Abstract
Introduction:
The efficacy and safety of statins for secondary prevention in patients who have experienced a cardioembolic stroke are not well-defined. However, previous observational data reported hyperlipidemia as a risk factor for both ischemic and bleeding complications in patients with AF and previous stroke. Based on these premises, we conducted a sub-analysis of the RAF and RAF-NOAC studies to evaluate the efficacy and safety of statins in secondary prevention in patients with acute ischemic stroke and AF.
Materials and methods:
We combined patient data from the RAF and RAF-NOAC studies, prospective observational studies conducted across Stroke Units in Europe, the United States, and Asia from January 2012 to June 2016. We included consecutive patients with AF who suffered an acute ischemic stroke with a follow-up of 90 days. Our outcomes were the combined endpoint, including stroke, transient ischemic attack, systemic embolism, symptomatic intracerebral hemorrhage, and major extracranial bleeding. Furthermore, both ischemic and hemorrhagic outcomes were evaluated separately.
Results:
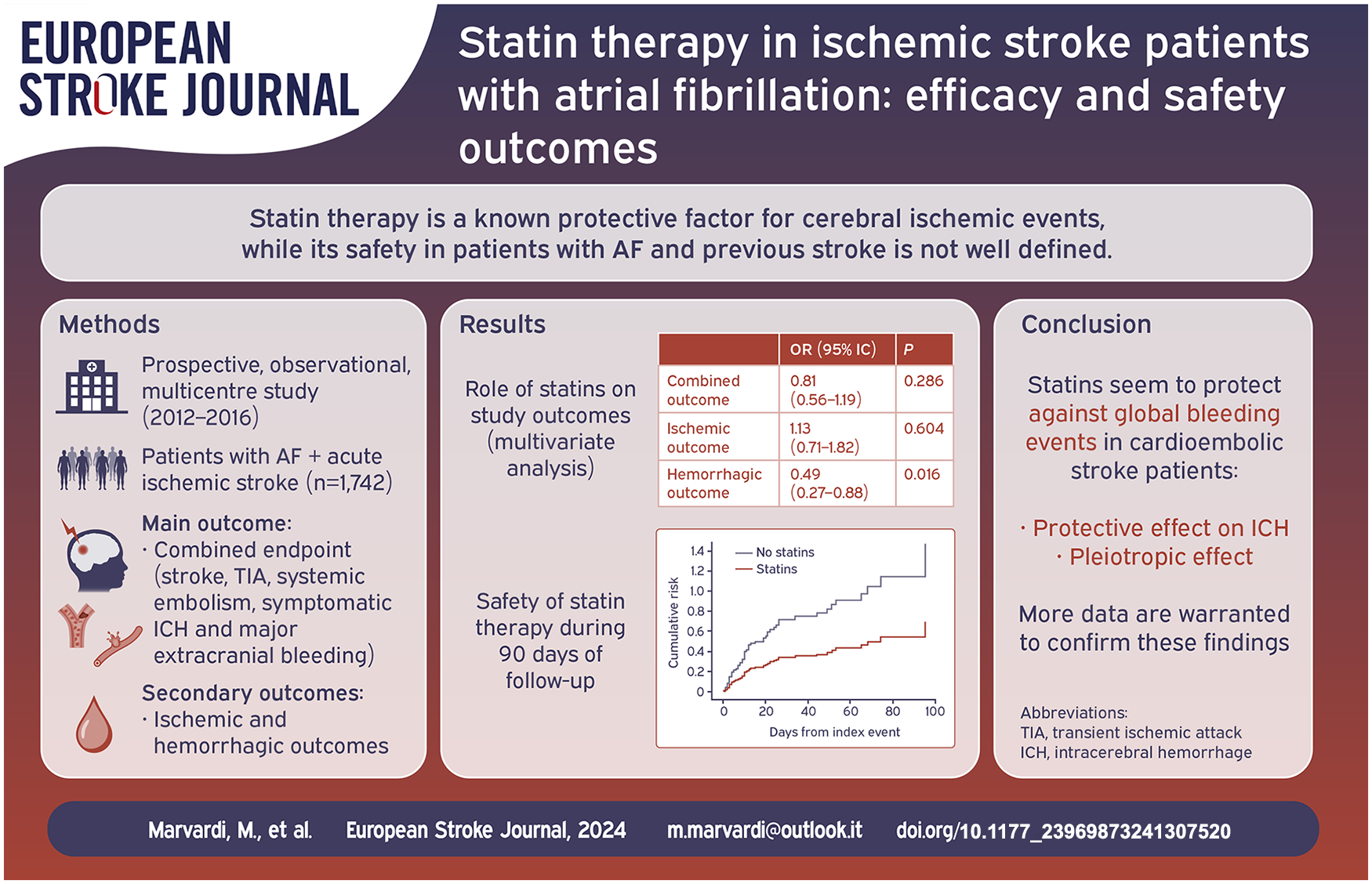
A total of 1742 patients were included (46% male), and 898 (52%) received statins after the index event, of whom 436 (48.6%) were already taking statins before the index event, 462 (51.4%) started treatment after. At multivariable analysis, statin use was statistically associated with age (OR 0.92, 95% CI 0.97–0.99, p = 0.001), male sex (OR 1.35, 95% CI 1.07–1.70, p = 0.013), anticoagulation (OR 2.53, 95% CI 1.90–3.36, p < 0.0001), hyperlipidemia (OR 5.52, 95% CI 4.28–7.12, p < 0.0001), paroxysmal AF (OR 1.40, 95% CI 1.12–1.75, p = 0.003), leukoaraiosis (OR 1.39, 95% CI 1.11–1.75, p = 0.004) and heart failure (OR 0.72, 95% CI 0.53–0.98, p = 0.034). Statin use was not associated with the combined outcome event (OR 0.84, 95% CI 0.58–1.23, p = 0.3) and ischemic outcome event (OR 1.17, 95% CI 0.73–1.88, p = 0.5) while was associated with a lower risk of hemorrhagic outcome event (OR 0.51, 95% CI 0.28–0.91, p = 0.02).
