Abstract
Introduction:
The impact of multiple feeding arteries on clinical outcomes of cerebral arteriovenous malformations (AVMs) is not well understood. This study aims to compare outcomes between AVMs with multiple versus single feeding arteries.
Patients and Methods:
Data from the Multicenter International Study for Treatment of Brain AVMs (MISTA) consortium were analyzed. Propensity score matching (PSM) was used to balance cohorts. Subgroup analysis was conducted for ruptured and unruptured AVMs and different treatment options, and multivariable logistic regression was applied to assess the impact of feeding artery origin.
Results:
Among 953 patients, 661(69.4%) had multiple feeding arteries, and 292 (30.6%) had a single feeding artery. After PSM, which included 422 matched patients (211 in each group), the differences in obliteration rates (68.7% vs 74.8%, OR 0.73, 95% CI: 0.48–1.12, p = 0.16) and symptomatic complications (15.6% vs 11.8%, OR 1.37, 95% CI: 0.78–2.41, p = 0.25) were not significant. Subgroup analysis comparing ruptured and unruptured AVMs and different treatment options showed no significant differences across all subgroups. Multivariable analysis identified PICA feeders as significantly associated with increased odds of all complications (OR 7.33, 95% CI: 2.14–25.1, p = 0.002).
Discussion and Conclusion:
AVMs with a single feeding artery were more likely to present with rupture, but no significant differences in obliteration rates or complications were observed between the groups after PSM. These findings suggest that while the number of feeding arteries may influence the initial presentation, it does not appear to impact overall treatment success or patient prognosis. Further prospective studies are needed to confirm these findings.
This is a visual representation of the abstract.
Introduction
Cerebral arteriovenous malformations (AVMs) are abnormal vascular networks composed of dilated and tortuous arteries and veins, lacking a capillary bed or containing some microvasculature within the nidus.1–4 Hemorrhage is the most common initial symptom, occurring in 42% to 72% of cases, followed by seizures, headaches, progressive neurological deficits, and pulsatile tinnitus.5–9
AVMs receive blood from one or more feeding arteries and include an intranidal network, along with one or several draining veins . 1 An AVM can be supplied by one or multiple feeding arteries, which may originate from the same vascular territory or from different vascular territories. 1
While identifying feeding arteries is routinely performed for treatment planning, detailed classification of their number, origin, and specific contributions to AVM angioarchitecture has not always been systematically documented.10–12 Knowledge of the numbers of feeders and location is clinically important. 1 The involvement of certain feeders can usually explain neurological signs and symptoms in patients. 1 In addition, feeders can be the site for interventions.4,13–16
AVMs with multiple feeders are often associated with more intricate angioarchitecture, which can complicate treatment and increases the risk of procedural complications.1,17–19
Despite some studies suggesting the safety of endovascular and surgical approaches in multiple feeders’ AVMs,1,20–22 studies comparing multiple and single feeders AVMs are scarce. Therefore, the present study aims to compare the outcomes of AVM patients with multiple feeders versus those with single feeders, utilizing data from a multicenter retrospective study.
Methods
Data coordination and patient sample
The Multicenter International Study for Treatment of Brain AVMs (MISTA) consortium combines retrospective databases from multiple academic institutions in North America and Europe.
Data were collected using a standardized datasheet, which recorded patient demographics (age, gender, race, smoking status, comorbidities, presence of hereditary disease, clinical presentation, modified Rankin Scale (mRS), and use of antithrombotic medications), AVM characteristics (side, size, location, Spetzler-Martin grade, nidus compactness, status of feeders and draining veins, associated aneurysms, other vascular anomalies, and prior treatment), procedural details (date of treatment, type of treatment, and details of each treatment), complications (type, timing, location, symptoms, duration, and need for additional treatment), angiographic outcomes (length of imaging follow-up, obliteration rate, and recurrence), and functional outcomes (length of clinical follow-up, mRS, need for retreatment, and mortality).
Institutional review board (IRB) approval was obtained at each participating site. No identifiable patient information was included, thus informed consent was not required. Each site designated a coordinator responsible for data collection and integrity. The data were pooled into a large, secure database at the main center, protected by a password-secured server and files. The data are available for further studies and contributing sites as deemed appropriate by the scientific steering committee.
The current study is a subanalysis of the MISTA consortium, which compared patients with multiple and single feeders. Both ruptured and unruptured AVMs were included. Only treated AVMs were analyzed, and cases managed with watchful waiting were excluded. All treatment options were included. The selection of treatment options was at the discretion of the treating physicians. The primary endpoint of this study was complete obliteration at the last follow-up. Secondary endpoints included symptomatic complications, permanent complications, functional outcomes measured by the mRS, and mortality.
Angiographic outcomes
Angiographic outcomes were assessed using digital subtraction angiography (DSA), MR angiography, or CT angiography. AVM obliteration after treatment, both immediately, prior to staged procedures, and during follow-ups, was categorized into four levels: 100%, 90%–99%, 50%–89%, and less than 50%. Other angiographic outcomes included AVM recurrence.
Functional outcomes and complications
Functional outcomes were assessed using mRS scores following each treatment stage and during follow-up. Independent functional status was defined by an mRS score of 0–2.
Symptomatic complications were those that caused symptoms or signs attributable to the complication and were deemed permanent if symptoms persisted for more than 6 months.
Statistical analysis
We conducted our statistical analysis using Stata (V.17.0; StataCorp). Descriptive statistics were utilized to summarize the baseline characteristics of cohorts with multiple feeding arteries and a single feeding artery. Continuous variables were represented by the median and interquartile range (IQR), while categorical data were expressed as frequencies and percentages. To compare categorical variables, we employed Pearson’s chi-square or Fisher’s exact tests as appropriate, and the Mann-Whitney U test was used for continuous variables.
Propensity score matching (PSM) was performed using logistic regression to calculate a propensity score (PS) for each patient based on clinically relevant and statistically significant variables. Specifically, the model included age, gender, race, hypertension, diabetes mellitus, history of smoking, family history, cardiovascular disease, underlying hereditary disease, antithrombotic medication, mRS at presentation, seizures, rupture status, side, location, eloquent location, deep location, nidus size, Spetzler-Martin grade, number and location of draining veins, venous stenosis, compacted nidus, nidal aneurysm, prior treatment, and current treatment. The final PSM model demonstrated a good fit based on the Hosmer-Lemeshow test (p = 0.853), indicating no significant differences between observed and expected frequencies. The model also showed good discriminatory power with an Area Under the ROC Curve (AUC) of 0.81.
We matched the multiple feeding arteries and single feeding artery cohorts in a 1:1 ratio without replacement, utilizing a caliper of 0.2 standard deviations of the logit of the propensity score through greedy matching. The PSMATCH2 package was used for the propensity score derivation. Primary and secondary outcomes between the cohorts, both before and after matching, were compared using univariable binary logistic regression, and linear regression analyses as appropriate. A Bonferroni correction was applied to adjust the p-value for multiple comparisons.
