Abstract
Introduction:
There is an unmet need for improved detection of intracranial aneurysms (IAs) and distinction between stable and unstable (high rupture risk) IAs. Within the IA wall, synthesis and degradation of type I collagen as the main molecular constituent balance each other to maintain IA stability. We hypothesized that collagen breakdown products could serve as molecular markers for IA presence and instability.
Patients and methods:
This prospective, cross-sectional, single-center study included patients with unstable (growing/symptomatic/ruptured) and stable IAs and controls. We determined C-telopeptide (CTx) and c-terminal telopeptide (ICTP) as breakdown products of type I collagen in arterial and venous blood.
Results:
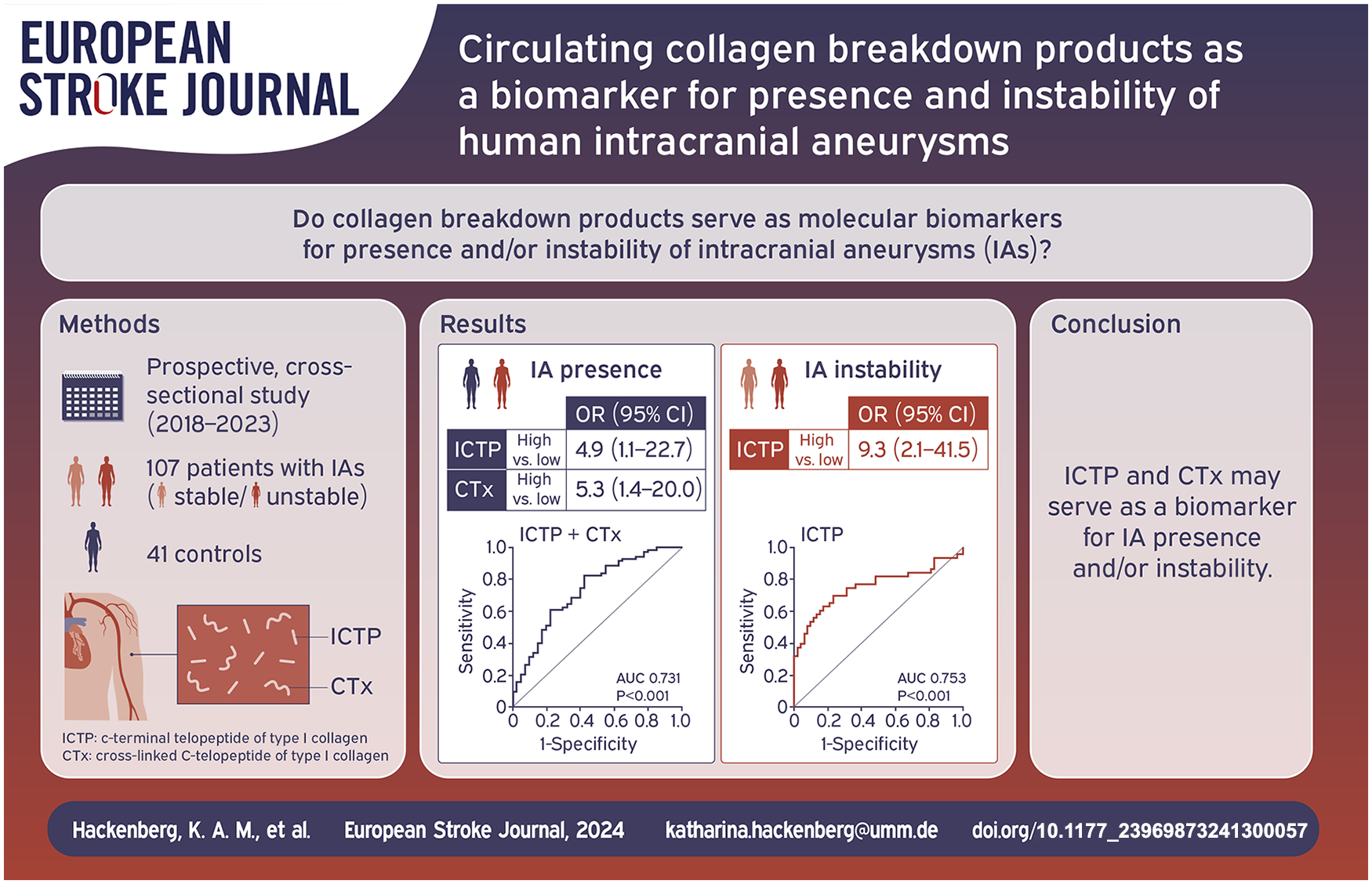
We included 107 participants with IAs (52 stable/44 unstable) and 41 controls. The correlation between intra-aneurysmal and venous levels was r = 0.63 (p < 0.001) for ICTP, r = 0.55 (p = 0.001) for CTx. The odds of harboring an IA were five times higher for participants with high compared to low venous levels of collagen breakdown products (ICTP: odds ratio (OR) 4.9 (95% CI 1.1–22.7); CTx: OR 5.3 (95% CI 1.4–20.0)). The OR for having an unstable IA was 9.3 (95% CI 2.1–41.5) for patients with high compared to low venous ICTP levels. The area under the curve for ICTP levels as a marker for IA instability was 0.75.
Discussion and conclusion:
Increased levels of venous collagen breakdown products, especially ICTP levels, could serve as a biomarker for IA presence and instability and complement current data for management of unruptured IAs on an individual patient level. Future studies with longitudinal data are needed to validate ICTP as a biomarker for high risk IAs.
Introduction
Unruptured intracranial aneurysms (IAs) have a prevalence of 3% in the adult population. 1 While the majority of IAs remain clinically silent, some IAs rupture and result in subarachnoid hemorrhage (SAH). 2 SAH still continues to have a poor prognosis with a combined risk of severe long-term dependency and case fatality between 25% and 50%. 