Discussion:
Statins protect cerebral arterial vessels (particularly small vessels) from subacute damage due to hypertension, diabetes, and other harmful agents (such as reactive oxygen species, proinflammatory cytokines, etc.) due to their systemic anti-inflammatory and endothelium-protective effects.
Conclusions:
Our data show that statins seem to protect against global bleeding events in cardioembolic stroke patients; this may be due to the pleiotropic effect of statins. More data are warranted to confirm these findings.
Background
While randomized controlled trials (RCTs) have evaluated statins for their potential in preventing recurrent acute ischemic stroke related to atherosclerosis, including large vessel occlusion (LVO) and small vessel disease (SVD), their effectiveness for ischemic stroke of cardioembolic origin remains unclear1–4 Although there is a well-documented link between high LDL cholesterol and increased risk of acute ischemic stroke due to LVO and SVD, the association with cardioembolic stroke is not yet clear.3–10 The RENo and RENo-ICH studies identified hyperlipidemia as a statistically significant risk factor for both ischemic and hemorrhagic recurrences in patients with cardioembolic stroke.11,12 Additionally, the RENo study revealed that approximately one-third of recurrent events were caused by LVO and SVD rather than cardioembolism. 11 Notably, statins have demonstrated efficacy in reducing the risk of such non-cardioembolic events.11,13,14
Beyond their role in lowering cholesterol, statins are known for their benefits in plaque stabilization, endothelial protection, and anti-inflammatory and anti-thrombotic effects – which may theoretically reduce the risk of acute ischemic stroke from any causes.15,16
Despite the benefits, there is a described risk of increased bleeding events, particularly intracerebral, reported mainly in secondary prophylaxis. Two large RCTs including patients with non-cardioembolic stroke, the Heart Protection Study (HPS) and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) Study, showed a reduction in the number of cerebral ischemic events in the group of statin-treated patients but a statistically significant increase of bleeding events was observed, also in secondary prophylaxis.8,9
Based on these premises, we conducted a sub-analysis of the RAF and RAF-NOAC studies to evaluate the efficacy and safety of statins in secondary prevention in patients with ischemic stroke and AF.
Methods
The data that supported the findings of this study are available from the corresponding author upon reasonable request.
We merged the datasets from the RAF and RAF-NOAC studies, both prospective observational studies.17,18 The RAF study was conducted from January 2012 to March 2014 in 29 Stroke Units, while the RAF-NOAC study spanned from April 2014 to June 2016 in 35 Stroke Units throughout Europe, the United States, and Asia.17,18 Both studies recruited consecutive patients who experienced an acute ischemic stroke and were diagnosed with AF, either prior to or following their stroke. Exclusion criteria of both RAF and RAF-NOACs studies were: (1) high risk of bleeding, defined as clinically significant liver disease (acute or chronic hepatitis, cirrhosis, or alanine aminotransferase level >3 times the upper limit of normal), creatinine clearance <30 ml/min (for apixaban, the threshold was 25 ml/min); (2) life expectancy of <3 to 6 months; (3) use of interacting medications; (4) uncontrolled hypertension.17,18
Ethical approval for both studies was obtained from local Institutional Review Boards as necessary.
The stroke severity was evaluated upon admission using the NIH Stroke Scale (NIHSS). All patients underwent either a non-contrast CT or an MR scan to rule out intracranial hemorrhage. Revascularization treatments, following the local standard protocols, were administered when indicated. Standard care in the Stroke Unit, along with monitoring and treatment, adhered to the international guidelines for acute ischemic stroke. Decisions regarding the selection and initiation timing of anticoagulants for secondary prevention were at the discretion of the attending physicians. The RAF study encompassed patients prescribed vitamin K antagonists or NOACs, whereas the RAF-NOAC study was exclusive to those treated with NOACs. Comprehensive medical history, clinical assessment, admission ECG, and 48-h ECG monitoring post-index stroke were conducted to identify non-valvular AF, which was then classified accordingly:
- Paroxysmal: associated with episodes terminating spontaneously within 7 days;
- Persistent: associated with episodes lasting more than 7 days or requiring pharmacological and/ or electrical cardioversion;
- Permanent: persisting for more than 1 year, either because cardioversion had failed or had not been pursued.
Risk factors
We collected data on established stroke risk factors, including age, gender, and medical histories of hypertension, diabetes mellitus, smoking, hyperlipidemia, ischemic heart disease, peripheral arterial disease, alcohol abuse, obesity, and prior stroke or transient ischemic attack (TIA). Hypertension was defined by a blood pressure reading of ⩾140/90 mmHg on at least two occasions prior to the stroke or current antihypertensive medication use. Diabetes mellitus was identified by fasting glucose levels ⩾ 126 mg/dl, postprandial levels ⩾ 200 mg/dl, HbA1c ⩾ 6.5%, or ongoing antidiabetic treatment. Hyperlipidemia at admission was defined by total cholesterol ⩾ 200 mg/dl, triglycerides ⩾ 140 mg/dl, or existing lipid-lowering therapy. Symptomatic ischemic heart and peripheral arterial diseases were confirmed through clinical history and diagnostic procedures.
Additionally, we evaluated white matter changes, categorizing leukoaraiosis (defined on the first computed tomography examination as ill-defined and moderately hypodense areas of ⩾5 mm) on initial CT or MR imaging as absent or present, regardless of severity. 19
Baseline measurements at admission for all patients included fasting serum glucose, cholesterol profiles (total, HDL, and LDL), platelet count, coagulation times, and blood pressure.
Physicians prescribed statins based on clinical judgment from among the seven commercially available in Europe, the United States, and Asia. We also documented the use of antiplatelet, anticoagulant, and thrombolytic agents before and during the study.
Outcomes
Primary study outcome at 90 days was defined as the composite of, ischemic stroke, transient ischemic attack, symptomatic systemic embolism, symptomatic intracerebral hemorrhage, and major extracranial hemorrhage. Additionally, ischemic and hemorrhagic events were analyzed separately.