A multivariable logistic regression model was conducted on the entire dataset (n = 953) to evaluate the association between the origin of arterial feeders and complications, including all complications, symptomatic complications, and permanent complications. The model was adjusted for age, mRS at presentation, number of feeders, venous stenosis, Spetzler-Martin grade, location, and nidus size (cm).
Missing data
Missing variables were detailed in Supplemental Figure 1. The missing data were assumed to be either completely at random or missing at random. To address the potential bias and reduction in statistical power resulting from excluding the missing data and given the significant amount of missing data (>5%), we performed multiple imputation using chained equations. 23 This process involved 50 imputations across 10 iterations. The imputation technique varied based on the type of missing variable: logistic regression was used for binary variables, predictive mean matching for continuous variables, and polynomial regression for categorical variables. We used R Studio version 4.2.2 to perform the multiple imputation.
In addition to the primary analysis, a sensitivity analysis was performed to evaluate the robustness of the results. We compared the results between the imputed outcomes and the non-imputed outcomes. The results were similar before and after imputations (Supplemental Table 1).
Results
Baseline characteristics
A total of 953 patients with arteriovenous malformations (AVMs) were included in the study, with 661 (69.4%) patients having multiple feeding arteries and 292 (30.6%) patients having a single feeding artery. The median age of the entire cohort was 42 years (IQR 26–57), with a median age of 41 years (IQR 26–56) for the multiple feeders group and 43 years (IQR 28–59) for the single feeder group (p = 0.17). The gender distribution was similar between the groups, with 48.5% males in the multiple feeders group and 52.0% in the single feeder group (p = 0.32).
Regarding AVM characteristics, the nidus size was significantly larger in the multiple feeders group, with a median of 2.5 cm (IQR 1.7–3.5) compared to 1.5 cm (IQR 1–2.2) in the single feeder group (p < 0.001). Grade 1 AVMs were present in 21.6% of the multiple feeders group versus 34.2% of the single feeder group, and grade 2 AVMs were present in 31.9% of the multiple feeders group versus 44.5% of the single feeder group (p < 0.001). Grade 3 AVMs were found in 28.4% of the multiple feeders group compared to 19.5% of the single feeder group (p < 0.001), and grade 4 AVMs were significantly more common in the multiple feeders group (16.6% vs 1.7%, p < 0.001; Table 1).
Comparison of baseline characteristics in patients with arteriovenous malformation according to number of feeding arteries before propensity score matching.
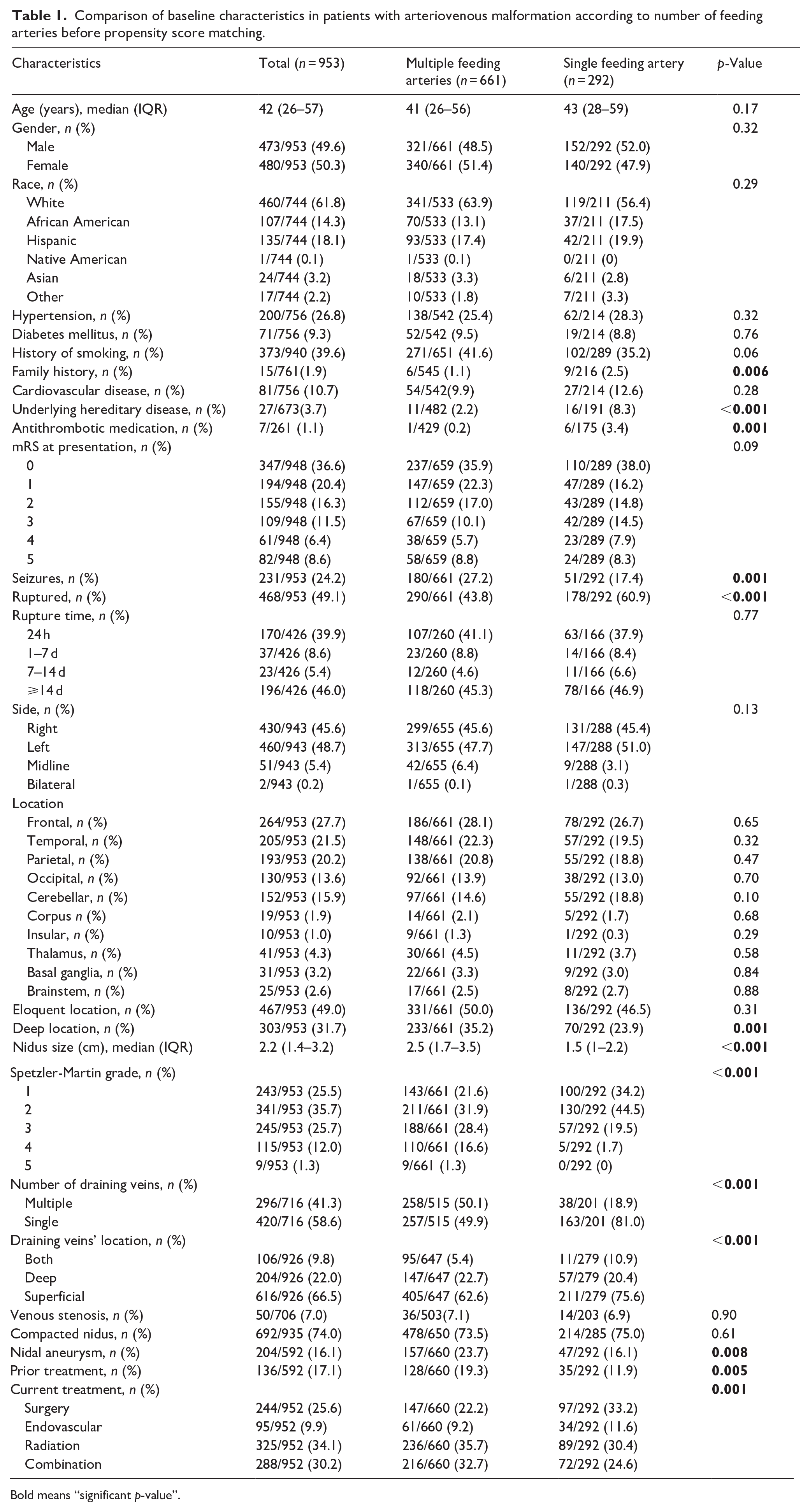
Bold means “significant p-value”.
Characteristics of feeding arteries
Feeders with a diameter greater than 1mm were observed in 66.5% of the patients, with 67.9% in the multiple feeders group and 52.6% in the single feeder group (p = 0.20). The middle cerebral artery (MCA) was the most common origin of feeders, identified in 26.6% of the patients, with a significantly higher prevalence in the single feeder group (38.8%) compared to the multiple feeders group (21.2%; p < 0.001).