3 Since SAH often occurs in young patients, with a peak age between 50 and 60 years, the burden of SAH on an individual and socioeconomic level is high.4,5 Possible ways to prevent SAH are improved detection of IAs and identification as well as preventive repair of those IAs that have a high risk of rupture. Aneurysms can only be diagnosed by neuroimaging, but at present it is not effective to screen for IAs on a broad scale in the general population. Many persons, even in high-risk groups, undergo screening without having an IA. 6 Thus, to increase the efficiency, an upstream examination in terms of a feasible and cost-effective biomarker is needed as a screening tool for downsizing the population that can benefit from neuroimaging. Furthermore, once IAs are detected, it is pivotal to distinguish between patients with stable and unstable IAs to better select patients for preventive aneurysm treatment.7–9 Currently available clinical and radiological risk factors for IA instability do not discriminate well enough between stable and unstable IAs.8,10,11 Thus, a more precise and personalized approach is required to reliably identify those few patients that are at high risk of rupture to advise them preventive repair.
Type I collagen is the major molecular constituent of the IA wall. 12 In patients with IAs and concomitant hypertension and/or smoking collagen age of IAs was younger, that is collagen turnover was more rapid.12,13 Within the IA wall ongoing collagen synthesis and degradation balance each other for maintaining IA stability.12,14 Aneurysm instability has been linked to rapid collagen turnover due to accumulation of proteases and subsequent collagen degradation.13,14 Assuming that rapid turnover of collagen results in increased breakdown of collagen in IAs, circulating collagen breakdown products from IAs could therefore be a marker for presence and instability of IAs.
We hypothesized that collagen breakdown products can be detected both from arterial blood within the aneurysm and from peripheral venipuncture in patients with IAs, and that levels of collagen breakdown products may predict IA presence and IA instability.
Patients and methods
The study was performed based on The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cross-sectional studies.
Written informed consent was obtained from all participants or, in comatose patients, from an authorized representative, according to the Declaration of Helsinki.