Stroke was defined as the sudden onset of a new focal neurological deficit of a vascular origin in a site consistent with the territory of a major cerebral artery, and categorized as either ischemic or hemorrhagic. Hemorrhagic transformations revealed on neuroimaging 24–72 h after onset were not considered outcome events, unless they were classified as being symptomatic. TIA was defined as a transient episode of neurological dysfunction caused by focal brain ischemia without acute infarction. Systemic embolism was defined as an acute vascular occlusion of an extremity or organ confirmed at imaging, surgery, or autopsy. Major extracerebral bleeding was defined as a reduction in the hemoglobin level of at least 2 g per deciliter, requiring a blood transfusion of at least 2U, or symptomatic bleeding in a critical area or organ. 20
Statistical analyses
The analysis was performed considering statin therapy after the index event: for patients with or without statins after the index event and with or without primary outcome event at 90 days, differences in clinical characteristics and risk factors were evaluated using the χ2 test of proportions (with a two-sided α level of 5%), by calculating odds ratio (OR) and 95% confidence intervals (CIs). Multivariable logistic regression analysis was performed to identify independent associations with statin use and outcome events. The variables included a priori in this analysis were the following: age, sex, oral anticoagulant therapy, diabetes, hypertension, hyperlipidemia, paroxysmal atrial fibrillation, history of stroke/TIA, alcoholism, heart failure, history of myocardial infarction, peripheral arterial disease, leukoaraiosis, and smoking (CHA2DS2-VASc and total cholesterol were excluded due to the concurrent presence within the multivariate analysis of the individual factors already included in the CHA2DS2-VASc score and hyperlipidemia). Subsequent analyses were conducted to identify independent predictive factors for both the Ischemic Outcome and the Hemorrhagic Outcome events, including the same predictive factors used previously. We conducted a sensitivity analysis that included the participating center as a random effect in the model.
Finally, we carried out Cox Regression analyses (Cox proportional hazard model) to study in detail the effect of statins over the 90 days of follow-up on the Hemorrhagic Outcome and symptomatic intracranial hemorrhage; the variables included in these two analyses were the same: statin therapy, hyperlipidemia, paroxysmal atrial fibrillation, and CHA2DS2-VASc ⩾ 4. The results of the Cox Regression are reported in Hazard Ratios (HR).
Results
In total, the study enrolled 1742 patients. After excluding 34 due to incomplete follow-up data, the analysis included 1708 participants. Among them, 811 (46.6%) were male and with a mean age of 76 ± 9.86.