Feeders from the posterior cerebral artery (PCA) were more common in the single feeder group (16.7%) compared to the multiple feeders group (8.9%; p < 0.001). The vertebrobasilar (VB) system contributed to feeders in 4.2% of the patients, with a higher prevalence in the single feeder group (6.9%) than in the multiple feeders group (3.0%; p < 0.001). The posterior inferior cerebellar artery (PICA) was a feeder in 1.5% of the patients, more frequently in the single feeder group (3.5%) than in the multiple feeders group (0.6%; p < 0.001; Table 2).
Characteristics of feeding arteries.

Bold means “significant p-value”.
Outcomes
Patients with a single feeding artery had a higher rate of complete obliteration at the last follow-up (81.3%) compared to those with multiple feeders (70.1%; OR 0.53, 95% CI: 0.37–0.76, p = 0.001; Figure 1). Functional outcomes, as measured by the mRS score, were generally similar between the groups at various time points, though patients with multiple feeders showed better outcomes at the last follow-up (88.6% vs 80.4%, OR 1.90, 95% CI: 1.27–2.83, p = 0.002).
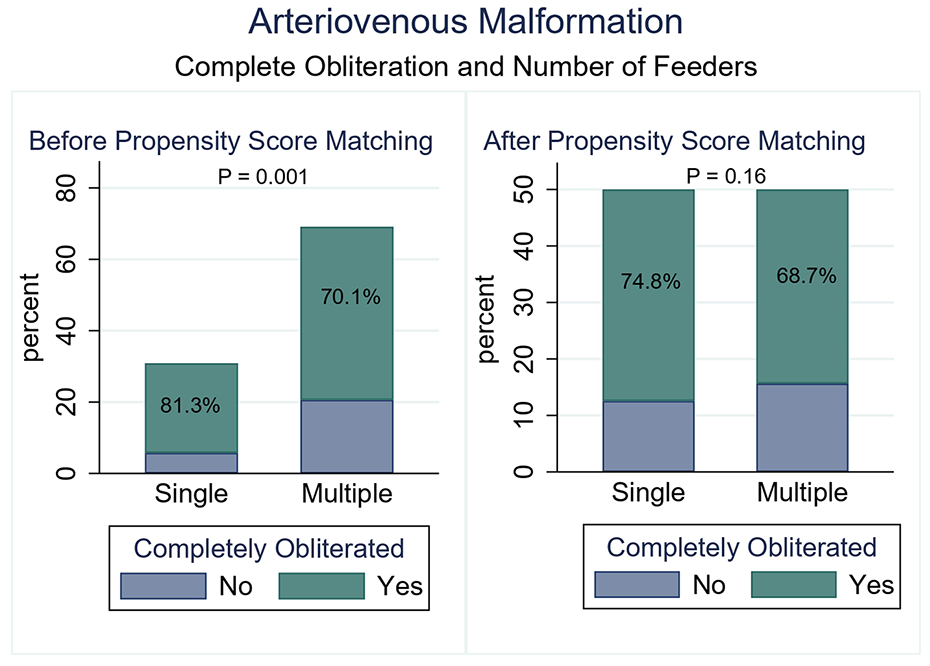
Comparison of complete obliteration rate at last follow-up between multiple and single feeders groups before and after PSM.
Symptomatic complications were more common in the multiple feeders group (19.2% vs 11.9%, OR 1.74, 95% CI: 1.16–2.61, p = 0.007; Table 3; Figure 2). No significant difference was found between the multiple and single feeders groups regarding hemorrhagic complications (4.3% vs 3.7%, OR 1.17, 95% CI: 0.57–2.37, p = 0.66) or mortality rates (1.2% vs 1.7%, OR 0.70, 95% CI: 0.22–2.17, p = 0.54).
Outcomes of patients included in the study before propensity score matching.

Bonferroni corrected p-value = 0.0033.
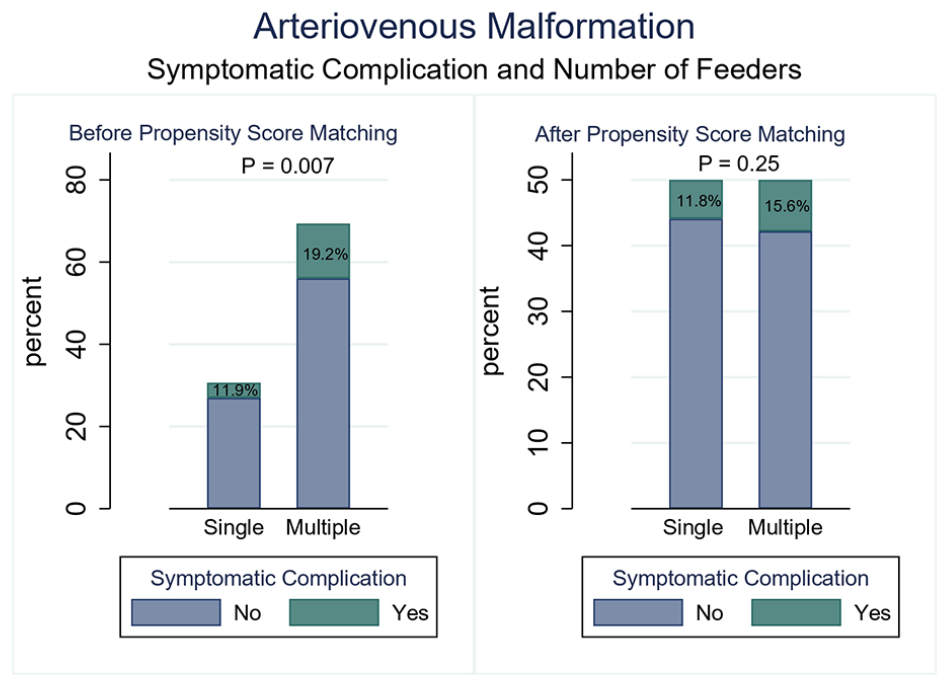
Comparison of symptomatic complication rate between multiple and single feeders groups before and after PSM.
Kaplan-Meier survival estimates were analyzed to assess the probability of patients remaining free from complete AVM obliteration over time, based on the origin of the feeders. The log-rank test indicated no significant difference between the different feeder origins (p = 0.22; Figure 3).
