Abstract
Introduction:
Seasonality in the incidence of stroke has been examined in numerous studies, yet data on whether it differs with age are limited. To fill this gap, we utilized a largescale dataset from Israel.
Patients and methods:
We retrieved data of all hospitalizations for ischemic stroke (IS), transient ischemic attack (TIA) and intra cerebral hemorrhage (ICH) from 2000 to 2020. We maintained separate datasets for IS/TIA and ICH, divided into five age groups: 18–49, 50–59, 60–69, 70–79, and 80+. We modeled the monthly incidence using a generalized additive model. The seasonal effect was defined by the rate ratio (RR) of each month compared to the annual mean.
Results:
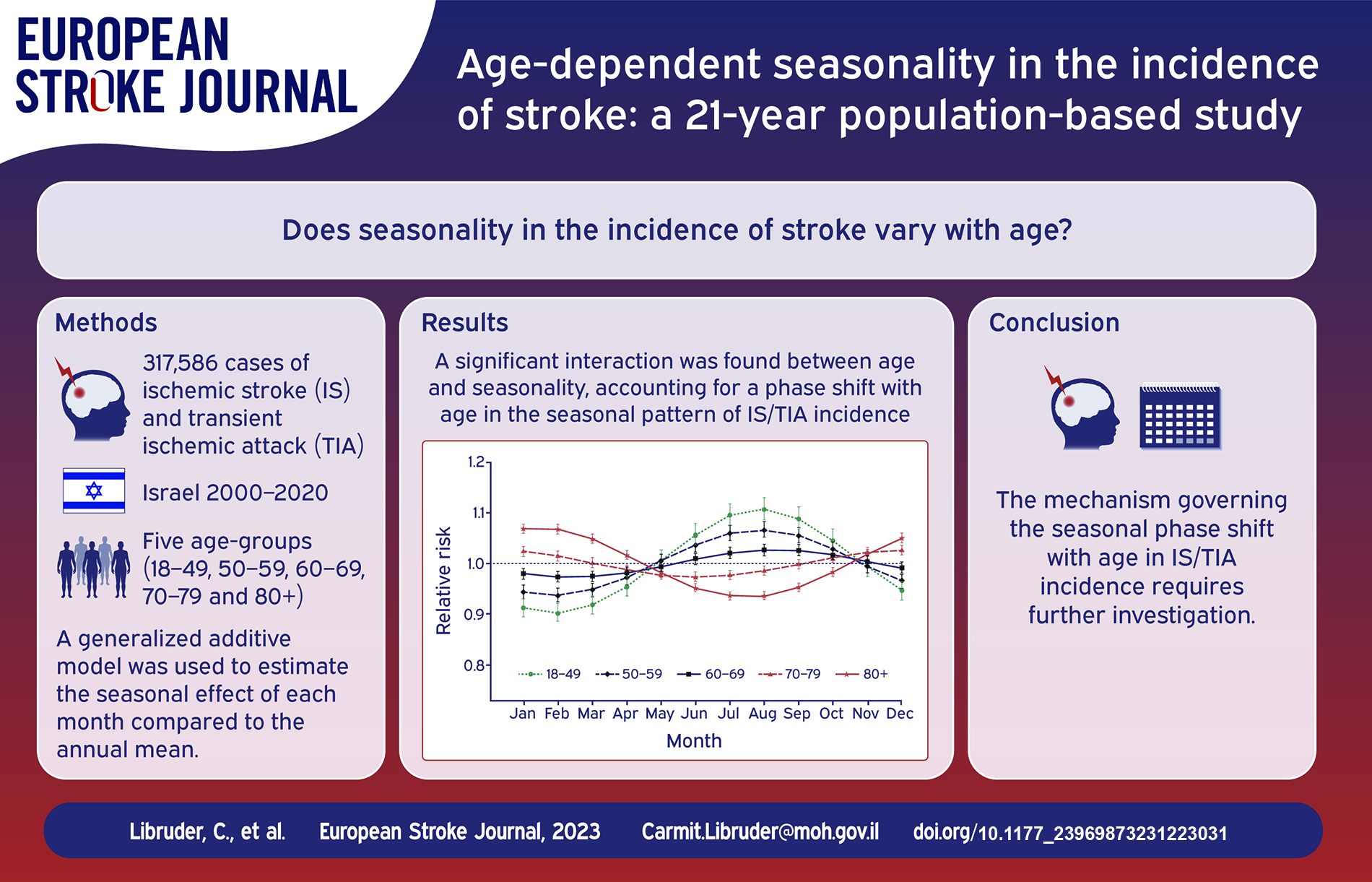
The analysis included 317,586 and 23,789 events of IS/TIA and ICH respectively. We found an interaction between age and seasonality, accounting for a phase shift with age in the seasonal pattern of IS/TIA incidence. For cases under 70 years, the peak was during summertime and the RRs increased with decreasing age, reaching 1.11 (95% CI 1.09–1.13) at the youngest age group. In contrast, among the elderly, a winter peak was observed and the RRs increased with age to 1.07 (95% CI 1.06–1.08) at the oldest age group. For ICH, a winter/autumn peak was identified and the RRs increased with age to 1.20 (95% CI 1.16–1.24).
