Abstract
Introduction:
The degree of culprit artery stenosis affects the risk of early neurological deterioration (END) after acute ischemic stroke (AIS). The TREND trial demonstrated the efficacy of tirofiban in preventing END in patients with AIS. We aimed to investigate whether the degree of intracranial artery stenosis affects the efficacy of tirofiban in preventing END in patients with AIS.
Patients and Methods:
We conducted a post hoc analysis of the TREND trial, which enrolled patients within 24 h of onset and randomly allocated to receive intravenous tirofiban or oral aspirin. We stratified the stenosis degrees into three subgroups: no stenosis, mild-to-moderate stenosis (stenosis <70%), and severe stenosis or occlusion (stenosis ⩾70%). The primary endpoint is END4 defined as an increase of the NIHSS ⩾4 within 72 h after randomization. Secondary outcomes include END2 (defined as an increase of NIHSS ⩾2) within 72 h after randomization, the proportion of mRS 0–1 and 0–2 at 90 days.
Results:
A total of 296 patients were analyzed. In patients with severe stenosis or occlusion, tirofiban significantly reduced the incidence of END4 (5.7% vs 30.8%, adjusted OR 0.156, 95% CI 0.028–0.873, adjusted p = 0.034), whereas its effects in preventing END4 were similar to those of aspirin in patients with no stenosis (2.4% vs 4.6%, adjusted OR 0.193, 95% CI 0.018–2.083, adjusted p = 0.175) or mild-to-moderate stenosis (2.9% vs 10.0%, adjusted OR 0.171, 95% CI 0.015–1.943, adjusted p = 0.155). The p value for interaction between stenosis subgroups and treatment was 0.513. Furthermore, tirofiban significantly reduced the incidence of END2 in patients with mild-to-moderate stenosis (5.9% vs 22.5%, OR 0.146, 95% CI 0.022–0.951, adjusted p = 0.044) and severe stenosis or occlusion (11.4% vs 43.6%, adjusted OR 0.140, 95% CI 0.036–0.540, adjusted p = 0.004). A significant improvement in favorable outcomes with a 90-day mRS of 0–1 was observed only in patients with mild-to-moderate stenosis (85.3% vs 70.0%, adjusted OR 4.617, 95% CI 1.077–19.798, adjusted p = 0.039).
Discussion and conclusion:
Tirofiban may significantly reduce the incidence of END in patients with severe arterial stenosis or occlusion. Further studies are required to confirm the effects of intracranial artery stenosis on the benefits of intravenous tirofiban.
Trial registration:
ClinicalTrials.gov; identifier: NCT04491695.
This is a visual representation of the abstract.
Introduction
Up to 40% of patients experience neurological deterioration in the early stages of acute ischemic stroke (AIS), which is strongly associated with unfavorable outcomes. 1 Notably, ischemic progression plays a pivotal role in this early neurological deterioration (END), accounting for almost over 70% of all the worsening causes.2,3 Previous studies demonstrated that patients with severe stenosis or occlusion have a heightened risk of 46%–65% of developing END compared to 8.5%–10% in those with normal arterial findings.4–6 Individuals with artery stenosis or occlusion are especially susceptible to the influences of in-situ thrombus extension and hemodynamic decline, ultimately leading to expansion infarction growth and neurological deterioration.7–9
To date, many studies have investigated the effects of aggressive antithrombotic therapy in preventing END after ischemic stroke.10–13 Tirofiban, a non-peptide selective inhibitor of the glycoprotein IIb/IIIa (GP IIb/IIIa) receptor on platelets, inhibits the final common pathway of platelet aggregation, impedes thrombus formation, 14 and it has been widely investigated in patients with AIS. Recently, the TREND trial demonstrated that intravenous tirofiban administered within 24 h of stroke onset could effectively prevent END compared to oral aspirin in patients with acute non-cardioembolic ischemic stroke with good safety profiles. 15 However, whether the culprit intracranial artery stenosis has any impact on the effects of tirofiban in preventing END remains unclear. As different degrees of stenosis are strongly associated with the risk of END and its characteristics, we hypothesized that the degree of intracranial artery stenosis influences the efficacy of tirofiban in preventing END. In this post hoc analysis of the TREND trial, we aimed to assess the influence of intracranial artery stenosis on the effectiveness of tirofiban in preventing END in patients with AIS.
Methods
Study design and patients
This study is a post hoc analysis of the TREND trial, an investigator-initiated, multicenter, prospective, randomized controlled, open-label trial with a blinded endpoint assessment that evaluated the safety and efficacy of intravenous tirofiban administered within 24 h of stroke onset in preventing END in patients with acute non-cardioembolic stroke, in comparison to oral aspirin. Between September 2020 and March 2023, the TREND study enrolled 425 patients from 10 comprehensive stroke centers across China. The participants were randomly assigned to either the tirofiban group or the aspirin group. Details of the trial design, study protocol, and statistical analysis plan have been published.15,16 Patients were included in this study if vessel imaging data were available that definitely demonstrated the degree of stenosis of the culprit artery. In addition, patients with uncertain infarction lesions, cardioembolic stroke, a baseline NIHSS score of less than 4 points, or those who underwent intravenous thrombolysis, anticoagulation therapy, or endovascular treatment within 72 h of randomization were excluded from the study.