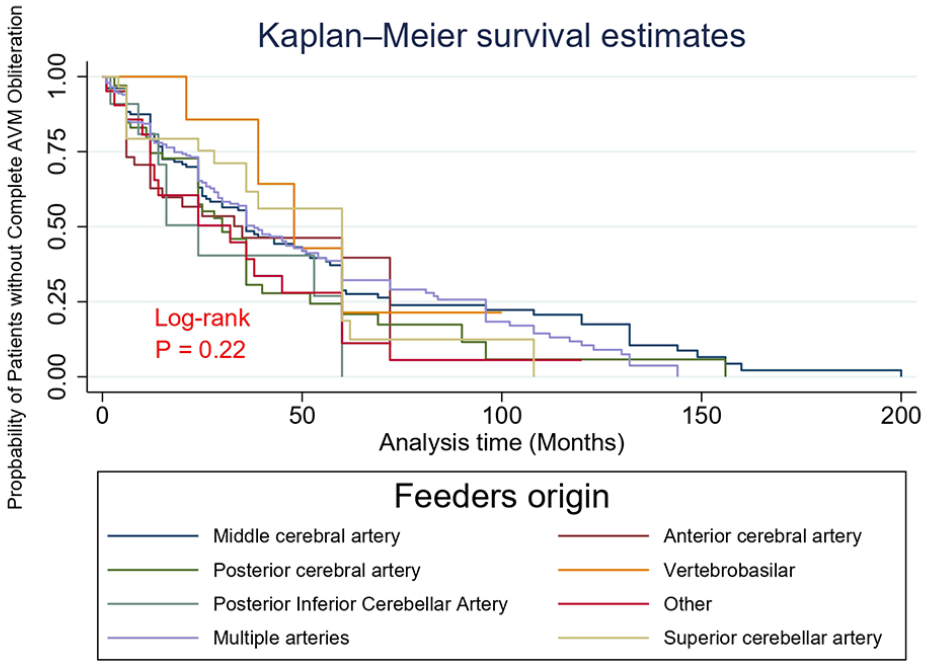
Kaplan-Meier curve comparing different feeders origin.
Propensity score matching
After propensity score matching, 422 patients were analyzed, with 211 in each cohort (Table 4). The rates of complete obliteration at the last follow-up were similar (68.7% for multiple feeders vs 74.8% for single feeders, OR 0.73, 95% CI: 0.48–1.12, p = 0.16; Figure 1). Functional outcomes, as measured by mRS scores at various follow-up points, showed no significant differences between the groups.
Comparison of baseline characteristics in patients with arteriovenous malformation according to number of feeding arteries after propensity score matching.
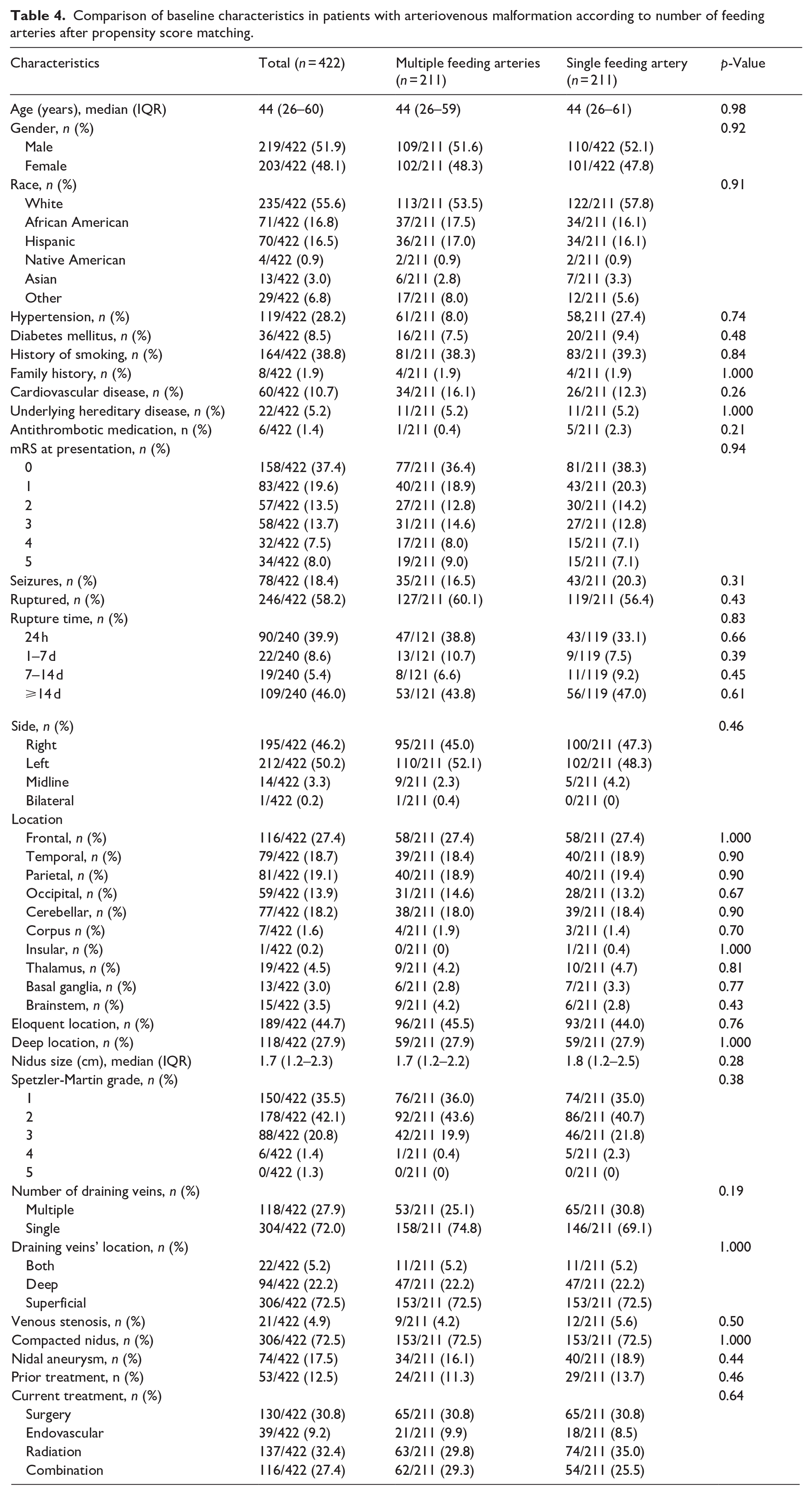
Symptomatic complications were not significantly different between the matched cohorts (15.6% for multiple feeders vs 11.8% for single feeders, OR 1.37, 95% CI: 0.78–2.41, p = 0.25; Table 5; Figure 2).
Outcomes of Patients Included in the Study after Propensity Score Matching.