Conclusions:
Our finding of age-dependent seasonal patterns in the occurrence of stroke, suggests closer monitoring of cardiovascular risk factors during wintertime among elderly individuals. The mechanism governing the seasonal phase shift with age in IS/TIA incidence, requires further investigation.
Introduction
According to the Global Burden of Disease study 2019, stroke ranks second among the causes of disability-adjusted life-years (DALYs) in adults aged 50 and above, despite declines in age-standardized incidence rates. 1 Of note, a decrease in the incidence of stroke is evident in the elderly population, 2 while rates are still increasing among young adults. 3
Seasonal variations in the incidence of stroke have been examined in numerous studies worldwide. Most studies reported a winter peak in the occurrence of stroke,4–12 others found no seasonal variation13–15 and fewer showed a summer peak 16 or an autumn peak. 17 Data on whether seasonality of stroke differs with age are limited. Most studies did not stratify the results by age groups. Those who did, may have lacked the power to reveal a significant seasonal trend in the young age categories, due to smaller numbers, 8 or used broad age categories (⩾70 vs <70), which may not adequately represent young adults. 7
The etiologies of ischemic cerebrovascular events, as well as the prevalence of cardiovascular risk factors, are known to differ with age18,19 and vary across different seasons.20,21 This may lead to seasonal variation in the occurrence of events according to age, and thus hold important epidemiological and practical implications to better understand and reduce the burden of stroke.
We utilized a largescale dataset from Israel in order to study and estimate seasonal variations in the incidence of stroke by age.
Methods
Data source and case definition
In this nationwide population-based study, we retrospectively retrieved data of all hospitalizations for stroke that occurred in Israel between January 1, 2000 and 31 December 2020, in individuals aged 18 and above. Data were obtained from the Israel National Hospital Discharge Registry (INHDR) of the Israeli Ministry of Health. All acute care hospitals in Israel report to the INHDR. The database is regularly updated via hospitals’ electronic medical records. Patient identification numbers are encrypted in order to protect patient’s privacy. The INHDR contains demographic and hospitalization data, including diagnoses coded according to the International Classification of Disease, 9th edition (ICD-9). 22 The relevant ICD-9 codes obtained included 431 for intra cerebral hemorrhage (ICH); 433.X1, 434.XX, 436 and 997.02 for ischemic stroke (IS) and 435.X for transient ischemic attack (TIA). In our main analysis, the two ischemic cerebrovascular events (IS and TIA) were combined together as they share similar pathophysiological mechanisms. 23
Cases of stroke that occurred within 90 days from a previous event for the same individual were removed from the datasets (6% and 7% of the cases in the IS/TIA and ICH datasets respectively), to exclude the effect of increased hazard for recurrent stroke events when estimating the seasonal effect.
Each reported case included the following details: main diagnosis, admission date, age at admission, gender and institute code. We associated each case with an administrative district in Israel (North, Center, South, Haifa, Tel-Aviv, and Jerusalem) according to the institute location.
Statistical analyses
We maintained two separate datasets for IS/TIA and ICH. Each dataset was divided into five age groups (18–49, 50–59, 60–69, 70–79, 80+) and was aggregated into time series of monthly incidence, according to the date of admission. The number of events per month was normalized for the month length by dividing the monthly number of events by the number of days in that month and multiplying by 30. For the calculation of incidence rates per 100,000 people, the size of the general population by age groups in the modeled period (as well as by gender and district for the supplemental tests) was obtained from reports of the Israeli Central Bureau of Statistics. 24
We modeled the monthly incidence of IS/TIA and ICH events using a generalized additive model (gam procedure in the mgcv package in R 25 ). The model includes a monthly seasonality and a smoothed non-parametric effect of trend in time (see Supplemental Material, page 1, for the detailed model). The monthly seasonal effect is presented throughout this work as the rate ratio (RR) of each month compared to the annual mean. To estimate and test age specific seasonality or trend we added interaction terms between age and these terms, as we explain in page 2 of the Supplemental Material. Additionally, we examined whether age specific seasonal effects were modified by gender (male/female), geographical area (according to the main districts of Israel) or time-period (comparing 2000–2009 to 2010–2019). Details are given in page 3 of the Supplemental Material.
The importance of the seasonality effect for the model fit was measured by the partial pseudo-R squared of McFadden 26 defined as the amount of reduction (%) in the deviance between the model with just the time trend and the model that includes also the seasonality.
The data underlying this article will be shared on reasonable request to the corresponding author.
Results
The analysis included 317,586 events of IS/TIA and 23,789 events of ICH. The mean age (± standard deviation) was 71.6 ± 13.4 and 70.3 ± 14.9 for IS/TIA and ICH cases respectively. The fraction of male cases was 53.1% and 57.7% for IS/TIA and ICH respectively. Table 1 summarizes the number of cases and the average monthly incidence rates per age group, in each dataset. As shown in Table 1, both the number of cases and the average monthly incidence rates increase with age. Figure 1 presents the monthly incidence rates of IS/TIA per age group during 2000–2020. It includes a smoothed line that indicates the possible seasonal effect. Figure S1 shows the incidence for the ICH data. While the incidence rates of IS/TIA show a steady decline with time in the older age groups (60+), they do not show a pattern in the younger age groups (18–59). For ICH cases, the incidence rates stay relatively stable in all age groups.
