Abstract
Introduction:
Developing an effective stroke prevention strategy is crucial for elderly atrial fibrillation (AF) patients with dementia. This is due to the limited and inconsistent evidence available on this topic. In this nationwide, population-based cohort study, we aim to compare the effectiveness and safety of direct oral anticoagulants (DOACs) and warfarin in AF patients with dementia.
Patients and methods:
We identified AF patients with dementia, aged 50 years or older, from Taiwan’s National Health Insurance Research Database between 2010 and 2019. The primary outcome was a composite of hospitalizations due to ischemic stroke, acute myocardial infarction, intracranial hemorrhage, or major bleeding, as well as all-cause mortality. We used 1:1 propensity score matching and Cox proportional hazard models to adjust for confounding factors when comparing outcomes between warfarin and DOAC (apixaban, dabigatran, edoxaban, or rivaroxaban) users or warfarin and each individual DOAC.
Results:
There were 2952 patients in the DOAC-warfarin matched cohort. The apixaban-, dabigatran-, edoxaban-, and rivaroxaban-warfarin matched cohorts had 2346, 2554, 1684, and 2938 patients, respectively. The DOAC group, when compared to warfarin, was associated with a lower risk of both the composite outcome (hazard ratio (HR), 0.81; 95% confidence interval (CI) 0.69–0.95) and ischemic stroke (HR 0.65; 95% CI 0.48–0.87). Apixaban (HR 0.79; 95% CI 0.66–0.94), dabigatran (HR 0.64; 95% CI 0.53–0.77), and rivaroxaban (HR 0.82; 95% CI 0.70–0.97) were also associated with a lower risk of the composite outcome.
Discussion and conclusion:
Compared to warfarin, DOACs, whether as a group or apixaban, dabigatran, or rivaroxaban individually, were associated with a reduced risk of the composite outcome in elderly patients with concurrent AF and dementia.

Introduction
Atrial fibrillation (AF) is the most common clinically significant cardiac arrhythmia, and its prevalence is increasing worldwide. 1 It is associated with a fivefold increased risk of ischemic stroke. 2 As a result, according to major guidelines,3–5 most AF patients may require lifelong use of oral anticoagulants (OACs) to reduce the risk of stroke. However, long-term treatment with OACs is usually complicated with intracranial hemorrhage (ICH), other major bleeding, or even death. This is especially true for elderly AF patients with dementia.6–10
The prevalence of AF rises with increasing age, 11 a factor inevitably linked to dementia. Furthermore, AF is associated with a more than twofold risk of dementia, 12 making it common in dementia patients. 8 However, even when diagnosed with AF, patients with dementia or cognitive impairment are less likely to start OAC treatment. 13 Only around a quarter of dementia patients with AF receive OACs. 8 A meta-analysis reveals that OAC usage reduces mortality in AF patients with dementia, although it does not affect the risk of ischemic stroke or major bleeding. 14 Thus, developing an effective stroke prevention strategy is essential for elderly AF patients with dementia.
OACs include traditional vitamin K antagonists like warfarin and direct oral anticoagulants (DOACs). DOACs, such as direct thrombin inhibitor dabigatran and factor Xa inhibitors, including apixaban, edoxaban, and rivaroxaban, have demonstrated similar or superior effectiveness to warfarin in preventing stroke or systemic embolism (SSE), with less major bleeding. 15 However, dementia patients were substantially under-represented in the large DOAC trial, 16 making the evidence for the effectiveness of DOACs in AF patients with dementia less clear.17–20
So far, very few retrospective observational studies have compared the effectiveness and safety of DOACs and warfarin in elderly AF patients with dementia.21,22 Their results were inconsistent and did not include data for edoxaban, the most recent DOAC on the market. It is crucial to investigate further the benefits of DOACs over warfarin for this particular group of patients. 23 Therefore, we conducted a nationwide, population-based cohort study to compare the effectiveness and safety of DOACS versus warfarin in AF patients with dementia.
Methods
Data source
The study adopted a retrospective population-based cohort design to analyze data from the Taiwan National Health Insurance Research Database (NHIRD) from 2009 to 2020. Taiwan’s National Health Insurance program started in 1995. By June 2009, it had contracted with 97% of medical providers, providing coverage for around 23 million beneficiaries, over 99% of the population. The NHIRD houses several data types from its beneficiaries, encompassing demographic information, service records, expenditure claims, and data from contracted pharmacies for reimbursement purposes.
