Abstract
Introduction:
Computed tomography perfusion (CTP) is variably considered to assess eligibility for endovascular thrombectomy (EVT) in acute ischemic (AIS) stroke patients. Although CTP is recommended for patient selection in later (6–24 h) time window, it is currently not recommended in the earlier (0–6 h) time window and the costs and health effects of including CTP for EVT selection remain unknown. We aim to estimate the costs and health effects of using CTP for EVT selection in AIS patients compared to conventional selection.
Patients and methods:
CLEOPATRA is a healthcare evaluation study using clinical and imaging data from multiple, prospective EVT trials and registries in both the earlier and later time windows. To study the long-term health and cost effects, we will construct a (“Markov”) health state transition model simulating the clinical outcome over a 5-year follow-up period for CTP-based and conventional selection for EVT. Clinical data acquired within the current study and estimates from the literature will be used as input for probabilities of events, costs, and Quality-Adjusted Life Years (QALYs) per modified Rankin Scale (mRS) subscore. Primary outcome for the cost-effectiveness analysis will be the Incremental Cost-Effectiveness Ratio (ICER) in terms of costs per QALY gained over the simulated follow-up period.
Study outcomes:
Outcome measures will be reported as cumulative values over a 5-year follow-up period.
Discussion:
This study will provide preliminary insight into costs and health effects of including CTP in the selection for EVT for AIS patients, presenting between 0 and 24 h after time last known well. The results may be used to develop recommendations and inform further implementation projects and studies.
Introduction and rationale
Computed tomography perfusion (CTP) is variably considered in the selection of anterior circulation large vessel occlusion (LVO) acute ischemic stroke (AIS) patients for endovascular treatment (EVT).1–3 In the earlier (0–6 h) time window, CTP-estimated core volume is currently not recommended for the selection for EVT.4,5 Although EVT showed improved outcomes compared to best medical management in the earlier time window regardless of CTP-estimated core volume, it remains unclear if CTP can reliably estimate the ischemic core in the first hours after stroke onset and whether EVT is favorable for patients with larger core volumes.2,3,6,7 In the later (6–24 h) time window, patient selection based on advanced imaging, such as CTP, is recommended after the added value of EVT was shown in patients with small, – mostly CTP-estimated – core volumes.2,3
It is unethical to randomize on the added value of CTP for the selection for EVT in the earlier time window as a beneficial treatment option could be withheld. Modeling studies can be used to simulate diagnostic workups (i.e. based on CTP or conventional imaging) and to deviate treatment decision thresholds based on CTP-estimated core volumes to assess our research question: What is the potential value of CTP as a diagnostic tool? In addition, cost-effectiveness modeling allows to include costs and health effects for the use of CTP for EVT selection in the long run. The primary objective of Cost-Effectiveness of CT Perfusion for Patients With Acute Ischemic Stroke (CLEOPATRA) is to estimate the costs and health effects of including CTP in the treatment selection for EVT for patients with LVO AIS compared to the conventional diagnostic workup in both the earlier and later time windows in the Netherlands.
Patients and methods
Data acquisition and inclusion criteria
CLEOPATRA is a healthcare evaluation study with a cost-effectiveness analysis based on a health state transition model using retrospective, multicenter, observational data from within the Dutch healthcare system. Patients enrolled in one of the trials from the Consortium for New Treatments of Acute Stroke (CONTRAST) consortium 8 (i.e. MR CLEAN-NO IV, MR CLEAN-MED, and MR CLEAN-LATE) and patients included in the MR CLEAN Registry will be included if EVT and CTP was performed between January 2018 and March 2022. Additionally, a local cohort of AIS patients from our comprehensive stroke center (Amsterdam University Medical Centers, Amsterdam, The Netherlands) will be included if EVT and CTP were both performed. Patients included in our local cohort were not included one of the abovementioned trials. All patients in the 0–6 h time window were treated with EVT. If eligible, patients received additional IVT prior to EVT. Patients enrolled in the MR CLEAN-LATE trial were randomized between EVT and best medical management. 9
Inclusion criteria are: ⩾18 years; AIS caused by a proximal occlusion of the anterior circulation demonstrated by CTA (intracranial carotid artery (ICA), middle (M1/M2) or anterior (A1/A2) cerebral artery); National Institutes of Health Stroke Scale (NIHSS) ⩾ 2; and ⩾8 cm brain coverage on CTP. Exclusion criteria are: pre-stroke disability (i.e. modified Rankin Scale (mRS) > 2), and presence of intracranial hemorrhage on CT or MRI. A detailed overview of the study-specific inclusion and exclusion criteria for the studies used as data source for CLEOPATRA is provided in Table 1.
Summary table. Data sources used in CLEOPATRA and their specifications.
