Abstract
Purpose:
Small subcortical infarction (SSI) accounts for approximately 25% of ischemic strokes and shares a comparable recurrence rate of cardiovascular events with other stroke subtypes. This study aimed to evaluate the efficacy and safety of various antiplatelet for secondary prevention of SSI by network meta-analysis (NMA).
Methods:
We systematically searched Medline, Embase, Cochrane Library, and Web of Science from inception to October 2024 for randomized controlled trials (RCTs). Efficacy outcomes included: incidence of major adverse cardiovascular events (MACEs), rates of any stroke and ischemic stroke recurrence. Safety outcomes included: incidence of intracranial hemorrhage, severe bleeding, any bleeding events, and mortality.
Findings:
A total of 24 RCTs involving 47,507 SSI patients were included in systematic review. The NMA included 19 RCTs (39,137 patients). The NMA demonstrated that Cilostazol showed the best efficacy in preventing MACEs (surface under the cumulative ranking curve (SUCRA): 90.0%). cilostazol significantly reduced the incidence of MACEs compared to aspirin (OR, 0.66; 95% CI, 0.49–0.89), ticlopidine (OR, 0.65; 95% CI, 0.43–1.00), dipyridamole (OR, 0.61; 95% CI, 0.42–0.90), vorapaxar (OR, 0.51; 95% CI, 0.35–0.74), Sarpogrelate (OR, 0.62; 95% CI, 0.40–0.97), and placebo (OR, 0.51; 95% CI, 0.37–0.71). Regarding safety, aspirin plus clopidogrel and vorapaxar was associated with a significantly increased risk of severe bleeding events compared to the control.
Discussion and Conclusions:
Cilostazol may be the most effective agent for preventing cardiovascular event recurrence. Aspirin plus clopidogrel and vorapaxar may be not recommended due to heightened bleeding risks.
Registration:
International prospective register of systematic reviews (PROSPERO) - CRD42024607819

Introduction
Small subcortical infarction (SSI), also known as lacunar infarction, is caused by occlusion of small penetrating arteries, which typically measure <20 mm in diameter and occur in the subcortical white matter, brainstem, basal ganglia, or thalamus.1,2 SSI accounting for approximately 25% of all acute ischemic strokes. 3 Despite their small size and often mild symptoms frequently overlooked by patients and clinicians, SSI can result in severe neurological deficits, gait disturbances, vascular cognitive impairment, and even mortality.4–6 Moreover, previous cohort studies have demonstrated that the recurrence rate of cardiovascular events in SSI is comparable to other ischemic stroke subtypes, 7 with reports suggesting the stroke recurrence rate of up to 20% during a 3-year follow-up period. 8 The underlying mechanisms include atherosclerotic occlusion at the orifice of the penetrating arteries, microatheroma in the proximal segment of arteries, and lipohyalinosis in the distal segment of arteries. 9 Antiplatelet therapy is currently the cornerstone for secondary prevention in SSI patients.
Randomized controlled trials (RCTs) investigating secondary prevention strategies for SSI or acute lacunar stroke have explored various antiplatelet therapies, yet the relative efficacy and optimal choice of these therapies remain uncertain. A meta-analysis published in 2015 concluded no significant differences between aspirin and other antiplatelet therapies in preventing recurrent strokes after acute lacunar infarction. 10 However, conventional meta-analysis has limitations, being restricted to direct comparisons between two interventions and unable to provide indirect comparisons or rank efficacy among multiple agents. Network meta-analysis (NMA), a methodology developed in recent years, enables the simultaneous integration of direct and indirect evidence, facilitating the comprehensive ranking of efficacy and safety among multiple interventions. 11 Previous NMAs on antiplatelet therapies for acute lacunar infarction were limited in scope, with a small number of included studies and antiplatelet agents, failing to incorporate several large RCTs specifically targeting SSI and omitting safety outcomes. 12
In this study, we systematically reviewed evidence from RCTs comparing antiplatelet therapies for secondary prevention in SSI or acute lacunar infarction. Using NMA, we aimed to identify the most effective antiplatelet therapies to reduce recurrent events in high-risk SSI patients.
Methods
This systematic review and network meta-analysis was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 13 The study protocol was registered on PROSPERO (Registration number: CRD42024607819). Ethical approval was not required as this study did not involve the collection of new demographic or biological data from humans or animals.
Search strategy
In October 2024, a systematic search was performed in Medline, Embase, Web of Science, and Cochrane Library databases with no publication date restrictions and limited to English-language articles. Keywords included terms related to SSI such as “minor stroke,” “moderate stroke,” “mild stroke,” “subcortical stroke,” “subcortical infarct*,” “lacunar infarct*,” “lacune,” “RSSI,” “SSI,”* and “SSSI,” etc. and antiplatelet therapies like “ASA,” “DAPT,” “platelet aggregation inhibitors,” “antiplatelet,” “dipyridamole,” “aspirin,” “clopidogrel,” “ticlopidine,” “prasugrel,” “ticagrelor,”* and “cilostazol,” etc. Randomized controlled trials (RCTs) were identified using terms such as “randomised controlled trial,” “randomized controlled trial,” “RCT,” “random*,” and “placebo,” etc. To cover studies including SSI patients as subgroups, broader ischemic stroke terms like “cerebral infarct*,” “brain infarct*,” “cerebrovascular disease,” “cerebrovascular accident,” and “ischemic stroke,” etc. were also included. Additionally, the reference lists of relevant reviews and included studies were screened for eligible trials. The search strategy for the MEDLINE database is summarized in Supplemental Table S1.
Inclusion and exclusion criteria
Two neurologists independently screened the literature in a two-step process: (1) title and abstract review to exclude irrelevant studies and (2) full-text review to identify eligible studies. Discrepancies were resolved by a senior neurologist. Inclusion criteria: (1) Patients with SSI or acute lacunar infarction (no diameter restriction). (2) Interventions involving antiplatelet agents, alone or in combination. (3) Comparators included placebo or other antiplatelet agents. (4) Study design: RCTs. Exclusion criteria: (1) Patients aged < 18 years. (2) Patients undergoing arterial thrombolysis, thrombectomy, stenting, endarterectomy, or other vascular procedures. (3) Stroke caused by systemic conditions (e.g. immune, oncological, infectious, or metabolic diseases). (4) Interventions or comparators involving anticoagulants or thrombolytic agents. (5) Conference abstracts without full-text data.
Outcomes
Efficacy outcomes: Incidence of major adverse cardiovascular events (MACEs) during treatment (stroke, vascular death, myocardial infarction, etc.). Stroke recurrence rate. Ischemic stroke recurrence rate. Safety outcomes: Incidence of intracranial hemorrhage. Severe bleeding requiring transfusion. Any bleeding events. All-cause mortality.
Data extraction and risk of bias assessment
Two researchers independently extracted data from eligible studies. For studies with multiple publications, report with the largest smaple size were included. Extracted data included: (1) Study characteristics: publication year, country, first author, sample size, study name, design, duration, diagnostic criteria, time to intervention, treatment details (drugs, doses, duration), follow-up duration, and outcome measures. (2) Patient characteristics: ethnicity, age, sex, comorbidities (e.g. hypertension, diabetes, dyslipidemia), history of stroke or TIA, cardioembolic stroke proportion, and baseline NIHSS scores. (3)Intervention and comparator group details: sample size and number of events, effect size estimates for interventions and comparators.