All diagnoses in the NHIRD were coded using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) until the end of 2015. From 2016 onward, the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) was used instead. 24 Patient privacy is maintained by encoding personal information and allocating anonymous identification numbers, which are used for data linkage. This database has been extensively documented and has served as a basis for numerous epidemiological studies published in peer-reviewed academic journals. 25 The study was approved by the Institutional Review Board of Ditmanson Medical Foundation Chia-Yi Christian Hospital (CYCH-IRB No: 2022009). Written informed consent was not required as all patient data from the NHIRD are anonymized.
Study population
We identified AF patients with dementia, aged 50 years or older, from the NHIRD between 2010 and 2019. AF was identified by ICD-9-CM code 427.31, and ICD-10-CM codes I48.0 to I48.2 or I48.9.26,27 Dementia was recognized by ICD-9-CM codes 290.0–290.4, 294.1, 331.0, 331.1, 331.2, and ICD-10-CM codes F01, F02, F03, G30, G31.0, G31.1. 28 A diagnosis was confirmed when relevant codes appeared in at least two outpatient department visits or at least once during hospitalization. The index date, defined as the start of OAC therapy following the diagnosis of AF and dementia, was noted for each patient.
Patients were excluded if they met any of the following criteria: (1) missing or invalid sex information, (2) valve replacement or valvuloplasty, (3) valvular heart disease, (4) end-stage renal disease, (5) OAC treatment within 365 days before AF diagnosis, and (6) OAC treatment for less than 90 days.
Baseline characteristics and comorbidities
We extracted patient information, including sex, age, comorbidities, and medications, from the NHIRD. Comorbidities were identified by finding corresponding diagnostic codes in at least one inpatient or two outpatient diagnoses recorded within 1 year before the index date.
The comorbidities included stroke, systemic embolism, myocardial infarction, peripheral artery disease, heart failure, hypertension, diabetes mellitus, renal failure, liver disease, parkinsonism, hypothyroidism, hyperthyroidism, chronic obstructive pulmonary disease, cancer, coronary artery disease, depression, and epilepsy (Supplemental Table 1). The medications taken into account included aspirin, clopidogrel, angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, statins, beta-blockers, class I or III antiarrhythmics, digoxin, diuretics, calcium channel blockers, antiepileptics, anti-parkinsonism drugs, antipsychotics, anxiolytics, hypnotics and sedatives, antidepressants, thyroid preparations, and antithyroid preparations.
The CHA2DS2-VASc (congestive heart failure, hypertension, age ⩾75 years, diabetes, stroke or transient ischemic attack, vascular disease, age 65–74 years, and sex category) score 29 was used to compute the risk of ischemic stroke risk. On the other hand, the modified HAS-BLED (hypertension, abnormal liver and/or kidney function, stroke history, bleeding history or predisposition, labile international normalized ratio, elderly, drug and alcohol use concomitantly) score 30 was used to calculate the risk of major bleeding.
Outcomes
The primary outcome was a composite outcome of hospitalizations due to ischemic stroke (ICD-9-CM codes 433 or 434, and ICD-10-CM code I63), acute myocardial infarction (ICD-9-CM code 410 and ICD-10-CM code I21), intracranial hemorrhage (ICD-9-CM codes 430–432, and ICD-10-CM codes I60 to I61), or major bleeding (Supplemental Table 2), as well as all-cause mortality. Secondary outcomes included individual elements of the primary outcome. All patients were traced from the day after the index date until they experienced the outcome, changed between oral anticoagulants, died, left the National Health Insurance program, or reached the end of the database (December 31, 2020), whichever came first.
Statistical analysis
We used 1:1 propensity score matching to adjust for confounding factors. A logistic regression analysis estimated the propensity score, modeling the probability of initiating warfarin versus DOACs (apixaban, dabigatran, edoxaban, or rivaroxaban) or each individual DOAC based on baseline characteristics (Table 1). We used t-tests and Chi-squared tests to examine the distribution of patient characteristics, comorbidities, and medications across different treatment pairs. Both crude and multivariable-adjusted hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated using Cox proportional hazard regression models. We conducted subgroup analyses to assess the impact of factors such as age, sex, history of stroke, and prior antiplatelet use on the outcome.
Characteristics of the warfarin-DOACs matched cohort.