This post hoc analysis was designed to investigate the influence of intracranial artery stenosis on the effects of tirofiban in preventing END in patients with AIS. This study was approved by the ethics committees of all the participating centers. The study is a post hoc analysis of an randomized controlled trial, and informed consent was obtained at the time the study was conducted. The data supporting the findings of this study are available from the corresponding author upon request.
Collected variables and stenosis classification
We examined the baseline variables gathered from the TREND study, including age; sex; medical histories such as hypertension, diabetes, dyslipidemia, ischemic stroke, and intracerebral hemorrhage; smoking status; previous antiplatelet therapy; baseline NIHSS score; blood pressure at admission; glucose levels at admission; time from stroke onset to randomization; infarct location; and stroke etiology. The stroke etiology was determined based on the criteria established by the Trial of Org 10172 in Acute Stroke Treatment (TOAST). 17 Vessel imaging examinations, including computed tomography angiography (CTA) and magnetic resonance angiography (MRA), were performed either prior to enrollment or during patients’ hospitalization.
The culprit vessel was determined by two independent physicians who were blinded to the patient’s baseline characteristics and treatment outcomes based on vascular imaging examinations combined with the infarct lesion and clinical features. In cases of any dispute, a third physician was consulted to make the final decision. The degree of intracranial vessel stenosis was measured using the Warfarin-Aspirin Symptomatic Intracranial Disease Study (WASID) method. 18 Patients were categorized into three groups: no stenosis, mild-to-moderate stenosis (<70%), and severe stenosis or occlusion (⩾70%).
Outcome assessment
The primary efficacy endpoint was END4, defined as an increase in the NIHSS score by 4 or more points at any time within 72 h of randomization compared to the score immediately before randomization.
The secondary efficacy endpoints included: END2 (defined as an NIHSS score increase of ⩾2 points within 72 h of randomization), early improvement (defined as an NIHSS score decrease of ⩾4 points within 72 h of randomization), and excellent functional outcome (mRS score of 0–1) and functional independence (mRS score of 0–2) at 90 days.
Safety endpoints included the incidence of symptomatic intracerebral hemorrhage (ICH) and any ICH within 72 h after randomization (according to the European Cooperative Acute Stroke Study III criteria 19 ), systemic bleeding within 72 h after randomization, and death from any cause within 90-day follow-up.
Statistical analysis
Baseline patient characteristics and clinical outcomes were compared between the tirofiban and aspirin treatment arms within each of three subgroups: patients with no stenosis, mild-to-moderate stenosis, and severe stenosis or occlusion. The baseline characteristics were summarized using descriptive statistics. Continuous data were presented with mean±SD or medians (IQR), and differences between groups were tested using two-sided t-tests or Mann–Whitney U Tests, as appropriate. Categorical data are summarized as frequency (%), with χ2 tests for comparisons. For unadjusted analysis, we used univariate binary logistic regression to compare the outcomes between the groups. In the adjusted analysis, multivariate analysis was performed to detect the impact of treatment on efficacy and safety outcomes after accounting for factors such as age, sex, infarction location, baseline NIHSS score, and pre-stroke antiplatelet therapy. We assessed interactions between treatment assignment and stenosis stratification by including the terms for treatment, subgroup, and treatment-by-subgroup interaction in the model with p < 0.05 for a statistically significant interaction effect. Furthermore, Kaplan-Meier failure curves were used to illustrate the cumulative risk of primary endpoint events within 7 days of randomization across different stratifications. Statistical analyses were conducted using R 4.2.3 for Kaplan-Meier failure curves, and SPSS version 26.0.
Results
Among the 425 patients randomized in the TREND study, 129 were excluded from the post hoc analysis, including 29 lacking vessel imaging examinations and 62 without a clear acute infarction lesion. A total of 296 patients were included in this post hoc analysis, with 152 in the tirofiban group and 144 in the aspirin group. There were 148 (50%) patients without stenosis (83 receiving tirofiban and 65 receiving aspirin), 74 (25%) with mild-to-moderate stenosis (34 receiving tirofiban and 40 receiving aspirin), and 74 (25%) with severe stenosis or occlusion (35 receiving tirofiban and 39 receiving aspirin). In this study, intracranial artery stenosis was primarily attributed to atherosclerosis, but one patient was suspected to have an immune-mediated cause of vascular occlusion. A patient flowchart of this study is shown in Figure 1. The baseline characteristics of patients with different stenosis degrees and treatment allocations are presented in Table 1.