Total number of events and average monthly incidence rates for IS/TIA and ICH per age group, 2000–2020.
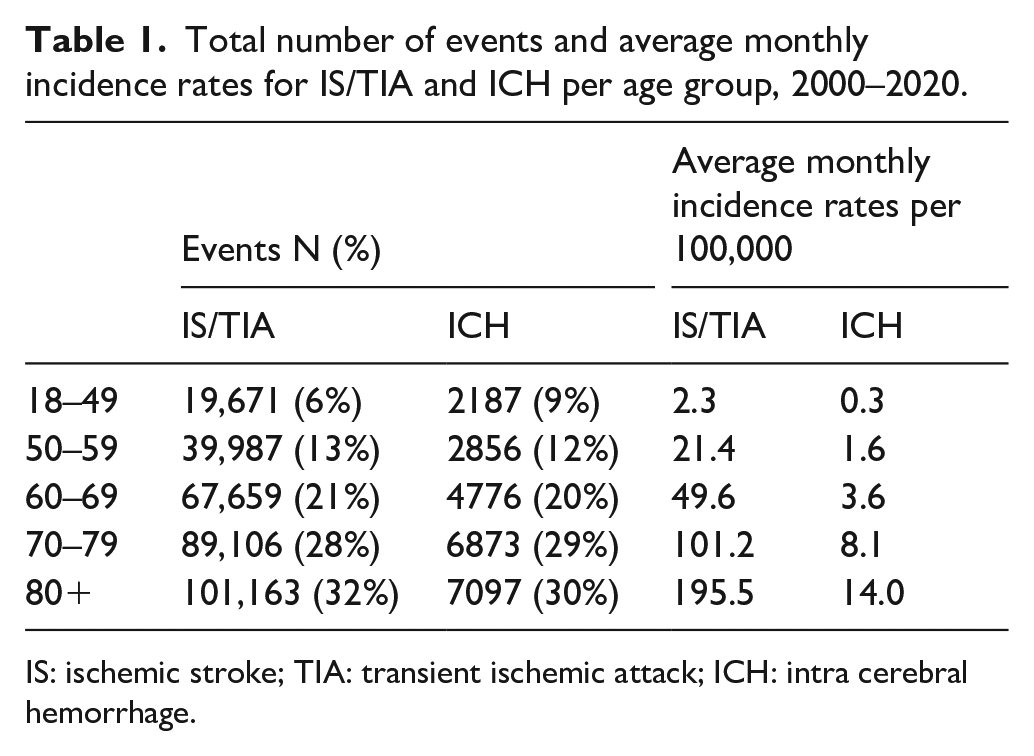
IS: ischemic stroke; TIA: transient ischemic attack; ICH: intra cerebral hemorrhage.
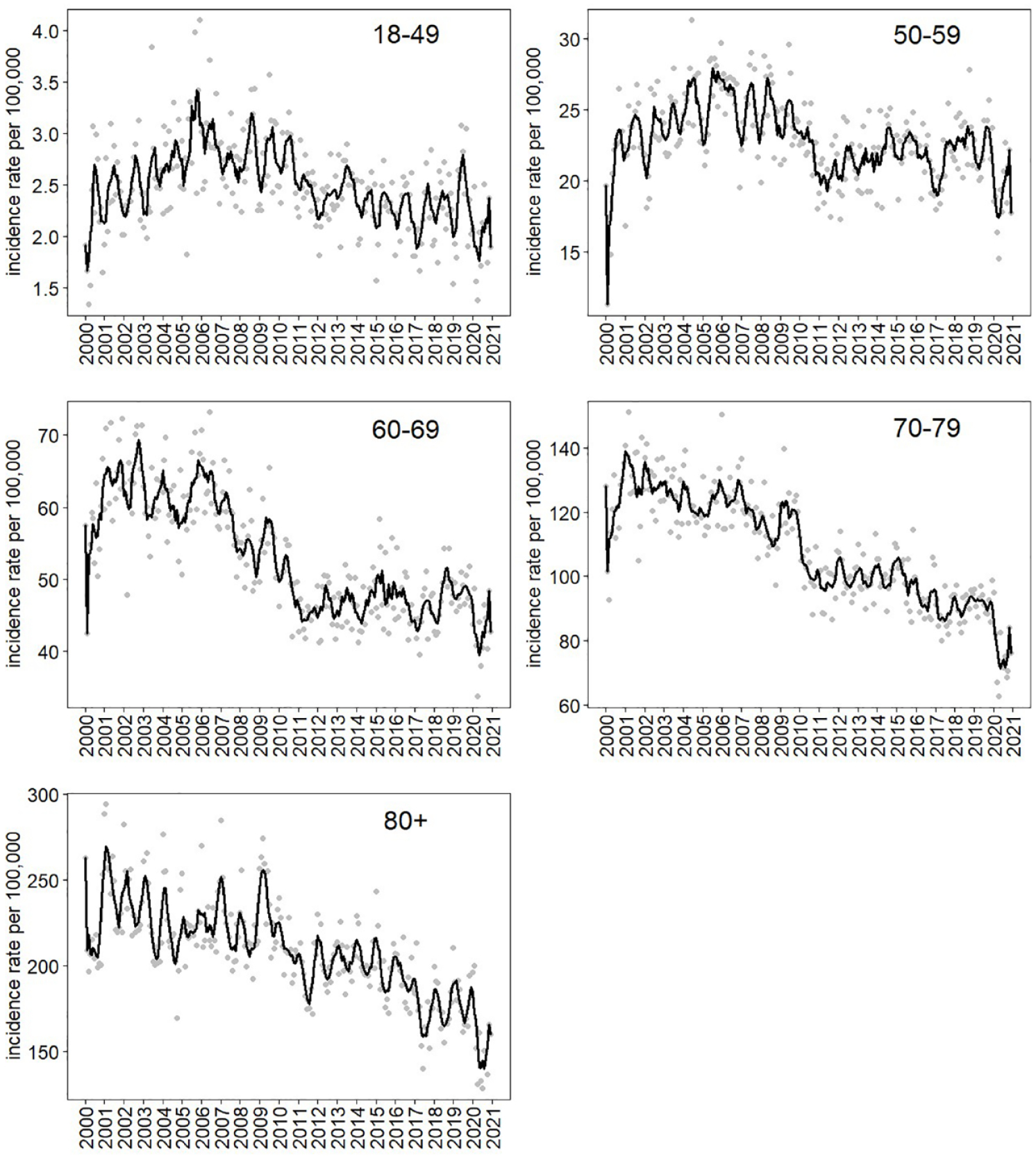
IS/TIA incidence rates per month by age groups, 2000–2020. Points indicate the monthly incidence rate per 100,000. Lines were obtained using a moving-average smoothing of the observed cases with a 3-month window. IS: ischemic stroke; TIA: transient ischemic attack.
Testing the age modification effect, our analysis supported modeling a separate seasonal effect and trend for each age group; that is, incorporating both age-seasonality and age-trend interaction terms (Table S1). Table 2 and Figure 2 present estimated measures related to the seasonal effects, obtained by fitting the model for each age group in each dataset. Table 2 details the maximal RRs (i.e. the height of the peak in the sinusoidal wave which measures the difference between the seasons) for the incidence of IS/TIA and ICH, and the seasonal peak month per each age group. Figure 2 plots the estimated monthly RRs for each age group in each dataset. All RRs reported are compared to the annual geometric mean.
The maximal monthly rate ratio (RR),* and the seasonal peak month for IS/TIA and ICH per age group.
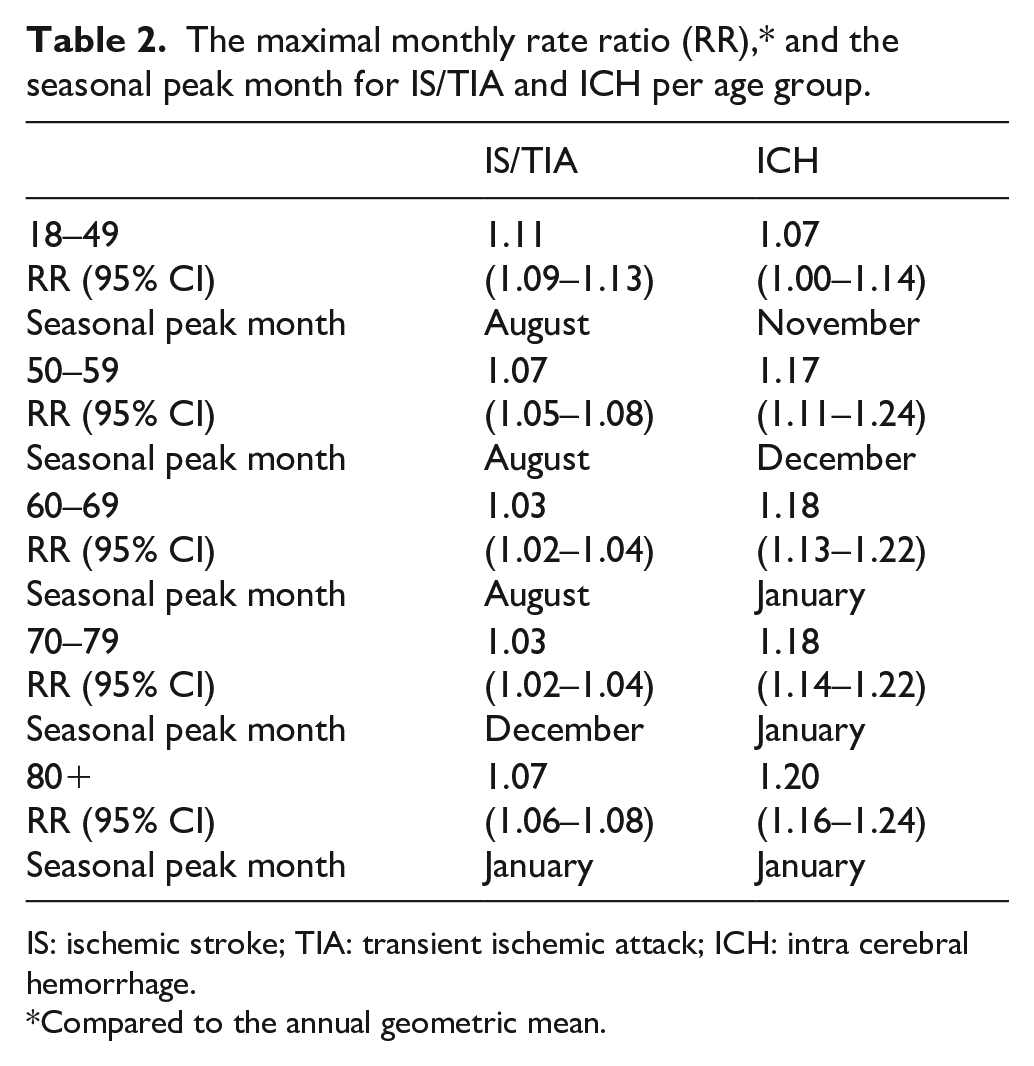
IS: ischemic stroke; TIA: transient ischemic attack; ICH: intra cerebral hemorrhage.