In this cohort of 1708 patients, 898 (52%) were prescribed statins after the index event, of whom 436 (48.6%) were already taking statins before the index event, while 462 (51.4%) started the treatment after. Atorvastatin was the most common, used by 28.5% of patients, followed by Simvastatin at 12.8% and Rosuvastatin at 5.6%. A smaller number (0.6%) were on Pravastatin. Fluvastatin and Pitavastatin were the least used, with only one patient on each. Data on statin usage was missing for 69 (4%). Only 13 (0.7%) of patients received Ezetimibe, excluded in the statistical analyses.
Regarding anticoagulant therapy, 1380 (79.2%) patients received treatment, 841 (48.3%) on NOACs, and 539 (30.9%) on Warfarin. The remaining 361 (20.7%) were not on oral anticoagulants, most commonly used subcutaneous low molecular weight heparin or, less frequently, antiplatelet drugs alone. In Table S1 are listed the main risk factors.
At 90 days, 120 deaths were reported (7%), while 548 patients (32,1%) reported a disability (mRS scale 3–5).
Predictors for statin therapy
The clinical characteristics of the patients treated and not treated with statins are listed in Table 1.
Univariate analysis: predictors for statin therapy.
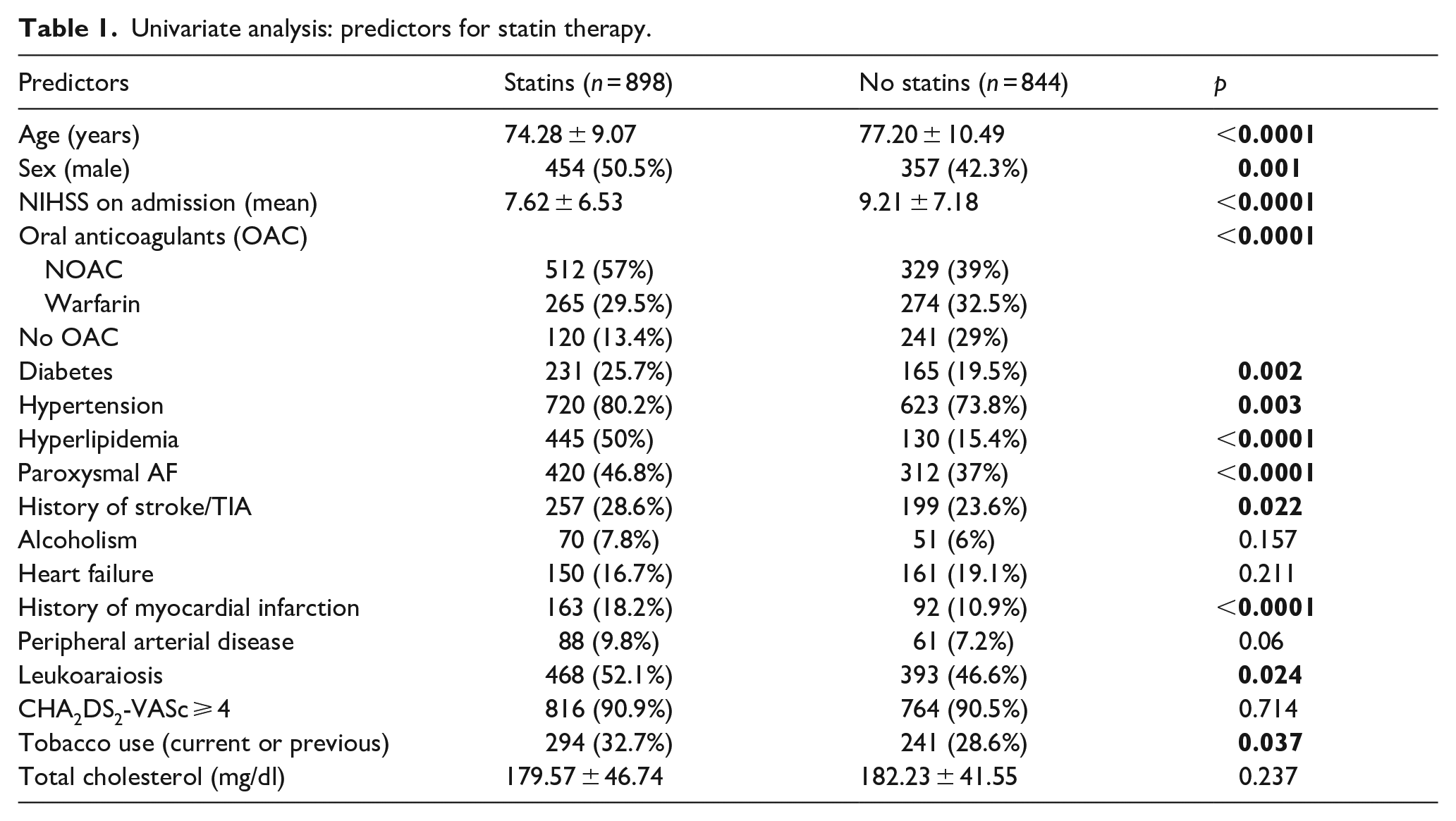
Statin therapy was more frequent in younger patients (74.28 ± 9.07 vs 77.20 ± 10.49, p < 0.0001), those taking oral anticoagulants (57% vs 39%, p < 0.0001), in patients with hyperlipidemia (50.0% vs 15.4%, p < 0.0001), in those with paroxysmal AF (46.8% vs 37.0%, p < 0.0001), and in those with a history of myocardial infarction (18.2% vs 10.9%, p < 0.0001). Statistical significance was also shown in diabetic patients (25.7% vs 19.5%, p = 0.002), hypertensive patients (80.2% vs 72.8%, p = 0.003), in those with a history of cerebral ischemic events (acute ischemic stroke/TIA, 28.6% vs 23.6%, p = 0.022), in those with leukoaraiosis on imaging studies (52.1% vs 46.6%, p = 0.024), and in smokers, current or past (32.7% vs 28.6%, p = 0.037).
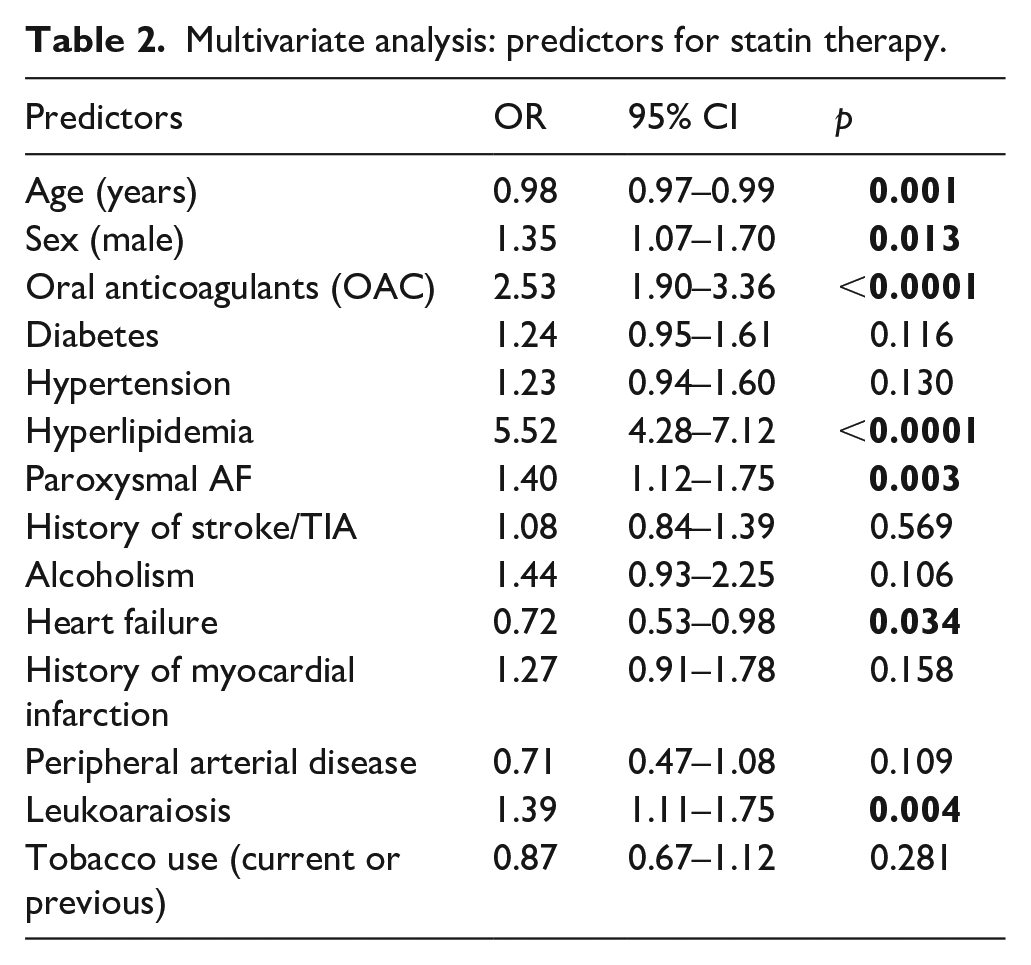
The multivariable analysis (Table 2) identified oral anticoagulation (OR 2.53, 95% CI 1.90–3.36, p < 0.0001) and hyperlipidemia (OR 5.52, 95% CI 4.28–7.12, p < 0.0001) as predictors of statin therapy. Conversely, age had an inverse association with statin use (OR 0.98, 95% CI 0.97–0.99, p = 0.001), as did heart failure (OR 0.72, 95% CI 0.53–0.98, p = 0.034). In male patients, there was an increase in the likelihood of statin therapy (OR 1.35, 95% CI 1.07–1.70, p = 0.013). Statins were also more commonly prescribed among patients with paroxysmal atrial fibrillation (OR 1.40, 95% CI 1.12–1.75, p = 0.003) and leukoaraiosis (OR 1.39, 95% CI 1.11–1.75, p = 0.004).