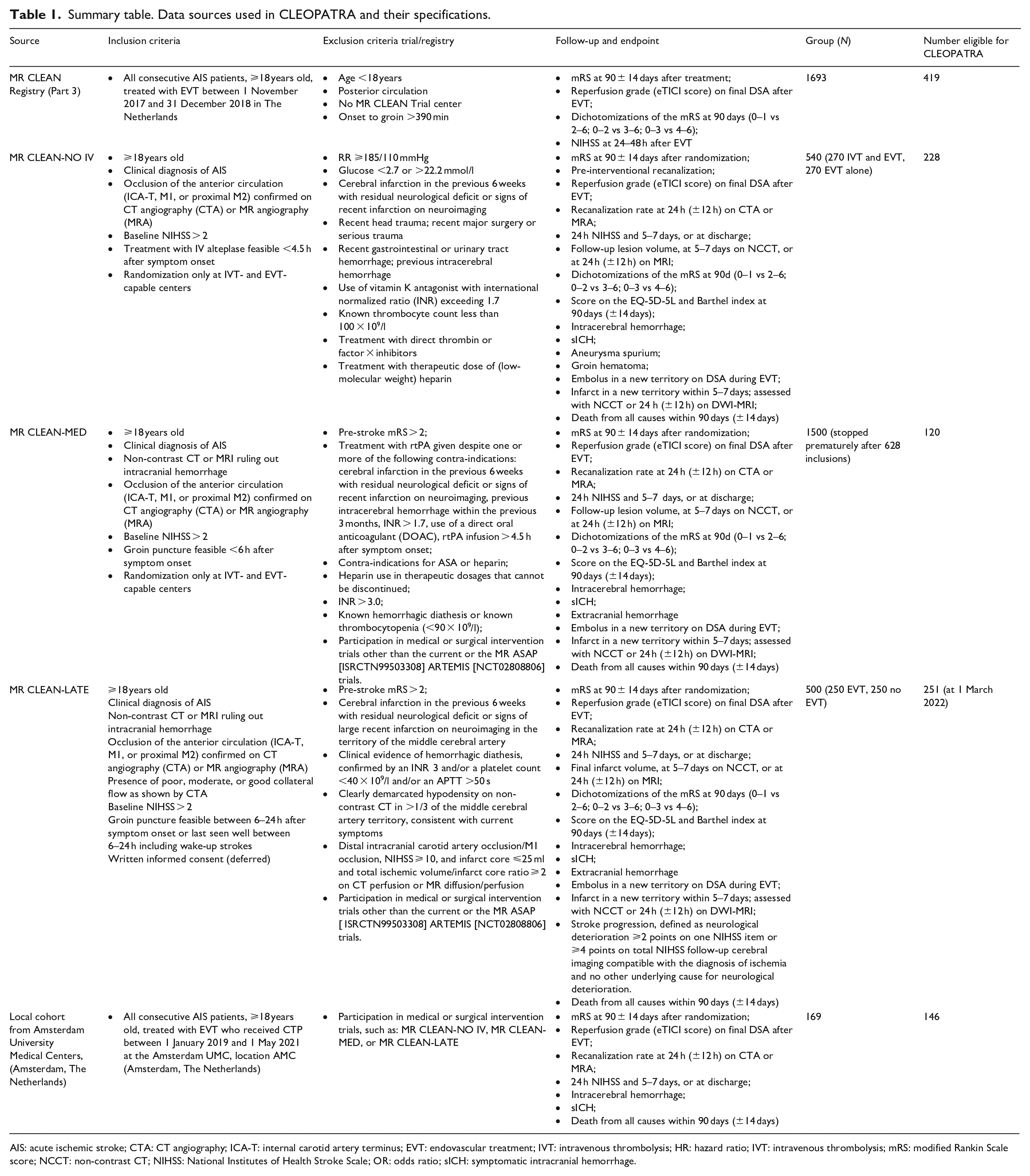
AIS: acute ischemic stroke; CTA: CT angiography; ICA-T: internal carotid artery terminus; EVT: endovascular treatment; IVT: intravenous thrombolysis; HR: hazard ratio; IVT: intravenous thrombolysis; mRS: modified Rankin Scale score; NCCT: non-contrast CT; NIHSS: National Institutes of Health Stroke Scale; OR: odds ratio; sICH: symptomatic intracranial hemorrhage.