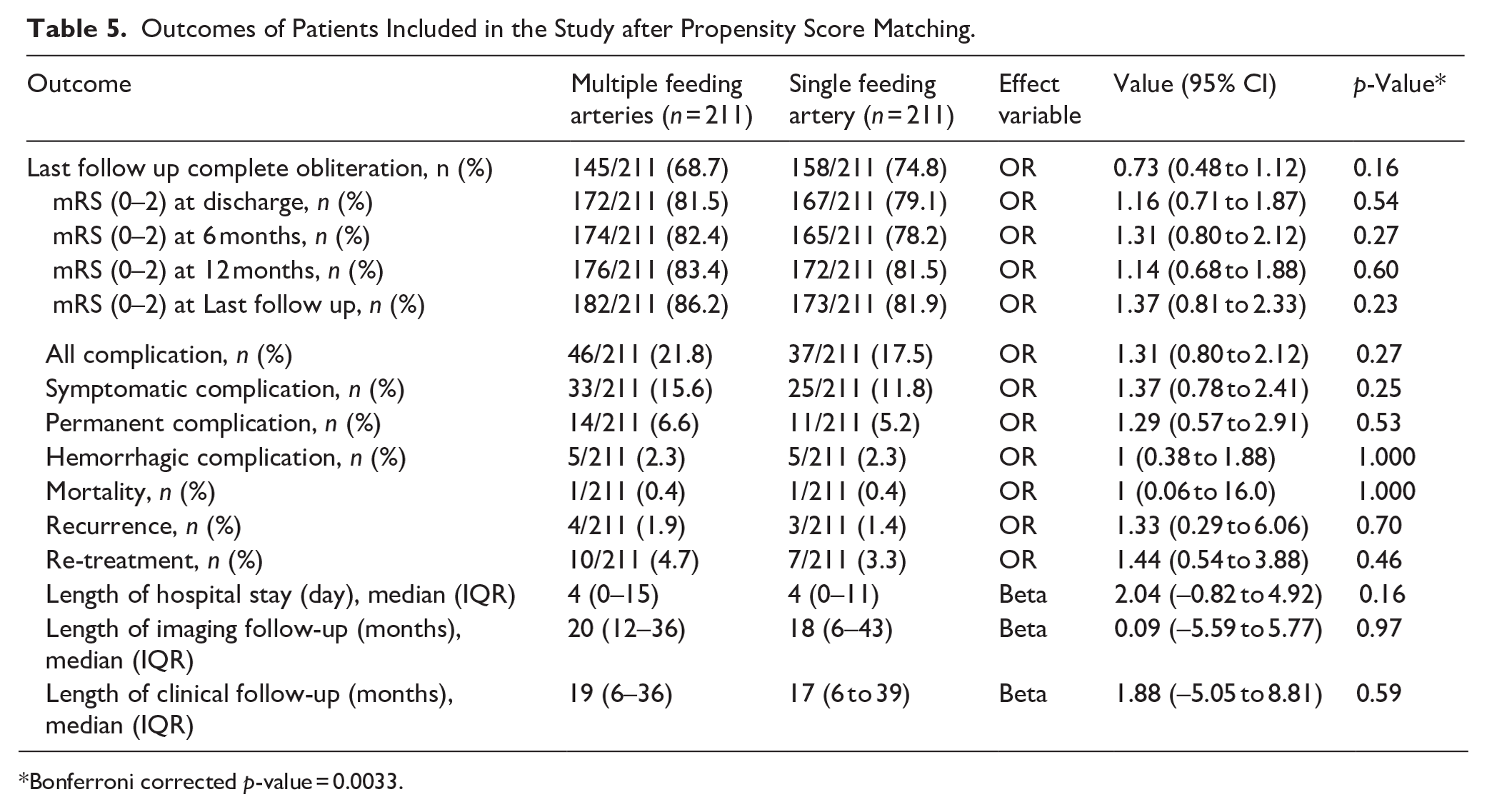
Bonferroni corrected p-value = 0.0033.
Subgroup analysis
Our sub-group analysis comparing ruptured and unruptured AVMs showed no significant differences between the two in terms of complete obliteration at last follow-up, mortality, complications, and functional outcomes (Figure 4). Similarly, sub-group analysis comparing different treatment options (surgery, radiosurgery, embolization, or combination) revealed no significant differences in terms of complete obliteration at last follow-up, symptomatic complications, or functional outcomes (Figure 5).
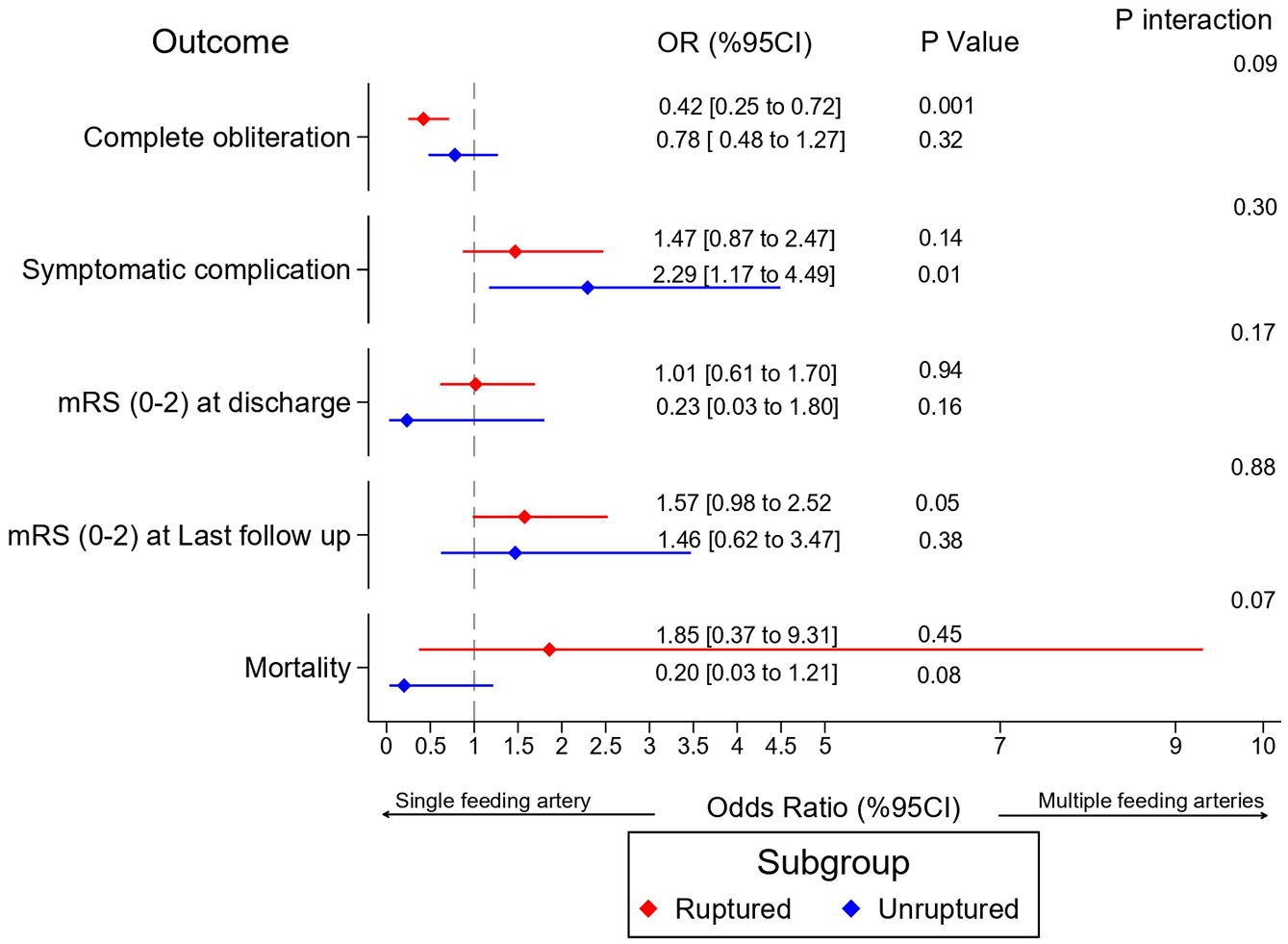
Sub-group analysis of ruptured and not ruptured AVMs.