Multivariate analysis: predictors for statin therapy.
Predictors for primary and secondary outcomes
A total of 134 patients (7.8%) experienced a primary outcome, whereas ischemic outcome was observed in 99 individuals (5.8%) and hemorrhagic outcomes in 70 individuals (4.1%). Symptomatic intracerebral hemorrhage alone was reported in 49 patients (2.9%).
In univariate analysis, statin therapy suggested a protective trend (44.5% vs 51.9%), but this did not reach statistical significance (p = 0.084) (Table S2).
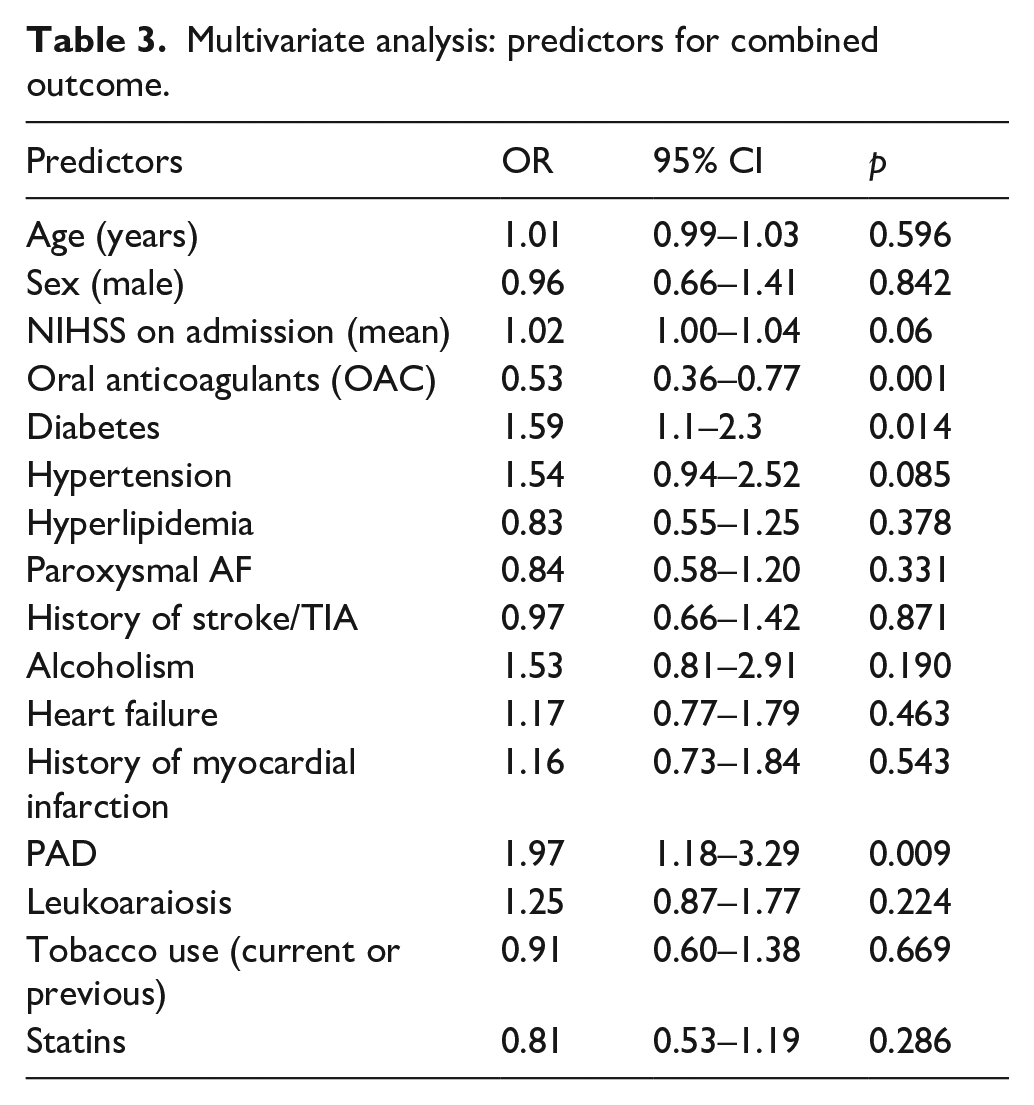
On multivariable analysis, statin use was associated with a reduced risk of primary outcome, but this association did not reach statistical significance (OR 0.81, 95% CI 0.53–1.19, p = 0.286) (Table 3).
Multivariate analysis: predictors for combined outcome.
Regarding secondary outcomes, the hemorrhagic event rate was lower in the statin group (2.9% compared to 5.4%, p = 0.010) (Table S3), as was the rate of symptomatic intracerebral hemorrhage (2.1% vs 3.7%, p = 0.043).
In Table 4 are reported the results of the multivariate analysis on the role of statins on the combined outcome: statin use was associated with a 49% reduction of total bleeding events, within 90 days (OR 0.51, 95% CI 0.28–0.91, p = 0.02) and this effect remains stable over the 90 days as shown in the Cox survival curve analysis (Figure 1). Regarding the symptomatic intracerebral hemorrhage outcome, statin therapy showed an early protective effect with a stable trend over the 90 days of follow-up, albeit in the absence of adequate statistical significance (p = 0.066) (Figure 2).
Multivariate analysis on the role of statins on the combined, ischemic and hemorrhagic outcome.
