Abstract
Introduction:
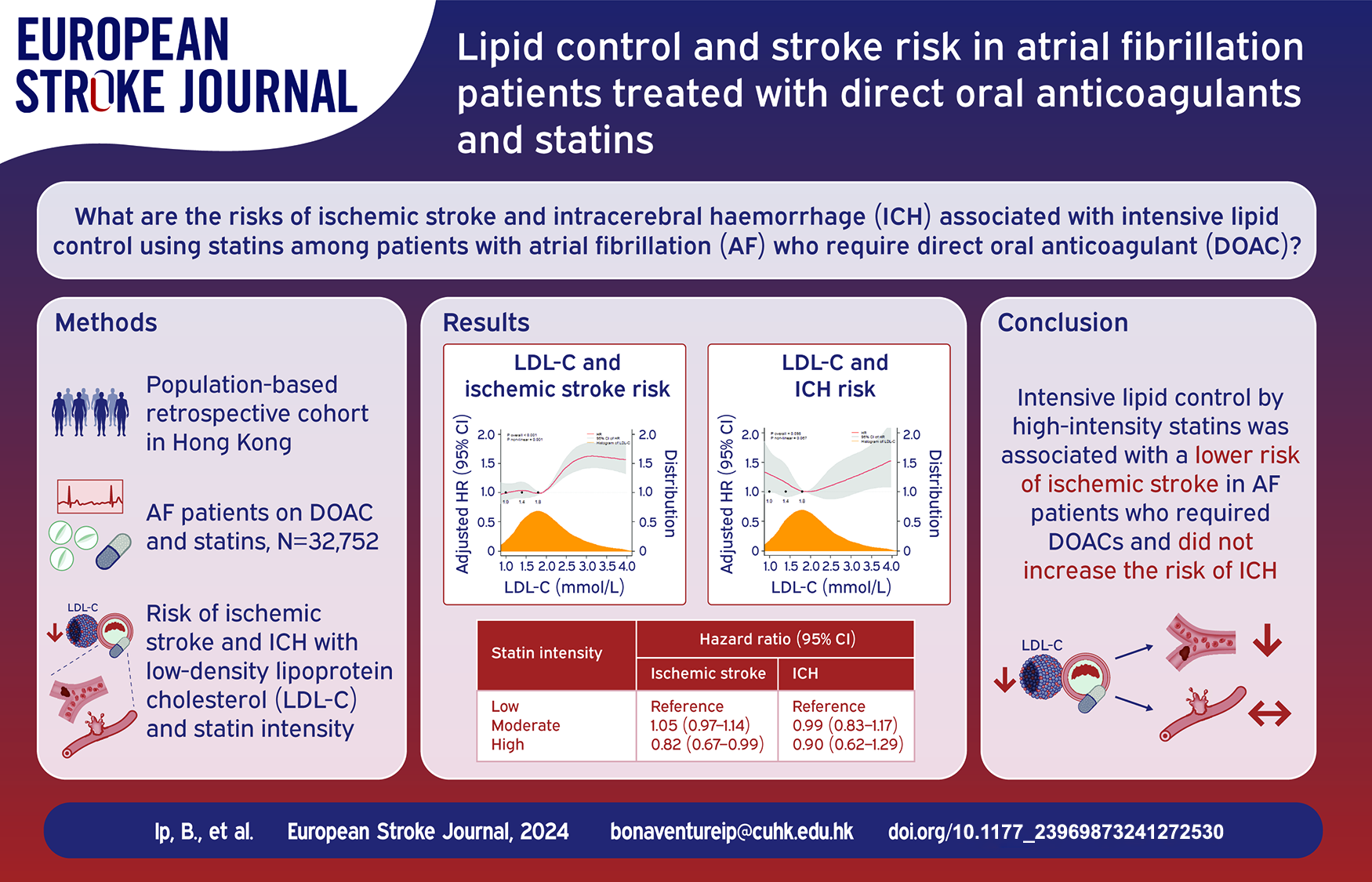
The risk of ischemic stroke and intracerebral hemorrhage (ICH) with intensive lipid control by statins among patients with atrial fibrillation (AF) who require direct oral anticoagulants (DOAC) is unclear. We aimed to determine the risks of ischemic stroke and ICH in AF patients treated with DOAC and statins.
Patients and methods:
In a population-based retrospective cohort study, we identified AF patients concurrently on DOAC and statins from 2015 to 2021 in Hong Kong. Primary outcome was ischemic stroke. Secondary outcomes were ICH and death. We correlated study outcomes with low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) as time-varying, continuous variables with restricted cubic spline. In secondary analyses, the risks of study outcomes with statin intensity (low, moderate, high) were determined by multivariable time-dependent marginal structural Cox models.