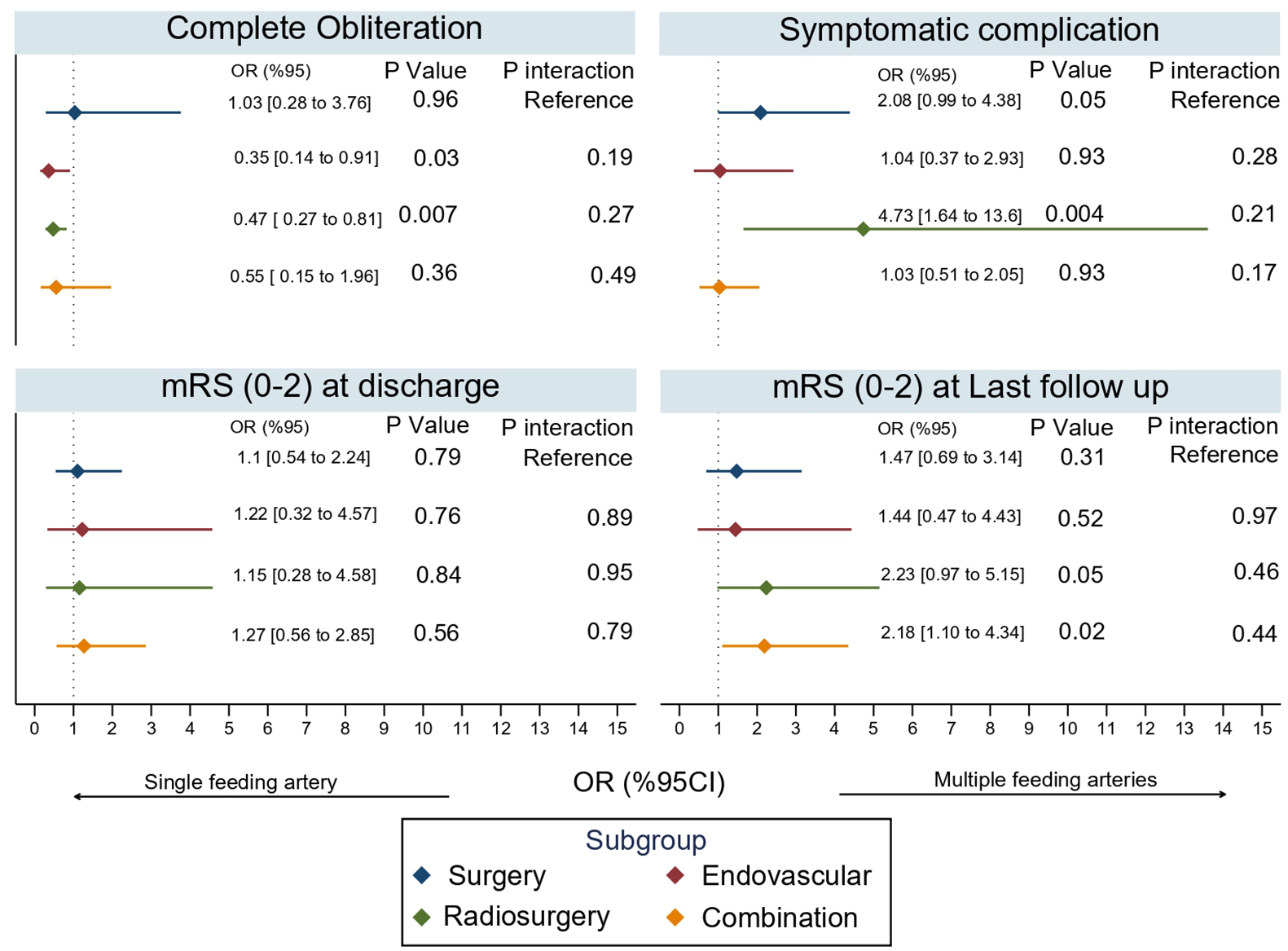
Sub-group analysis of different treatment types.
Multivariable logistic regression for feeders origin and complications
After adjusting for various factors, feeders originating from the PICA were associated with significantly higher odds of all complications (OR: 7.33, 95% CI: 2.14–25.1, p = 0.002). PICA feeders also had higher odds of symptomatic complications (OR: 4.27, 95% CI: 1.21–15.0, p = 0.02) and permanent complications (OR: 6.20, 95% CI: 1.51–25.3, p = 0.01; Table 6).
Feeders origin and complication regression model.

OR [CI], p-Value.
MCA: middle cerebral artery; ACA: anterior cerebral artery; PCA: posterior cerebral artery; VB: vertebrobasilar; PICA: posterior inferior cerebellar artery; SCA: superior cerebellar artery.
Bonferroni corrected p-value = 0.016.
All estimates were adjusted for age, mRS at presentation, number of feeders, venous stenosis, Spetzler-Martin Grade, location, nidus size (cm).
Discussion
In this multicenter retrospective study, we compared the outcomes of AVM patients with multiple feeding arteries to those with a single feeding artery. Patients with multiple feeders generally had more complex AVMs, evidenced by larger nidus sizes and higher Spetzler-Martin grades, though AVM patients with single feeding artery presented with more ruptured AVMs. AVMs with multiple feeders had lower obliteration rates and higher symptomatic complication rates. After propensity score matching, the differences in obliteration rates and complications were not significant. These changes are likely attributable to the covariate adjustments made during propensity score matching, which reduced baseline variability and imbalances between the groups. Subgroup analysis comparing ruptured and unruptured AVMs and different treatment options showed no significant differences across all subgroups.