The quality of included RCTs was assessed independently by two reviewers using the Cochrane Risk of Bias (ROB) tool. Seven domains were evaluated: Blinding of participants and personnel. Blinding of outcome assessors. Random sequence generation. Allocation concealment. Incomplete outcome data. Selective reporting. Other biases. Studies scoring ⩾4 on these criteria were considered high-quality.
The Confidence in Network Meta-Analysis (CINeMA) method was used to assess evidence confidence of NMA by evaluating the credibility of each comparison across six dimensions (within-study bias, reporting bias, indirectness, imprecision, heterogeneity, incoherence).14,15
Statistical analysis
Network meta-analysis was performed using the STATA 14.2 network package.16,17 The conventional meta-analysis was conducted using RevMan 5.4 software. Odds ratio (OR) with 95% confidence intervals (CIs) were calculated to compare the efficacy and safety of interventions. A network diagram illustrated the direct comparisons between drugs. The effect sizes were pooled using the random-effects model, with I² and chi-square tests used to assess the heterogeneity among studies. Rankings of efficacy and safety were determined using the surface under the cumulative ranking curve (SUCRA), where higher values indicated better efficacy and safety, range from 0% to 100%. 18 the presence of inconsistency was assessed by comparing whether there were significant differences in direct and indirect comparison results in each closed loop evidence. Depending on the results, consistent or inconsistent models were used for direct, indirect, and mixed comparisons. Sensitivity analyses were performed by excluding specific category of studies. Statistical significance was set at a two-sided p < 0.05.
Results
Literature screening
A total of 5301 studies were identified from four databases, covering publications from January 1977 to February 2024. No additional studies were included through manual search of references in related studies and reviews. After removing duplicates, 4169 studies underwent initial screening. Based on titles and abstracts, irrelevant studies were excluded, leaving 108 studies for full-text and Supplemental Material review. Among these, 2 studies were published in Chinese, 52 were conference abstracts, 12 were observational studies, and 6 included interventions involving anticoagulants. Four papers reporting on two studies, we included the papers with the largest sample sizes. Eleven studies lacked sufficient data for inclusion. Ultimately, 24 randomized controlled trials (RCTs) were included in the systematic review.19–41 The CHANCE-2 trial comparing clopidogrel and ticagrelor was excluded from the NMA due to its focus on patients with CYP2C19 loss-of-function (LOF) alleles, which differed significantly from the populations of other studies. 21 In addition, the outcomes of the IST study 38 were not consistent with the NMA and were not included. The intervention and control groups in the LACI-2, 19 CSPS.com, 20 and Shimizu et al. 23 studies all used aspirin or clopidogrel as the routine background antiplatelet therapy. However, the proportion and dosage of aspirin or clopidogrel were not provided. Therefore, these studies cannot be included in the NMA. For these three studies, a conventional meta-analysis were conducted to combine the effect sizes. The flowchart of study selection is presented in Figure 1.
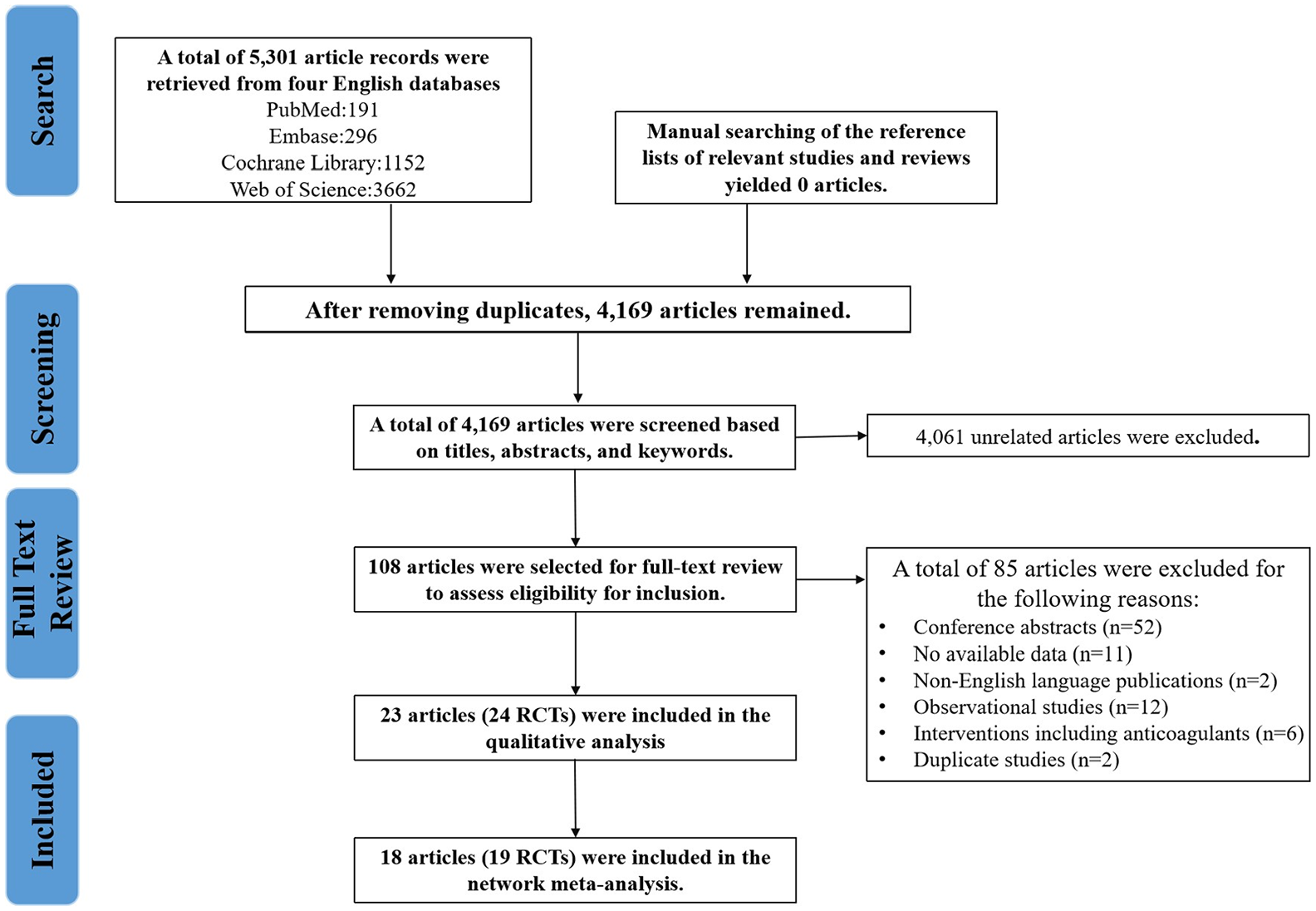
Flowchart of study selection.
Characteristics of included studies and risk of bias assessment
The systematic review included 47,507 patients with SSI/lacunar stroke. A total of 39,137 patients were included in the NMA. The characteristics and ROB score of the included studies are summarized in Table 1. Interventions and controls involved: aspirin (12 studies, 19,916 participants), clopidogrel (seven studies, 10,024 participants), ticagrelor (one study, 1077 participants), cilostazol (two studies, 1269 participants), ticlopidine (three studies, 1401 participants), dipyridamole (one study, 651 participants), aspirin plus clopidogrel (three studies, 3277 participants), aspirin plus dipyridamole (four studies, 6672 participants), terutroban (one study, 9556 participants), vorapaxar (one study, 1131 participants), Prasugrel (one study, 583 participants), Sarpogrelate (one study, 484 participants), Cilostazol plus Aspirin (one study, 100 participants), Cilostazol plus Aspirin/Clopidogrel (three studies, 806 participants), Aspirin/Clopidogrel (three studies, 808 participants), and placebo (six studies, 5523 participants).