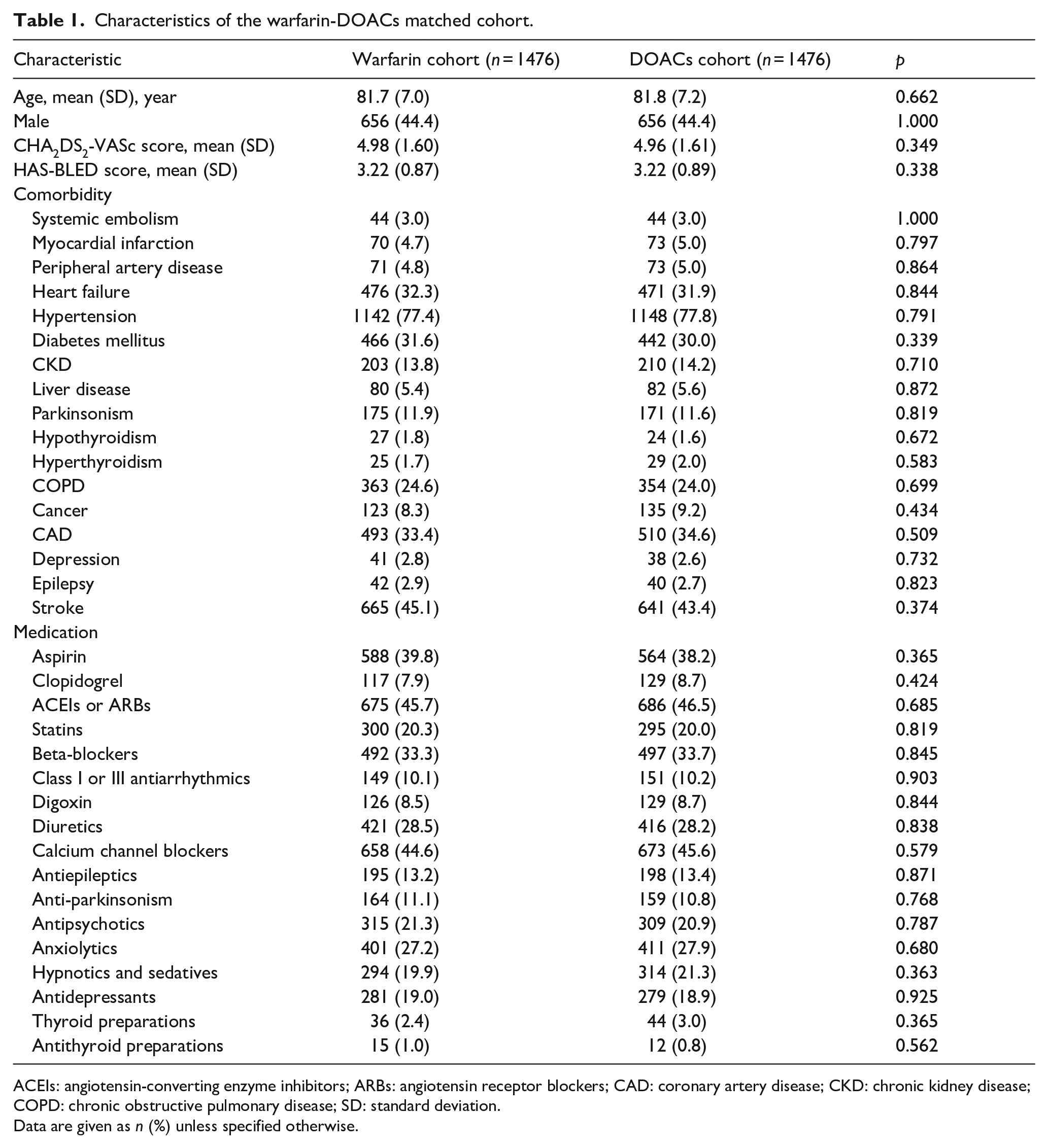
ACEIs: angiotensin-converting enzyme inhibitors; ARBs: angiotensin receptor blockers; CAD: coronary artery disease; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; SD: standard deviation.
Data are given as n (%) unless specified otherwise.
Statistical analyses were conducted using SAS 9.4 for Windows (SAS Institute, Inc., Cary, NC, USA). A two-tailed p-value less than 0.05 was considered statistically significant.
Results
From the NHIRD, a total of 16,362 patients aged 50 years or older with concurrent AF and dementia were identified. After excluding those who did not meet the study criteria (Figure 1), the study population comprised 6993 patients. Of these, 1477 patients were on warfarin, while 5516 patients used DOACs. The baseline characteristics before propensity score matching are presented in Supplemental Table 3.