Study population
This cross-sectional study prospectively included eligible patients with IAs admitted to a single academic medical center between October 2018 and June 2023. Inclusion criteria were an age of 18 years or older and the presence of IAs. Exclusion criteria were pre-existing conditions that are associated with more rapid bone turnover (osteoporosis, bone metastases), renal failure, ongoing hyperthyroidism, connective tissue diseases and permanent intake of corticosteroids, that could interfere with the bone metabolism, thus confound blood type I collagen breakdown products. Participants served as controls, if they were at least 18 years of age, if they did not meet the exclusion criteria and were either admitted to our department for conditions other than IAs or further participants without a known IA during the same time period (Supplemental Table S1).
Biosampling and biomarker analysis
Detailed venous and arterial sampling is described in Supplemental Methods. 15
Serum was separated by centrifugation at 3000g at room temperature for 30 min and stored in aliquots in a freezer at −80°C until analysis.
The collagen breakdown products cross-linked C-telopeptide of type collagen I (CTx) and c-terminal telopeptide of type I collagen (ICTP) were measured in duplicates using immunoassays 16 (detailed methods in Supplemental Methods).
Definitions and clinical data acquisition
The presence of an IA was defined as a saccular dilation at a bifurcation of cerebral arteries. Aneurysm growth was defined as an increase of IA diameter of 1 mm or larger in any direction in comparison to preceding imaging. 17 Patients with unruptured IAs receive annual follow-up imaging according to our institutional protocol. Stable IAs were defined as unruptured IAs without change in morphology and aneurysm growth for at least 1 year. Aneurysm instability was defined as (a) growth or change in morphology on follow-up imaging or (b) symptomatic IA with presence of clinical symptoms due to mass effect of the IA or (c) IA rupture. Ruptured IAs were defined by presence of SAH in neuroimaging or lumbar puncture. Further definitions (SAH severity, hypertension, smoking, familial history, previous SAH, irregularity) are described in Supplemental Methods.17,18 In case of multiple IAs further analyses were performed with the largest IA as the index IA.
Clinical data acquisition was performed by a study nurse, who was blinded to data analyses.
Statistical analyses
For statistical analyses the SPSS statistics software package 25 (IBM Corporation, Armonk, New York, USA) and SAS statistics software package 9.4 (SAS Institute Inc., Cary, North Carolina, USA) were used. For data visualization the libraries pandas, seaborn, matplotlib based on Python 3.12 were used (Python Software Foundation, Wilmington, Delaware USA). Data are reported as median (interquartile range). To identify differences in levels of collagen breakdown products regarding IA presence or instability the Mann-Whitney test was used. To identify relations between collagen breakdown products of different locations and/or clinical, morphological parameters correlation analyses were performed using Pearson correlation (r). To identify biomarkers for IA presence and (in)stability we performed logistic regression analyses with the covariates sex, age, hypertension, and smoking as known biological risk factors for IA pathogenesis and possible confounders for IA presence and instability. Logistic regression analyses were performed with continuous levels of collagen breakdown products and according to quartiles with the lowest quartile as reference. Receiver operating characteristic (ROC) analysis was performed to quantify the predictive accuracy using the area under the curve (AUC) for collagen breakdown products as predictors for IA presence and instability. Missing data assumed as missing completely at random were handled with pairwise deletion. The value for statistically significant differences was set at probability (p) value < 0.05.
Results
A total of 148 participants (107 patients with IAs and 41 controls) were included (Table 1). Out of the 107 patients with IAs, 52 patients presented with stable IAs and 44 patients with an unstable IA, of which 35 presented with SAH. Out of the 11 unstable unruptured IAs, 9 were growing and 2 were symptomatic IAs (symptoms are described in Supplemental Methods). Out of the 72 patients with unruptured IAs 11 patients did not fulfill the criteria of IA stability or instability and were omitted from the analyses for IA instability. Intra-aneurysmal blood samples were available from 39 patients, femoral arterial blood samples from 56 patients. Venous blood samples were collected from all participants.
Characteristics of controls and patients.