Characteristics of included studies.
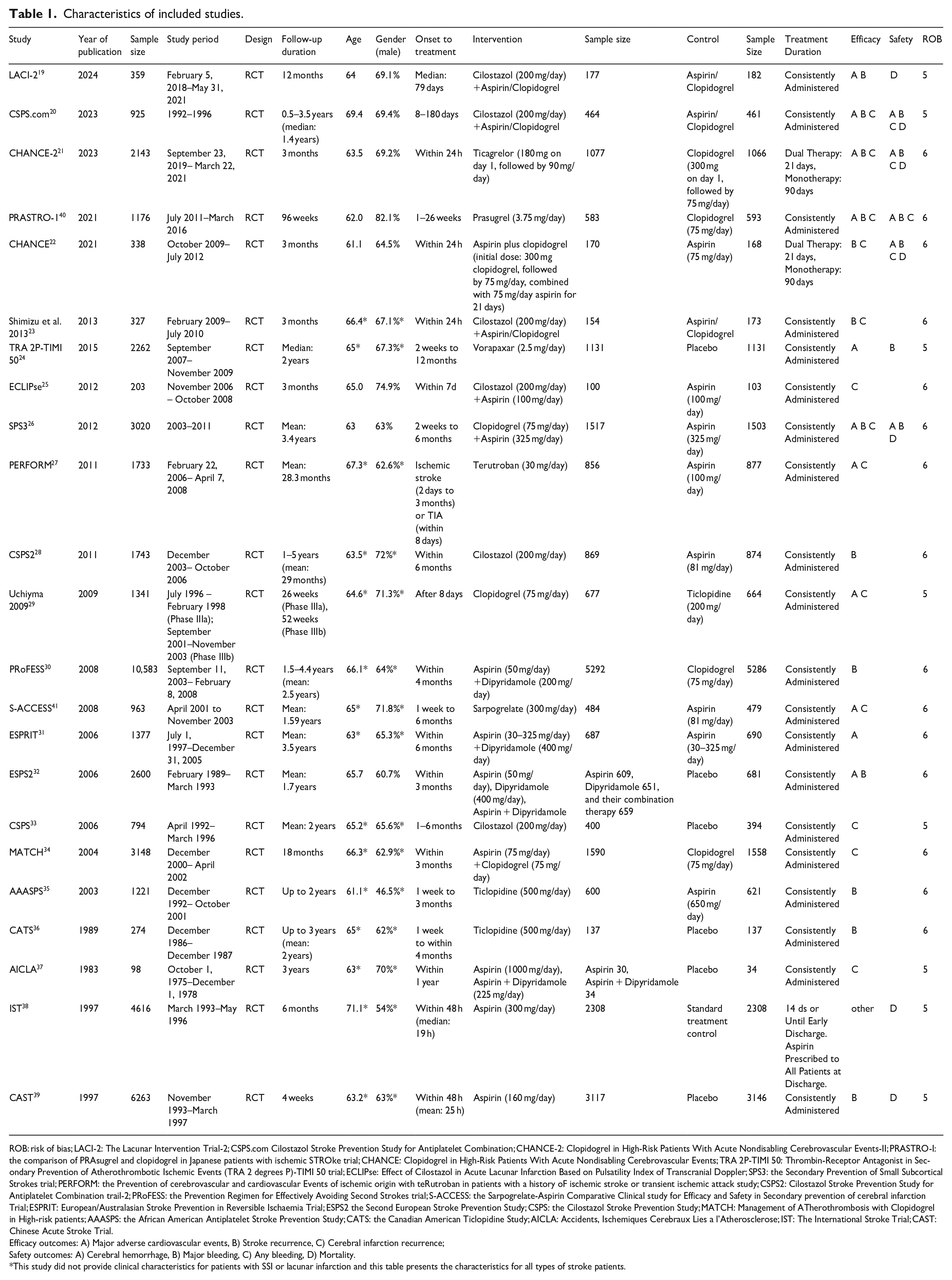
ROB: risk of bias; LACI-2: The Lacunar Intervention Trial-2; CSPS.com Cilostazol Stroke Prevention Study for Antiplatelet Combination; CHANCE-2: Clopidogrel in High-Risk Patients With Acute Nondisabling Cerebrovascular Events-II; PRASTRO-I: the comparison of PRAsugrel and clopidogrel in Japanese patients with ischemic STROke trial; CHANCE: Clopidogrel in High-Risk Patients With Acute Nondisabling Cerebrovascular Events; TRA 2P-TIMI 50: Thrombin-Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2 degrees P)-TIMI 50 trial; ECLIPse: Effect of Cilostazol in Acute Lacunar Infarction Based on Pulsatility Index of Transcranial Doppler; SPS3: the Secondary Prevention of Small Subcortical Strokes trial; PERFORM: the Prevention of cerebrovascular and cardiovascular Events of ischemic origin with teRutroban in patients with a history oF ischemic stroke or transient ischemic attack study; CSPS2: Cilostazol Stroke Prevention Study for Antiplatelet Combination trail-2; PRoFESS: the Prevention Regimen for Effectively Avoiding Second Strokes trial; S-ACCESS: the Sarpogrelate-Aspirin Comparative Clinical study for Efficacy and Safety in Secondary prevention of cerebral infarction Trial; ESPRIT: European/Australasian Stroke Prevention in Reversible Ischaemia Trial; ESPS2 the Second European Stroke Prevention Study; CSPS: the Cilostazol Stroke Prevention Study; MATCH: Management of ATherothrombosis with Clopidogrel in High-risk patients; AAASPS: the African American Antiplatelet Stroke Prevention Study; CATS: the Canadian American Ticlopidine Study; AICLA: Accidents, Ischemiques Cerebraux Lies a l’Atherosclerose; IST: The International Stroke Trial; CAST: Chinese Acute Stroke Trial.
Efficacy outcomes: A) Major adverse cardiovascular events, B) Stroke recurrence, C) Cerebral infarction recurrence;
Safety outcomes: A) Cerebral hemorrhage, B) Major bleeding, C) Any bleeding, D) Mortality.
This study did not provide clinical characteristics for patients with SSI or lacunar infarction and this table presents the characteristics for all types of stroke patients.
Three RCTs selected SSI/lacunar infarction patients as the study population,19,25,26 while in the remaining studies, the comparison of interventions for SSI patients was conducted as a subgroup of the overall participants. Most studies excluded infarctions caused by large artery stenosis and cardioembolic events. Seven studies used the TOAST classification to define SSI,20,24,25,30,34,35,40 while three studies used the National Institute of Neurological Disorders and Stroke (NINDS) criteria (Supplemental Table S2).23,28,41 Twelve studies focused on East Asian populations,20–23,25,28,29,33,39–41 while others included Caucasian, Hispanic, African American, and Latino populations. The average or median age of participants was above 60 years in all studies, and most had a male predominance. Cardioembolic strokes were excluded in the majority of studies. Most trials were multicenter, with 19 studies involving non-acute stroke patients (time from stroke onset to intervention >7 days) and 5 involving acute stroke patients. Follow-up durations ranged from 4 weeks to 5 years. All included studies were assessed as having a low risk of bias (Table 1).