Results:
We identified 32,752 AF patients co-prescribed with DOAC and statins. Lower LDL-C (
Discussion and conclusion:
Intensive lipid control by high-intensity statins was associated with a lower risk of ischemic stroke in AF patients who required DOACs and did not appear to increase the risk of ICH.
Introduction
Intensive lowering of low-density lipoprotein cholesterol (LDL-C) with statins effectively reduces atherothrombotic cardiovascular events.1,2 However, the efficacy and safety of intensive lipid control in patients with atrial fibrillation (AF) remains uncertain.3,4 Although integrated cardiovascular optimization in addition to anticoagulation in AF patients lowered composite cardiovascular outcomes,5–7 the net benefit of lipid control specifically for ischemic stroke prevention among AF patients was not well established. The association between lipid control and ischemic stroke risk among AF patients had been inconsistent according to current evidence.4,7–9
On the other hand, excessive lipid lowering or pleiotropic antithrombotic property of statins might increase the risk of intracerebral hemorrhage (ICH), especially in patients with a history of ischemic stroke and anticoagulant users.10,11 Multiple studies suggested statins may increase the risk of ICH among anticoagulation recipients.3,12 As AF patients who require direct oral anticoagulant (DOAC) frequently have concurrent cardiovascular risks that require aggressive lipid control, 13 it is imperative to elucidate the effects of intensive lipid lowering on ischemic and haemorrhagic stroke in AF patients taking DOAC. We aimed to determine the risks of ischemic stroke and ICH associated with the degree of lipid control and statin intensity among AF patients treated with DOACs.
Methods
Study design and data source
In this population-based retrospective cohort study, we identified all DOAC recipients receiving care by public hospitals or outpatient clinics under the Hong Kong Hospital Authority from January 1st 2015 to December 31st 2021. We retrieved data from the territory-wide Clinical Data Analysis and Reporting System (CDARS) of the Hospital Authority, which is the sole public healthcare provider in Hong Kong responsible for almost 90% of in-patient services of the 7-million population. 14 The coding accuracy of CDARS for the diagnoses of ischemic stroke, ICH and AF had been validated in a previous population-based study, with positive predictive values of 90%, 95%, and 95% respectively. 15 The local institutional review board approved the study (Joint CUHK-NTEC CREC Ref No. 2021.349) and waived the need for written informed consent given the de-identified data. We followed the STROBE reporting guideline.
Data collection
We identified AF patients on apixaban, dabigatran, edoxaban or rivaroxaban, with co-prescription of statins for more than 6 months. Patients with mitral stenosis, left ventricular thrombus, valvular replacement, atrial septal defect, myeloproliferative disorders, active malignancies were excluded via