Numbers are given in n (%) or median (interquartile range).
ACA: anterior cerebral artery; ACOM: anterior communicating artery; IA: intracranial aneurysm; ICA: internal carotid artery; MCA: middle cerebral artery; PCOM: posterior communicating artery; SAH: subarachnoid hemorrhage.
Collagen breakdown products in arterial and venous blood
The correlation between intra-aneurysmal ICTP levels and femoral arterial ICTP levels was r = 0.90 (p < 0.001), and venous ICTP levels r = 0.63 (p < 0.001; Supplemental Figure S1). The correlation between intra-aneurysmal CTx levels and femoral arterial CTx levels was r = 0.93 (p < 0.001), and venous CTx levels r = 0.55 (p = 0.001; Supplemental Figure S1).
Distributions of intra-aneurysmal, femoral arterial and venous ICTP and CTx levels are shown in Supplemental Figure S2.
Distributions of venous collagen breakdown products regarding potential confounders for IA presence and instability are shown in Supplemental Figures S3 to S5.
Collagen breakdown products and IA presence
Median venous ICTP levels were in patients with IAs 4.49 (3.36–6.39) µg/l and in controls 3.95 (2.71–4.71) µg/l (p = 0.002; Figure 1(a)). Median venous CTx levels were in patients with IAs 0.31 (0.20–0.49) ng/ml and in controls 0.21 (0.11–0.30) ng/ml (p < 0.001; Figure 1(b)).
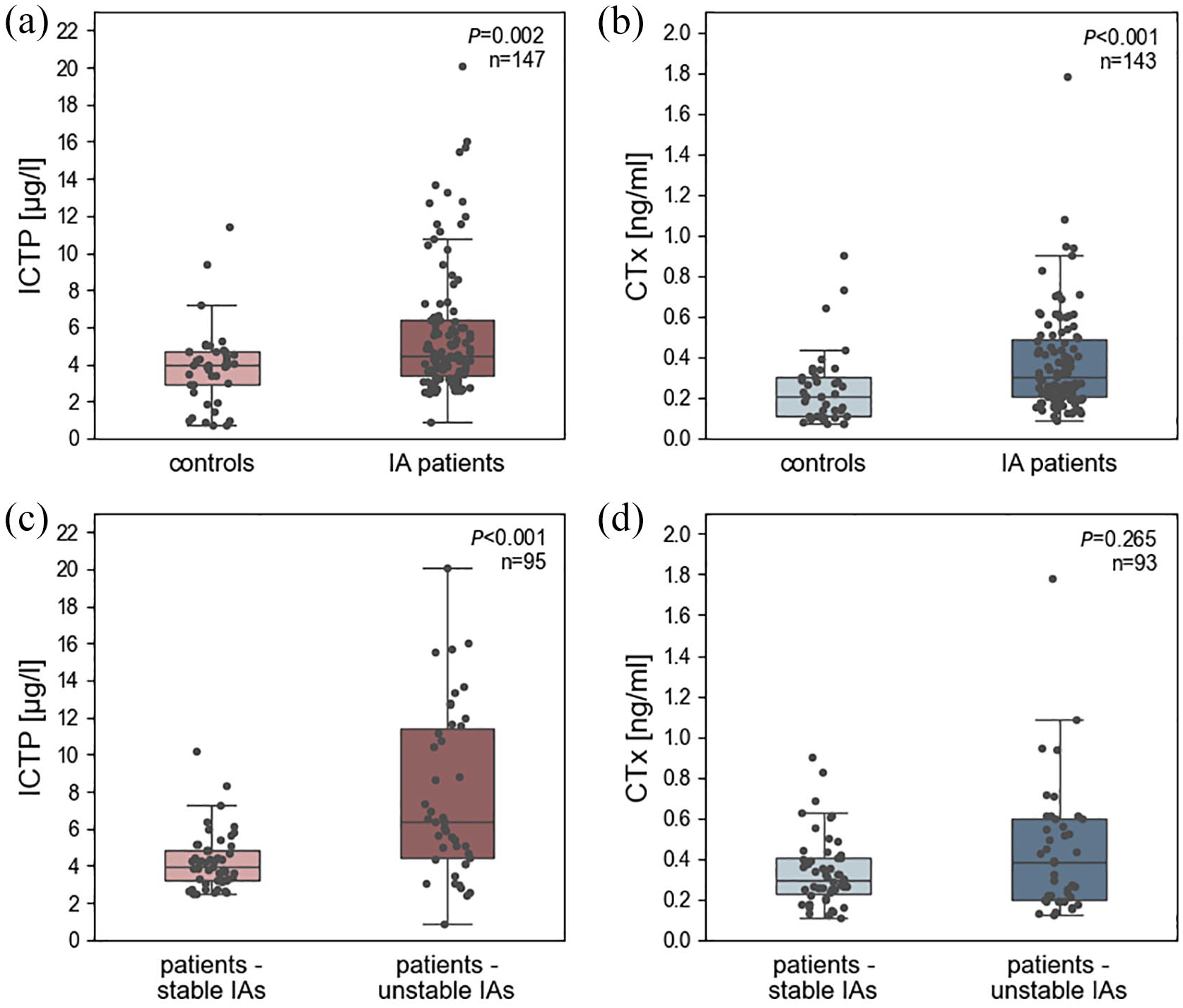
