Abstract
Introduction:
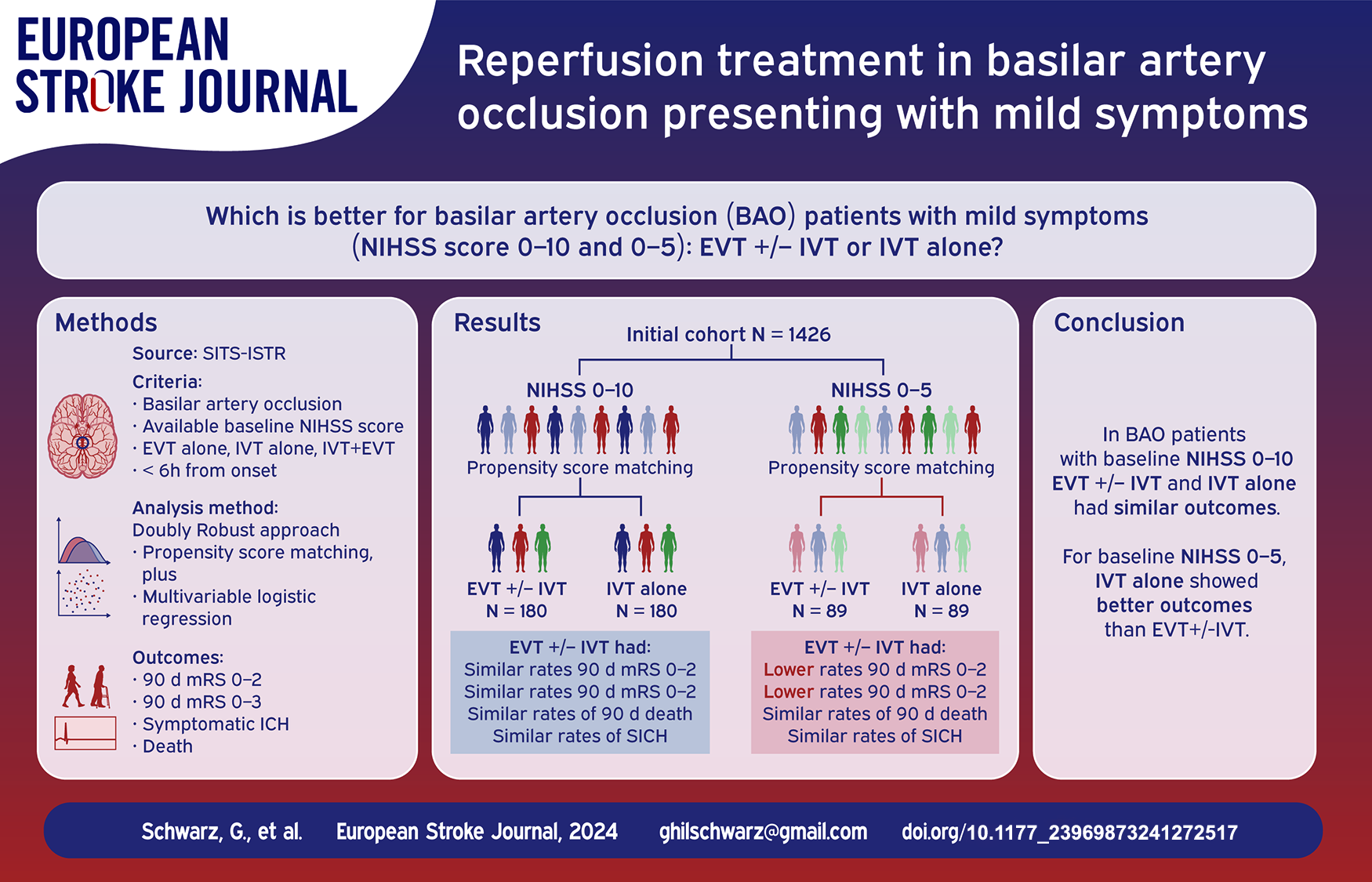
Endovascular treatment (EVT) improves outcomes for basilar artery occlusion (BAO) with moderate-to-severe symptoms. However, the best treatment for mild symptoms (NIHSS score 0–10 and 0–5) remains unclear. This study compared EVT ± IVT to IVT alone in BAO patients with mild symptoms.
Patients and Methods:
From the SITS-International Stroke Treatment Register, we included BAO patients with available baseline NIHSS score, treated by EVT, IVT, or both within 6 h of symptom onset from 2013 to 2021. Using the Doubly Robust approach (propensity score matching plus multivariable logistic regression), we analyzed efficacy (3-month mRS) and safety (SICH and 3-month death) outcomes for EVT ± IVT versus IVT alone in BAO patients with NIHSS scores 0–10 and 0–5.