Ethical considerations
After approval from the institutional review board, patients or representatives were given the possibility to refuse to provide consent as part of the deferred consent procedure from the CONTRAST consortium (i.e. the MR CLEAN-NO IV, MR CLEAN-MED, MR CLEAN-LATE trials).8–11 Patients in the MR CLEAN Registry were provided with a written explanation of the study. 12 In the written explanation for all of the abovementioned trials, patients or their representatives are informed that the acquired data can be re-used by future studies. Patients included in our local cohort were not asked for written consent, since it was expected that a large part of the patients would have deceased or would not be reachable by mail. Data of the latter patients were entered fully anonymously in the study base. This was decided in consultation with the legal consultant of the Amsterdam University Medical Centers, location AMC. Model consent forms from the trials used as data source for CLEOPATRA can be accessed on https://www.contrast-consortium.nl. Final decisions on protocol changes, publication, and reporting will be made by the CLEOPATRA PIs.
Health care problem analyzed
In CLEOPATRA, the conventional diagnostic workup (i.e. baseline non-contrast CT (NCCT) and CT angiography (CTA); control arm) will be compared with CTP-based patient selection for EVT (intervention arm). In the intervention arm, patients received best medical management if they were excluded from EVT based on CTP-based selection. In the control arm, all patients received EVT in the earlier time window and no patients received EVT in the later time window, according to the current AHA/ESMINT guidelines.4,5 Since only a small proportion of the patients included in CLEOPATRA were randomized for EVT (i.e. the 6–24 h time window patients enrolled in the MR CLEAN-LATE trial), we will simulate the counterfactual clinical outcomes for patients in both time windows. More specifically, we will use odds ratios (ORs) for EVT treatment effect and the ORs for treatment effect modification by other variables (e.g. collateral status, time from onset to randomization) to compute the 90-day mRS of patients who underwent EVT as if they did not undergo EVT and vice versa. The ORs for the earlier time window will be derived from the HERMES collaboration and the MR CLEAN trial.6,13 The ORs for the later time window will be based on analyses from the MR CLEAN-LATE trial (Table 2). 11
Data sources for model input parameter estimation.
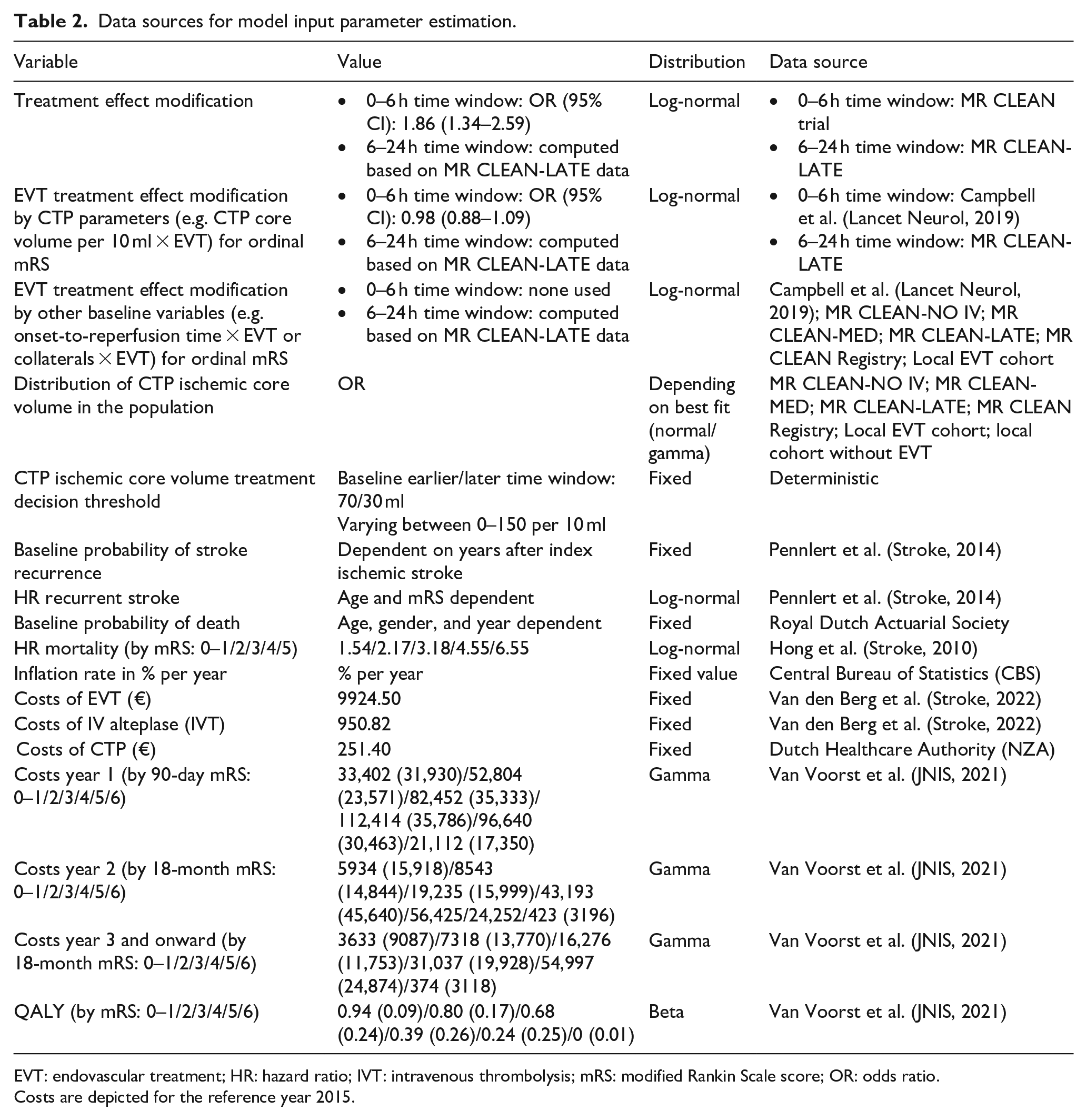
EVT: endovascular treatment; HR: hazard ratio; IVT: intravenous thrombolysis; mRS: modified Rankin Scale score; OR: odds ratio.