Distribution of collagen breakdown products regarding IA presence and IA instability. Distribution of (a) venous ICTP levels in patients with IAs compared to controls, (b) venous CTx levels in patients with IAs compared to controls, (c) venous ICTP levels in patients with unstable IAs compared to patients with stable IAs, and (d) venous CTx levels in patients with unstable IAs compared to patients with stable IAs.
The adjusted odds of harboring an IA increased by 32% per 1 µg/l increase in ICTP levels (odds ratio (OR) 1.32 (95% CI, 1.1–1.6) per 1 µg/l unit change) and by 34% per 0.1 ng/ml increase in CTx levels (OR 1.34 (95% CI, 1.03–1.7) per 0.1 ng/ml unit change; Supplemental Table S2). For participants with venous ICTP levels in the highest quartile the adjusted OR for having an IA was 4.9 (95% CI 1.1–22.7) and for participants with venous CTx levels in the highest quartile the OR for IA presence was 5.3 (95% CI 1.4–20.0) compared to the lowest quartile each (Table 2).
Association of collagen breakdown products with IA presence or IA instability according to quartiles.

aOR: adjusted odds ratio; CTx: cross-linked c-telopeptide of type collagen I; IA: intracranial aneurysm; ICTP: c-terminal telopeptide of type I collagen; OR: odds ratio.
Adjusted for covariates sex, age, arterial hypertension, and smoking.
ROC analysis demonstrated an AUC of 0.73 for venous ICTP and CTx levels as predictors for IA presence (Figure 2).