Results:
1426 patients were included. For NIHSS scores 0–10 (180 matched, 1:1 ratio), outcomes were similar between EVT ± IVT and IVT alone groups. For NIHSS scores 0–5 (89 matched, 1:1 ratio), EVT ± IVT was associated with worse outcomes compared to IVT alone (mRS 0–2, aOR 0.20 [95% CI 0.06–0.61]; p = 0.005; mRS 0–3, aOR 0.27 [95% CI 0.08–0.89]; p = 0.031), but safety outcomes were similar.
Discussion:
In early-treated BAO patients with mild symptoms, defined as NIHSS 0–10, there were no significant differences in outcomes between EVT ± IVT and IVT alone. However, for very mild symptoms, defined as NIHSS 0–5, IVT alone was associated with better outcomes compared to EVT ± IVT.
Introduction
Basilar artery occlusion (BAO) is potentially one of the most devastating neurological conditions, with high risks of disability and mortality.1–3 Endovascular thrombectomy (EVT) has become the standard of care for large vessel occlusion (LVO) in the anterior circulation; however, the value of EVT in BAO patients had remained uncertain because these patients were excluded from most pivotal endovascular thrombectomy trials.4–7 The Basilar Artery Occlusion Endovascular Intervention versus Standard Medical Treatment (BEST) trial 8 and the Basilar Artery International Cooperation Study (BASICS) trial, 9 did not show a clinical benefit from EVT (vs best medical management [BMM]) in early-presenting BAO patients. Two more recent trials, ATTENTION (Endovascular Treatment For Acute Basilar Artery Occlusion: A Multicentre Randomized Clinical Trial) 10 and BAOCHE (Basilar Artery Occlusion Chinese Endovascular Trial) 11 reported better outcomes with EVT than with BMM for BAO patients with moderate-to-severe deficit, up to 24 h from stroke onset. Therefore, EVT should be considered for BAO patients presenting with non-mild symptoms. BAO strokes typically present with moderate-to-severe symptoms. However—due to better collateral supply and lower thrombus load—some patients only have mild symptoms. Patients with low baseline NIHSS scores were excluded from both the ATTENTION (NIHSS < 10 excluded) and the BAOCHE (NIHSS < 6 excluded) trials. These trials evaluated the efficacy of EVT in a different therapeutic window (up to 24 h) compared to BEST and BASICS and included a significantly different (lower) percentage of IVT in the BMM group. Given these differences among the four available RCTs, it remains difficult to derive definitive conclusions about the optimal treatment for patients with BAO and mild symptoms, and the benefit of EVT in this patient group remains uncertain. A post hoc subgroup analysis of the BASICS trial 9 and other observational studies12,13 suggest that the higher the NIHSS score, the greater the benefit from EVT. Similarly, a subgroup meta-analysis 14 of patients with low NIHSS score (defined as NIHSS < 10) included in the BASICS and BAOCHE trials showed that the treatment effect of EVT was not significant in these subgroups. Overall, these data suggest that EVT should be used in BAO presenting with moderate-to-severe deficit, while patients with BAO and mild symptoms have better outcomes with intravenous thrombolysis (IVT) alone.
There is no consensus on how to define mild symptoms in BAO, and different cutoffs have been adopted in available clinical trials: NIHSS 0–5 versus 0–10. The NIHSS cutoff of less than 10 is more frequently used in BAO; however, the NIHSS may underestimate posterior circulation symptoms, 15 and an NIHSS score of 6–10 can be highly disabling in BAO. Whether there is a difference in the role of reperfusion therapy between these two cutoffs is not yet known.
Using data from an international multicenter registry, we aimed to compare the efficacy and safety of EVT ± IVT versus IVT alone in early-treated (<6 h from symptoms onset) BAO patients with low baseline NIHSS score, defined as 0–10 (primary analysis) and as 0–5 (secondary analysis).
Methods
Data availability
Access to the anonymized Safe Implementation of Treatments in Stroke-International Stroke Thrombolysis Register (SITS-ISTR) data will be available from the corresponding author upon reasonable request from qualified researchers, contingent on approval by the SITS Scientific Committee.