We collected the baseline demographics, blood pressure, laboratory results, and prescriptions of antiplatelet agents, statins, antihypertensive drugs, cytochrome P450 or P-glycoprotein (CYP/P-gp) modulators, and non-steroidal anti-inflammatory drugs (NSAID) (Table 1). The 10-year risk of atherosclerotic cardiovascular disease (ASCVD), CHADS2-Vasc and HAS-BLED scores at the commencement of DOAC-statin co-prescription were calculated.13,16
Baseline characteristics of study population.
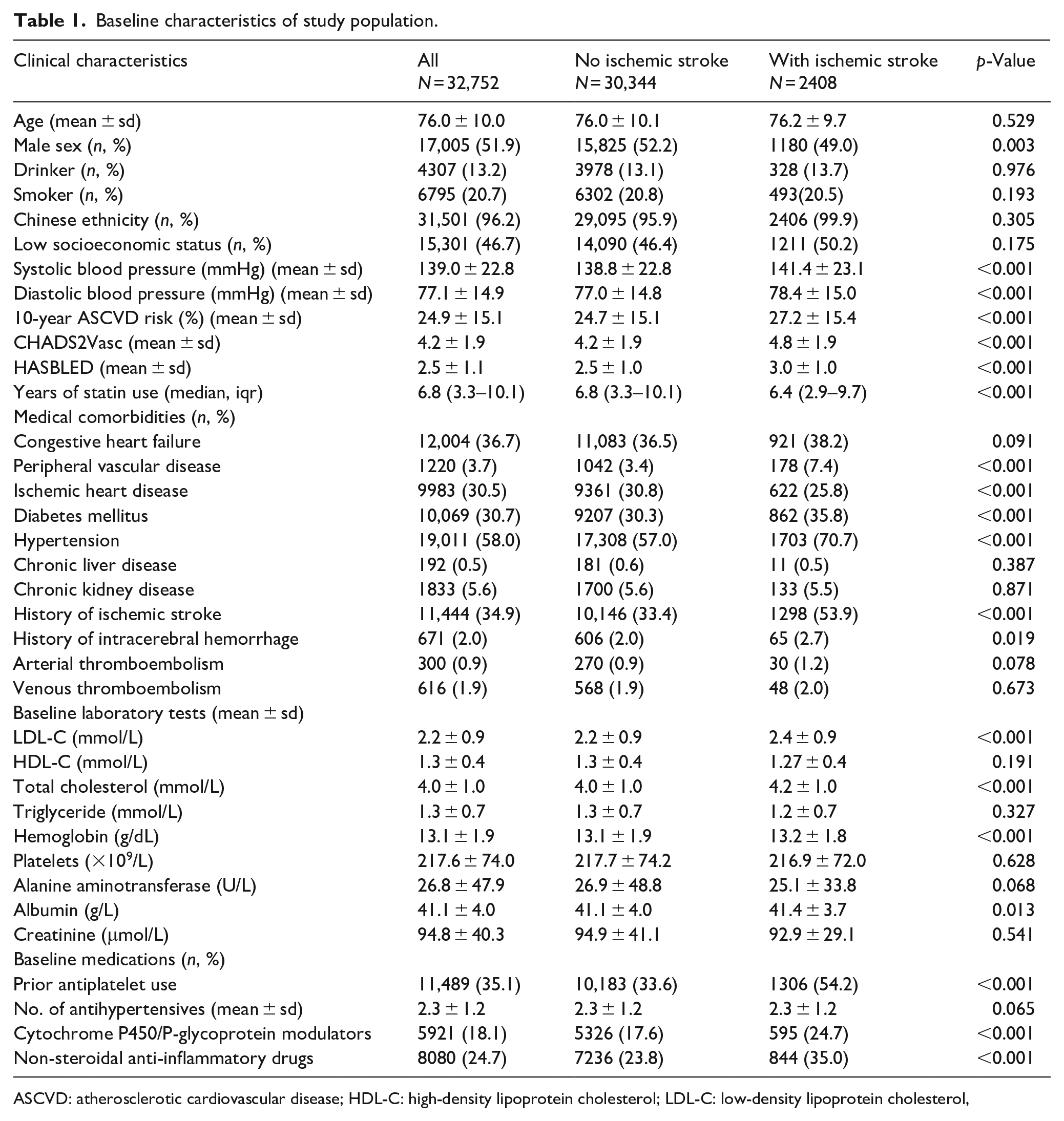
ASCVD: atherosclerotic cardiovascular disease; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol,
Lipid profile and statin intensity
LDL-C was calculated by the Friedewald 17 equation, except in individuals with triglyceride >4.5 mmol/L [400 mg/dL], in which a direct LDL-C level was measured. We only included lipid profiles measured in out-patient clinics to avoid reduced cholesterol levels due to acute phase responses. 18 Changes of lipid levels over time were subjected to a time-dependent model (see Statistical Analyses). We categorized statin intensity into low-, moderate-, and high-intensity regimens (Supplemental Methods). 19
Outcomes
Primary outcome was the first acute ischemic stroke during DOAC-statin treatment. Secondary outcomes were ICH and all-cause mortality. Subdural hemorrhage, subarachnoid hemorrhage, or intracranial hemorrhage secondary to traumatic brain injury, vascular anomalies, or brain tumors were excluded.
Follow-up period
The follow-up period was determined as the interval between the date of first DOAC/statin co-prescription and the date of the outcome events, if any. In patients without outcome events, the follow-up period was defined as the time interval between the date of first DOAC/statin co-prescription and the earliest date among study end; death; or discontinuation of DOAC or statin.
Statistical analyses
We performed data analysis by R software version 4.2.2. Continuous variables were expressed in mean ± standard deviation or median (interquartile range [IQR]) as appropriate, while categorical variables were presented as number (percentage). We used Chi-square test or Fisher’s exact test to compare categorical parameters and Student
In the primary analysis, we used restricted cubic spline (RCS) with 5 knot to account for the continuous and time-dependent properties of LDL-C and HDL-C levels, as well as their potential non-linear relationships with study outcomes.
21
A sensitivity analysis with a marginal structural cause-specific hazard model was used to determine the association between study outcomes and an LDL-C level of <1.8 mmol/L [70 mg/dL], a common target for primary and secondary cardiovascular protection3,22; and a high-density lipoprotein cholesterol level (HDL-C) of >1.0 mmol/L [40 mg/dL]. Cause-specific hazard model is an extension of Cox proportional hazard model to handle competing risks of the study outcomes. For all-cause mortality, marginal structural Cox model was used. Marginal structural model was aimed to estimate the effect of a time-dependent exposure on a clinical outcome using observational data in the presence of other time-dependent covariates that may simultaneously affect the exposure and clinical outcome.