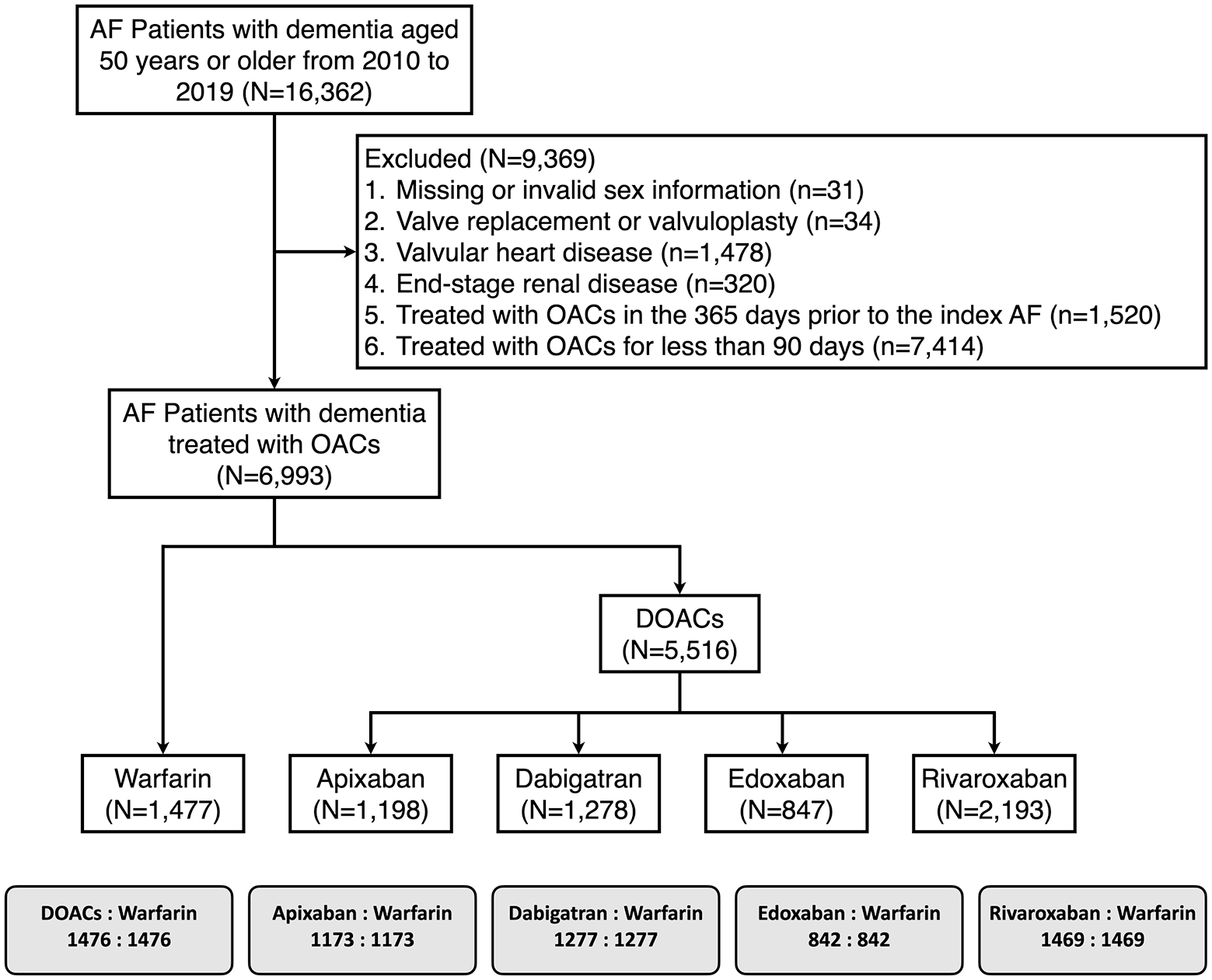
Flowchart of the study population.
After conducting propensity score matching, we obtained five cohorts for our final data analysis. These include DOAC-warfarin, apixaban-warfarin, dabigatran-warfarin, edoxaban-warfarin, and rivaroxaban-warfarin, consisting of 2952, 2346, 2554, 1684, and 2938 patients, respectively. Table 1 shows the characteristics of the warfarin-DOACs cohort after propensity matching. The baseline characteristics were similar between the warfarin and DOAC matched cohorts (Table 1). The characteristics of the matched cohorts between warfarin and individual DOAC are listed in Supplemental Tables 4 to 7.
Supplemental Table 8 and Figure 2(a) display crude event numbers, crude incidence rates, unadjusted and multivariable-adjusted HRs, and 95% CIs. Compared to warfarin, the group treated with DOACs showed significantly lower risks for both the composite outcome (HR, 0.81 [95% CI, 0.69–0.95]) and ischemic stroke (HR, 0.65 [95% CI, 0.48–0.87]). Risks for acute myocardial infarction, ICH, major bleeding, and all-cause mortality were similar in both treatment groups.