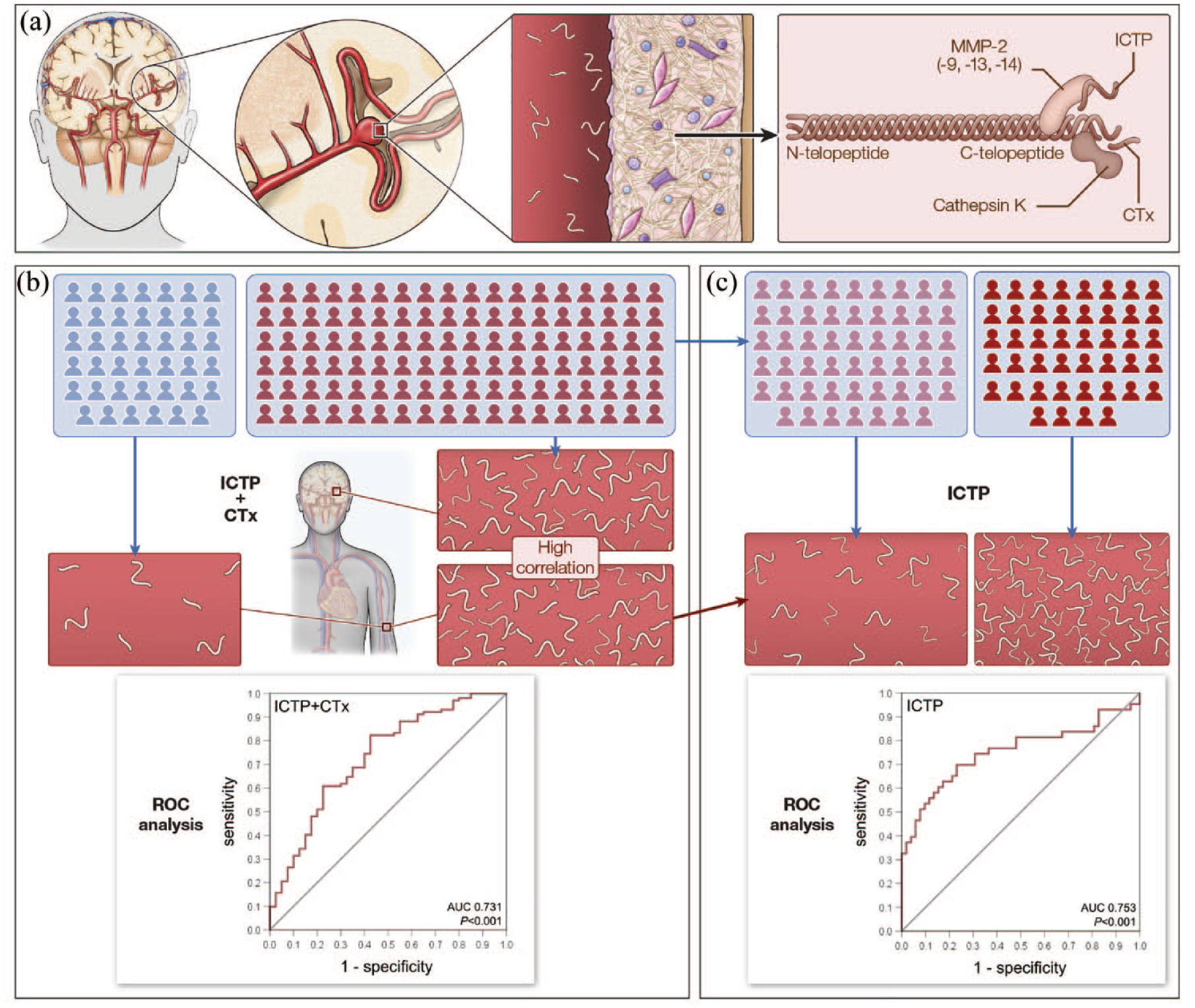
Collagen breakdown products CTx and ICTP as markers for IA presence and/or IA instability: (a) Unruptured IA of the bifurcation of the middle cerebral artery. On a molecular level, synthesis and degradation of type I collagen in the IA wall balance each other for maintaining wall stability.12-14 Collagen breakdown products ICTP and CTx, which are degraded on the c-telopeptide of type I collagen, accumulate in the blood. 16 While ICTP is predominantly generated by MMP-2, CTx is degraded by cathepsin K, 16 (b) To establish a biomarker for IA presence, blood samples were drawn from controls (blue persons) and patients with IAs (red persons). Intra-aneurysmal samples were collected from patients with IAs before aneurysm repair. Correlations were found between intra-aneurysmal and venous levels of ICTP and CTx each in patients with IAs. Venous levels of ICTP and CTx were both higher in patients with IAs compared to controls. ROC analysis demonstrated an AUC of 0.73 for venous ICTP and CTx levels markers for IA presence (n = 142), and (c) To establish a biomarker for IA instability, patients with IAs were divided into patients with stable IAs (light red persons) and unstable IAs (dark red persons). Venous ICTP levels were higher in patients with unstable IAs compared to stable IAs. ROC analysis demonstrated an AUC of 0.75 for ICTP levels as a predictor for IA instability (n = 95).