23
Details on the estimation of marginal structural model were included in the Supplemental Methods. The marginal structural cause-specific hazard model was estimated using a time-dependent inverse probability of treatment and censoring-weighted Cox model.
24
Deaths due to non-ischemic-stroke-related and non-ICH-related causes were considered as competing events for the development of ischemic stroke and ICH, respectively. Secondary analyses were conducted as follows: (1) We evaluated the association between study outcomes and statin intensity in a time-dependent manner similar to the primary analysis. (2) We compared the difference in ischemic stroke below versus above an LDL-C cutoff at 1.8 mmol/L (70 mg/dL) in a time-dependent manner, stratified by the most likely causative ischemic stroke mechanisms.
25
(3) We performed subgroup analyses in patients with a history of ischemic stroke. All analyses were adjusted for age, sex, drinking and smoking status, liver and renal function, history of hypertension, diabetes mellitus, congestive heart failure, ischemic heart disease, arterial or venous thromboembolism, intracerebral hemorrhage, concurrent antiplatelet, antihypertensives, CYP/P-gp modulators and NSAIDs. All statistical tests were two-sided. Statistical significance was taken as
Results
We identified 32,752 AF patients who received concurrent DOAC and statin therapy from January 1st 2015 to December 31st 2021 (Chart 1), among which 11,444 patients had a history of ischemic stroke and 671 patients had a history of ICH prior to DOAC-statin co-prescription. Median duration of statin use was 6.8 years. At study baseline, 13,902 (42.4%) were on low-intensity statin; 16,720 (51.1%) on moderate-intensity statin; and 2130 (6.5%) on high-intensity statin. The median (IQR) follow-up duration was 1.5 (0.5–3.3) years. During the 6-year study period, the mean interval of lipid profile test per patient was 6 ± 1.7 months. 2408 (7.4%) developed ischemic stroke, 625 (1.9%) developed ICH, and 6168 (18.8%) died. Univariate testing suggested that patients who developed ischemic stroke while on DOAC had higher prevalence of hypertension, diabetes mellitus, peripheral vascular disease, and prior ischemic stroke and ICH (Table 1).
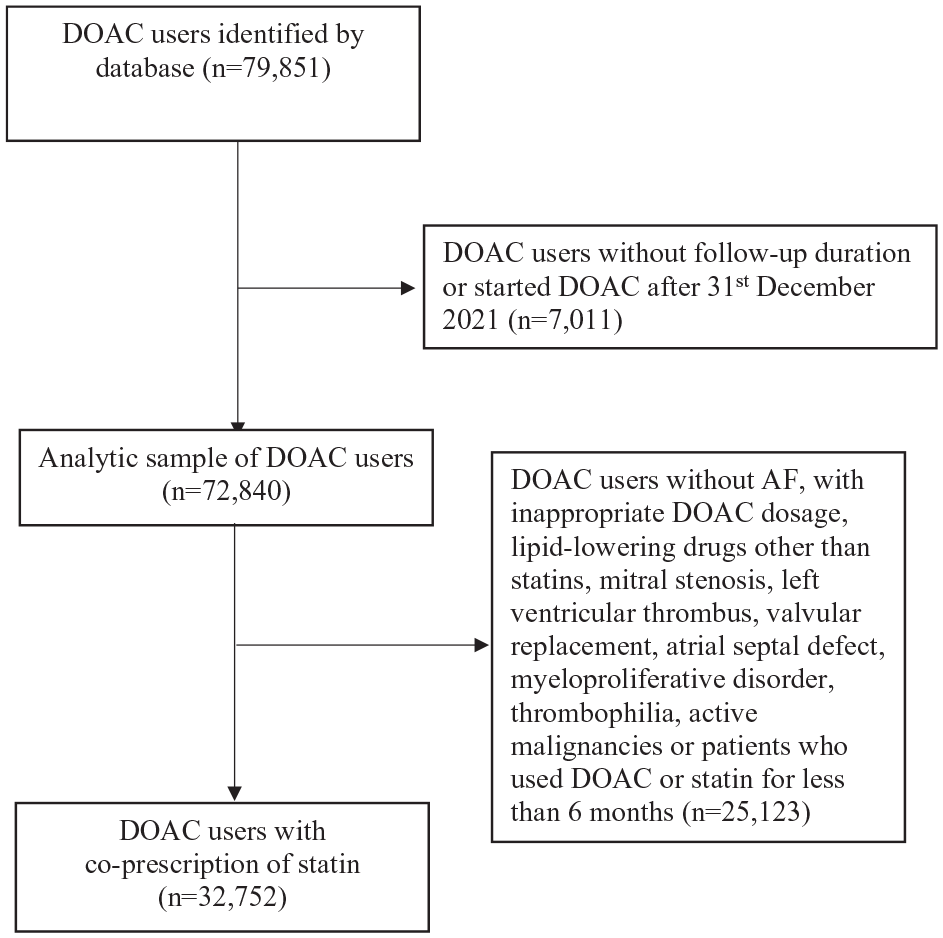
Study flow diagram.