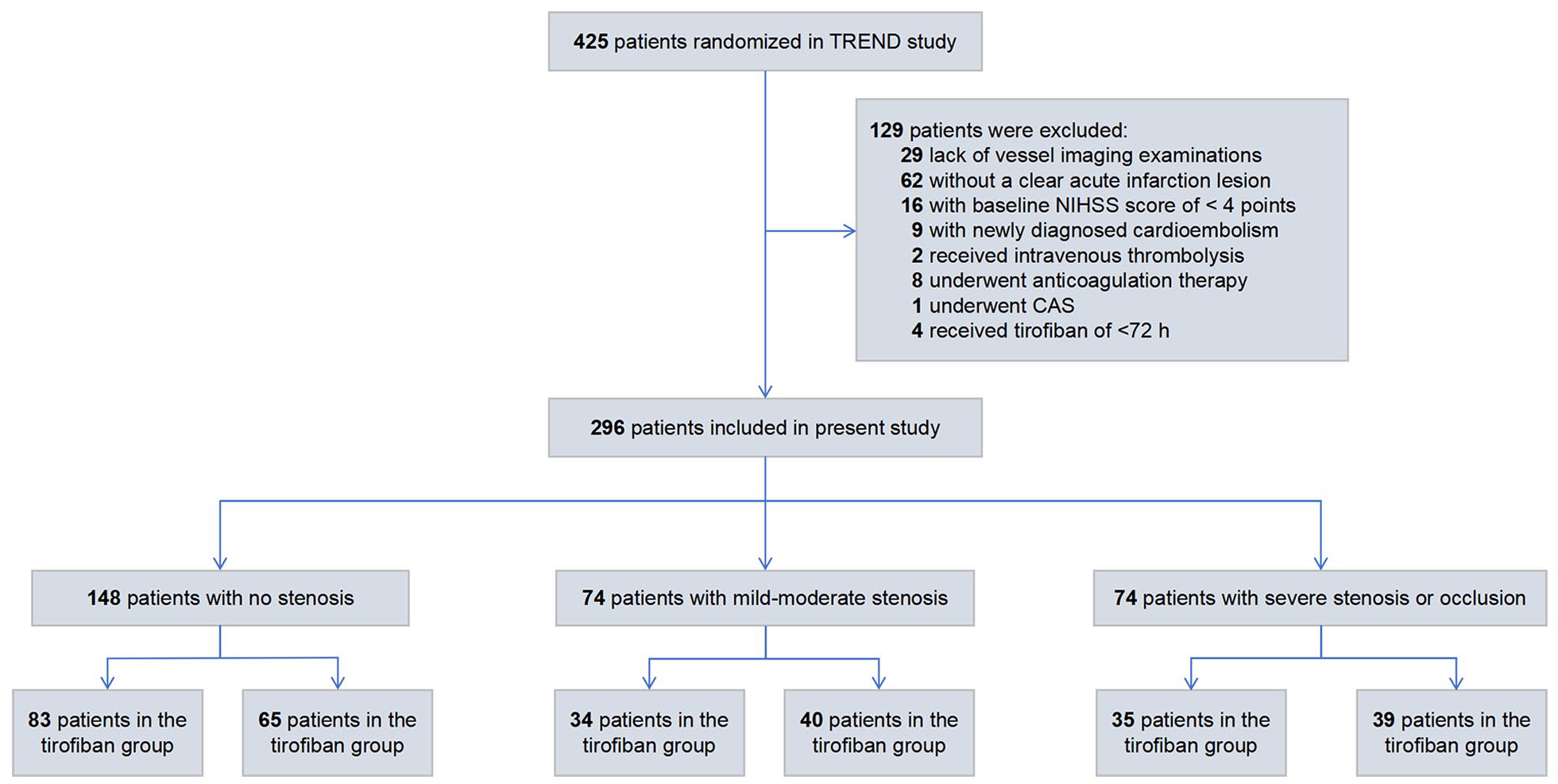
Patient flow chart.
Baseline characteristics of patients with different stenosis stratifications.