Compared to the annual geometric mean.
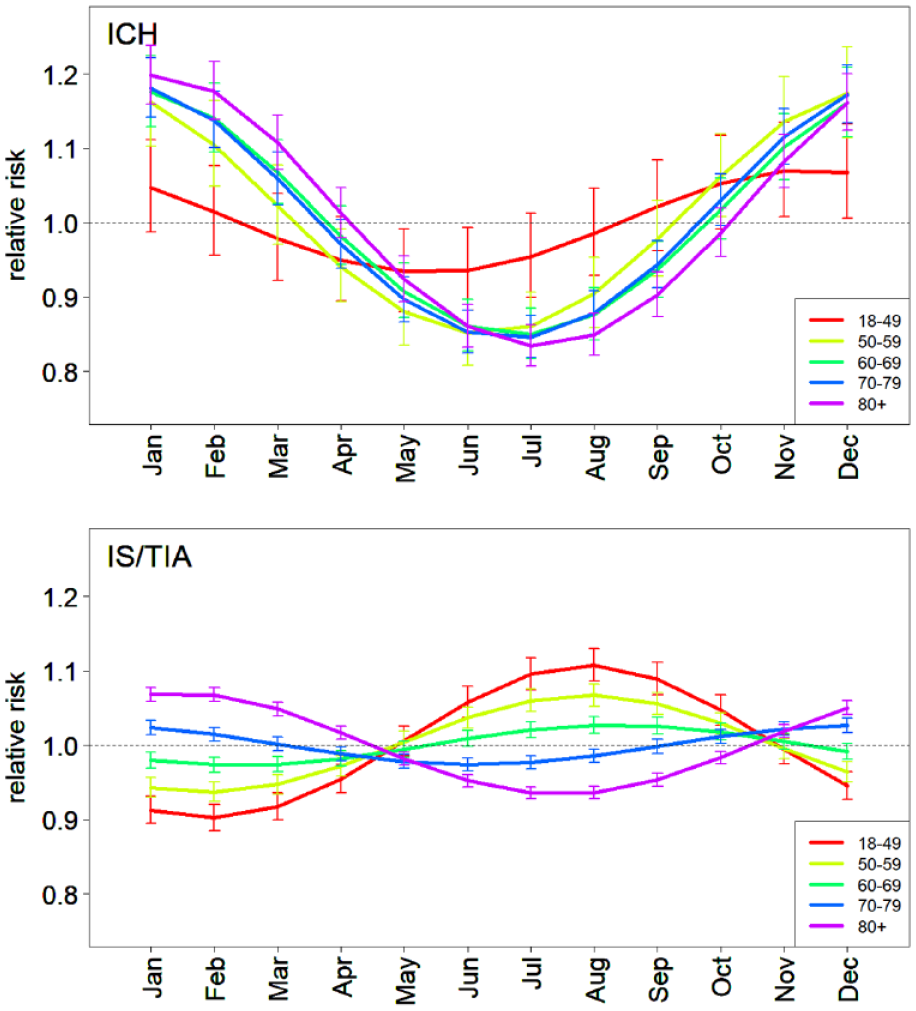
Estimated seasonal effect per month by age groups, 2000–2020. Lines depict the estimated rate ratio of each month (compared to the annual geometric mean). Bars indicate 95% confidence intervals. ICH: intra cerebral hemorrhage; IS: ischemic stroke; TIA: transient ischemic attack.
We found a phase shift with age in the seasonal effect of IS/TIA incidence. For people under 70 years old, the incidence peak is during summertime in Israel (August) while above 70 years of age the peak is during wintertime (December–January). The estimated seasonal effect indicates an increased risk of 11% (RR = 1.11; 95% CI 1.08–1.13) for the youngest age group (18–49). The effect gradually decreases with age, so for the 60–69 age group it is only 3% (RR = 1.03; 95% CI 1.01–1.04), and then increases again after the phase shift to an increased risk of 7% (RR = 1.07; 95% CI 1.06–1.08) in the 80+ age group.