Microcatheterization of feeding arteries is a critical step in the treatment of AVMs, particularly for endovascular embolization, as it allows precise delivery of embolic materials to the nidus. The ability to safely navigate these arteries depends on their anatomy, diameter, and tortuosity, which are often more complex in AVMs with multiple feeders.4,13–15,24 Furthermore, understanding the anatomy and number of feeders is essential for planning microsurgical approaches, as these factors influence the surgical strategy and risk of complications.1,4,10,11,24–28
High blood flow velocity and volume in feeding arteries can lead to structural changes in the vessel walls, known as high-flow angiopathy.1,24,29–32 These changes can include splitting or destruction of the internal elastic lamina, hyperplasia, fibrosis, or loss of smooth muscle cells, and endothelial cell proliferation. 1 Such changes often result in enlargement and tortuosity of the feeding arteries, as observed in many patients and reported by others. 1 These altered feeders can sometimes compress neural structures, causing neurological symptoms. 33
Studies and clinical practice have shown that AVMs with multiple feeders tend to have higher flow dynamics, which can result in increased pressure within the nidus and a greater likelihood of complications. 19 This is why some studies suggested the use of staged approaches in these kinds of AVMs. 19 While our study initially showed significantly higher rates of symptomatic complications in AVM patients with multiple feeders compared to those with single feeders, this difference was no longer significant after controlling for covariates through PSM. This suggests that, although the number of feeders is important for treatment planning and understanding AVMs, other factors such as size, location, nidus complexity, and high grade may play a more critical role in determining long-term efficacy and safety.
Lv et al. did a study to assess AVM characteristics in patients presented with hemorrhage and those with no hemorrhage. 21 They found single arterial feeders to be associated with higher hemorrhagic presentations. 21 Similarly, Chen et al. showed a significant high rate of ruptured AVMs in single feeders. 22 Our study aligning with this showed that single feeders have a significantly higher patients presenting with ruptured AVMs compared to multiple feeders. This could explain the findings observed before PSM where good functional outcomes were found to be significantly higher in the multiple feeders group.
Valavanis and Yaşargil investigated the relationship between number of feeders and obliteration rates and showed the number of feeders not to be a major determinant of obliteration rates. 34 Our study aligns with this finding. No significant differences were observed between the multiple and single feeders groups after PSM in terms of obliteration rates or complications. This supports the idea that factors other than the number of feeders, which influence AVM complexity, may have a more significant impact on outcomes.
Milatović et al. identified the anatomic features of the feeding arteries of AVMs. 1 They found that feeders commonly originate from MCA, followed by ACA and PCA. Our study aligns with this and emphasize the fact that the anatomical location of certain feeders’ origins can be associated with higher complication rates. 1 This why it’s important to identify both the anatomy and number of feeders in AVMs. 1 We found that feeders originating from PICA can have a significantly higher rates of complications compared to other locations. This could be explained by the fact that PICA lies close to the medulla and lower cranial nerves increasing the risk of damaging these structures during treatment, potentially leading to severe neurological deficits. 35 Additionally, lesions involving the PICA territory are often located in the posterior fossa, a region with limited space and a high density of critical neurovascular structures. 35 This confined space can complicate surgical access, increasing the risk of hemorrhage, and limit the ability to effectively manage complications once they arise. 35 Furthermore, the PICA territory is more prone to ischemia because of the limited collateral blood supply, which can exacerbate the severity of complications following interventions. 35
Limitations
This study has several limitations that must be considered. First, the retrospective nature of the data collection may introduce selection bias and affect the generalizability of the results. Second, the data were pooled from multiple academic institutions in North America and Europe, which might have differences in treatment protocols, patient management, and reporting practices. Third, despite the use of propensity score matching (PSM) to balance the cohorts and reduce confounding, residual confounding cannot be entirely excluded. The matching process relies on the measured variables, and any unmeasured confounders could still influence the outcomes. Additionally, while PSM minimizes confounding and enhances comparability, it may limit the generalizability of the findings to the overall population, as the matched cohort represents a specific subset of patients. This is particularly evident in the follow-up durations, where substantial changes in effect sizes were observed between the unmatched and matched analyses, reflecting the reduced variability in the matched dataset. Lastly, the classification of AVMs into multiple and single feeders was based on the data available, which might not account for all nuances in the angioarchitecture of AVMs. The complexity of AVM angioarchitecture and its impact on outcomes might be oversimplified by this binary classification.
Conclusion
This multicenter retrospective study reveals that cerebral AVMs with single feeding artery are more likely to present with rupture. However, no significant differences in obliteration rates or complications were observed between the multiple and single feeders groups after PSM. These findings suggest that factors other than the number of feeders may play a more significant role in influencing outcomes. Future prospective studies are necessary to validate these findings and refine management strategies for AVM patients.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251319924 – Supplemental material for Outcomes of arteriovenous malformation patients with multiple versus single feeders: A multicenter retrospective study with propensity-score matching
Supplemental material, sj-docx-1-eso-10.1177_23969873251319924 for Outcomes of arteriovenous malformation patients with multiple versus single feeders: A multicenter retrospective study with propensity-score matching by Basel Musmar, Nimer Adeeb, Hammam Abdalrazeq, Joanna M Roy, Stavropoula I Tjoumakaris, Hamza Adel Salim, Douglas Kondziolka, Jason Sheehan, Christopher S Ogilvy, Howard Riina, Sandeep Kandregula, Adam A Dmytriw, Kareem El Naamani, Ahmed Abdelsalam, Natasha Ironside, Deepak Kumbhare, Cagdas Ataoglu, Muhammed Amir Essibayi, Abdullah Keles, Sandeep Muram, Daniel Sconzo, Arwin Rezai, Omar Alwakaa, Salem M Tos, Ufuk Erginoglu, Johannes Pöppe, Rajeev D Sen, Christoph J Griessenauer, Jan-Karl Burkhardt, Robert M Starke, Mustafa K Baskaya, Laligam N Sekhar, Michael R Levitt, David J Altschul, Malia McAvoy, Assala Aslan, Abdallah Abushehab, Christian Swaid, Adib A Abla, Saman Sizdahkhani, Sravanthi Koduri, Elias Atallah, Spyridon Karadimas, M Reid Gooch, Robert H Rosenwasser, Christopher Stapleton, Matthew Koch, Visish M Srinivasan, Peng R Chen, Spiros Blackburn, Joseph Cochran, Omar Choudhri, Bryan Pukenas, Darren Orbach, Edward Smith, Pascal J Mosimann, Ali Alaraj, Mohammad A Aziz-Sultan, Aman B Patel, Hugo H Cuellar, Michael Lawton, Bharat Guthikonda, Jacques Morcos and Pascal Jabbour in European Stroke Journal
Supplemental Material
sj-jpg-2-eso-10.1177_23969873251319924 – Supplemental material for Outcomes of arteriovenous malformation patients with multiple versus single feeders: A multicenter retrospective study with propensity-score matching
Supplemental material, sj-jpg-2-eso-10.1177_23969873251319924 for Outcomes of arteriovenous malformation patients with multiple versus single feeders: A multicenter retrospective study with propensity-score matching by Basel Musmar, Nimer Adeeb, Hammam Abdalrazeq, Joanna M Roy, Stavropoula I Tjoumakaris, Hamza Adel Salim, Douglas Kondziolka, Jason Sheehan, Christopher S Ogilvy, Howard Riina, Sandeep Kandregula, Adam A Dmytriw, Kareem El Naamani, Ahmed Abdelsalam, Natasha Ironside, Deepak Kumbhare, Cagdas Ataoglu, Muhammed Amir Essibayi, Abdullah Keles, Sandeep Muram, Daniel Sconzo, Arwin Rezai, Omar Alwakaa, Salem M Tos, Ufuk Erginoglu, Johannes Pöppe, Rajeev D Sen, Christoph J Griessenauer, Jan-Karl Burkhardt, Robert M Starke, Mustafa K Baskaya, Laligam N Sekhar, Michael R Levitt, David J Altschul, Malia McAvoy, Assala Aslan, Abdallah Abushehab, Christian Swaid, Adib A Abla, Saman Sizdahkhani, Sravanthi Koduri, Elias Atallah, Spyridon Karadimas, M Reid Gooch, Robert H Rosenwasser, Christopher Stapleton, Matthew Koch, Visish M Srinivasan, Peng R Chen, Spiros Blackburn, Joseph Cochran, Omar Choudhri, Bryan Pukenas, Darren Orbach, Edward Smith, Pascal J Mosimann, Ali Alaraj, Mohammad A Aziz-Sultan, Aman B Patel, Hugo H Cuellar, Michael Lawton, Bharat Guthikonda, Jacques Morcos and Pascal Jabbour in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in the studies involving human participants were per the Institutional Review Board (IRB) ethical standards and national research committee and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
The study protocol was reviewed and approved by the Institutional Review Board. Following institutional guidelines, all protected health information was removed, and individual patient consent was not required in the analysis of the case series.