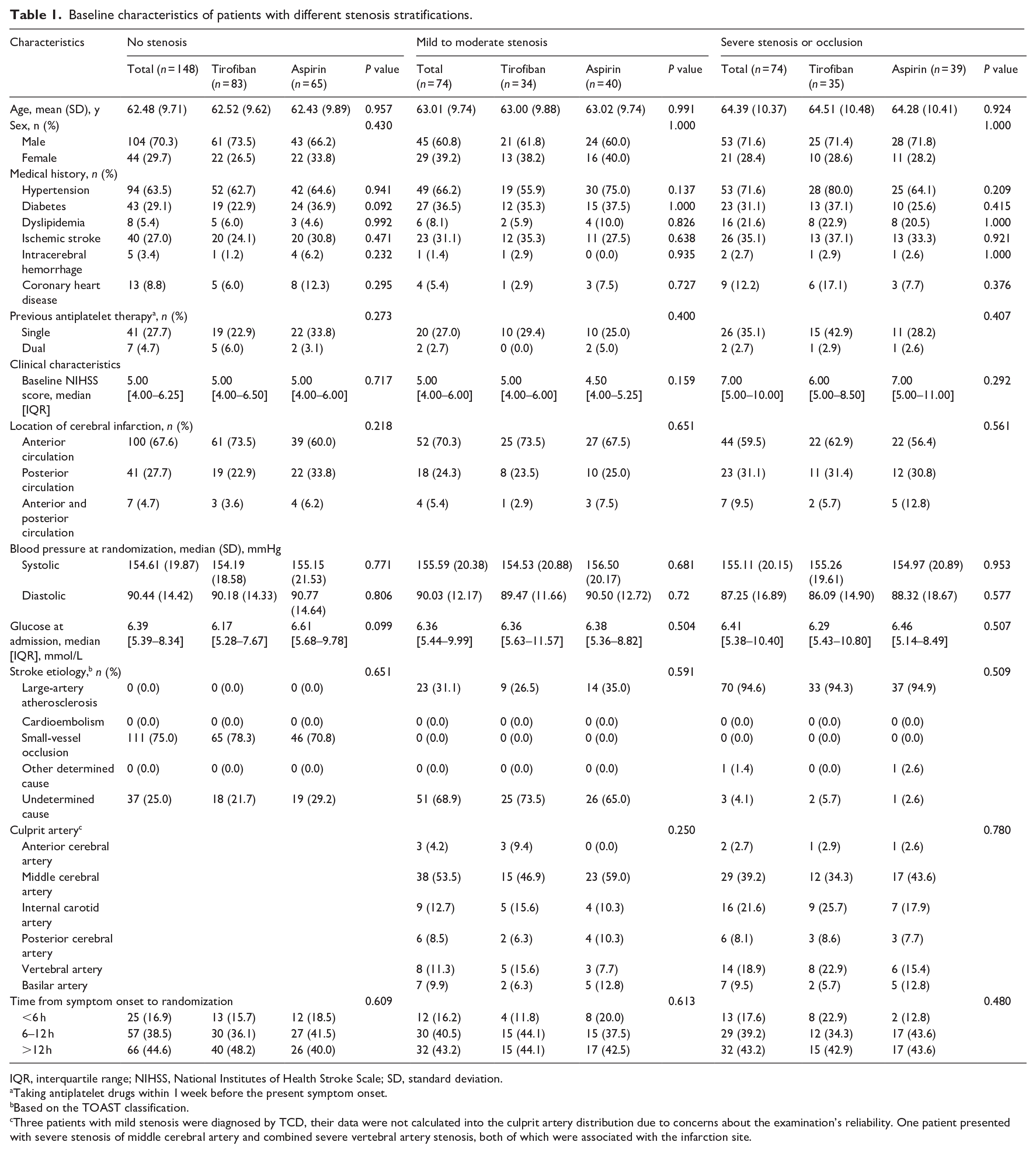
IQR, interquartile range; NIHSS, National Institutes of Health Stroke Scale; SD, standard deviation.
Taking antiplatelet drugs within 1 week before the present symptom onset.
Based on the TOAST classification.
Three patients with mild stenosis were diagnosed by TCD, their data were not calculated into the culprit artery distribution due to concerns about the examination’s reliability. One patient presented with severe stenosis of middle cerebral artery and combined severe vertebral artery stenosis, both of which were associated with the infarction site.
Primary efficacy outcome
A total of 24 patients experienced an END4. Among the patients with no stenosis, 2 of 83 (2.4%) in the tirofiban group and 3 of 65 (4.6%) in the aspirin group experienced END4. Among the patients with mild-to-moderate stenosis, 1 of 34 (2.9%) in the tirofiban group and 4 of 40 (10.0%) in the aspirin group experienced END4. Among patients with severe stenosis or occlusion, two of 35 (5.7%) receiving tirofiban and 12 of 39 (30.8%) receiving aspirin experienced END4.
In the adjusted analysis, tirofiban significantly reduced the incidence of END4 in patients with severe stenosis or occlusion (adjusted OR 0.156; 95% CI 0.028–0.873; adjusted p = 0.034; Figure 2). This effect was less pronounced in those with mild-to-moderate stenosis (adjusted OR 0.171; 95% CI 0.015–1.943; adjusted p = 0.155; Figure 2) and those without stenosis (adjusted OR 0.193; 95% CI 0.018–2.083; adjusted p = 0.175; Figure 2). The subgroup×treatment arm interaction was not significant (p for interaction = 0.513; Figure 2).