Study design and inclusion criteria
Based on Safe Implementation of Treatment in Stroke (SITS) International Register, we included treated patients (IVT alone, EVT alone or EVT + IVT) fulfilling the following criteria: (1) diagnosis of acute ischemic stroke (AIS) between January 2013 and January 2021, (2) known symptoms onset (or last seen well) and onset-to-treatment <6 h, (3) evidence of BAO, based on baseline computed tomography angiography or baseline magnetic resonance angiography and (4) available baseline NIHSS score. Onset-to-treatment was defined as onset-to-IVT for patients receiving IVT only and for EVT + IVT; onset-to-EVT was used for patients receiving EVT only. In cases treated with EVT + IVT and non-available onset-to-IVT time, onset-to-EVT was adopted to define onset-to-treatment. In the SITS-ISTR, stroke etiology is coded as follows: large artery atherosclerosis is determined by significant stenosis on radiological examinations (CT, MR, or angiography), or by ICD-10 diagnosis I633 or I630 if no radiological data is available. Cardiac embolism is identified by detecting atrial fibrillation from onset to the 3-month follow-up, or by ICD-10 diagnosis I634 if no data on atrial fibrillation is available. If no etiological data is available for a patient, or if multiple possible etiologies are possible, patients are categorized as unknown etiology. This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 16
Outcomes
We defined primary efficacy outcome as 90-day modified Rankin Scale (mRS) score 0–2; for patients with pre-stroke mRS > 2 primary efficacy outcome was considered achieved in case of return to pre-stroke mRS. We defined secondary efficacy outcome as 90-day mRS 0–3; for patients with pre-stroke mRS > 3 secondary efficacy outcome was considered achieved in case of return to pre-stroke mRS. Safety outcomes were symptomatic intra-cranial hemorrhage (SICH, per modified SITS-MOST definition 17 ) and death by 3-month follow-up.
Statistical analysis
To account for potential confounding factors when comparing efficacy and safety outcome in groups of interest, we adopted Doubly Robust models,18,19 which combine two consecutive estimation techniques: the propensity score model (exposure model) and univariable/multivariable regression model (outcome model). To compare efficacy and safety of EVT ± IVT versus IVT alone in early-time-window BAO patients presenting with NIHSS score 0–10 (primary analysis), we first performed propensity score matching (PSM; exposure model) including only patients presenting with NIHSS 0–10 and onset-to-treatment <6 h. PSM (EVT ± IVT vs IVT alone as response variable), was based on the following prespecified baseline variables, as covariates: age, gender, baseline NIHSS score, pre-stroke mRS, arterial hypertension, hyperlipidemia, diabetes, atrial fibrillation, antithrombotic drug, onset to treatment time. The corresponding propensity score was calculated for each subject and a nearest neighbor matching algorithm with a 1:1 allocation was used to match patients in the two treatment groups (EVT ± IVT vs IVT alone) within 0.2 × SD of the logit of the propensity score. To assess the effectiveness of the PSM, standardized mean differences (SMD) were calculated for each variable. After PSM, we assessed via univariable (unadjusted) and multivariable (adjusted) logistic regression analysis (outcome model) the odds ratio of efficacy and safety outcomes in matched groups. Multivariable analysis was adjusted for the following pre-specified baseline variables: age, gender, baseline NIHSS score, pre-stroke mRS, arterial hypertension, baseline mean arterial pressure, hyperlipidemia, diabetes, baseline glucose level, atrial fibrillation, antithrombotic drug and onset to treatment time. As secondary analysis we re-performed the analysis adopting an NIHSS score 0–5 as cutoff to define mild symptoms.
Considering that the NIHSS score range of 0–10 includes potential variability in symptom severity, we performed a supplementary analysis using the same Doubly Robust approach as the previous analyses. In this analysis, we replaced the “NIHSS score” covariate in the PSM with the “disabling symptoms” covariate to account for different levels of baseline severity, even within the same score range. Disabling symptoms were defined as: 2 points in dysarthria (NIHSS item 10), or 3 points in visual field (NIHSS item 3), or at least one score >2 points in strength items (NIHSS items 5A, 5B, 6A, or 6B), or >1 point in the level of consciousness item (NIHSS item 1A), or >1 point in the gaze deviation item (NIHSS item 2). We also performed a selection-bias-analysis on patients with available baseline NIHSS score, assessing baseline differences between patients with available versus non-available 90-day mRS. Moreover, to more specifically account for the etiology of the ischemic event, we conducted an additional (in Supplementary) multivariate logistic regression analysis (after PSM) following the previously reported multivariate analyses: this further analysis was also adjusted for the etiology of the index event (categorized as LAA vs all other causes).
The significance level was set at p = 0.05. Statistical analysis was performed using STATA 18. For propensity score matching we used R (r-project.org/).
Standard protocol approvals, registrations, and Consent
Requirements for ethical approval and patient consent for participation in the SITS International Thrombectomy Register differed among participating countries. Ethical approval and patient consent were obtained in countries that required this, while other countries approved the register for conduct as an anonymized audit. The SITS Register was approved by the Research Ethics Committee in Stockholm, Sweden any subsequent amendment by the Swedish National Ethics Review Authority.
Results
We included 1426 patients with available baseline NIHSS score and onset-to-treatment <6 h (Table 1): mean age was 69.5 years (SD 13.2) and median NIHSS score was 13 (IQR 7–23). 507 (35.6%) patients presented with NIHSS score 0–10 and 259 (18.2%) with NIHSS 0–5. The median onset-to-treatment time was 175 min, and 47.8% (681 patients) received EVT + IVT. Overall, 42.9% (417 patients) achieved an mRS of 0–2, 52.0% (506 patients) achieved an mRS of 0–3, and 34.6% (337 patients) died within 90 days. The study flow-chart is presented in Figure 1. Differences between patients with- versus without available 90-day mRS (selection bias analysis) are reported in Table S1.