Primary analysis
RCS revealed a non-linear relationship between LDL-C and ischemic stroke (
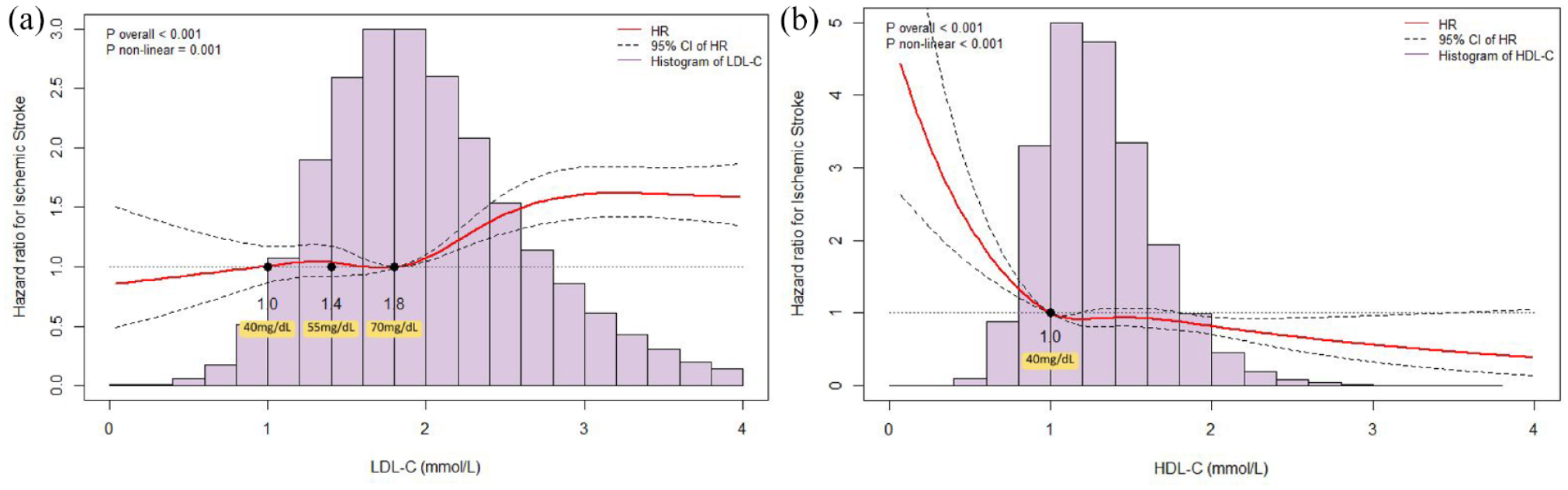
The association of: (a) low-density lipoprotein cholesterol (LDL-C) and (b) high-density lipoprotein cholesterol (HDL-C) with the development of ischemic stroke by restricted cubic spline.
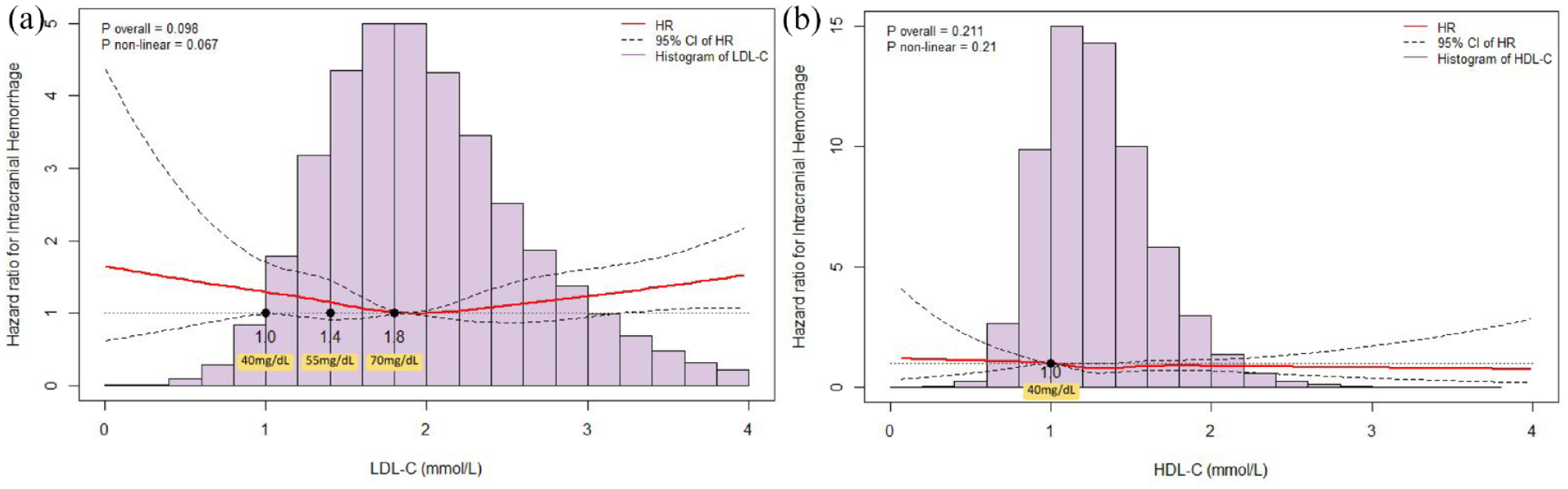
The association of: (a) low-density lipoprotein cholesterol (LDL-C) and (b) high-density lipoprotein cholesterol (HDL-C) with the development of intracerebral hemorrhage by restricted cubic spline.