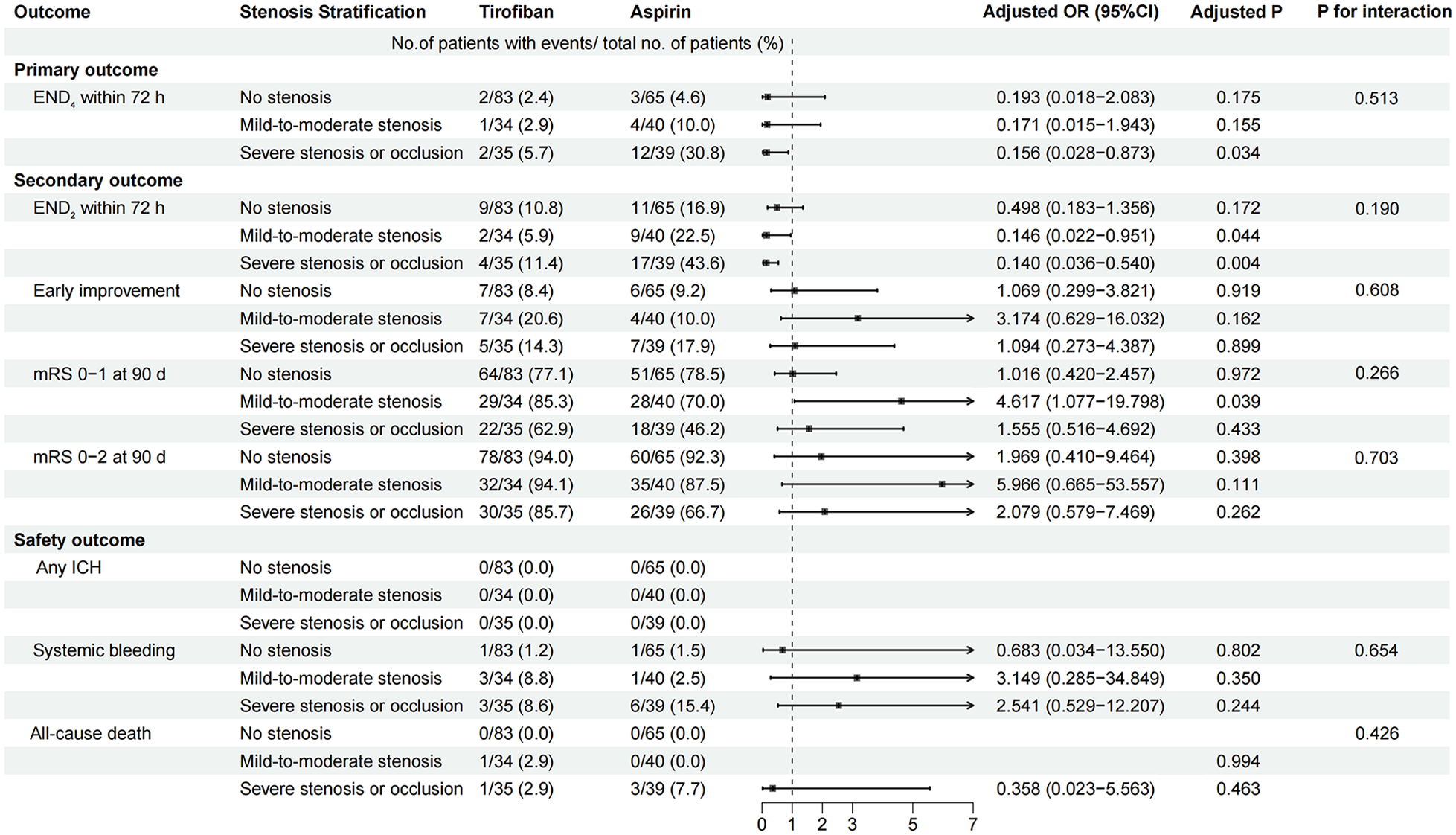
Association of treatment with clinical outcomes stratified by stenosis degrees. Adjusted for age, sex, infarction location, baseline NIHSS score, and pre-stroke antiplatelet therapy.
The cumulative risk of END4 among the different stenosis subgroups in the two treatment assignments is illustrated in Figure 3. There was variation in the cumulative risk of END4 within 7 days among patients belonging to the different stenosis severity subgroups (p = 0.00035, Figure 3(a)). Specifically, among patients with severe stenosis or occlusion, patients treated with tirofiban exhibited a significantly lower cumulative risk of END4 development within 7 days than those receiving aspirin therapy (p = 0.006, Figure 3(d)).