Baseline, treatment characteristics and outcomes in the entire cohort.

AIS: acute ischemic stroke; BAO: basilar artery occlusion; EVT: endovascular thrombectomy; ICH: intracranial hemorrhage; IVT: intravenous thrombolysis; mRS: modified Rankin Scale; mTICI: modified Thrombolysis in Cerebral Infarction scale; NIHSS: National Institutes of Health stroke scale; SICH: symptomatic intracranial hemorrhage.
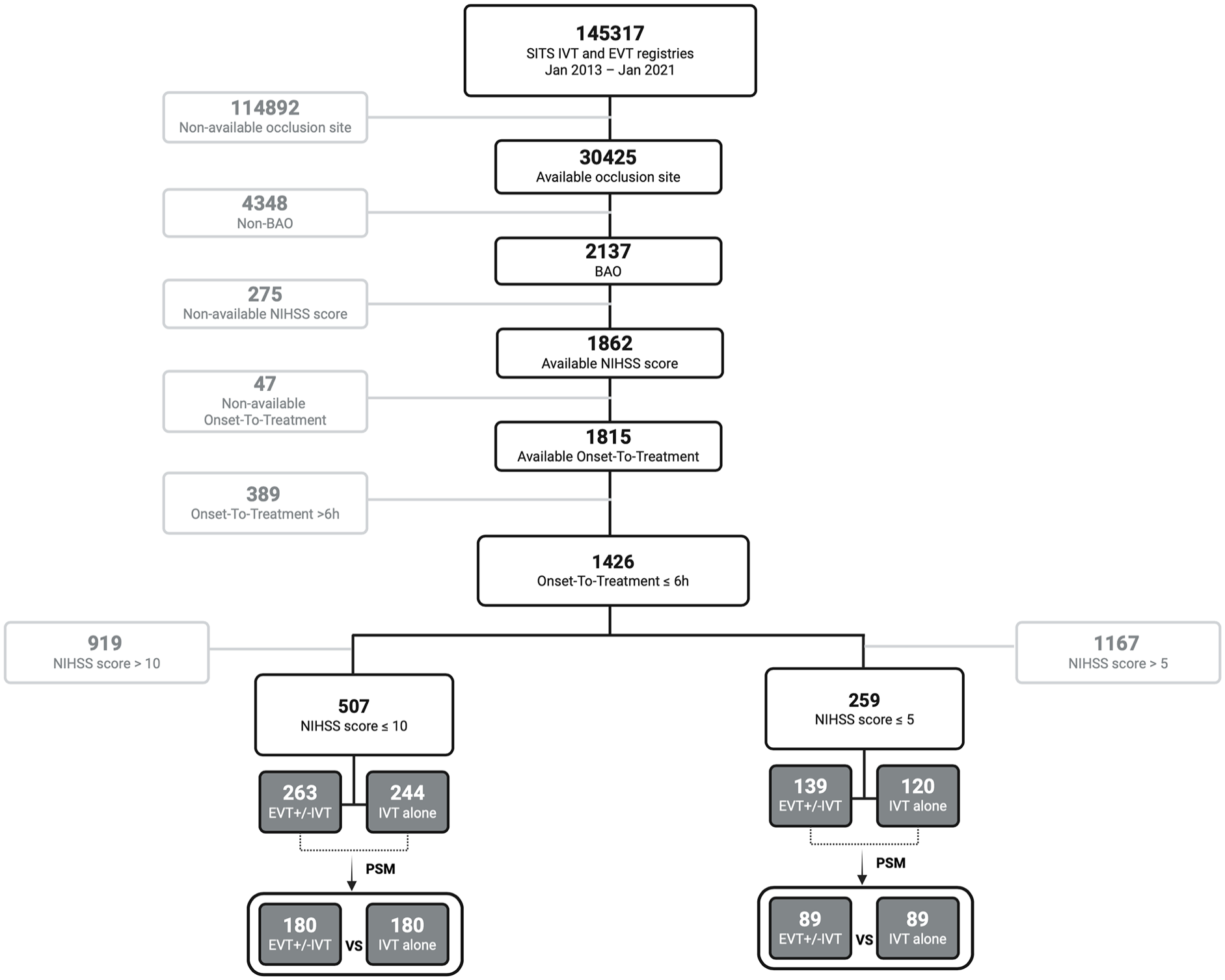
Study flow-chart.