Major adverse cardiovascular events
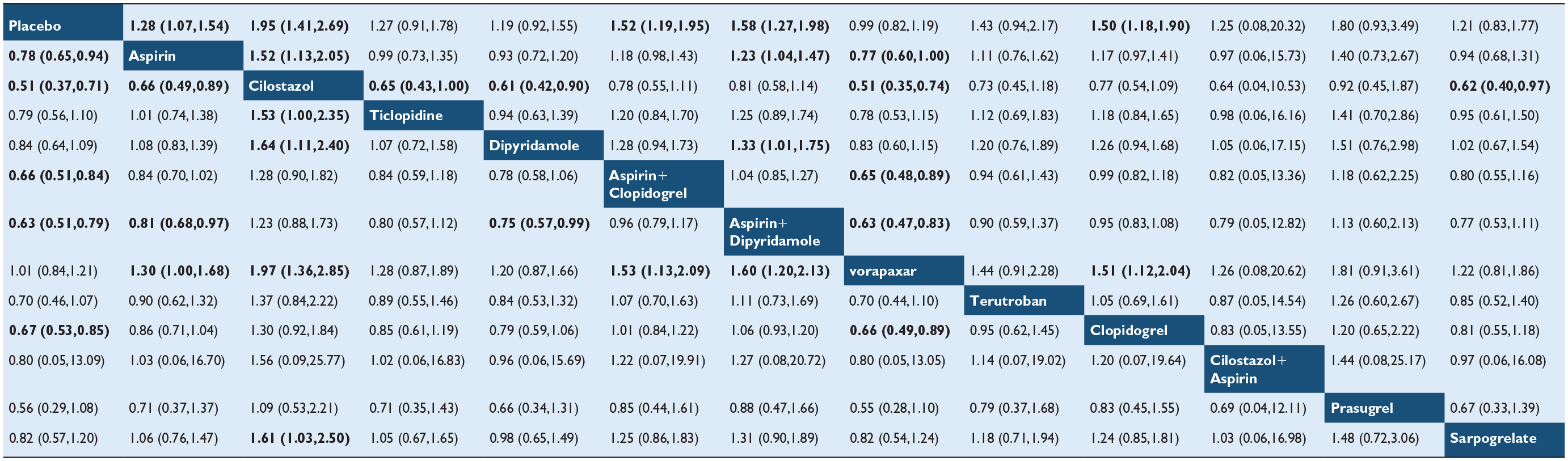
Nineteen studies,22,24–37,39–41 comprising 39,137 participants, reported MACEs as outcomes (including studies using recurrent stroke or ischemic stroke recurrence as only outcome). A total of 3315 events were recorded during follow-up. The evidence network is shown in Figure 2(a). Pairwise comparisons (Table 2) revealed that cilostazol significantly reduced the incidence of MACE compared to aspirin (OR, 0.66; 95% CI, 0.49–0.89), ticlopidine (OR, 0.65; 95% CI, 0.43–1.00), dipyridamole (OR, 0.61; 95% CI, 0.42–0.90), vorapaxar (OR, 0.51; 95% CI, 0.35–0.74), Sarpogrelate (OR, 0.62; 95% CI, 0.40–0.97), and placebo (OR, 0.51; 95% CI, 0.37–0.71).
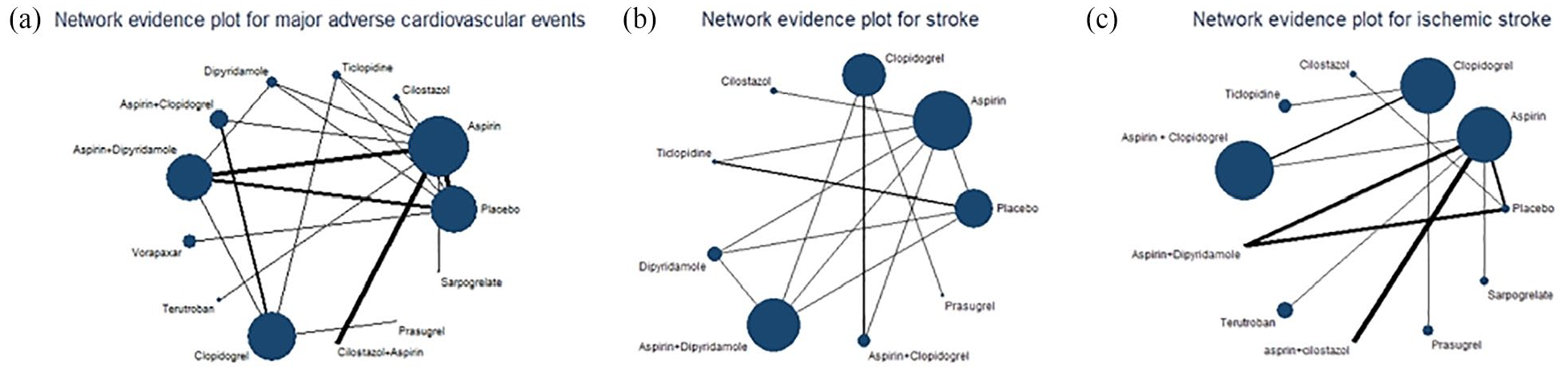
Network evidence plot for the outcomes of major adverse cardiovascular events recurrence (a), stroke recurrence (b), and ischemic stroke recurrence (c).
Effect sizes of pairwise comparisons of interventions for the outcome of major adverse cardiovascular event recurrence.
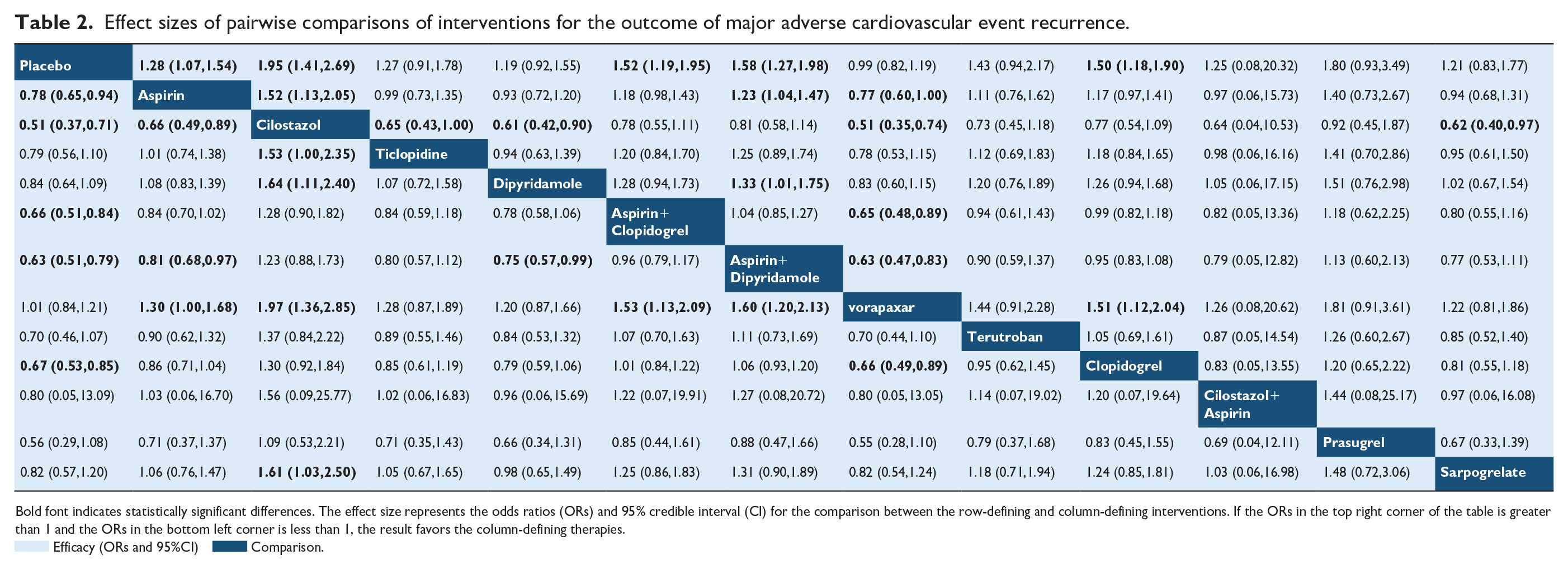
Bold font indicates statistically significant differences. The effect size represents the odds ratios (ORs) and 95% credible interval (CI) for the comparison between the row-defining and column-defining interventions. If the ORs in the top right corner of the table is greater than 1 and the ORs in the bottom left corner is less than 1, the result favors the column-defining therapies.