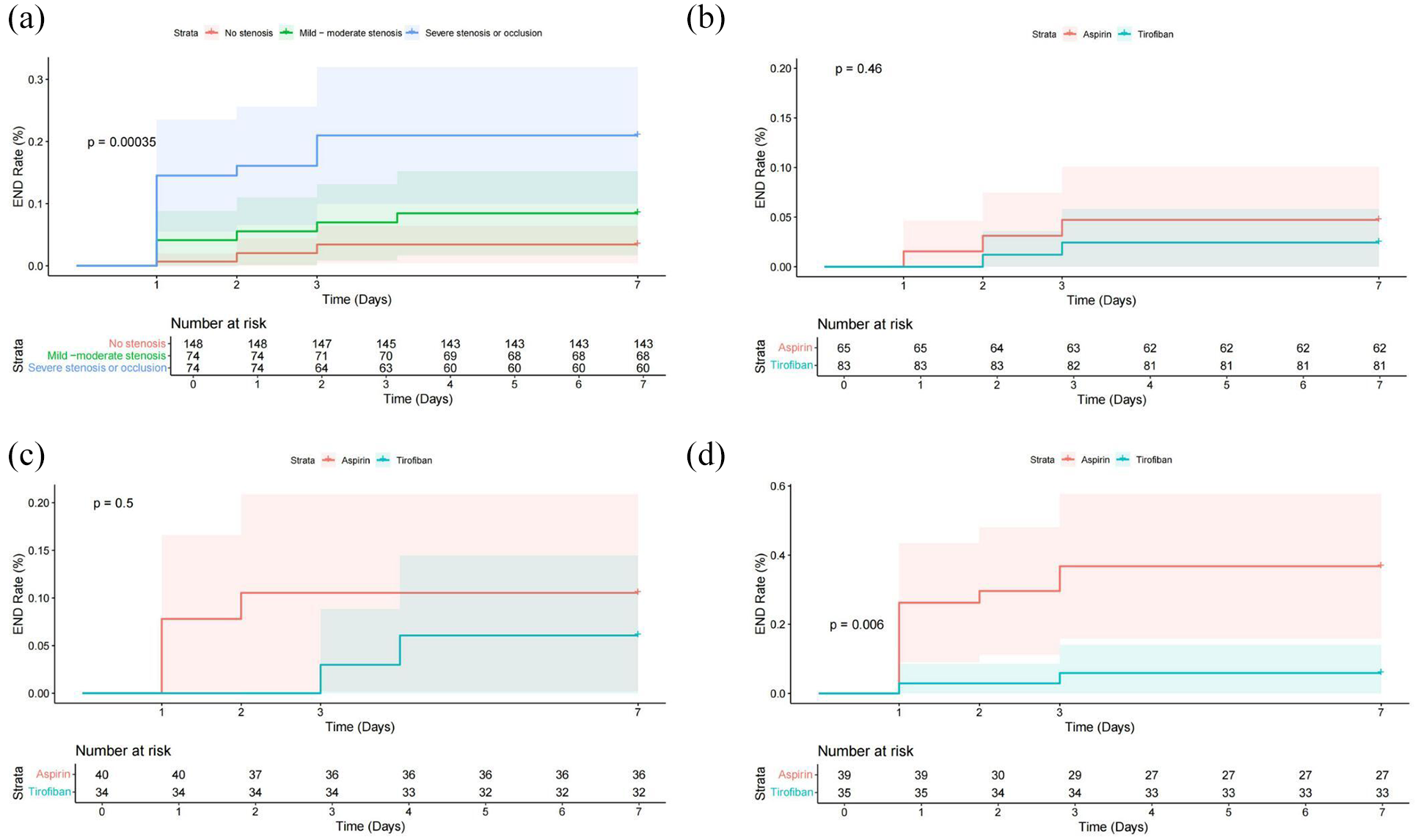
Cumulative risk of early neurological deterioration in patients with different stenosis: (a) total cohort, (b) no stenosis, (c) mild-to-moderate stenosis, and (d) severe stenosis/occlusion
Secondary efficacy outcomes
If END was defined as an increase in the NIHSS score of no less than 2 points within 72 h of onset, supportive evidence for tirofiban treatment was observed in patients with severe stenosis or occlusion (11.4% vs 43.6%, adjusted OR 0.140; 95% CI 0.036–0.540; adjusted p = 0.004; Figure 2) and mild-to-moderate stenosis (5.9% vs 22.5%, adjusted OR 0.146; 95% CI 0.022–0.951; adjusted p = 0.044; Figure 2). Among patients with mild-to-moderate stenosis, 29 of 34 patients (85.3%) in the tirofiban group and 28 of 40 patients (70.0%) in the aspirin group achieved an mRS score of 0–1 at 90 days (adjusted OR 4.617; 95% CI 1.077–10.798; adjusted p = 0.039; Figures 2 and 4). Regarding the outcomes of early improvement and the proportion of mRS 0–2 at 90 days, intravenous tirofiban and oral aspirin had similar effects in patients of all subgroups (p > 0.05, Figures 2 and 4).