Costs are depicted for the reference year 2015.
The predictive effect of CTP-estimated core volume on functional outcome (mRS) in the earlier time window will be based on previously determined treatment effect modification estimates regarding the mRS at 90 days. The predictive effect of CTP on functional outcome (mRS) in the later time window will be derived from models for individual absolute treatment benefit of EVT that will be developed in the ongoing MR CLEAN-LATE trial. 11 Treatment benefit will be expressed as the shift in the distribution of the mRS at 90 days with and without treatment. Model performance will be evaluated with discrimination and calibration. We will seek to validate the model in international datasets of studies of thrombectomy versus no thrombectomy in the later time window.
Model design
For both time windows, a separate health state transition (“Markov”) model will be constructed to simulate 5-year modified Rankin Scale (mRS) follow-up for selection strategies with (control arm) and without CTP (intervention arm). TreeAge Pro (TreeAge Pro 2019, version R2.1; TreeAge, Williamstown, MA, USA) will be used to perform a baseline simulation. More fine-grained simulations and sensitivity analyses will be performed in Python 3.9. The source code will be made available. The 5-year follow-up period was chosen based on the availability of cohorts for evaluating the forecasted mRS up to 5 years. 14 Moreover, simulating effects beyond 5 years would introduce an exponentially increasing error in the mRS forecasts which becomes even more prominent due to the high baseline age (median MR CLEAN-NO IV cohort included in CLEOPATRA: 71 (IQR 61–79) years old) and concurrent relatively short life expectancy of the selected population. The model will exist of a short-term part simulating mRS at 90 days and a long-term part simulating the yearly mRS decay. Similar to our previous work, mRS subscores will be used as health states with related costs and QALYs per unit of time per health state. 14 Cohorts will be simulated based on patient-level micro-simulations to relate baseline characteristics to cost-effectiveness outcomes. The health state transition model is depicted in Figure 1.
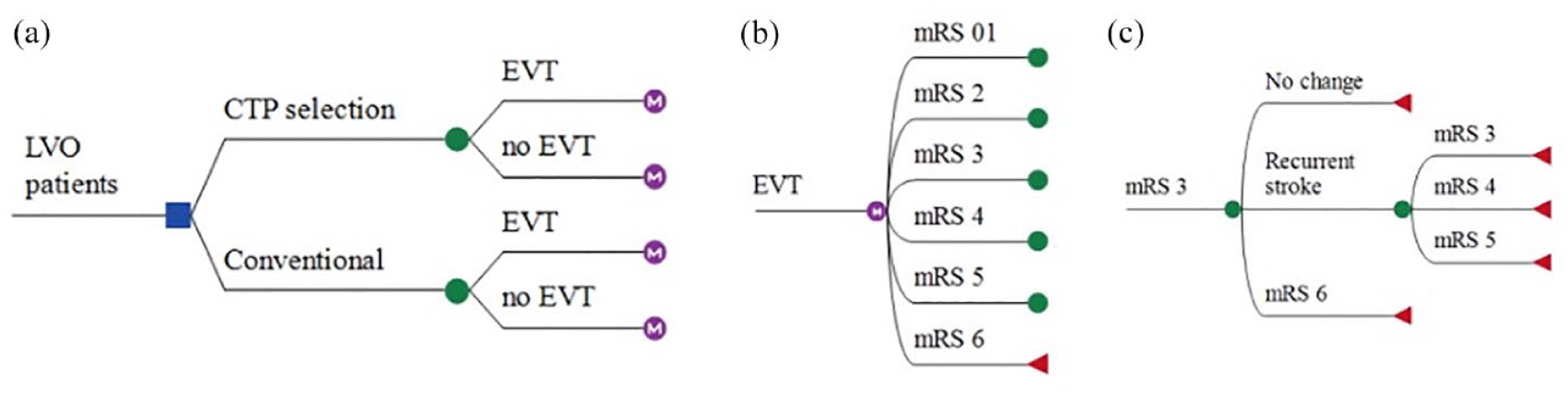
Markov diagram. (a) Patients with large vessel occlusion stroke receive either conventional imaging (NCCT and CTA) or CTP for selection for endovascular treatment. (b) Ninety-day mRS will be simulated for both the EVT and no EVT arms. (c) The yearly mRS will be simulated where mRS can stay unchanged or could change due to recurrent stroke or death (mRS 6). An example is given for a 90-day mRS of 3. The mRS could only stay equal or increase (i.e. worse functional health state).