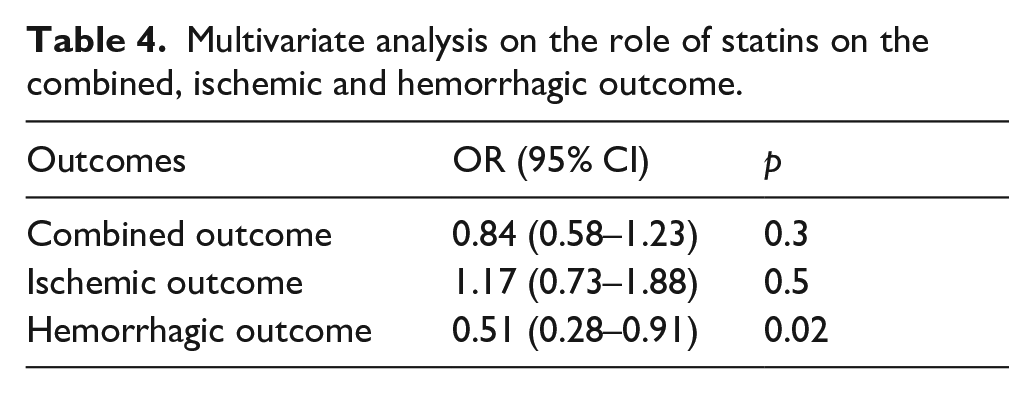
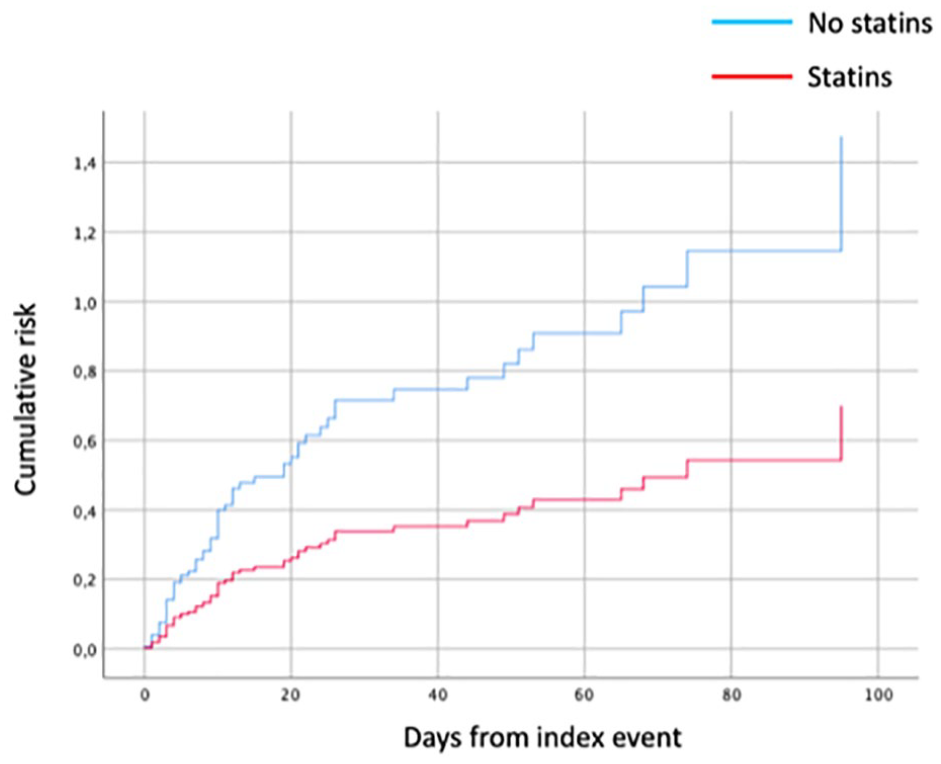
Cox regression: statins as an independent predictive factor for hemorrhagic outcome.
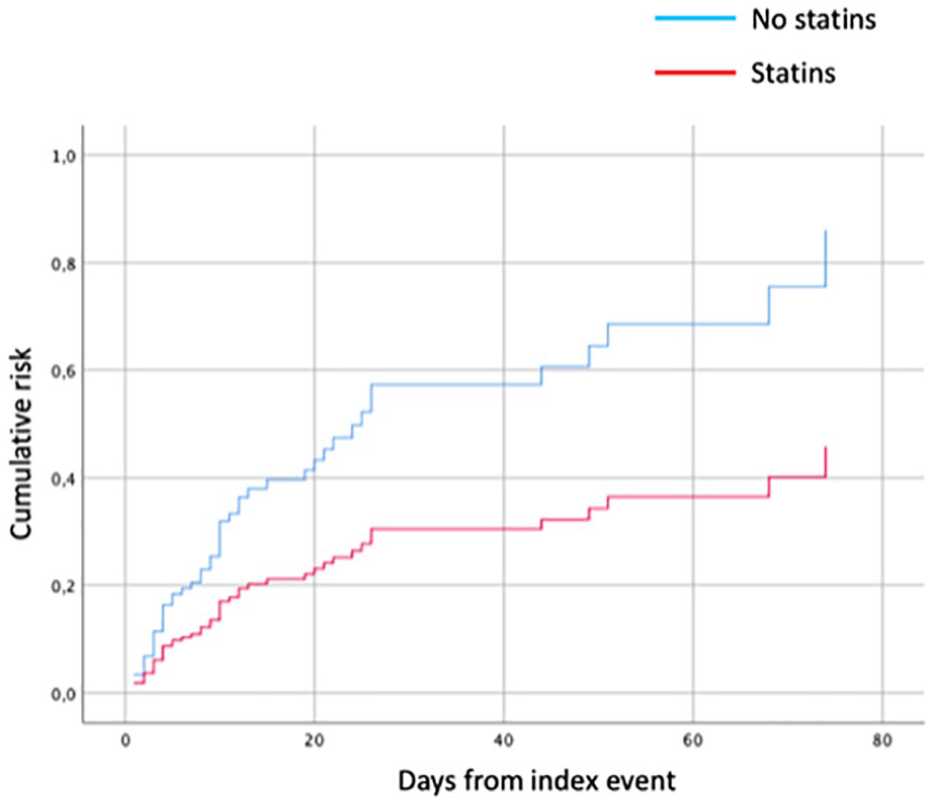
Cox regression: statins as an independent predictor for symptomatic intracranial hemorrhage.