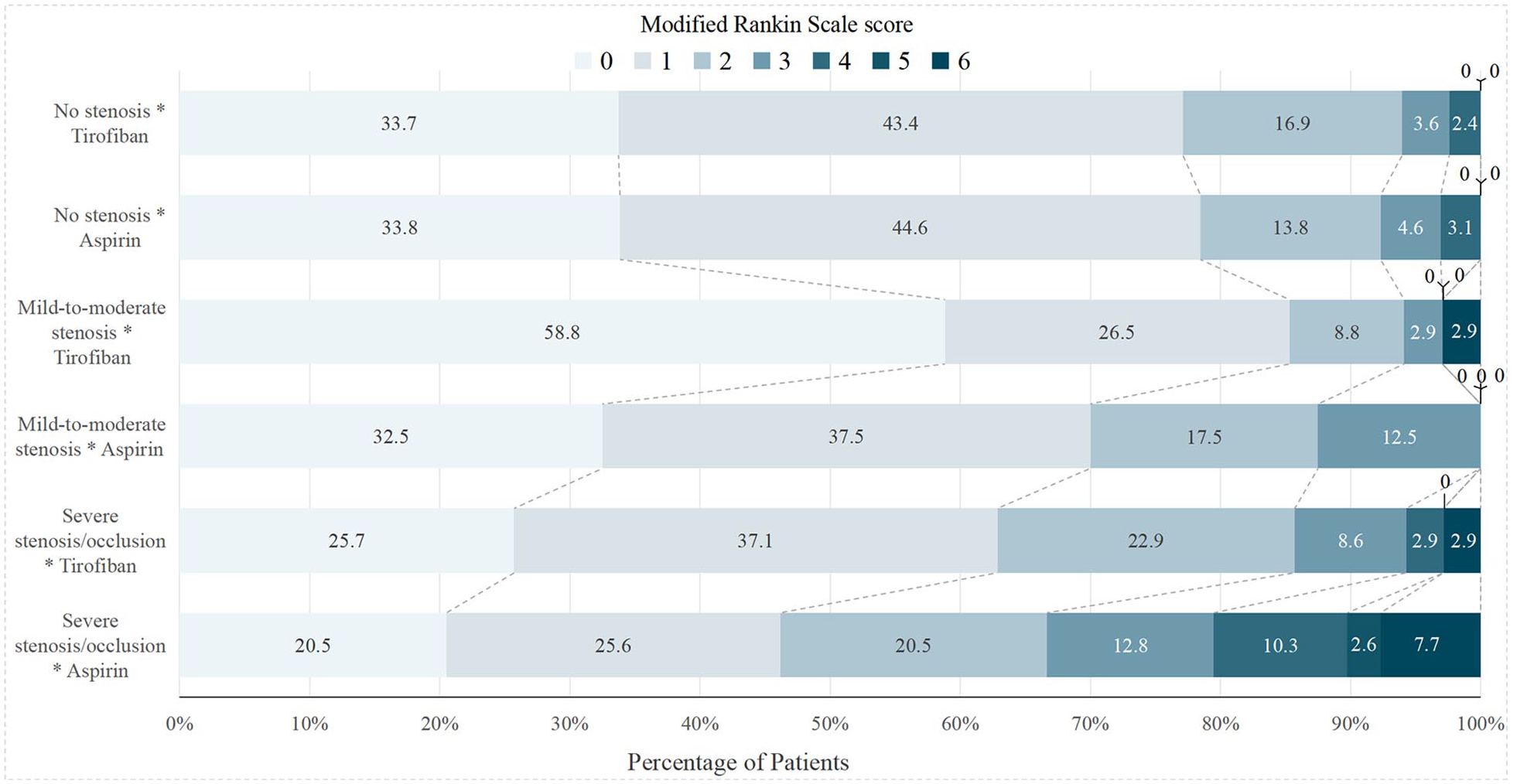
Distribution of mRS score at 90 days. Score range from 0 to 6, with 0 indicating no symptoms, 1, no clinically significant disability, 2, slight disability, 3, moderate disability, 4, moderately severe disability, 5, severe disability, and 6, death.
Safety outcomes
None of the patients in either treatment group experienced ICH. Patients with different degrees of stenosis in the tirofiban group and aspirin group had similar rates of systemic bleeding (1.2% vs 1.5% in patients with no stenosis, adjusted p = 0.802; 8.8% vs 2.5% in patients with mild-to-moderate stenosis, adjusted p = 0.350; and 8.6% vs 15.4% in patients with severe stenosis or occlusion, respectively; adjusted p = 0.244, Figure 2). One patient with mild stenosis who received tirofiban treatment and four patients with occluded arteries (one in the tirofiban group and three in the aspirin group) died during the 90-day follow-up period. Mortality did not differ significantly between the treatment groups for any of the stenosis subtypes.
Discussion
In this post hoc analysis of the TREND trial, we found that intravenous tirofiban significantly reduces the risk of END among AIS patients with culprit artery severe stenosis or occlusion, with no significant benefit of tirofiban in other stenosis stratification were observed. In addition, among patients with culprit artery stenosis of less than 70%, intravenous tirofiban can also lower the risk of END (if defined as ⩾2-point NIHSS score increase) and improve the proportion of favorable outcomes. However, there is no statistically significant difference in treatment effects across subgroups with different degrees of stenosis.