Effect estimates and their use
Table 2 shows the values and estimated distributions of input parameters extracted from the collected data in the current study and from the literature.
Short-term model: The OR for shift on mRS at 90 days for EVT together with the treatment effect modification of CTP parameters will be used to adjust the observed 90-day mRS values per patient for each diagnostic protocol. If other baseline variables have a statistically significant EVT effect modification, they will also be included in the model.
Thus, based on the observed mRS, treatment allocation (EVT vs best medical management), and observed clinical and CTP measures, the 90-day mRS of a counterfactual treatment allocation can be estimated. In each simulation, four arms will be created: (1) treatment with EVT based on CTP selection, (2) no EVT based on CTP selection, (3) treatment with EVT based on conventional diagnostics, and (4) no EVT based on conventional diagnostics.
Long-term model: Based on the mRS of the previous period, yearly death and stroke recurrence will be simulated. For each simulation period, the probability of death by age, year, and gender will be multiplied with a mortality Hazard Rate (HR) based on mRS to compute the death rate per mRS state.15,16 For survivors, the probability of stroke recurrence by year after index stroke is multiplied with age and a mRS-dependent HR to compute the stroke recurrence rate per mRS state. In case of stroke recurrence, the mRS could deteriorate to mRS 5 or stay equal based on probabilities from the MR CLEAN trial control arm. 13 Patients surviving without recurrent stroke remain in the same mRS state.
Costs and QALYs
Per mRS based health state, costs in euros (€) and utility (QALYs) will be computed per year based on previously collected data. 15 A healthcare payer perspective will be adopted for cost computation with reference year 2023. Computations of costs per mRS, healthcare expenses, and QALYs per mRS subscore have been described previously. 17 Acute care costs include: costs for IVT, EVT, CTA and NCCT imaging, and personnel costs. These costs will be derived from the work by Van den Berg et al. 18 Additionally, costs of CTP will be estimated based on expert opinion with the following arguments: 10% additional costs for acute care personnel (€94), €129 for the CTP acquisition, 19 and €20 for the CTP software license costs per patient. Acute care costs will be inflated by 42% to attribute to overhead costs as is the Dutch standard for cost pricing. 20 Similar to previous studies by van Voorst et al. 14 and van den Berg et al., 18 follow-up costs and index hospital admission costs beyond interventions were combined into groups representing costs made in (1) the first year post-stroke, (2) the second year post-stroke, and (3) the third year and onward. These costs include: in-hospital care, outpatient clinic visits, rehabilitation, formal homecare, and long-term institutionalized care. For the third year and onward, follow-up costs are equal to costs of the second year minus the rehabilitation costs, as it was assumed that this care discontinued. For a more fine-grained description of the costs, we refer to previously published data.14,18 The simulated mRS score over a period of 5 years will be used to compute cumulative costs and QALYs discounted to present values with annual compound interest rates of 1.5% and 4%, respectively. 20 Costs of healthcare will increase with forecasted inflation rates adapted from the Dutch Ministry of Health, Welfare, and Sport. 21 Similarly, the present value of historical cost estimates will be adjusted with historical inflation rates to current values. 20
Simulations and sensitivity analyses
Baseline and one-way sensitivity analysis
We will perform a baseline simulation with mean values of the model input parameters (Table 2). Additionally, we will perform one-way sensitivity analyses to analyze the effect of a 10% change of these parameters on outcome measures relative to the baseline simulation.
Probabilistic sensitivity analysis
A probabilistic sensitivity analysis (PSA) will be performed with 10,000 Monte-Carlo resamples of model input parameters from corresponding distributions (Table 2). A second PSA will be performed where each of the 10,000 iterations a cohort of 100 patients is resampled with replacement from the available data; varying the baseline characteristics each iteration. Outcomes of the PSA will be reported as median values with interquartile ranges (IQR). Outcomes of baseline, one-, and two-way sensitivity analyses will be reported as single values.