We performed a sensitivity analysis to evaluate the association between statin use and outcome events including in the model the participating centers as a random effect. The analysis confirmed the main results regarding the association between statins and outcomes (Table S4).
Furthermore, in patients receiving oral anticoagulants, an additional multivariable model was performed to evaluate the association between the type of anticoagulant (NOACs vs vitamin K antagonists) and clinical outcomes. The results are listed in Table S5.
Discussion
In this observational study, which included a cohort of patients with AF in secondary prevention for ischemic stroke, statin therapy showed a statistically significant protective effect against global bleeding events during the 90 days following the index cerebral ischemic event. This correlation is probably due to a protective effect of statins against intracerebral hemorrhage. Whereas, for the combined outcome, statin therapy showed a protective trend, albeit without adequate statistical significance.
Our findings diverge from those of the Heart Protection Study and the SPARCL Trial, which both observed a statistically significant increase in bleeding events, particularly intracerebral hemorrhage.8,9 A subsequent meta-analysis which included data from 24 RCTs, including HPS and the SPARCL Trial, supported a trend toward increased bleeding events in patients undergoing secondary prophylaxis, with RR of 1.73 (95% CI 1.19–2.50). 6 This analysis suggested that reduced LDL cholesterol could contribute to the increased risk of bleeding. However, it is important to note that data from the HPS and SPARCL trials predominantly influenced this result.8,9 Specifically, in the HPS, the incidence of ICH was 32 cases (21 in the Simvastatin group vs 11 in the placebo group). Given the small sample size, this did not confer statistical significance (RR 1.91, 95% CI 0.92–3.95), suggesting that the association may be less robust than initially perceived. 8 In a sub-analysis of the SPARCL trial, which pooled patients who had an ICH, it was observed that ICH risk was higher in those having a hemorrhagic stroke as the entry event (HR 5.65, 95% CI 2.82–11.30, p < 0.001) without statistical interactions between statin therapy. 21 Consequently, the patients identified as having the highest risk for developing intracranial bleeding were those with a history of a previous bleeding event.
Data from comprehensive national registries in Denmark,22–24 South Korea, 25 and China, 26 encompassing nearly 1.5 million patients, consistently reported the safety of statins in preventing cerebral hemorrhagic events, both as primary and secondary prophylaxis.22–24,26 Furthermore, the findings suggest that both the duration and the dosage of statin therapy contribute to a protective effect against intracerebral hemorrhage. Patients with prolonged statin use and those on higher dosages had a lower risk of ICH, challenging the previously hypothesized biological mechanism linking LDL reduction to an increased bleeding risk. 27 These results are in line with those of an extensive meta-analysis published in 2012 for a total of 31 RCTs with 182,803 patients. 28 A similar meta-analysis by Hackman et al., which included published and unpublished data from 23 randomized trials and 19 observational studies, reported that statins were not associated with an increased risk of ICH neither in randomized trials (RR 1.10, 95% CI 0.86–1.41) or cohort studies (RR 0.94, 95% CI 0.81–1.10), both in primary (48% of patients included in RCTs) and in secondary (52% of patients included in RCTs) stroke prevention.29,30
A recent Japanese study examining a patient cohort similar to ours investigated the impact of statins on individuals with AF who were also on oral anticoagulants. The Authors observed that the group treated with statins, compared to those not on statins, had a higher presence of paroxysmal AF, hypertension, diabetes, and dyslipidemia. 31 Notably, the statin group demonstrated a reduced risk of major bleeding. Furthermore, this group also showed decreased risks in all-cause mortality, ischemic events, and various types of strokes. 31
It is plausible that statins protect cerebral arterial vessels (particularly small vessels) from subacute damage due to hypertension, diabetes, and other harmful agents (such as reactive oxygen species, proinflammatory cytokines, etc.) due to their systemic anti-inflammatory and endothelium-protective effects. 15
The reduction of cellular infiltration and metalloproteinase synthesis and the increase in collagen synthesis guarantee greater stability of the atheromatous plaque preventing plaque ulceration or, if it were already present, prevents its rupture with consequent acute thrombosis and vessel occlusion.
Anti-inflammatory effects are mainly due to the reduction of the synthesis of pro-inflammatory cytokines, in particular the C-Reactive Protein, and the NF-kB complex. The systemic anti-inflammatory effect consequently reduces the oxidation of LDL, which underlies atherogenesis.
Statins also have an important protective effect on the vascular endothelium: they stimulate the production and release of nitric oxide by endothelial cells, which stimulates vasodilation and protects against endothelial damage thanks to its antioxidant properties.
Finally, statins enhance fibrinolysis by stimulating the production and release of t-PA, inhibit platelet aggregation by blocking the release of thromboxane in the blood stream and inhibit the extrinsic pathway of coagulation by reducing the expression of the tissue factor; this effect is added to that of any other antithrombotic ongoing therapies, and prevents thrombosis on plaque with occlusion of the vessel lumen which is the basis of major cardiovascular events.
The cumulative effects of statins may serve as a preventive measure against the onset or progression of cerebral small vessel disease. By safeguarding the integrity of small cerebral vessels, statins potentially mitigate the risk of ischemic events caused by thrombotic occlusions and hemorrhagic events due to the rupture of compromised vessels. Building on the same biological hypothesis, it is plausible that administering statins during the acute phase of an ischemic stroke could lower the likelihood of hemorrhagic transformation.
We did not perform logistic regression for the symptomatic intracranial hemorrhage outcome, due to the small sample size.
This study has several limitations:
- it is an observational study, based on data collected from databases of previous studies, in which neither the use nor the dosage of statins were randomized;
- a possible presence of a possible indication bias, where clinicians may have preferentially prescribed statins to patients perceived to be at lower risk for bleeding. This bias is partially evident in the observed association between OAC use and statin therapy, suggesting that an assessment of bleeding risk may have influenced the choice of treatment. This indication bias could impact our findings by potentially underestimating the bleeding risk in the statin-treated group compared to those who did not receive statins.