While the point estimates are similar across subgroups, the significant p value observed in the severe stenosis or occlusion subgroup provides indicative evidence of a treatment benefit specifically within this population, which could hold clinical significance. 20 The possible explanations are as follows: On the one hand, stroke etiology may be one of the driving factors behind these differences in treatment efficacy, particularly in the group of patients without arterial stenosis, where the majority had small vessel disease as the cause of their stroke. Within this group, artery-to-artery embolism and distal small vessel pathology may be the primary stroke mechanisms, and antiplatelet therapy may not work. On the other hand, in patients with AIS secondary to intracranial artery stenosis, rupture of atherosclerotic plaques and release of tissue factor from the endothelial surface trigger platelet adhesion and aggregation, ultimately leading to the formation of in-situ thrombosis that extends beyond the existing stenosis and intensifies luminal narrowing to a critical level. 21 This thrombosis can either obstruct the stenosed artery or detach completely or partially, causing distal embolism. 22 Furthermore, in cases of severe vascular stenosis, progressive narrowing diminishes antegrade blood flow, resulting in hypoperfusion, which is exacerbated in cases of hypovolemia or hypotension.23–26 Consequently, this scenario leads to a heightened incidence of neurological deterioration and stroke recurrence, which may benefit more from aggressive antiplatelet therapy.27,28
The clinical benefit findings of this study were consistent with those of the ESCAPITST trial, which investigated intravenous tirofiban for 48 h or oral aspirin 100 mg/day in non-cardioembolic moderate-to-severe stroke patients within 12 h of symptom onset who did not receive revascularization therapy and found that tirofiban significantly increased the proportion of patients with favorable outcomes among those with severe stenosis or occlusion (59.7% vs 34.3%, p = 0.002), but had no significant effect on patients with mild stenosis.29,30 In our study, this proportion of patients with mRS scores of 0 and 1 was higher in the tirofiban group than in the aspirin group (62.9% vs 46.2%), although the difference was not statistically significant. Furthermore, among patients with culprit artery stenosis of less than 70%, the proportion of favorable outcomes significantly improved.
This study had several limitations. First, this study enrolled patients with baseline NIHSS scores of 4–20, and the benefit of intravenous tirofiban was only detected in patients with severe stenosis or occlusion; therefore, the benefits of intravenous tirofiban cannot be extrapolated to the entire patient population. Second, the study design did not mandate a repeat brain CT scan during or after the intervention unless neurological deterioration was observed, potentially underestimating the actual risk of intracranial hemorrhage. In addition, the relatively small sample size and low incidence of primary endpoint events across the subgroups resulted in extremely wide 95% CIs for some of our key findings, which reflect the uncertainty associated with our estimates. Furthermore, it is noteworthy that even though there was no statistically significant difference in the effectiveness of tirofiban among non-severe stenosis subgroups, the OR values approached those of the severe stenosis or occlusion patients after adjustment. This necessitates consideration of both the rarity of the endpoint events and the selection bias inherent in post hoc subgroup analyses, which may have diluted the differences in efficacy among different subgroups. Therefore, our findings should be considered exploratory and hypothesis-generating rather than definitive. Larger studies to improve the accuracy of statistical estimates in the future and further validate and expand our findings are needed.
Conclusion
In patients with acute non-cardioembolic ischemic stroke, intravenous tirofiban within 24 h of onset is significantly associated with a reduced incidence of END in those with severe artery stenosis or occlusion. Further studies are required to confirm the effects of intracranial artery stenosis on the efficacy of intravenous tirofiban.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251319151 – Supplemental material for Effects of tirofiban in preventing neurological deterioration in acute ischemic stroke with intracranial artery stenosis: A post hoc analysis of the TREND Trial
Supplemental material, sj-docx-1-eso-10.1177_23969873251319151 for Effects of tirofiban in preventing neurological deterioration in acute ischemic stroke with intracranial artery stenosis: A post hoc analysis of the TREND Trial by Jing Wang, Yue Qiao, Sijie Li, Chuanhui Li, Chuanjie Wu, Pingping Wang, Ting Yang, Xunming Ji, Qingfeng Ma and Wenbo Zhao in European Stroke Journal
Footnotes
Acknowledgements
We thank all study participants and their relatives, and all the investigators of the TREND trial.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Beijing Natural Science Fund for Outstanding Young Scholars (JQ22020), the National Natural Science Fund of China (82422024), and Beijing Nova Program (Z201100006820143).
Ethical approval and informed consent
This study was approved by the ethics committees of Xuanwu Hospital, Capital Medical University, and all the participating centers. The study is a post hoc analysis of an Randomized Controlled Trial, and informed consent was obtained at the time the study was conducted.
Guarantor
Wenbo Zhao
Contributorship
Drs Wenbo Zhao, Xunming Ji, and Qingfeng Ma contributed to the conception, study design, and critical review of the manuscript. Drs Jing Wang, Yue Qiao contributed to data acquisition and analysis, drafted the manuscript and revision of the manuscript, and contributed equally to this work. Drs Jing Wang, Sijie Li, Chuanhui Li, Chuanjie Wu, Pingping Wang, and Ting Yang contributed to drafting a significant portion of the article or figures. Drs Wenbo Zhao and Qingfeng Ma contributed with critical comments during the article revision. All authors approved the submitted article.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