The obtained seasonal effect of ICH shows a peak at wintertime (December-January) in people aged 50 and above, and an autumn peak (November) at the youngest age group (18–49). The estimated seasonal effect indicates an increased risk of 7% (RR = 1.07; 95% CI 1.0–1.12) for the youngest age group and more than twice that for the older age groups (50+), reaching an increased risk of 20% (RR = 1.20; 95% CI 1.16–1.24) in the 80+ age group.
Figure 3 presents the obtained model fit of the IS/TIA data for each age group. It depicts the observed numbers of monthly cases and the expected numbers according to the model. The fit of the ICH model is given in Figure S2. The amount of variance explained by seasonality in each model for each age group is given in Table 3. For ages 80+ the seasonality has a major influence for both types of strokes (25%–27%). In the younger age groups (18–49 and 50–59) the seasonality has a major effect only for IS/TIA. In the middle age groups the seasonality has an effect only in ICH.
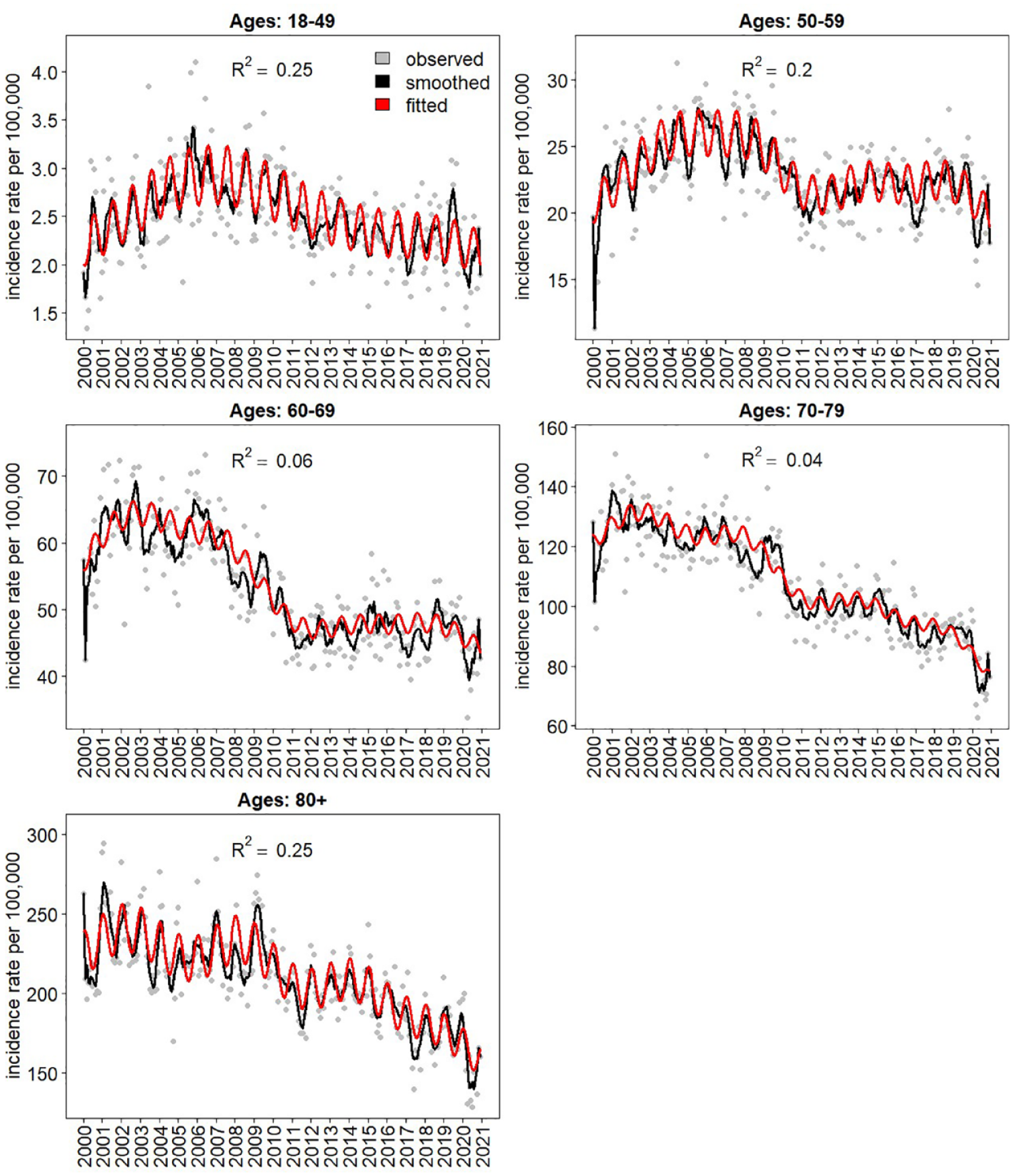
IS/TIA Model fit by age categories, 2000–2020. Points indicate the number of cases per month in the datasets. Lines show the expected number according to the model. IS: ischemic stroke; TIA: transient ischemic attack.
Amount of deviance (%) explained by the seasonality effect for models of IS/TIA and ICH per age group (partial pseudo-R squared of McFadden).