Collagen breakdown products and IA instability
Median venous ICTP levels were in patients with unstable IAs 6.35 (4.38–11.59) µg/l and in patients with stable IAs 3.92 (3.21–4.88) µg/l (p < 0.001; Figure 1(c)).
The adjusted odds of harboring an unstable IA increased by 47% per 1 µg/l increase in ICTP levels (OR 1.47 (95% CI, 1.2–1.8) per 1 µg/l unit change; Supplemental Table S2). For patients with venous ICTP levels in the highest quartile the adjusted OR for having an unstable IA was 9.3 (95% CI 2.1–41.5) compared to the lowest quartile (Table 2).
ROC analysis demonstrated an AUC of 0.75 for venous ICTP levels as a predictor for IA instability (Figure 2).
Median venous CTx levels were in patients with unstable IAs 0.38 (0.20–0.60) ng/ml and in patients with stable IAs 0.30 (0.23–0.40) ng/ml (p = 0.265; Figure 1(d)).
Discussion
Our study on the collagen breakdown products ICTP and CTx in patients with IAs found an association between levels within IAs, from the femoral artery and venous levels of ICTP and CTx each. Venous ICTP and CTx levels were higher in patients with IAs compared to controls. The odds of harboring an IA were nearly five times higher for participants with high compared to low collagen breakdown products. In patients with IAs ICTP levels were higher in patients with unstable IAs compared to stable IAs. The odds of having an unstable IA were nine times higher for patients with high compared to low ICTP levels.
Previous studies aimed to identify high-risk groups for IAs and for SAH in the general population, 6 but biomarkers to complement screening tools or rupture risk assessment are needed to guide IA management on an individual patient level. Recent studies utilized molecular targets (e.g. circular non-coding RNA, circular neutrophil RNA, glycosylated proteins, microRNA-126),19–22 but these biomarkers are currently not readily available as standard laboratory assay. Other studies identified potentially predictive biomarkers using intra-aneurysmal or femoral arterial blood,23,24 which require invasive procedures that are not part of routine clinical practice, particularly for screening purposes. Thus, collagen breakdown products determined in venous blood might be a promising diagnostic and prognostic biomarker.
There are different explanations for our findings: Type I collagen is the major constituent of the IA wall.12,14 Since ongoing collagen remodeling seems to play a major role in balancing between IA stability and instability,25,26 accumulating collagen breakdown products from the vascular wall are the most likely source of higher venous CTx and ICTP levels in patients with IAs compared to controls. Before rupture many macrophages infiltrate the IA wall and secrete proteases which degrade the collagen matrix leading to younger collagen age and higher collagen turnover.12–14,25,26 This is probably reflected by even higher ICTP levels in patients with unstable IAs. An explanation for the difference in CTx and ICTP levels in patients with unstable IAs might be that they are generated by different proteases, ICTP mainly by MMP-2 and CTx by cathepsin K, 16 and that MMPs produced by macrophages seem to be rather associated with IA instability.24,27,28 Studies on patients with cardiovascular disease and risk factors reported that venous ICTP levels in serum were higher. 29 Since we adjusted for cardiovascular risk factors as potential confounders, an unspecific association of ICTP and IA presence or instability in our study is unlikely.
Strengths of our study are: First, this is one of the largest prospective studies on human blood biomarkers of patients with stable and unstable IAs. Second, we simultaneously collected and correlated target blood biomarkers in multiple different locations in the vasculature (intra-aneurysmal, femoral arterial, peripheral venous). Third, we prospectively assessed and included patients with unstable IAs at high risk for rupture, which are a rare patient population. Fourth, the analysis per patient requires about 13 min for CTx and ICTP each and costs (including immunoassays and working hours of staff) about EUR 26 for CTx and EUR 31 for ICTP and thereby are easily retrievable and cost-effective.