 Efficacy (ORs and 95%CI)
Efficacy (ORs and 95%CI)  Comparison.
Comparison.
The SUCRA rankings indicated cilostazol as the most effective (90.0%), followed by prasugrel (75.8%), aspirin plus dipyridamole (74.8%), aspirin plus clopidogrel (67.5%), clopidogrel (64.2%), terutroban (56.8%), Cilostazol + Aspirin (47.3%), ticlopidine (41.1%), aspirin (40.9%), Sarpogrelate (35.3%), dipyridamole (32.4%), vorapaxar (12.1%), and placebo (11.8%; Figure 3, Table 3). The CHANCE-2 study found that ticagrelor was more effective than clopidogrel in preventing MACE in patients with CYP2C19 LOF alleles (HR, 0.58; 95% CI, 0.41–0.82). 21
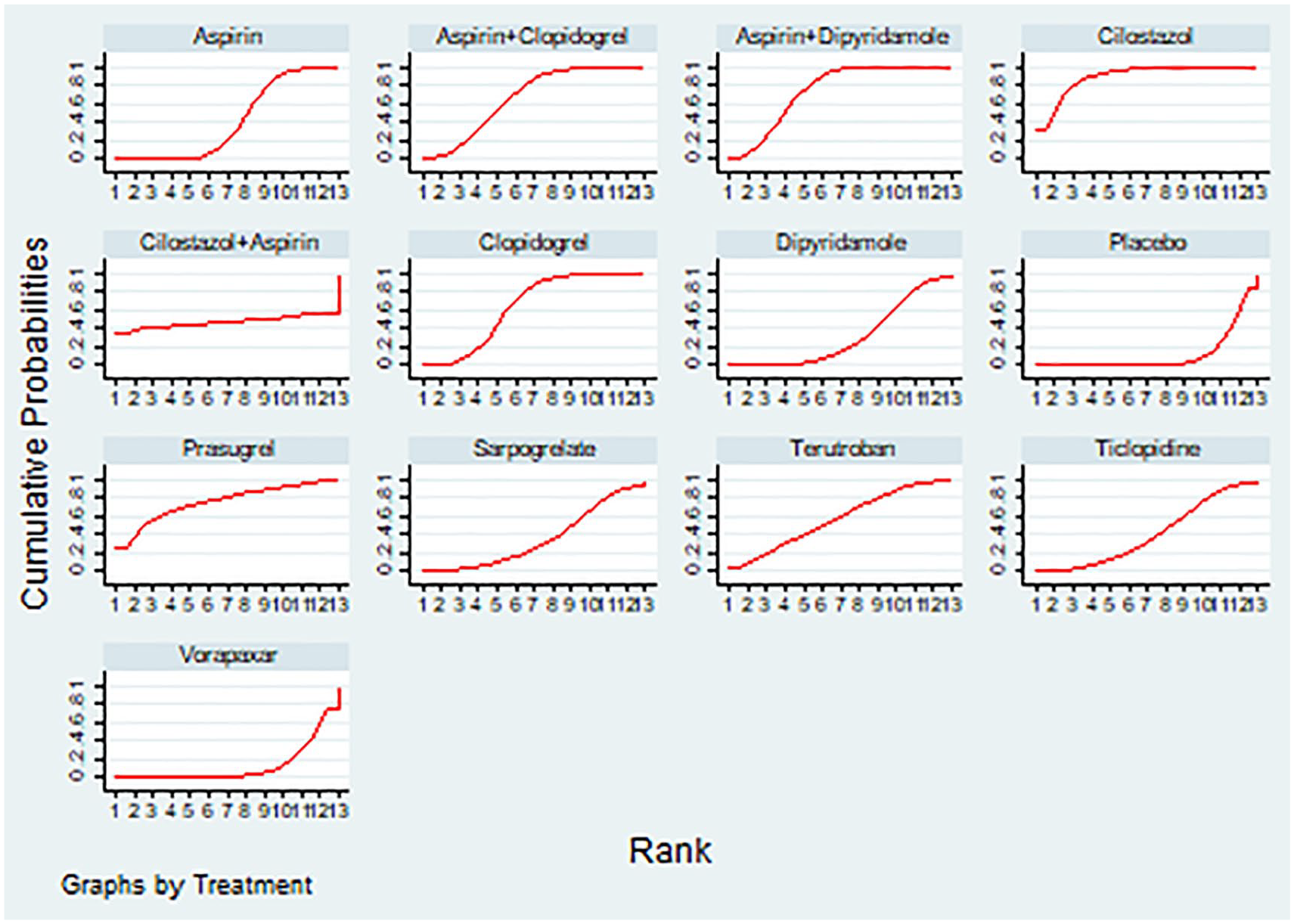
Cumulative ranking curve of interventions for the outcome of major adverse cardiovascular events recurrence.
Efficacy ranking of interventions for the outcome of major adverse cardiovascular event recurrence.