Low levels of LDL-C and HDL-C were associated with increased risk of mortality in RCS (
Secondary analysis
Statin intensity
In the multivariable marginal structural cause-specific hazard model, high-intensity statin was independently associated with a lower risk of ischemic stroke (wCSHR 0.82, 95% CI 0.67–0.99,
Association of statin intensity and study outcomes.
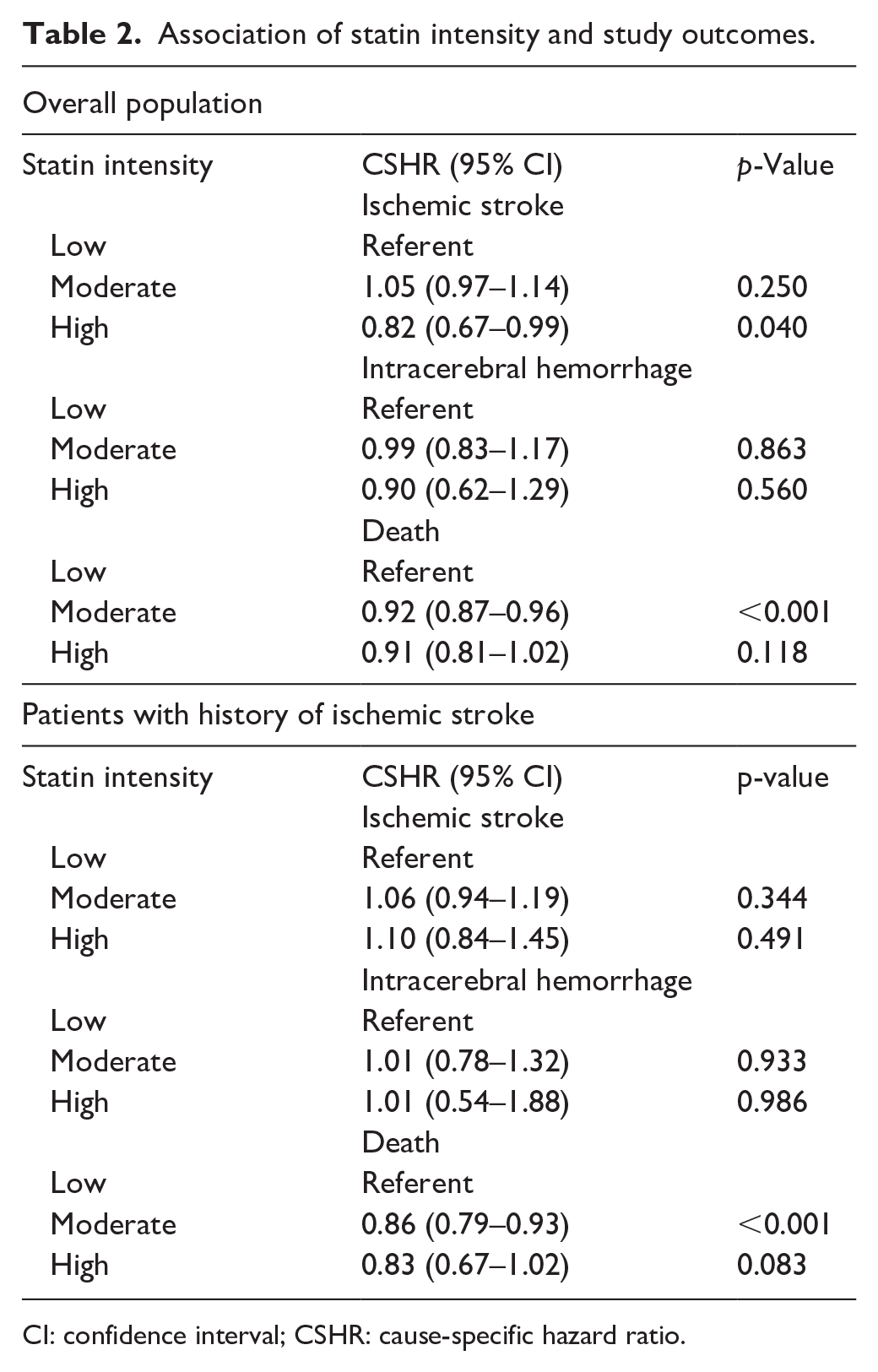
CI: confidence interval; CSHR: cause-specific hazard ratio.