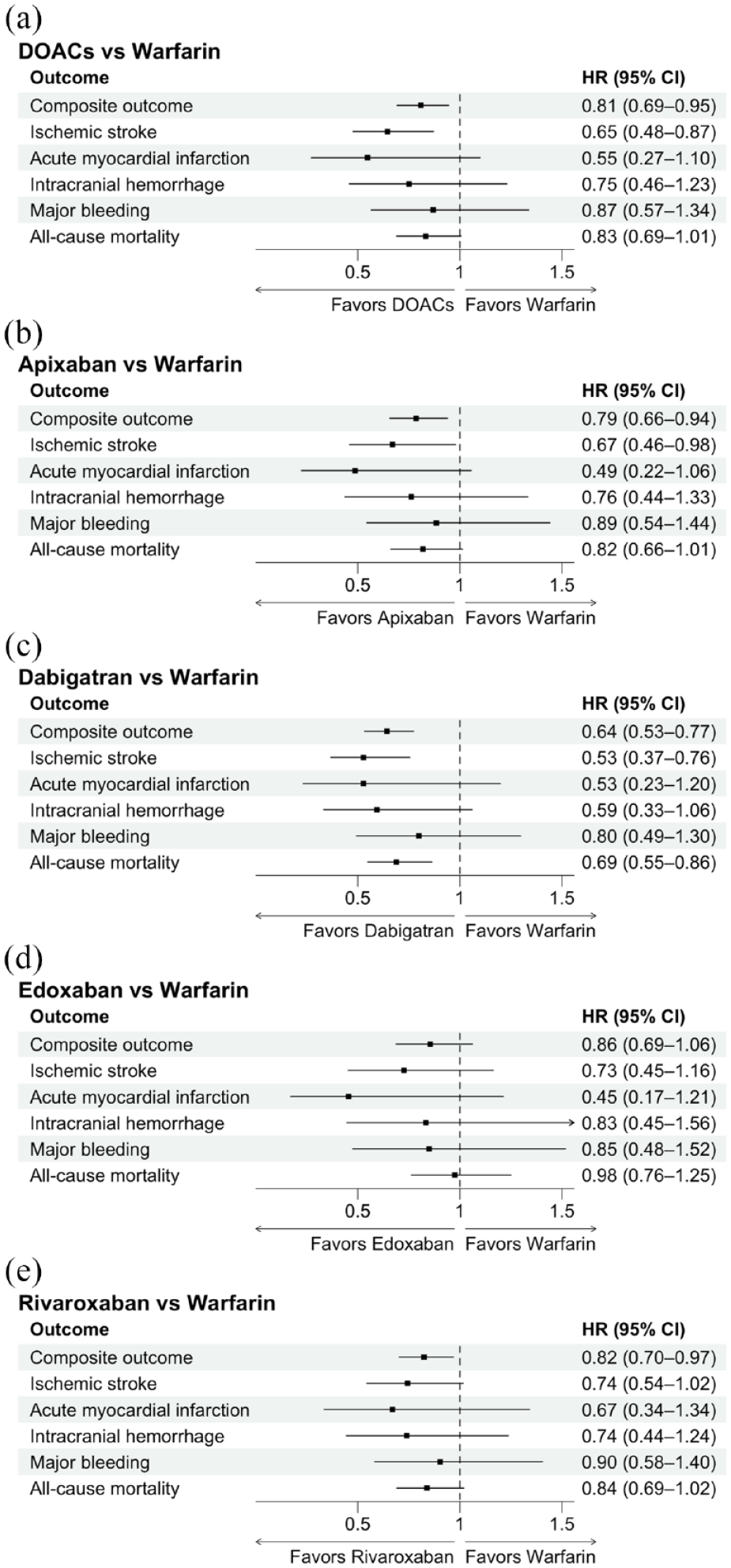
Forest plots for outcomes between oral anticoagulant treatment-matched cohorts, including direct oral anticoagulants versus warfarin (a), apixaban versus warfarin (b), dabigatran versus warfarin (c), edoxaban versus warfarin (d), and rivaroxaban versus warfarin (e).
Supplemental Tables 9–12 list crude event numbers, crude incidence rates, unadjusted and multivariable-adjusted HRs, and 95% CIs for the apixaban-, dabigatran-, edoxaban-, and rivaroxaban-warfarin matched cohorts, respectively. Apixaban (HR, 0.79 [95% CI, 0.66–0.94]), dabigatran (HR, 0.64 [95% CI, 0.53–0.77]), and rivaroxaban (HR, 0.82 [0.70–0.97]) were associated with lower risks than warfarin for the composite outcome (Figure 2(b), (c) and (e)). However, edoxaban (HR, 0.86 [0.69–1.06]) showed a risk comparable to warfarin (Figure 2(d)). Regarding the individual components of the outcome, dabigatran had lower risks for ischemic stroke (HR, 0.53 [95% CI, 0.37–0.76]) and all-cause mortality (HR, 0.69 [95% CI, 0.55–0.86]) than warfarin. Apixaban also exhibited a lower risk for ischemic stroke (HR, 0.67 [95% CI, 0.46–0.98]) than warfarin.