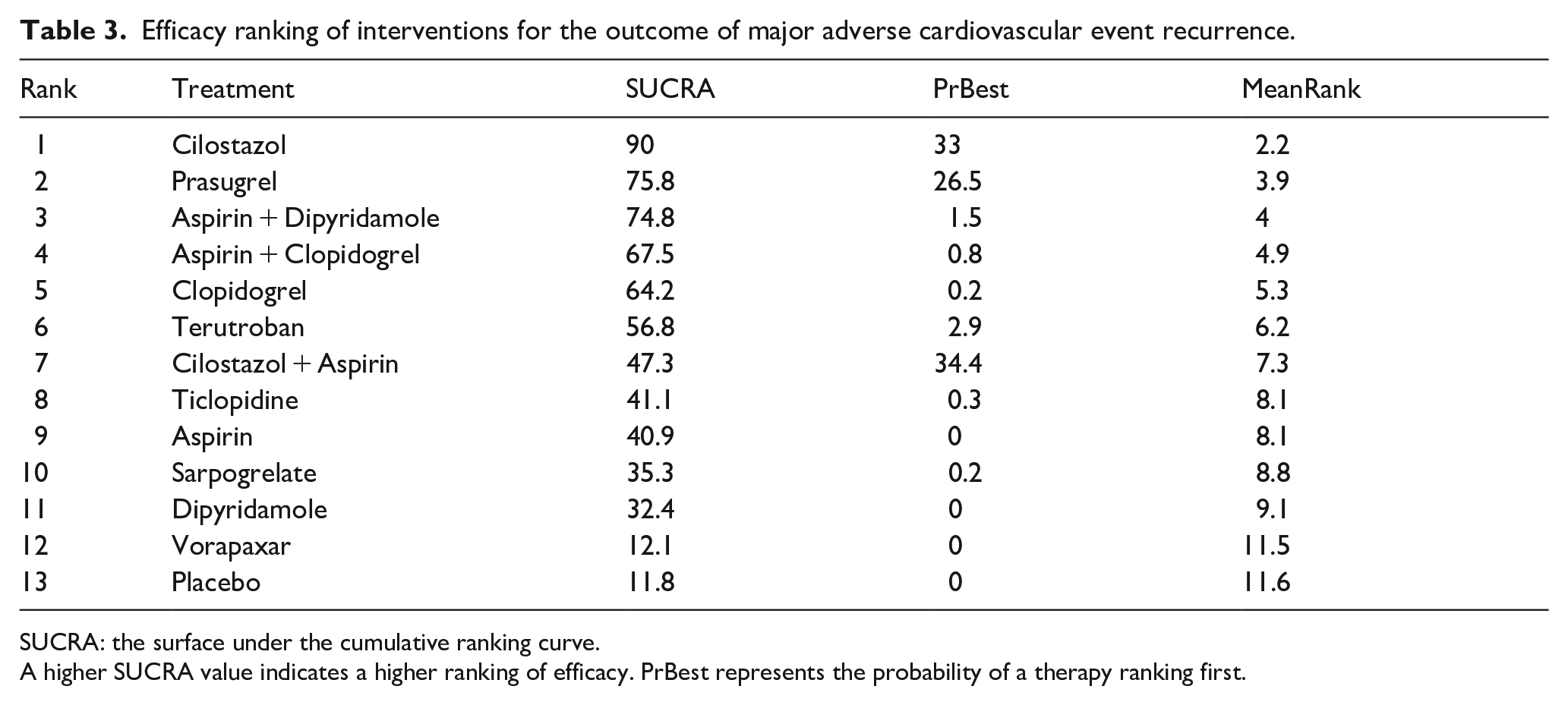
SUCRA: the surface under the cumulative ranking curve.
A higher SUCRA value indicates a higher ranking of efficacy. PrBest represents the probability of a therapy ranking first.
The meta-analysis of three studies19,20,23 shows that, compared with the group receiving standard background mono-antiplatelet therapy (aspirin/clopidogrel), the intervention group with cilostazol added significantly reduced the incidence of MACE (OR, 0.57; 95% CI, 0.35–0.93; Supplemental Figure S1(a)).
No significant inconsistencies were detected in loop-specific tests (Supplemental Table S3).
Stroke recurrence
Nine studies (27,213 participants, 1935 events) examined stroke recurrence.22,26,28,30,32,35,36,39,40 The evidence network is shown in Figure 2(b). Pairwise comparisons (Supplemental Table S4) indicated that aspirin plus dipyridamole was significantly more effective than aspirin (OR, 0.70; 95% CI, 0.49–1.00) and dipyridamole (OR, 0.70; 95% CI, 0.49–1.00) mono-antiplatelet therapy in reducing stroke recurrence. Cilostazol is superior to aspirin in reducing the incidence of stroke (OR, 0.68; 95% CI, 0.48–0.96).
Aspirin plus dipyridamole ranked highest in SUCRA (80.7%), followed by Cilostazol (79.7%), prasugrel (76.9%), clopidogrel (69.4%), aspirin plus clopidogrel (49.8%), ticlopidine (31.1%), dipyridamole (30.8%), aspirin (28.2%), and placebo (3.4%; Supplemental Table S5, Supplemental Figure S2).
The intervention group with cilostazol plus aspirin/clopidogrel did not show a significant difference compared to the control group in preventing stroke recurrence (Supplemental Figure S1(b); three studies 19,20,23; OR, 0.73; 95% CI, 0.32–1.70).
The CHANCE-2 study showed ticagrelor outperformed clopidogrel in reducing stroke recurrence in patients with CYP2C19 deficiency (HR, 0.55; 95% CI, 0.38–0.78). 21 No inconsistencies were found (loop-specific: Supplemental Table S6).
Ischemic stroke recurrence
Eleven studies22,25–27,29,33,34,37,40,41 (12,814 participants, 1,042 events) focused on ischemic stroke recurrence. The evidence network is shown in Figure 2(c). Pairwise comparisons (Supplemental Table S7) showed that cilostazol was superior to placebo (OR, 0.48; 95% CI, 0.27–0.84). Aspirin plus clopidogrel outperformed placebo (OR, 0.48; 95% CI, 0.27–0.84), Sarpogrelate (OR, 0.57; 95% CI, 0.36–0.89) and aspirin (OR, 0.76; 95% CI, 0.59–0.97). Aspirin plus dipyridamole was superior to placebo (OR, 0.17; 95% CI, 0.03–0.88). Clopidogrel was superior to sarpogrelate (OR, 0.60; 95% CI, 0.36–0.99).
Prasugrel ranked highest in SUCRA (76.6%), followed by dual antiplatelet therapies (aspirin plus clopidogrel, 72.4%; aspirin plus dipyridamole, 72.3%), clopidogrel (65.7%), terutroban (55.1%), ticlopidine (51.2%), aspirin plus cilostazol (50.1%), aspirin (42.9%), cilostazol (33.1%), Sarpogrelate (24.5%), and placebo (6.1%; Supplemental Table S8, Supplemental Figure S3).
The combined results of the two studies20,23 did not show that the intervention group with cilostazol significantly reduced the incidence of ischemic stroke (Supplemental Figure S1(c); OR, 0.53; 95% CI, 0.24–1.14).
The CHANCE-2 study confirmed superiority of ticagrelor over clopidogrel in reducing ischemic stroke recurrence in patients with CYP2C19 deficiency (HR, 0.55; 95% CI, 0.38–0.79). 21 The evidence network contained only one loop (aspirin, aspirin plus dipyridamole, placebo), precluding inconsistency testing (As no direct and indirect comparisons exist simultaneously among all medications).
Evidence confidence rating
The results of evidence confidence rating using the CINeMA method showed that the confidence of 9 comparisons with MACEs as the outcome were rated as “moderate” (Aspirin vs Aspirin plus Dipyridamole, Aspirin vs Cilostazol, Aspirin plus Dipyridamole vs Placebo, Cilostazol vs Placebo, Aspirin plus Dipyridamole vs Vorapaxar, Aspirin plus clopidogrel vs Placebo, Cilostazol vs Dipyridamole, Cilostazol vs Vorapaxar, Clopidogrel vs Placebo), while the remaining comparisons were rated as “very low.” Comparisons with recurrent stroke or ischemic stroke as the outcome were rated as “very low.” The main concern arises from imprecision. The heterogeneity of some comparisons was assessed as “major concern” (Supplemental Tables S9–S11). The contribution of each included study to the network evidence comparisons indicates that some comparison results were significantly influenced by one or two studies (Supplemental Tables S12–S14).