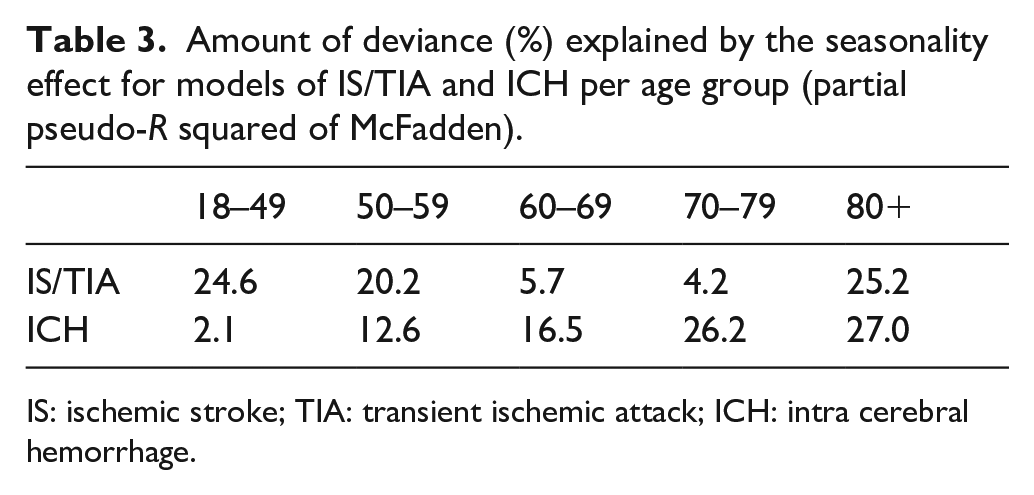
IS: ischemic stroke; TIA: transient ischemic attack; ICH: intra cerebral hemorrhage.
We performed several sensitivity analyses to assess the robustness of our results. For each dataset, we tested the modification effect of gender, district (as a proxy for differences in climate) and period (2000–10 vs 2011–20) on the age-specific seasonal effects. All interactions were found non-significant, except for period in the IS/TIA dataset (Table S2). The similar patterns of the estimated age specific seasonal effects for each category of gender, district and period are shown in Figures S3–6. The differences between the two periods in the IS/TIA dataset (Figure S6) are in the phase and amplitude on the middle groups, but the overall pattern of a monotonic effect by age remains unchanged.
Repeating the main analysis using a finer categorization of age groups to 5-year intervals resulted in a similar monotonic seasonal effect (Figure S7).
Discussion
In this 21-year nationwide population-based study, we found an interaction between age and seasonality in the incidence of stroke. To our knowledge, our study is the first largescale study to report such interaction. As the analysis revealed, an interesting phase shift with age occurred in the seasonal pattern of IS/TIA incidence. Among the elderly (70+) a winter peak and a summer trough were identified, while among younger adults an unexpected opposite pattern of a summer peak and a winter trough was observed. In addition, the seasonal effects were strongest amongst the extreme ages (i.e. oldest and youngest) and gradually decreased until the pattern reversed at around 70 years of age. For ICH incidence, a seasonal pattern of a winter/autumn peak and a summer trough was identified, and the seasonal effect increased with age.
Various studies from different regions of the world support a winter peak in the incidence of stroke.4–12 Seasonality in several cardiovascular risk factors may explain this winter peak, specifically in the elderly population. Fibrinogen, for instance, is an independent risk factor for cardiovascular disease. 27 A seasonal pattern was found for fibrinogen, with higher plasma fibrinogen levels in the cold months compared to the summer months, 28 and the seasonal variation was more pronounced with increasing age. 29 Additionally, winter respiratory infections are associated with increased levels of fibrinogen among older adults 30 and a positive association was found between winter respiratory infections and the incidence of stroke. 31
Seasonality was also found in some of the conventional cardiovascular risk factors, including hypertension and increased cholesterol levels. Blood pressure (BP) exhibits a seasonal variation, with higher levels during winter compared to summer, and the difference in BP levels between the seasons appears to be larger in treated hypertensive patients and in older individuals. 32 Seasonal pattern characterized by higher levels in winter and lower levels in summer was also observed for triglycerides and cholesterol levels, and the excess risk of mortality from cardiovascular disease in winter increased with age. 21 Behavioral changes across seasons may affect seasonality of conventional cardiovascular risk factors. Physical activity tends to decrease during the winter months compared to the summer and spring, independently of the countries’ climate. 33 Additionally, an elevated consumption of fat and cholesterol was noted in the winter. 34
Atrial fibrillation (AF) also constitutes an established risk factor for IS. 35 In an Israeli study, AF episodes recurred more frequently during the cold months than during the summer months. 36 A marked seasonal variation in the rate of AF among stroke patients, with a winter peak and a summer trough was also demonstrated in an Argentinean hospital-based stroke registry, 37 and in cohorts from Denmark and New Zealand. 38 The risk for ischemic cerebrovascular events among AF patients was found to increase with age and was most prominent among people aged 80 and above. 39 Along with the increasing prevalence of AF with age, 40 it may play an important role, particularly in the increased risk for IS/TIA among the elderly during wintertime.