Specific sensitivity analysis: Treatment effect modification, treatment effect, and decision threshold
Geographical variations in the EVT treatment effect and variations in effect modification by CTP-estimated core volume represent the current statistical uncertainties on the added value of CTP-estimated core volume. Furthermore, different potential core volume decision thresholds for EVT likely affect the cost and health effects of to including CTP for the selection for EVT. Therefore, we will perform PSAs varying the decision threshold, EVT treatment effect, and CTP-estimated core volume EVT effect modification jointly to describe various scenarios. Furthermore, we will perform sensitivity analyses to determine the EVT treatment effect modification by CTP for of all separate data sources used in CLEOPATRA separately. Please also see Figure 2 for all data sources used in CLEOPATRA.
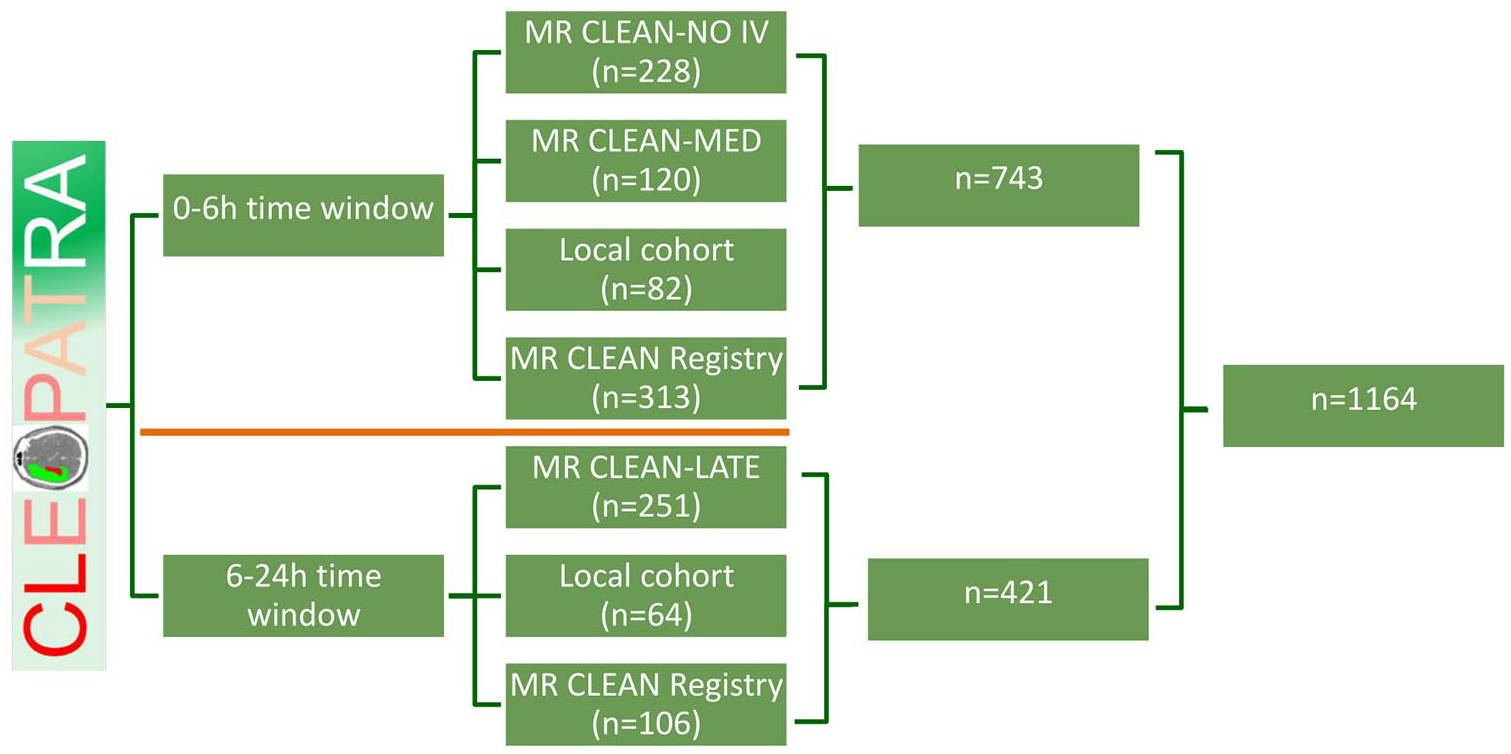
Schematic representation of the data acquisition and overview of total inclusion count per data source until March 1, 2022. Data sources are divided per time window (0–6 h vs 6–24 h) by the orange line.
Outcome measures
Outcome measures will be reported as cumulative values over the simulated 5-year follow-up period. Primary outcome will be the Incremental Cost-Effectiveness Ratio (ICER); which captures the costs (Cintervention and Ccontrol) per QALY (Qintervention and Qcontrol) gained.