Figure 3 shows the results of the subgroup analysis. No significant interaction was seen in the effect of DOACs versus warfarin on our primary or secondary outcomes across different subgroups based on age, sex, history of stroke, and prior antiplatelet use. For the composite outcome (Figure 3(a)), DOACs were associated with a significantly lower risk than warfarin in patients under 75 years old, women, and those with a history of stroke or prior antiplatelet use. DOACs were also linked to a significantly lower risk of ischemic stroke in both younger (50–75 years) and older (75 years and over) patients, women, and those with a history of stroke or prior antiplatelet use (Figure 3(b)). The risk of major bleeding was significantly lower with DOACs than warfarin in younger patients (Figure 3(e)). Meanwhile, DOACs were associated with a significantly lower risk of all-cause mortality in men (Figure 3(f)).
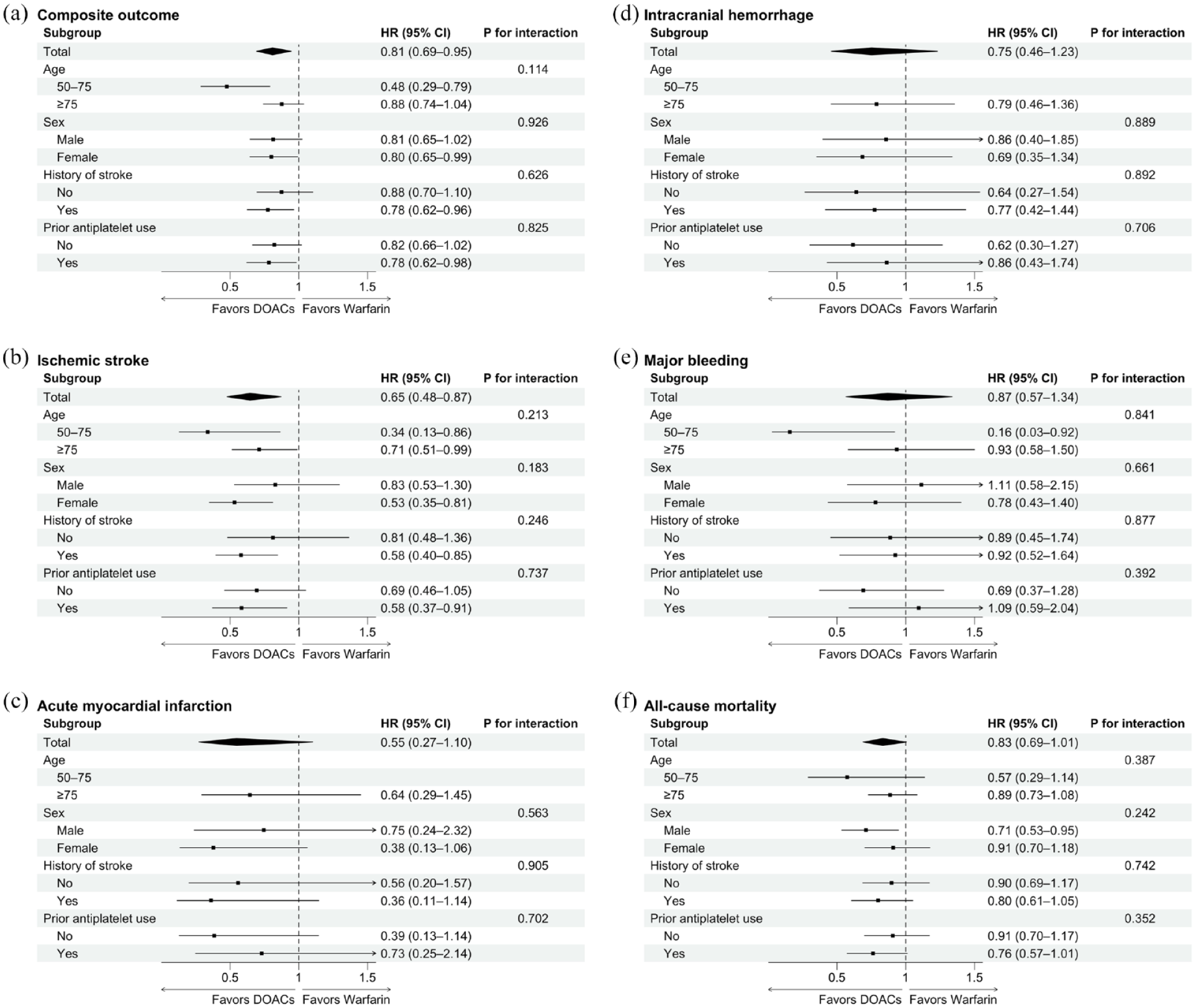
Forest plots for outcomes between direct oral anticoagulants and warfarin, including the composite outcome (a), ischemic stroke (b), acute myocardial infarction (c), intracranial hemorrhage (d), major bleeding (e), and all-cause mortality (f). Due to the small event number, a multivariable adjusted hazard ratio was not available for acute myocardial infarction and intracranial hemorrhage in the younger (50–75 years) subgroup.