Etiologies of ischemic stroke while on DOAC
During the 6-year study period, incidences of ischemic stroke due to cardioembolism (4.4% vs 2.2%, risk difference 2.2%, 95% CI 1.8–2.6), small artery occlusion (1.4% vs 0.7%, risk difference 0.7%, 95% CI 0.48–0.95) and intracranial artery atherosclerosis (1.3% vs 0.7%, risk difference 0.63%, 95% CI 0.39–0.85) were significantly higher in patients with time-dependent LDL-C ⩾ 1.8 mmol/L versus those < 1.8 mmol/L (eTable 4).
Subgroup with history of ischemic stroke
Among 11,444 AF patients with a history of ischemic stroke, 1298 (11.3%), 259 (2.3%), and 2695 (23.5%) developed recurrent ischemic stroke, ICH, and mortality respectively. LDL-C lowering was associated with reduced risk of recurrent ischemic stroke (RCS:
Discussion
In this study of intensive lipid control with statins in AF patients treated with DOAC, the major findings were: (1) LDL-C lowering was associated with a reduced risk of ischemic stroke; (2) LDL-C lowering was not associated with ICH; and (3) high-intensity statin was associated with a diminished risk of ischemic stroke without increasing the risk of ICH or mortality.
Optimal lipid-lowering treatment in anticoagulated AF patients had been contentious. Statins exhibit pleiotropic antiplatelet and anticoagulant effect in a dose-dependent manner, 26 and low lipid levels were associated an increased risk of ICH. 10 In a Mendelian randomization study, LDL-C-reducing genetic variants of HMGCR gene that mimics the mechanism of statins predicted ICH. 27 Yet, clinical studies that explored the relationships among statins, lipid levels, DOAC and cerebrovascular events had mixed findings. A recent cohort study showed that LDL-C lowering may reduce the risk of ischemic stroke in a population of mostly non-anticoagulated AF patients, 9 while two other cohort studies failed to demonstrate such a finding among anticoagulated AF patients.4,8 On the other hand, statins increased major bleeding risk among dabigatran users in one study, 12 but was associated with lower ICH risk when co-prescribed with DOACs in two other studies.28,29 Of note, the time-dependent models that treated lipid levels as time-varying continuous variables in our study may evaluate cardiovascular outcomes in DOAC users more accurately than investigations that did not consider the time-dependent changes in lipid levels and statin intensities during the follow-up period. 30
The lower ischemic stroke risk in association with LDL-C control was driven in part by the reduction in atherothrombotic strokes, which are common competing stroke mechanisms among AF patients with ischemic stroke despite DOAC. 31 Our study also provided important safety data that intensive LDL-C lowering by statins did not increase the risk of ICH. Yet, the increased mortality at very low LDL-C levels warrants further investigation. Although an increased death rate was reported with low LDL-C in observational studies, 32 subjects in randomized studies had reduced cardiovascular deaths with intensive lipid lowering. 33 As low LDL-C levels could be a proxy of frailty and malnutrition that contributed to mortality, 34 the inclusion of these patients may have confounded our comparisons. In addition, the risk difference in mortality for patients with LDL-C <1.8 mmol/L versus ⩾1.8 mmol/L was small and non-statistically significant in the sensitivity analysis. Therefore, judicious interpretation of the study results is required, especially among patients with a substantial ASCVD risk, such as concurrent ischemic heart disease. Future prospective studies are needed to elucidate the association between mortality and very low LDL-C. Moreover, whether low LDL-C levels attained through PCSK9 inhibition would potentiate ICH or death in DOAC users should be subjected to further research. The risk of ischemic stroke with low HDL-C levels was consistent with that reported in the general population. 35
High-intensity statin therapy was independently associated with lower risk of ischemic stroke without increasing mortality or ICH, while moderate-to-high intensity statins were associated with lower risk of mortality. These observations could be related to pleiotropic effects of statins independent of cholesterol lowering, including endothelial function improvement and anti-inflammatory properties. 36 Thus, patients on moderate-to-high intensity statins might have a lower risk of atherothrombotic stroke and cardiovascular death. Importantly, the previously reported risk of ICH associated with statins was not observed in our cohort. 11 To date, only cellular studies suggested DOACs may preserve blood-brain barrier integrity, which may counteract the pro-hemorrhagic effect of statins. 37 Alternatively, healthy user effect and immortal time bias may have contributed to the phenomenon. Further studies may reveal the possible mechanisms behind our observation. As ischemic stroke despite DOAC use portends poor prognosis especially among those with competing atherothrombotic stroke etiologies linked to dyslipidemia,31,38 intensive statin therapy among AF patients with high concurrent ASCVD risks may mitigate the risk of developing this high-risk condition without excessive risk of ICH as observed in the current study.