The following secondary outcome measures will be reported as cumulative values over the 5-year simulation period: costs and QALYs of CTP based EVT selection (Cintervention; Qintervention) and conventional EVT selection (Ccontrol; Qcontrol), and Net Monetary Benefit (NMB) at a willingness to pay (WTP) of €80,000 per QALY.
Outcomes of the PSA will be reported as median values with interquartile ranges. Outcomes of baseline, and one-way sensitivity analyses will be reported as single values.
CTP processing requirements and quality assessment
CTP data will be processed using Siemens syngo.via (version VB40, Siemens Healthcare, Forchheim, Germany). After processing, all results will be visually checked for obvious artifacts or erroneous core/penumbra volume estimations by two interventional neuroradiologists (>10 and >20 years of experience, respectively). For patients with an estimated CTP ischemic core volume >30 ml, manual artifact removal for the at the most cranial and caudal (i.e. at the level of the skull base) slices of the CTP scan is allowed. The used CTP processing software and CTP acquisition requirements are provided in Table O1 and Table O2 of the Supplemental Material.
Recorded baseline parameters
Age, sex, pre-stroke mRS, NIHSS at baseline, IVT administration, interhospital transfer, time from onset to imaging, time from onset to groin puncture, ASPECTS, collateral status, previous stroke, hypertension, history of diabetes mellitus, history of atrial fibrillation, previous myocardial infarction, blood pressure, weight, and height.
Other study parameters
Procedural data recorded in the context of the MR CLEAN Registry, MR CLEAN-NO IV, MR CLEAN-MED, and MR CLEAN-LATE trials include: time from symptom onset to ER or the time since last seen well to ER, time of arrival at the emergency department, and time of CTP acquisition. We recorded if – and when – patients received intravenous alteplase (IVT). Additionally, the time of groin puncture, time of first attempt, time of any additional attempts, time of recanalization, and time of end of procedure were noted.
Data monitoring body
Not required due to the retrospective nature of CLEOPATRA.
Trial status
As of this writing, a total of 17 centers have been initiated in the Netherlands. The first patient was included in November 2019. A full list of participating centers can be found at the study registration page (https://www.zorgevaluatienederland.nl/cleopatra). Until March 2022, 1164 patients have been included (743 in the 0–6 h time window vs 421 in the 6–24 h time window). For a schematic overview of all data sources and number of inclusions per data source, please see Figure 2 or visit the study registration page. The current article is based on protocol version 1.0 dating from May 2021.
Data management
All CLEOPATRA data will be entered into Castor EDC, a web-based database. Subject records will be coded by a unique study number. The local investigators will keep a list shadowing codes and names. Unique documents with identifying information will be stored separately from the study database in digital files, categorized by study number on a secure drive system, only accessible to the study coordinator. For patients with missing 90-day mRS data, the 90-day mRS score will be imputed with single imputation to obtain reliable population mRS estimates. Multiple imputation will also be performed for optimization of the prediction models. The database will be closed within 3 months after the last scheduled follow-up date of the last included patient. The end of the study is defined as the last data entry of the last patient. The investigator will publish the results of the study within 1 year after the end of the study.
Discussion
The costs and health effects of including CTP to the selection for EVT have rarely been studied. 17 Although CTP core volume is associated with clinical outcome after 3 months in the earlier time window, no EVT treatment effect modification by CTP could be established.
CLEOPATRA is a nationwide, multicenter healthcare evaluation study aiming to obtain parameter estimates as input for a cost-effectiveness model. For both earlier and later time windows, we will calculate the effect of including CTP-estimated core volume as selection criterion for EVT in AIS patients compared to the conventional diagnostic workup in terms of costs and health effects in the Dutch healthcare system. By adapting these input variables to local circumstances, the model could also be used to estimate cost-effectiveness in other regions.