Guarantor
Pascal Jabbour
Contributorship
Basel Musmar, Nimer Adeeb, Hammam Abdalrazeq, Joanna M Roy, Stavropoula I Tjoumakaris, Hamza Adel Salim, Douglas Kondziolka, Jason Sheehan, Christopher S Ogilvy, Howard Riina, Sandeep Kandregula, Adam A Dmytriw, Kareem El Naamani, Ahmed Abdelsalam, Natasha Ironside, Deepak Kumbhare, Cagdas Ataoglu, Muhammed Amir Essibayi, Abdullah Keles, Sandeep Muram, Daniel Sconzo, Arwin Rezai, Omar Alwakaa, Salem M Tos, Ufuk Erginoglu, Johannes Pöppe, Rajeev D Sen, Christoph J Griessenauer, Jan-Karl Burkhardt, Robert M Starke, Mustafa K Baskaya, Laligam N Sekhar, Michael R Levitt, David J Altschul, Malia McAvoy, Assala Aslan, Abdallah Abushehab, Christian Swaid, Adib A Abla, Saman Sizdahkhani, Sravanthi Koduri, Elias Atallah, Spyridon Karadimas, M Reid Gooch, Robert H Rosenwasser, Christopher Stapleton, Matthew Koch, Visish M Srinivasan, Peng R Chen, Spiros Blackburn, Joseph Cochran, Omar Choudhri, Bryan Pukenas, Darren Orbach, Edward Smith, Pascal J Mosimann, Ali Alaraj, Mohammad A Aziz-Sultan, Aman B Patel, Hugo H Cuellar, Michael Lawton, Bharat Guthikonda, Jacques Morcos, Pascal Jabbour contributed to the conception, and design of the work.
Basel Musmar, Nimer Adeeb, Hammam Abdalrazeq, Joanna M Roy, Stavropoula I Tjoumakaris, Hamza Adel Salim, Douglas Kondziolka, Jason Sheehan, Christopher S Ogilvy, Howard Riina, Sandeep Kandregula, Adam A Dmytriw, Kareem El Naamani, Ahmed Abdelsalam, Natasha Ironside, Deepak Kumbhare, Cagdas Ataoglu, Muhammed Amir Essibayi, Abdullah Keles, Sandeep Muram, Daniel Sconzo, Arwin Rezai, Omar Alwakaa, Salem M Tos, Ufuk Erginoglu, Johannes Pöppe, Rajeev D Sen, Christoph J Griessenauer, Jan-Karl Burkhardt, Robert M Starke, Mustafa K Baskaya, Laligam N Sekhar, Michael R Levitt, David J Altschul, Malia McAvoy, Assala Aslan, Abdallah Abushehab, Christian Swaid, Adib A Abla, Saman Sizdahkhani, Sravanthi Koduri, Elias Atallah, Spyridon Karadimas, M Reid Gooch, Robert H Rosenwasser, Christopher Stapleton, Matthew Koch, Visish M Srinivasan, Peng R Chen, Spiros Blackburn, Joseph Cochran, Omar Choudhri, Bryan Pukenas, Darren Orbach, Edward Smith, Pascal J Mosimann, Ali Alaraj, Mohammad A Aziz-Sultan, Aman B Patel, Hugo H Cuellar, Michael Lawton, Bharat Guthikonda, Jacques Morcos, Pascal Jabbour were involved in the acquisition of data, and data analysis and interpretation.
Basel Musmar, Nimer Adeeb, Hammam Abdalrazeq, Joanna M Roy, Stavropoula I Tjoumakaris, Hamza Adel Salim, Douglas Kondziolka, Jason Sheehan, Christopher S Ogilvy, Howard Riina, Sandeep Kandregula, Adam A Dmytriw, Kareem El Naamani, Ahmed Abdelsalam, Natasha Ironside, Deepak Kumbhare, Cagdas Ataoglu, Muhammed Amir Essibayi, Abdullah Keles, Sandeep Muram, Daniel Sconzo, Arwin Rezai, Omar Alwakaa, Salem M Tos, Ufuk Erginoglu, Johannes Pöppe, Rajeev D Sen, Christoph J Griessenauer, Jan-Karl Burkhardt, Robert M Starke, Mustafa K Baskaya, Laligam N Sekhar, Michael R Levitt, David J Altschul, Malia McAvoy, Assala Aslan, Abdallah Abushehab, Christian Swaid, Adib A Abla, Saman Sizdahkhani, Sravanthi Koduri, Elias Atallah, Spyridon Karadimas, M Reid Gooch, Robert H Rosenwasser, Christopher Stapleton, Matthew Koch, Visish M Srinivasan, Peng R Chen, Spiros Blackburn, Joseph Cochran, Omar Choudhri, Bryan Pukenas, Darren Orbach, Edward Smith, Pascal J Mosimann, Ali Alaraj, Mohammad A Aziz-Sultan, Aman B Patel, Hugo H Cuellar, Michael Lawton, Bharat Guthikonda, Jacques Morcos, Pascal Jabbour drafted the work, and revised it critically for important intellectual content.
All authors gave final approval of the version to be published and agree to be accountable for all aspects of the work.
ORCID iDs
Data sharing
The relevant anonymized patient-level data are available on reasonable request from the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