Discussion
In elderly patients with AF and dementia, DOACs were linked to a 19% and 35% reduction in the risks of the composite outcome and ischemic stroke, respectively, compared to warfarin. Individual DOACs, such as apixaban, dabigatran, and rivaroxaban, were associated with a lower risk of the composite outcome. Edoxaban, however, had a statistically similar risk for the composite outcome compared to warfarin. Importantly, apixaban and dabigatran were linked to a lower risk of ischemic stroke than warfarin, and dabigatran also showed a lower risk of all-cause mortality compared to warfarin.
DOACs, which require less strict adherence than warfarin, seem to be a more suitable choice for elderly patients with concurrent AF and dementia. This group is known for low medication adherence, and DOACs could potentially offer better clinical outcomes than warfarin. However, the literature search found only three articles that addressed this issue,6,21,22 and their results showed significant heterogeneity. 14
A UK population-based study of 2399 patients with AF and dementia found that DOAC-treated patients had a significantly reduced risk of ICH but significantly increased risks of gastrointestinal bleeding and all-cause mortality compared to those treated with warfarin. 22 A population-based study using data from three large US claims databases included more than 90,000 people with AF and dementia. 21 However, no direct comparison was made between users of warfarin and those of DOACs as a group.
Finally, in a study recruiting 1064 elderly AF patients taking OAC treatment, cognitive impairment was found to be independently linked to major bleeding. There was no significant difference observed between individuals taking warfarin or DOACs, 6 indicating no benefits of DOACs compared to warfarin. Hence, current research does not conclusively favor DOACs. More evidence is needed to compare the use of DOACs and warfarin in this vulnerable population.
Our study addresses this knowledge gap and suggests that compared to warfarin, DOACs may reduce the composite outcome of ischemic stroke, acute myocardial infarction, intracranial hemorrhage, major bleeding, or all-cause mortality. This beneficial result appears to be primarily due to a decreased risk of ischemic stroke. Hence, DOACs may be a preferable choice over warfarin for AF patients with dementia, as they could improve ischemic stroke prevention and clinical outcomes.
We discovered that apixaban and dabigatran, unlike edoxaban and rivaroxaban, were linked to a reduced risk of ischemic stroke compared to warfarin. These findings align with a study that examined the relationship between DOAC concentration and clinical outcomes. 31 The study found that, in general AF patients, a low trough blood concentration of DOACs predicted SSE. It also found that once-daily DOAC dosing (edoxaban and rivaroxaban) had more than a three-fold risk of low trough concentration than twice-daily dosing (apixaban and dabigatran). Missing a single dose equates to zero exposure to DOAC in a day, 32 which could further lower the trough concentration and endanger the continuity of anticoagulation action, 33 resulting in an increased risk of SSE.
In the large US population-based study mentioned above, 21 Lin et al. compared the risk of various outcomes between matched cohorts of warfarin-apixaban, dabigatran-apixaban, and rivaroxaban-apixaban pairs. They found that, compared to warfarin, apixaban was associated with significantly reduced risks of the composite outcome (ischemic stroke or major bleeding), major bleeding, and all-cause mortality in AF patients with dementia. Apixaban was also associated with significantly lower risks of the composite outcome and major bleeding compared to dabigatran or rivaroxaban. By pooling the results from the three claims databases in this US study and our own results using the random-effects meta-analyses, we found that apixaban users had significantly lower risks of ischemic stroke, major bleeding, and all-cause mortality than warfarin users (Supplemental Figure 1).
Notably, edoxaban demonstrated a similar risk of ischemic stroke and the composite outcome as warfarin according to our real-world data. The Edoxaban Low-Dose for Elder Care Atrial Fibrillation Patients trial 34 found that a very low dose of edoxaban (15 mg daily) was more effective than a placebo (not warfarin) at reducing the risk of SSE in elderly patients who could not tolerate standard dosing anticoagulants. This could lead physicians to prescribe a similarly low dose of edoxaban to elderly AF patients with dementia in real-world practice. Nevertheless, we cannot exclude the possibility that the smaller number of participants may have reduced the statistical power needed to detect true differences.
