artery
IVT Intravenous thrombolysis
LVO Large vessel occlusion
MCA Middle cerebral artery
mRS modified Rankin Scale
NIHSS National Institutes of Health Stroke Scale
PCA Posterior cerebral artery
RCT Randomized controlled trial
RR Risk ratio
sICH Symptomatic intracranial hemorrhage
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Not applicable. Our research is a meta-analysis, and all data used were extracted from openly accessible published journal articles.
Informed consent
Not applicable.
Guarantor
Corresponding author Wenmiao Luo is willing to take full responsibility for the article, including the accuracy and appropriateness of the reference list.
Contributorship
Study conception and design: WML, YG, and JWW. Acquisition, analysis, and interpretation of data: all authors. Manuscript drafting: ZNY and WTZ. Critical revision for important intellectual content: all authors. Final approval of the manuscript: all authors. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