Table 2 shows the baseline characteristics and outcomes for patients with NIHSS scores of 0–10 treated within 6 h. After PSM, 180 patients were included in each treatment group (EVT ± IVT vs IVT alone). The groups had similar baseline characteristics, with SMD for all covariates below 0.12, indicating successful matching. The median onset to IVT time was shorter in the EVT ± IVT group (130 min) compared to the IVT alone group (165 min, p < 0.001). At 90 days, there were no significant differences in favorable outcomes (mRS 0–2) or mortality rates between the groups. Intracranial hemorrhage rates were also similar across both groups. Table 3 displays both the adjusted (multivariable) and unadjusted (univariable) logistic regression models for primary analysis after PSM. When using an NIHSS score cutoff of 10, there was no significant association found between the treatment groups and outcomes in either the unadjusted or adjusted logistic regression analyses. In the supplementary analysis that accounts for symptoms-related disability within the 0–10 NIHSS score range (after PSM, 167 patients in each treatment group), a lower rate of mRS 0-3 (aOR 0.50 [95% CI 0.25–0.99]; p = 0.047) was found in the EVT ± IVT group (Supplemental Table S2).
Baseline, treatment characteristics and outcome in early-treated mild (NIHSS 0–10) BAO patients and differences between EVT ± IVT versus IVT alone, after PSM.
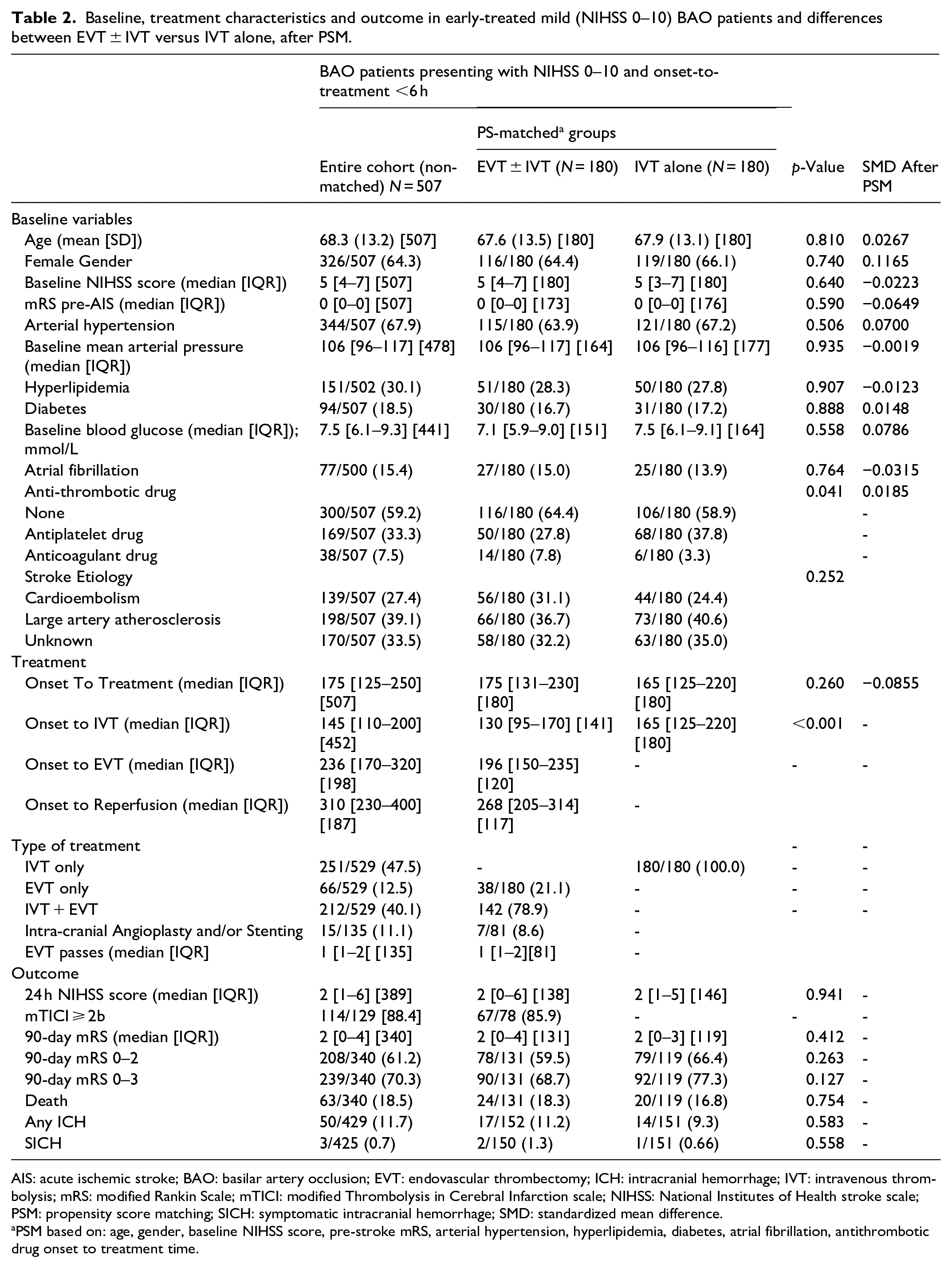
AIS: acute ischemic stroke; BAO: basilar artery occlusion; EVT: endovascular thrombectomy; ICH: intracranial hemorrhage; IVT: intravenous thrombolysis; mRS: modified Rankin Scale; mTICI: modified Thrombolysis in Cerebral Infarction scale; NIHSS: National Institutes of Health stroke scale; PSM: propensity score matching; SICH: symptomatic intracranial hemorrhage; SMD: standardized mean difference.