Despite dementia patients with AF being less likely to start, adhere to, or continue using OACs,13,35,36 a 39.8% increase in OAC use was observed from 2000 to 2015. 23 This increase can be attributed to the growing usage of DOACs instead of warfarin. 23 Our findings support this practice, as DOACs were associated with a lower risk of ischemic stroke and all-cause mortality in AF patients with dementia, compared to warfarin. This correlation remained consistent across different age groups, sexes, and regardless of a history of prior stroke or prior antiplatelet use. Therefore, having dementia should not deter AF patients from receiving guideline-directed DOAC treatment.
Selecting a DOAC for this specific population is challenging due to limited evidence on which DOAC is more suitable. Our study suggests that apixaban, dabigatran, and rivaroxaban are associated with a lower risk of the composite outcome. Additionally, our findings, along with Lin et al.′s study, support the use of apixaban in elderly AF patients with dementia. However, the current study did not make direct comparisons between individual DOACs, so our findings cannot guide the choice of DOACs. Given the existing evidence, more studies are needed to investigate the optimal DOAC treatment in AF patients with dementia.
This study had several limitations. First, since edoxaban was the most recent DOAC to enter the market, its sample size was smaller than other individual DOAC-warfarin cohorts (Figure 1). This could have resulted in insufficient statistical power to detect true differences in outcomes between edoxaban and warfarin users. Second, despite being a nationwide, population-based study, the sample size remained relatively small after propensity score matching. This limitation prevented further exploration of the outcomes between standard and reduced doses of each DOAC compared to warfarin.
Third, while propensity score matching was used to balance the baseline characteristics between DOACs and warfarin cohorts, the impact of unmeasured confounders, such as creatinine clearance, cannot be completely ruled out. Fourth, since this study was based on claims data, we did not control for patient compliance, anticoagulant doses, and the international normalized ratio for warfarin users, although these are important determinants of the anticoagulation effect. Finally, patients with dementia were identified using diagnostic codes. We cannot exclude the possibility of case misclassification. However, a systematic review indicates that using diagnostic codes to identify dementia cases has a high positive predictive value and reasonable sensitivity. 28
Conclusions
Compared to warfarin, DOACs, whether as a group or apixaban, dabigatran, or rivaroxaban individually, were associated with a lower risk of the composite outcome, including ischemic stroke, acute myocardial infarction, intracranial hemorrhage, major bleeding, and all-cause mortality, in elderly patients with concurrent AF and dementia. Given the increasing incidences of both AF and dementia in our aging society, further studies are needed to confirm these observations in this specific population.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241274598 – Supplemental material for Comparative effectiveness and safety of direct oral anticoagulants and warfarin in atrial fibrillation patients with dementia
Supplemental material, sj-docx-1-eso-10.1177_23969873241274598 for Comparative effectiveness and safety of direct oral anticoagulants and warfarin in atrial fibrillation patients with dementia by Chen-Wen Fang, Cheng-Yang Hsieh, Hsin-Yi Yang, Ching-Fang Tsai and Sheng-Feng Sung in European Stroke Journal
Footnotes
Acknowledgements
The authors thank the help from the Health and Welfare Data Science Center of Taiwan’s Ministry of Health and Welfare for maintaining and processing the data and facilitating the extraction of data. This study is based on data from the National Health Insurance Research Database provided by Taiwan’s Ministry of Health and Welfare. The interpretation and conclusions contained herein do not represent the position of Taiwan’s Ministry of Health and Welfare.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science and Technology Council [grant number NSTC 112-2221-E-705-001-MY2].
Ethical approval
Ethical approval for this study was obtained from the Institutional Review Board of Ditmanson Medical Foundation Chia-Yi Christian Hospital (approval number: 2022009).
Informed consent
Informed consent was not sought for the present study because the Institutional Review Board of Ditmanson Medical Foundation Chia-Yi Christian Hospital waived the requirement for informed consent as all patient data from the National Health Insurance Research Database are anonymized.
Guarantor
S.F.S.
Contributorship
C.W.F, C.Y.H., and S.F.S conceived the study. H.Y.Y. and C.F.T. acquired the study data. H.Y.Y. and C.F.T. performed statistical analysis. C.W.F. and C.Y.H. drafted the manuscript. S.F.S. revised the manuscript. All authors reviewed and approved the final manuscript.
Trial registration
Not applicable because this study was not a clinical trial.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