Summary and conclusions
CLEOPATRA aims to estimate the costs and health effects of using CTP for EVT selection in AIS patients compared to conventional EVT selection in the earlier and later time windows in the setting of the Dutch healthcare system.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221092535 – Supplemental material for Cost-effectiveness of CT perfusion for patients with acute ischemic stroke (CLEOPATRA)-Study protocol for a healthcare evaluation study
Supplemental material, sj-docx-1-eso-10.1177_23969873221092535 for Cost-effectiveness of CT perfusion for patients with acute ischemic stroke (CLEOPATRA)-Study protocol for a healthcare evaluation study by Miou S Koopman, Jan W Hoving, Henk van Voorst, Jasper D Daems, Daan Peerlings, Erik Buskens, Hester F Lingsma, Henk A Marquering, Hugo WAM de Jong, Olvert A Berkhemer, Wim H van Zwam, Marianne AA van Walderveen, Ido van den Wijngaard, Aad van der Lugt, Diederik WJ Dippel, Albert J Yoo, Bruce CV Campbell, Wolfgang G Kunz, Charles BLM Majoie and Bart J Emmer in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873221092535 – Supplemental material for Cost-effectiveness of CT perfusion for patients with acute ischemic stroke (CLEOPATRA)-Study protocol for a healthcare evaluation study
Supplemental material, sj-docx-2-eso-10.1177_23969873221092535 for Cost-effectiveness of CT perfusion for patients with acute ischemic stroke (CLEOPATRA)-Study protocol for a healthcare evaluation study by Miou S Koopman, Jan W Hoving, Henk van Voorst, Jasper D Daems, Daan Peerlings, Erik Buskens, Hester F Lingsma, Henk A Marquering, Hugo WAM de Jong, Olvert A Berkhemer, Wim H van Zwam, Marianne AA van Walderveen, Ido van den Wijngaard, Aad van der Lugt, Diederik WJ Dippel, Albert J Yoo, Bruce CV Campbell, Wolfgang G Kunz, Charles BLM Majoie and Bart J Emmer in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BJE reports grants from LtC (ZonMW and TKI-PPP of Health Holland). WHvZ reports personal fees from Codman and from Stryker. DWJD and AvdL report grants from the Dutch Heart Foundation, AngioCare, Medtronic/Covidien/EV3, MEDAC/LAMEPRO, Penumbra, Top Medical/Concentric, Stryker, and Cerenovus; consultation fees from Stryker, Bracco Imaging, and Servier, received by the Erasmus University Medical Centre outside this project. CBLMM reports grants from TWIN, during the conduct of the study and grants from CVON/Dutch Heart Foundation, European Commission, Dutch Health Evaluation Program, and from Stryker outside this project (paid to institution) and is shareholder of Nicolab. HAM is co-founder and shareholder of Nicolab. All other contributors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Leading the Change (LtC) (grant number 80-85009-98-2015). LtC is financed by “Health Insurers Netherlands” (Dutch: “Zorgverzekeraars Nederland”) and supports various healthcare evaluations in The Netherlands as part of the “Healthcare Evaluation Netherlands” (Dutch: “Zorgevaluatie Nederland”). LtC was not involved in the design of the project, but progress of the project is continuously monitored by LtC. The MR CLEAN Registry was funded and carried out by the Erasmus University Medical Center, the Academic Medical Center Amsterdam, and the Maastricht University Medical Center. The MR CLEAN Registry was additionally funded by the Applied Scientific Institute for Neuromodulation (TWIN). The CONTRAST consortium is supported by Netherlands Cardiovascular Research Initiative, an initiative of the Dutch Heart Foundation, by the Brain Foundation Netherlands and powered by Health~Holland, Top Sector Life Sciences and receives unrestricted funding from Medtronic and Cerenovus. The primary trial sponsor of CLEOPATRA is Amsterdam UMC, location AMC, Meibergdreef 9, 1105 AZ Amsterdam, The Netherlands.’
Ethical approval
The CLEOPATRA study protocol was reviewed by the Medical Ethical Committee Board of the Amsterdam University Medical Centers, location AMC, and informed consent was waived (reference: W19_281#19.334) as retrospective, observational studies do not fall under the scope Medical Research Involving Human Subject Act (WMO). CLEOPATRA is conducted according to the principles of the Declaration of Helsinki (October 2013), according to ICH-GCP principles and in accordance with the Medical Research Involving Human Subjects Act (WMO).
Informed consent
All patients included in the MR CLEAN-NO IV, MR CLEAN-MED, and MR CLEAN-LATE have given written informed consent in the context of the study concerned. The patients or their representatives had been given the opportunity to refuse participation. In this written explanation, patients or their representatives are informed that the acquired data can be re-used by future studies. Patients included in the MR CLEAN Registry were provided with a written explanation of the study. The patients or their representatives will be given the opportunity to refuse participation in the MR CLEAN Registry. In that case, all data will be deleted from the database and clinical material will be destroyed. Patients included in our local cohort were not be asked to give written consent, since it was expected that a large part of the patients would have deceased or would not be reachable by mail. Data of patients included in our local cohort were entered fully anonymously in the study base. This was decided in consultation with the legal consultant of the Amsterdam University Medical Centers, location AMC. Model consent forms from the data sources used in CLEOPATRA can be accessed on ![]() .
.
Guarantor
BJE.
Author contributions
MSK, JWH, HvV, BJE, CBLMM, EB, HFL, and DWJD were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. MSK, JWH, and HvV wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Authorship
Trial registration
Data availability
Individual patient data cannot be made available under Dutch law since we did not obtain patient approval for sharing individual patient data. All syntax files and output of statistical analyses are available on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