PSM based on: age, gender, baseline NIHSS score, pre-stroke mRS, arterial hypertension, hyperlipidemia, diabetes, atrial fibrillation, antithrombotic drug onset to treatment time.
Primary analysis: unadjusted and adjusted logistic regression analysis for different efficacy and safety outcomes in early-treated BAO patients with NIHSS 0–10: EVT ± IVT versus IVT alone (after PSM; 360 patients).
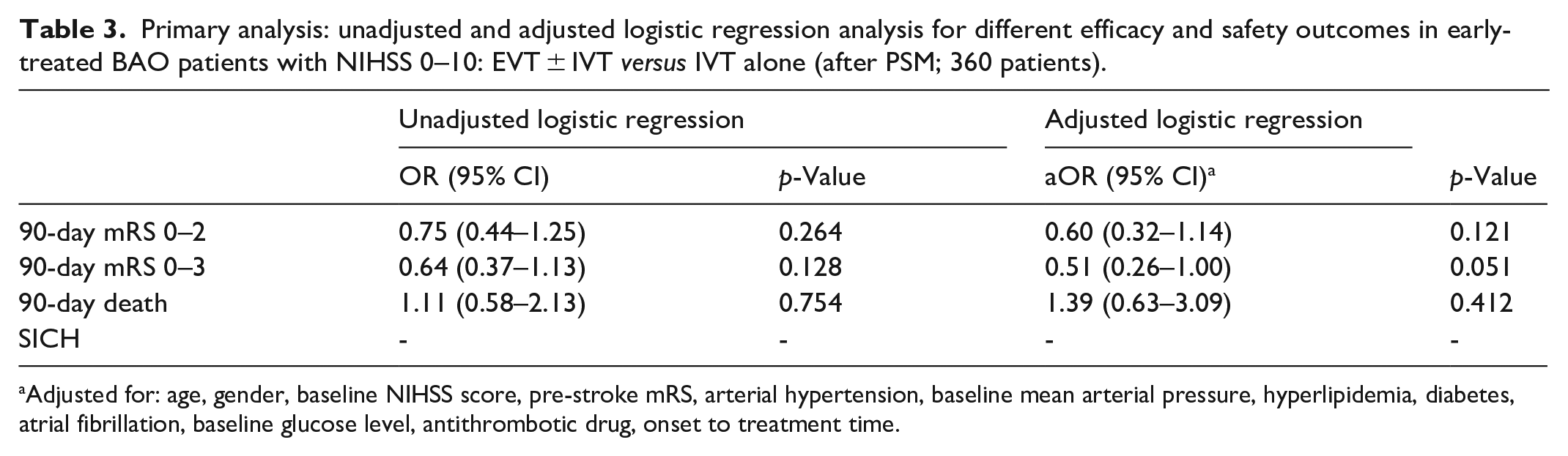
Adjusted for: age, gender, baseline NIHSS score, pre-stroke mRS, arterial hypertension, baseline mean arterial pressure, hyperlipidemia, diabetes, atrial fibrillation, baseline glucose level, antithrombotic drug, onset to treatment time.
The baseline characteristics and outcomes for patients with NIHSS scores of 0–5 treated within 6 h are shown in Table 4. After PSM, 89 patients were included in each treatment group (EVT ± IVT vs IVT alone). Both groups had similar baseline characteristics, with all covariates showing SMD less than 0.16, indicating that the PSM was successful in balancing the groups. The EVT ± IVT group had a significantly lower median onset to IVT time (135 min) compared to the IVT alone group (165 min, p = 0.001). At 90 days, the EVT ± IVT group had lower rates of favorable outcomes (mRS 0–2) compared to the IVT alone group (56.3% vs 76.8%, p = 0.018). Mortality rates and intracranial hemorrhage incidence were not significantly different between the groups. Table 5 presents both adjusted (multivariable) and unadjusted (univariable) logistic regression models for the secondary analysis, with significantly lower rates of the primary efficacy outcome (mRS 0–2) (aOR 0.20 [95%CI 0.06–0.61]; p = 0.005) and the secondary efficacy outcome (mRS 0–3) (aOR 0.27 [95% CI 0.08–0.89]; p = 0.031) in the EVT ± IVT group compared to the IVT alone group. No differences in mortality rates were observed between the two groups.