- the duration and intensity of the therapy, the type of statin and the LDL cholesterol levels at entry (index cerebral ischemic event) and at the time of the study outcome events were not considered;
- except for anticoagulants, additional medications that patients were taking besides statins were not included. As a result, we have no information on drug interactions that can potentially enhance or decrease the effects of statins;
- the 90-day follow-up is probably too limited to make conclusions, further studies are needed, both to confirm the safety of statin therapy in the acute and subacute phase, and subsequently to verify whether safety is maintained over a medium-long period of time;
- MACE (major adverse cardiovascular events) are not included in the primary outcome, as secondary. This is in line with other RCTs where the risk-benefit ratio for early versus late introduction of anticoagulation in cardioembolic stroke was studied.32,33
- finally, there are no complete data on the cholesterol levels and subtypes, limiting our study population’s lipid profile.
However, this study has a large sample size which gives significant statistical value to the results of the analyses. It also has a prospective design and reflects the real-life experiences of many centers located on different continents (Europe, North America, and Asia).
Conclusions
In this observational study, including patients with AF in secondary prevention for ischemic stroke, statin therapy showed a statistically significant protective effect against global bleeding events (intracranial and extracranial) during the 90 days following the index cerebral ischemic event. This correlation is mainly due to the protective effect of statins on an intracranial hemorrhage.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241307520 – Supplemental material for Statin therapy in ischemic stroke patients with atrial fibrillation: Efficacy and safety outcomes
Supplemental material, sj-docx-1-eso-10.1177_23969873241307520 for Statin therapy in ischemic stroke patients with atrial fibrillation: Efficacy and safety outcomes by Michele Marvardi, Maurizio Paciaroni and Valeria Caso in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241307520 – Supplemental material for Statin therapy in ischemic stroke patients with atrial fibrillation: Efficacy and safety outcomes
Supplemental material, sj-docx-2-eso-10.1177_23969873241307520 for Statin therapy in ischemic stroke patients with atrial fibrillation: Efficacy and safety outcomes by Michele Marvardi, Maurizio Paciaroni and Valeria Caso in European Stroke Journal
Footnotes
Acknowledgements
The Authors thank the RAF and RAF NOAC consortia for their continuous support. The Authors thank ARS UMBRIA for the unrestricted support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Paciaroni received honoraria as a member of the speaker bureau of Sanofi-Aventis, Bristol Meyer Squibb, Daiiki Sankyo and Pfizer. C. Becattini received honoraria as a member of the speaker bureau of Bristol Meyer Squibb and Bayer. G. Agnelli received honoraria as a member of the speaker bureau of Boehringer Ingelheim and Bayer. P. Michel received Research Grant by Swiss National Science Foundation and Swiss Heart Foundation; he received speaker fees by Bayer, Boehringer Ingelheim, Covidien, St. Jude Medical; he received honoraria as advisory relationship by Pierre-Fabre, Bayer, Bristol Meyer Squibb, Amgen, and Boehringer Ingelheim. J. Putaala received honoraria for lectures related to atrial fibrillation and anticoagulants for Orion Pharma, Bristol Meyer Squibb, Pfizer, Bayer, and Boehringer Ingelheim. T. Tatlisumak received honoraria as consultant or advisory relationship by Lundbeck and Boehringer Ingelheim. G. Tsivgoulis has received funding for travel or speaker’s honoraria from Bayer, Pfizer, and Boehringer Ingelheim. He has served on scientific advisory boards for Bayer, Boehringer Ingelheim, and Daiichi Sankyo. D. Toni has received personal fees from Abbott, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Medtronic, and Pfizer. V. Caso received honoraria as a member of the speaker bureau of Boehringer Ingelheim, Bayer, and Daiichi Sankyo (all fees were paid to Associazione Ricerca Stroke, Umbria). She received honoraria as consultant or advisory board member of Boehringer Ingelheim, Bayer, Daiichi Sankyo, and Pfizer. G. Ntaios reports speaker fees/advisory board/ research support from Bayer, Pfizer, Boehringer Ingelheim and Elpen. All fees are paid to his institution. S. Sacco has received personal fees as speaker or advisor from Abbott, Allergan, Astra Zeneca, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Teva and research grants from Allergan, Novartis, and Uriach. J. Dawson as received honoraria as speaker from Medtronic, Daicchi Sankyo, BMS, Pfizer. Advisory board Astra Zeneca. L. Pantoni recieved consultation fees from Amicus and PIAM. M. Del Sette has received honoraria for speaking from Bayer and Boehringer Ingelheim. M. Zedde received speaking and consulting fees from Daiichi Sankyo, Amicus Therapeutics, Sanofi Genzyme, Abbott, and Takeda. M. Cappellari has received consulting fees from Boehringer Ingelheim, Pfizer – BMS, and Daiichi Sankyo. Y. Flomin has received personal fees from Boehringer Ingelheim, Bayer and Takeda, grants, personal fees and nonfinancial support from Pfizer, personal fees and nonfinancial support from Sanofi Genzyme. R. Ornello has received nonfinancial support from Novartis, Allergan, and Teva. S. Giannopoulos has received funding for travel from Bayer and speaker’s honoraria from Pfizer. The other authors report no conflicts.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Each participating institution obtained ethical approval from their local IRBs before collecting data for the stroke data-base This ensured that all data collected initially met the ethical guidelines and standards required for research involving human subjects. While the initial data collection had comprehensive ethical approval, some specific subanalyses conducted as part of this study required separate ethical submissions. This additional step was necessary due to the nature of the subanalyses, which might have involved new research questions or methods that were not covered under the initial ethical approval.
Informed consent
All subjects provided informed consent for data collection in the respective stroke registry when present. Some centers collected the data only to participate in the RAF studies, so dedicated informed consent was needed.
Guarantor
Prof. Valeria Caso, Senior Author S. Maria della Misericordia, Perugia.
Contributorship
Michele Marvardi wrote the manuscript, Valeria Caso and Maurizio Paciaroni revised it, the remainent authors collected the data and revised the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