Our study has several limitations. First, due to the observational nature of our study, treatment effects or causality could not be established. Despite covariate adjustment in the multivariable models, unmeasured confounding factors such as drug adherence, physical activity and obesity might have introduced bias. Future randomized controlled trials are needed to substantiate our findings. Second, correlation between ICH etiologies and the degree of lipid control was not feasible due to missing ICH locations in 40% of patients who developed the event. Since emerging studies suggested the potential ICH risk with very low LDL-C was driven by cerebral amyloid angiopathy, 39 further studies should determine whether such a risk could be observed among DOAC users. Third, very low LDL-C levels might reflect malnourishment and frailty, contributing to mortality. Fourth, hypertension and diabetes were also associated with ischemic stroke during DOAC usage.5,38 Although adjusted statistically, the possible confounding effect of these comorbidities on study outcomes should be evaluated in future studies. Last, our study results may not be applicable to other ethnicities as Chinese ethnicity is an independent risk factor of ICH. 40
In conclusion, intensive lipid control by high-intensity statins was associated with a lower risk of ischemic stroke in AF patients who required DOACs and did not appear to increase the risk of ICH.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873241272530 – Supplemental material for Lipid control and stroke risk in atrial fibrillation patients treated with direct oral anticoagulants and statins
Supplemental material, sj-pdf-1-eso-10.1177_23969873241272530 for Lipid control and stroke risk in atrial fibrillation patients treated with direct oral anticoagulants and statins by Bonaventure Ip, Terry Yip, Trista Hung, Tsz-Fai Yam, Carly Yeung, Ho Ko, Grace Wong, Xinyi Leng, Vincent Mok, Yannie Soo, David Seiffge, Ashkan Shoamanesh and Thomas Leung in European Stroke Journal
Footnotes
Acknowledgements
The authors acknowledge the contributions from Dr Robert Dan, Mrs Sophia Dan, Ms Anki Miu and Kwok Tak Seng Centre for Stroke Research and Intervention.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.I., C.Y., T.H., T.Y., C.Y., H.K., G.W., X.L., V.M., Y.S., T.L. report no disclosures. D.S. reports research funding from the Swiss Heart Foundation, the Swiss National Science Foundation, the Bangerter-Rhyner Foundation, and Portola/Alexion Pharmaceuticals. All outside the submitted work. A.S. reports research support from the National Institutes of Health, Canadian Institutes for Health Research, Heart and Stroke Foundation of Canada, Marta and Owen Boris Foundation, Daiichi Sankyo, Bristol Myers Squibb, Bayer AG, and Servier Canada, and advisory/speakers bureau honoraria from Daiichi Sankyo, Bayer AG, Takeda Pharmaceutical Company, and Servier Canada. All outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Joint CUHK-NTEC ethnics committee approved the study (CREC Ref No. 2021.349).
Informed consent
Not required given the deidentified data.
Guarantor
B.I. takes full responsibility for the article, including for accuracy and appropriateness of the reference list.
Contributorship
B.I. researched literature, conceived the study and wrote the first draft of the manuscript. C.Y. was involved in data analysis. T.H. was involved in gaining ethical approval and data acquisition. T.Y. was involved in data analysis. C.Y. was involved in data acquisition and data analysis. H.K. was involved in data acquisition and data analysis. G.W. was involved in data acquisition and data analysis. X.L. was involved in data acquisition and data analysis. V.M. reviewed and edited the manuscript. Y.S. reviewed and edited the manuscript. D.S. reviewed and edited the manuscript. A.S. reviewed and edited the manuscript. T.L. reviewed and edited the manuscript. All authors reviewed the manuscript and approved the final version of the manuscript.
Data availability statement
Anonymized data not published within this article will be made available by request from any qualified investigator.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