Safety outcomes
Studies addressing safety outcomes included hemorrhagic stroke (five studies),20–22,26,40 hemorrhagic stroke plus severe bleeding (six studies),20–22,24,26,40 any bleeding events (four studies),20–22,40 and all-cause mortality (five studies).19–22,26 The total sample sizes and event counts were: hemorrhagic stroke (7638, 47 events), hemorrhagic stroke plus severe bleeding (9844, 279 events), any bleeding (4562, 173 events), and all-cause mortality (6723, 200 events).
Compared to aspirin monotherapy, aspirin plus clopidogrel increased the risk of severe bleeding (OR, 1.92; 95% CI, 1.38–2.68) and mortality (OR, 1.47; 95% CI, 1.09–1.98). Vorapaxar also elevated the risk of severe bleeding compared to placebo (OR, 2.35; 95% CI, 1.46–3.79). In patients with CYP2C19 deficiency, ticagrelor increased the risk of any bleeding events compared to clopidogrel (OR, 3.09; 95% CI, 1.85–5.16), but no significant differences were observed for severe bleeding (OR, 0.99; 95% CI, 0.29–3.43), mortality (OR, 1.49; 95% CI, 0.25–8.91), or hemorrhagic stroke (OR, 0.99; 95% CI, 0.14–7.04; Supplemental Figure S4).
Other outcomes
Three studies19,22,38 explored efficacy in preventing post-stroke disability (defined as a modified Rankin Scale score >1). The LACI-2 trial showed cilostazol reduced post-stroke disability compared to control (HR, 0.31; 95% CI, 0.14–0.72). 19 The CHANCE study 22 reported that aspirin combined with clopidogrel did not reduce the incidence of disabling stroke compared to aspirin alone. The IST study 38 indicated no significant difference in the rates of death or dependency within a 6-month follow-up period between the aspirin and control group.
Sensitivity analysis
The sensitivity analysis was performed by excluding RCTs conducted in Asian countries, with MACEs as the outcome (10 studies included in NMA, 26,311 participants, 2654 events).24,26,27,30–32,34–37 The evidence network is shown in Supplemental Figure S5. Pairwise comparisons revealed that aspirin, clopidogrel, aspirin plus clopidogrel, and aspirin plus dipyridamole were significantly more effective than placebo (aspirin: OR, 0.72; 95% CI, 0.57–0.93; clopidogrel: OR, 0.63; 95% CI, 0.48–0.83; aspirin plus clopidogrel: OR, 0.62; 95% CI, 0.46–0.82; aspirin plus dipyridamole: OR, 0.60; 95% CI, 0.46–0.77). The combination of aspirin and dipyridamole was significantly superior to aspirin alone (OR, 0.82; 95% CI, 0.69–0.99) and dipyridamole alone (OR, 0.82; 95% CI, 0.69–0.99). Clopidogrel (OR, 1.60; 95% CI, 1.10–2.34), aspirin plus clopidogrel (OR, 1.64; 95% CI, 1.11–2.42), and aspirin plus dipyridamole (OR, 1.69; 95% CI, 1.17–2.43) were significantly more effective than vorapaxar (Supplemental Table S15). The efficacy ranking based on SUCRA values was as follows: Aspirin plus Dipyridamol (85.8%), Aspirin plus clopidogrel (79.0%), Clopidogrel (73.1%), Terutroban (66.3%), Ticlopidine (46.7%), Aspirin (46.8%), Dipyridamol (33.3%), Vorapaxar (10.2%), Placebo (8.8%; Supplemental Table S16). Statistical tests did not reveal significant inconsistency (Supplemental Table S17).
The LACI-2 study conducted in the UK compared the efficacy of adding cilostazol to standard antiplatelet therapy (aspirin/clopidogrel) for secondary prevention. The results showed no significant difference in the incidence of composite outcome of cardiovascular events (HR, 0.77; 95% CI, 0.57–1.05) and the incidence of stroke or TIA (HR, 1.35; 95% CI, 0.51–3.57) between the cilostazol addition group and the non-addition group. 19
Discussion
This systematic review and network meta-analysis revealed that cilostazol, prasugrel, clopidogrel, aspirin plus clopidogrel, and aspirin plus dipyridamole are relatively effective antiplatelet therapies for preventing adverse cardiovascular events, stroke, and ischemic stroke in patients with SSI/lacunar infarction. Compared to aspirin monotherapy, the combination of aspirin and clopidogrel was associated with a higher risk of major bleeding and mortality. In patients with SSI/lacunar infarction carrying the CYP2C19 LOF allele, ticagrelor demonstrated superior efficacy over clopidogrel in preventing cardio-cerebrovascular events, with no significant difference in the risk of major bleeding, although minor bleeding events were more frequent in the ticagrelor group. 21
The included studies were similar in terms of study population selection, demographic characteristics, and the definition of SSI. Furthermore, most studies excluded SSI patients caused by large artery atherosclerosis or cardioembolic events. Statistical tests did not reveal significant inconsistency between direct and indirect comparisons (although many interventions only had indirect comparison results). These similarities support the transitivity of the network evidence comparisons. However, there were differences in the dosage selection of interventions, such as aspirin, between some studies. Inconsistencies in the time from onset to enrollment, follow-up duration, and the country and ethnicity of the study populations may also affect the transitivity of the study results.
SUCRA analysis indicated that cilostazol was the first and second effective antiplatelet agent for preventing recurrent MACEs and stroke. However, cilostazol ranked lower in efficacy when ischemic stroke recurrence was used as the endpoint. This discrepancy arises from the difference between the CSPS2 study 28 (with any stroke as the outcome) and the CSPS study 33 (with ischemic stroke as the outcome). Although both studies support the significantly better efficacy of cilostazol, the control group in CSPS2 was aspirin, while CSPS was placebo, and the former had a larger sample size (1731 vs 794). In the NMA of ischemic stroke recurrence, the significant advantage of placebo does not improve ranking of cilostazol. Two studies are all included in the NMA of MACEs, thus the ranking of cilostazol in this outcome is relatively more comprehensive and reliable. However, the CSPS 33 and CSPS228 study exclusively enrolled East Asian populations. Given that only one study conducted in non-Asian countries (LACI-2) assessed the efficacy of adding cilostazol to standard antiplatelet therapy (aspirin/clopidogrel), 19 with a negative result, the applicability of cilostazol in non-Asian populations remains limited. Additionally, CSPS228 study reported significantly lower rates of intracranial hemorrhage or major bleeding requiring hospitalization with cilostazol compared to aspirin but higher incidences of adverse effects such as headache. While cilostazol shows promise, its first-class efficacy should be interpreted cautiously. Direct comparisons between cilostazol and agents like prasugrel, clopidogrel or ticagrelor in SSI patients, especially in non-Asian populations, warrant further large-scale randomized controlled trials.
The efficacy ranking of cilostazol plus aspirin for MACE and stroke recurrence outcomes is lower than that of cilostazol monotherapy. This may be due to the limited evidence from a small sample study (ECLIPse 25 ) that included 203 participants, which directly compared the two treatments. The lack of statistically significant differences between the two therapies could be attributed to the relatively limited recurrence events. However, the CSPS.com 20 and LACI-219 studies reported no significant difference in bleeding and mortality events between cilostazol plus aspirin/clopidogrel and aspirin/clopidogrel monotherapy, indicating that dual antiplatelet therapy with cilostazol has good safety for secondary prevention of SSI. Therefore, whether aspirin/clopidogrel combined with cilostazol is the best therapy superior to cilostazol monotherapy warrants further validation.