Baseline, treatment characteristics and outcome in early-treated mild (NIHSS 0–5) BAO patients and differences between EVT ± IVT versus IVT alone, after PSM.
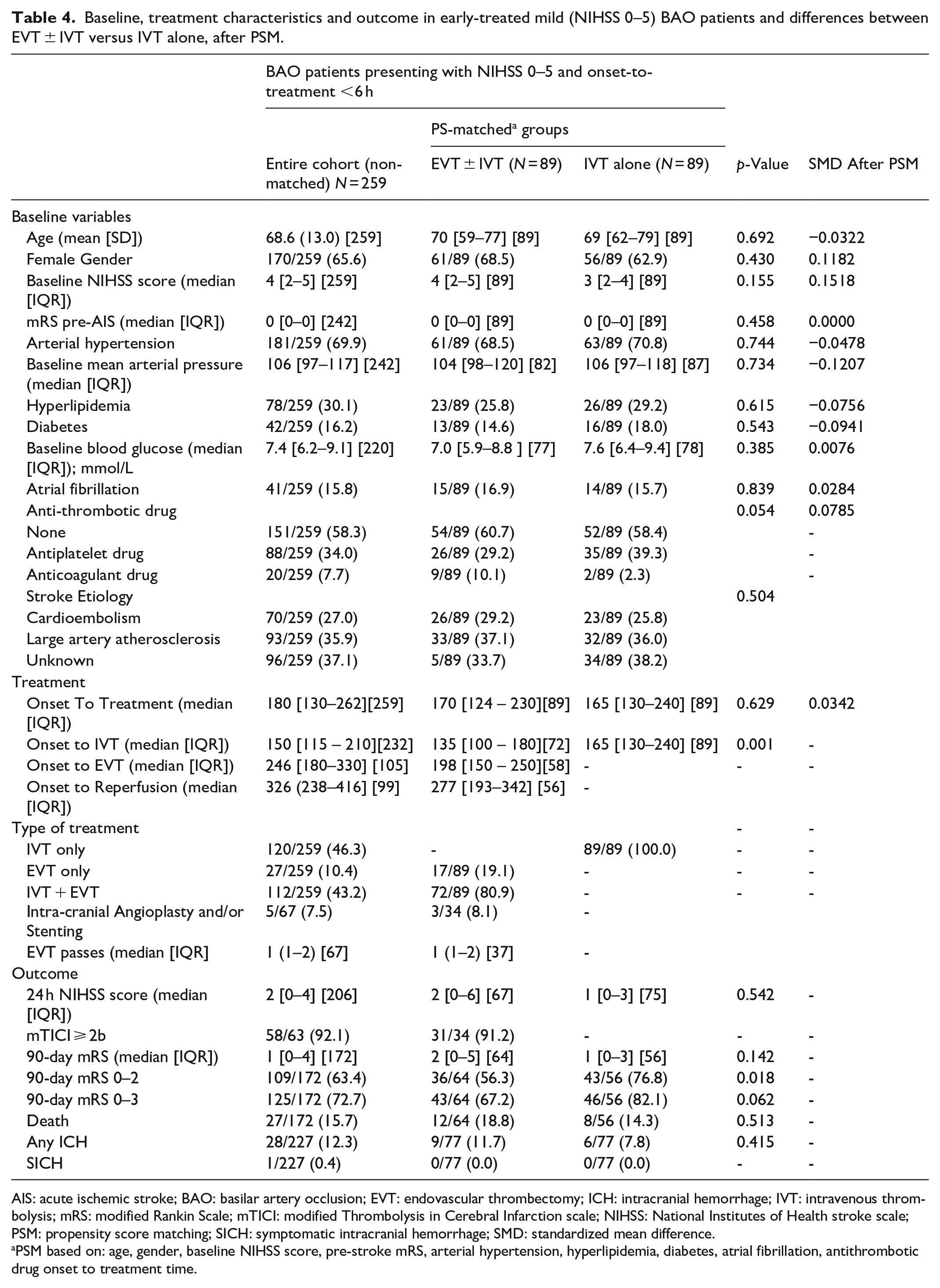
AIS: acute ischemic stroke; BAO: basilar artery occlusion; EVT: endovascular thrombectomy; ICH: intracranial hemorrhage; IVT: intravenous thrombolysis; mRS: modified Rankin Scale; mTICI: modified Thrombolysis in Cerebral Infarction scale; NIHSS: National Institutes of Health stroke scale; PSM: propensity score matching; SICH: symptomatic intracranial hemorrhage; SMD: standardized mean difference.