Our study also holds limitations: First, almost 80% of the unstable IAs in our population were ruptured IAs. Theoretically, systemic critical illness and/or lung injury from mechanical ventilation could induce higher levels of collagen breakdown products. 30 However, we did not observe any associations between ICTP levels and SAH severity or mechanical ventilation (Supplemental Figures S6 and S7). Due to the rather small sample size in the subgroups the results have to be interpreted with caution, but the ICTP levels seem to be similar in patients with good and poor grade SAH. Second, despite the strength of including unstable unruptured IAs, they only represent 20% of all unstable IAs. Further studies with a higher amount of these patients are needed. Third, we did not collect repeated blood samples in the same patients over time. To understand longitudinal trends of ICTP expression on an individual patient level, we currently investigate these trends with a subset of patients in the ongoing PROTECT-U trial. 31 Fourth, markers such as ICTP and CTx may underlie a circadian variation with a nocturnal peak, 32 which could confound measurement results. Since all blood sampling was performed between 9 am and 10 pm, this should play a minor part. Finally, we excluded participants with diseases and medication that interfere with bone turnover, mainly patients with osteoporosis. Thus, postmenopausal female smokers, who are a high-risk group for harboring IAs and having osteoporosis, were partially not included. Although primary prevention of osteoporosis in risk groups is increasingly performed, in future studies we will also assess patients with osteoporosis. This will help understanding how absolute CTx and ICTP levels differ in patients with IAs and concomitant osteoporosis and will increase the external validity of our results.
Conclusions
Venous concentrations of breakdown products of the main constituent in IAs – type I collagen – are elevated in patients with IAs compared to controls and in patients with unstable compared to stable IAs. ICTP and CTx are easily retrievable and cost-effective and might serve as a biomarker for IA presence, once cut-off values are established in cohorts of high-risk persons for harboring IAs. However, by serial investigations of ICTP levels in patients with unruptured IAs that undergo a strategy with follow up imaging, current decision systems or guidelines for unruptured IAs8,9,11 could be complemented to enable management of IAs on an individual patient level and according to actual biological data.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241300057 – Supplemental material for Circulating collagen breakdown products as a biomarker for presence and instability of human intracranial aneurysms
Supplemental material, sj-docx-1-eso-10.1177_23969873241300057 for Circulating collagen breakdown products as a biomarker for presence and instability of human intracranial aneurysms by Katharina AM Hackenberg, Peter Richter, Svetlana Hetjens, Rita Dreier, Thomas Ratliff, Oluwadamilola Akanji, Judith Dremel, Amr Abdulazim, Ibrahim Al Masalmeh, Mansour Alzghloul, Eva Neumaier-Probst, Christoph Groden, Sherry H-Y Chou, Gabriel JE Rinkel and Nima Etminan in European Stroke Journal
Footnotes
Acknowledgements
We are very grateful to all staff members of the Departments of Neurosurgery and Neuroradiology who helped collecting blood samples. For processing the blood samples, we are very thankful to our laboratory assistant Elena Maier. We thank Professor Carmen Wängler and Professor Björn Wängler and their laboratory team members for the assistance and provision of their laboratory for performing the radioactive immunoassays. We also thank Gereon Hackenberg for his support in data visualization and Professor Jörg Krebs for critical input during the interpretation of the results.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the ethical committee of the Medical Faculty Mannheim of the University of Heidelberg (ID 2018-619N-MA).
Informed consent
Written informed consent was obtained from all participants or, in comatose patients, from an authorized representative, for their anonymized information to be published in this article and according to the Declaration of Helsinki.
Guarantor
KH
Contributorship
NE, KH, and GR researched literature and conceived the study. KH, PR, RD, TR, OA, JR, AA, IA, MA, EN, and CG were involved in gaining ethical approval, patient recruitment, blood sampling and data analyses. KH, SH, and SC performed statistical analyses. KH, SC, NE, and GR were involved in data interpretation. KH wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
Anonymized data not published within this article will be made available by reasonable request from any qualified investigator.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