An unexpected finding of our analysis is the phase shift with age in IS/TIA incidence, accounting for a summer peak among young individuals. Of note, as the mean age for a first IS/TIA event is typically older than 65 years, and most cases occur in older individuals, an overall analysis diverts the seasonal pattern to that of the older population. This may explain the finding of a winter peak in most studies. However, two studies that included a relatively young population support our finding of a summer peak among young IS cases. A study conducted in Qatar on a population with a mean age of 54.4 years found an increase in IS incidence during the summer months compared to the winter months, while a higher incidence rate of ICH was observed in the winter months, compared to the hot season. 16 Another study conducted in Bangladesh on a population with a mean age of 59.9 also reported a higher incidence of IS during summer, compared with wintertime, and a higher incidence of hemorrhagic strokes during winter, compared with summertime. 41 In contrast to our results, in a largescale study from Japan that stratified the results by two age groups – older and younger than 70 years of age, a seasonal pattern of a winter peak was demonstrated in both age groups. However, this age-stratified analysis included all strokes, though the seasonal variation was less evident for IS than for ICH. 7 It cannot be ruled out that stratifying the analysis for IS alone by younger age groups would have revealed a different effect. As shown in our study, the effect for the youngest age group was stronger than that for the 60–69 age group (Figure 2).
The mechanism involved in the summer peak among young adults is yet unknown. One possible explanation is a stronger activation of the coagulation system during summer in young adults. While thrombin generation parameters are associated with a first IS in young individuals, 42 higher baseline levels of thrombin generation markers were found during summer in young healthy adults, 43 suggestive of a potential increase in the risk for IS among the young during summertime.
Another possible explanation is that different etiologies for IS/TIA among the young may be responsible for the phase shift seen in this age group. Ischemic cerebrovascular events among young adults demonstrate different distribution of stroke subtypes compared to the elderly and hold different mechanisms. 18 A largescale study from Japan that explored IS seasonality according to subtypes supports this assumption. In that study, cardio-embolic strokes that occurred more frequently in winter were more common among the elderly (age > 75), while lacunar strokes that occurred more frequently in summer were more common among younger adults. 20 Studies exploring seasonality by IS subtypes are scarce. More largescale studies are needed to explore the effect of different IS subtypes and their associations with age and seasonality.
Unlike the summer peak observed in the incidence of young IS/TIA cases, an autumn/winter peak was observed among young ICH cases. Hypertension, which is an established risk factor for stroke and the most frequent risk factor for ICH, 44 holds a stronger association with ICH than IS, specifically among younger adults. 45 As BP levels show a seasonal variation, with elevated levels during winter, 32 this may partially explain the winter peak among young ICH cases.
Strengths and limitations
One of the main strengths of our study is that it is based on a largescale nationwide population-based dataset. The robustness of our results was confirmed in several sensitivity analyses, supporting our unexpected finding of age-dependent seasonality in the incidence of stroke. Nonetheless, our study has several limitations. First, as we lacked information regarding IS subtypes, we were unable to examine whether different IS etiologies lead to differences in seasonal patterns. Second, our data lacks information regarding environmental factors such as temperature or air-pollution. To account for climate differences, we compared the seasonal effects obtained by fitting each district of Israel separately, and found no major spatial differences. Third, as our study included only hospitalized patients, we missed cases of fatal strokes that did not reach hospital and minor strokes/TIAs that were not diagnosed or admitted. However, those are the minority of cases. Finally, Israel has three distinct climate regions: a Mediterranean climate, a hot desert climate, and a narrow transitional strip characterized by semi-arid climate. 46 Our findings might not be generalized to other regions with different climates. As indicated in the Eurowinter Study, the increase in winter mortality with fall in temperature was higher in regions characterized by milder winter climates. 47
Conclusions
Our study suggests closer monitoring and tighter control of cardiovascular risk factors during the winter months, specifically in elderly individuals, to decrease the incidence of stroke. Further studies are needed to explore seasonality of IS/TIA events among young adults, in order to confirm our novel finding of a phase shift with age in the seasonal pattern of IS/TIA events. Further establishing this finding, and in particular its mechanism, is of utmost importance, especially in view of the growing incidence of IS events among the young and the socioeconomic implications of long-term disability in young individuals.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231223031 – Supplemental material for Age-dependent seasonality in the incidence of stroke: A 21-year population-based study
Supplemental material, sj-docx-1-eso-10.1177_23969873231223031 for Age-dependent seasonality in the incidence of stroke: A 21-year population-based study by Carmit Libruder, Rami Yaari, Ronen Fluss, Yael Hershkovitz, Amit Ram, David Tanne, Amit Huppert and Inbar Zucker in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Approval of an ethics committee was not required for the current analyses, which were conducted on fully anonymized datasets.
Informed consent
The Israeli Ministry of Health collects hospitalization data (individual-based but anonymized) for regulatory purposes, in accordance with Israeli regulations; the requirement for written informed consent is thus waived.
Guarantor
CL.
Contributorship
All authors contributed to the study conception and design. Material preparation was performed by YH, AR and IZ. Data analysis was performed by RY, RF and AH. The first draft of the manuscript was written by CL and RY, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