Due to concerns that the time from onset to enrollment may affect the efficacy of dual antiplatelet therapy, studies enrolling patients in the acute phase (the CHANCE and ECLIPSE trials) were excluded. A network meta-analysis was conducted on dual antiplatelet therapy studies enrolling patients in the chronic phase, with MACEs as the outcome. The results of the efficacy ranking and pairwise comparisons showed no significant differences compared to previous results (Supplemental Tables S18 and S19). In view of concerns regarding bleeding risk, dual antiplatelet therapy (DAPT) of aspirin plus dipyridamole or plus clopidogrel does not appear to be the optimal choice for secondary prevention in SSI patients. Although the PRoFESS trial did not report bleeding outcomes in the SSI/lacunar infarction subgroup treated with aspirin plus dipyridamole, it showed an increased risk of major bleeding (HR, 1.15; 95% CI, 1.00–1.32) and intracranial hemorrhage (HR, 1.42; 95% CI, 1.11–1.83) in the overall ischemic stroke population compared to clopidogrel monotherapy. 30 The ESPRIT study reported higher rates of drug discontinuation due to adverse effects (mainly headaches) in the aspirin plus dipyridamole group than aspirin group (34.5% vs 13.4%), but there was no significant difference in major bleeding between the two groups. 31 MATCH revealed that combining aspirin with clopidogrel increased the risks of fatal bleeding, intracranial hemorrhage, major bleeding, and minor bleeding compared to aspirin monotherapy in secondary prevention following ischemic stroke or transient ischemic attack. 34 SPS3 further directly highlighted an elevated risk of major bleeding and mortality with aspirin plus clopidogrel compared to aspirin alone in SSI patients. 26 Considering that ischemic stroke patients often share risk factors of hemorrhagic stroke, DAPT of aspirin plus dipyridamole or plus clopidogrel should be used with caution for secondary prevention.
Although prasugrel ranks among the top for multiple outcomes, its direct comparison with other antiplatelet agent in network evidence is limited to the PRASTRO-1 study. 40 This study showed that the incidence of adverse outcomes with prasugrel was lower than that with clopidogrel, but no statistically significant difference was observed between the two. In pairwise comparisons of therapies, prasugrel showed no significant difference compared to placebo in outcome of MACE and stroke recurrence. In contrast, clopidogrel has been supported by multiple studies demonstrating its efficacy. Therefore, the favorable ranking of prasugrel is likely due to the upward influence of clopidogrel, which is also ranked highly. Its efficacy ranking has not been fully validated and is less robust than that of clopidogrel. On the other hand, the activation of clopidogrel depends on hepatic metabolism via the CYP2C19 enzyme.42,43 SSI patients carrying CYP2C19 LOF alleles exhibit poorer responses to clopidogrel. 21 Subgroup analysis from SPS3 also showed increased stroke recurrence risk in patients with these alleles receiving clopidogrel and aspirin compared to those with rapid metabolizer genotypes. 44 As these LOF alleles are more prevalent in Asians than in Caucasians, genetic testing for CYP2C19 polymorphisms is recommended in Asian SSI patients considering clopidogrel.45,46 Ticagrelor, which does not require hepatic activation, 47 could be a suitable alternative, especially for patients with genetic impairments, though it carries a higher risk of minor bleeding.
The causes of SSI include perforating artery microatherosclerosis, parent artery atherosclerotic occlusion at the perforating artery orifice, small vessel lipohyalinosis, and other factors. 9 The efficacy and safety of antiplatelet drugs may vary depending on the underlying mechanisms of SSI. Subgroup analysis of the CHANCE-2 study found that SSI patients without responsible intracranial artery stenosis benefited more from antiplatelet therapy. 21 However, most previous RCTs did not further analyze based on the underlying causes of SSI, and future RCTs are expected to elucidate.
This study has several limitations. First, this study is a sparse network. The majority of comparisons, as assessed using the CINeMA approach, were evaluated to low-confidence evidence. Many comparisons were significantly influenced by one or two included studies, which compromised the generalizability and robustness of the results. Second, evidence from trials on cilostazol in SSI patients in non-Asian countries is limited, and its use in these patients should be carefully evaluated. Third, the timing of treatment initiation varied across studies, and the efficacy of treatment may differ between the acute and chronic phases of cerebral infarction. Additionally, heterogeneity in follow-up durations may introduce bias through cumulative outcome differences between interventions. Fourth, funnel plots were not applicable for detecting publication bias, as many comparisons were based on single studies or only indirect comparisons. Additionally, this study included only published research, excluding data from ongoing trials and conference abstracts, which may have introduced publication bias in the outcomes. Fifth, few RCTs specifically addressed the safety of antiplatelet therapies in SSI patients, precluding a comprehensive network comparison of evidences and narrowing the scope of conclusions. Finally, a few included studies defined outcomes differently; for instance, the CAST study combined death and nonfatal stroke as the primary endpoint, potentially including nonvascular deaths. However, as the follow-up was limited to 4 weeks, the impact of such biases was likely minimal.
Conclusion
Overall, cilostazol demonstrated the best efficacy and safety profile for preventing recurrent cardio-cerebrovascular events in patients with SSI/lacunar infarction. However, its use in non-Asian countries should be approached with caution. Dual antiplatelet therapies (aspirin plus clopidogrel, aspirin plus dipyridamole) also showed good efficacy but were associated with higher risks of severe bleeding and mortality. Among single-agent therapies, clopidogrel may serve as an alternative for patients intolerant to cilostazol. The comparison between prasugrel and other antiplatelet drugs awaits validation through further larger-scale RCTs. Routine CYP2C19 gene test is recommended for clopidogrel users. In patients with CYP2C19 LOF, ticagrelor may offer superior efficacy. However, it is important to note that ticagrelor may increase the risk of mild bleeding. In non-Asian countries, clopidogrel or ticagrelor may be better options. The efficacy of cilostazol in these patients requires further investigation.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251331470 – Supplemental material for Efficacy and safety of antiplatelet therapy for secondary prevention of small subcortical infarction: A systematic review and network meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873251331470 for Efficacy and safety of antiplatelet therapy for secondary prevention of small subcortical infarction: A systematic review and network meta-analysis by Xiao Feng, Junyong Du, Tong Qu, Shabei Xu and Wenhao Zhu in European Stroke Journal
Footnotes
Acknowledgements
The authors wish to extend their sincere appreciation to all contributors and collaborators involved in this research endeavor.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hubei Province (2021CFB382) and National Natural Science Foundation of China (92248304).
Informed consent
Not applicable. The manuscript we submitted is a systematic review and network meta-analysis, which involves collecting and analyzing data from previously published randomized controlled trials (RCTs). Our study does not involve any new demographic and/or biological data from human or animal subjects.
Ethical approval
Not applicable.
Guarantor
Wenhao Zhu.
Contributorship
FX: Responsible for study design, drafting the manuscript, data collection, and data analysis. DJY, QT: Responsible for data collection and analysis. XSB: Responsible for study design, data analysis, data interpretation, and manuscript revision. ZWH: Responsible for study design, data collection, data analysis, data interpretation, and manuscript revision. All authors have commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Availability of data and materials
Upon reasonable request, the corresponding author will provide access to the datasets generated and/or analyzed during this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.