PSM based on: age, gender, baseline NIHSS score, pre-stroke mRS, arterial hypertension, hyperlipidemia, diabetes, atrial fibrillation, antithrombotic drug onset to treatment time.
Secondary analysis: unadjusted and adjusted logistic regression analysis for different efficacy and safety outcomes in early-treated BAO patients with NIHSS 0–5: EVT ± IVT versus IVT alone (after PSM).
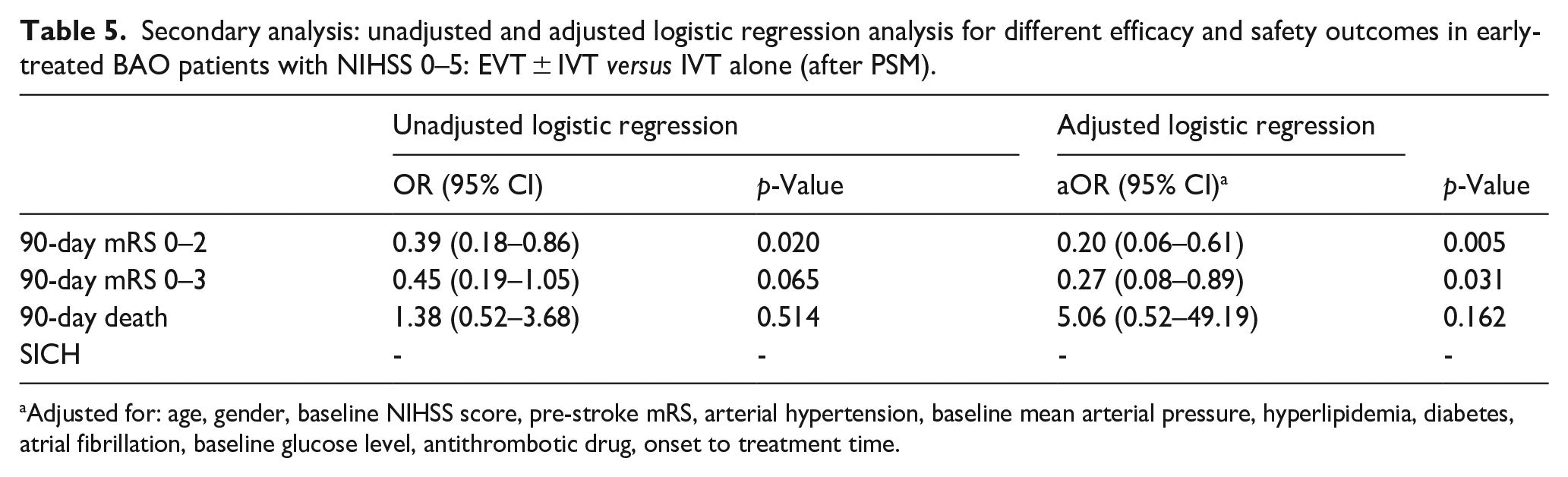
Adjusted for: age, gender, baseline NIHSS score, pre-stroke mRS, arterial hypertension, baseline mean arterial pressure, hyperlipidemia, diabetes, atrial fibrillation, baseline glucose level, antithrombotic drug, onset to treatment time.
In Supplemental Table S3, we report the type of treatment by year in the initial cohort of 1426 patients.
Discussion
Our multi-center register-based study on consecutive propensity-score-matched treated BAO patients shows that among patients with mild BAO defined as NIHSS 0–10, no significant differences in outcomes were observed between those treated with EVT ± IVT and those treated with IVT alone. However, for patients with very mild BAO defined as NIHSS 0–5, those treated with EVT ± IVT had significantly lower rates of mRS 0–2 and mRS 0–3 compared to those treated with IVT alone.
The BEST and BASICS trials, despite including patients with severe deficits on average (median NIHSS 32 and 22 in the treatment groups, respectively), also included patients with BAO and low baseline NIHSS. Notably, the BASICS study had a high percentage of IVT treatments in the BMM group (nearly 80%). Overall, the results from these two trials did not demonstrate the effectiveness of EVT for BAO. The subgroup analysis of the BASICS trial suggested that patients presenting with minor neurological deficits (NIHSS < 10) may not benefit from EVT. Subsequent ATTENTION and BAOCHE trials excluded BAO patients presenting with NIHSS < 10 and <6, respectively, and confirmed an overwhelming benefit of EVT in patients with non-mild BAO, also in extended-time-window. It is noteworthy that in these latter two studies, the percentage of IVT in the BMM group was very low (21% in BAOCHE and 34% in ATTENTION). Considering the differences among the four available RCTs, including varying time criteria (early- vs late-time window), different NIHSS score cutoffs, and differing percentages of IVT in the BMM group, it is challenging to obtain clear data regarding the optimal therapy for patients with BAO and mild symptoms.
In the early time window, the American Heart Association/American Stroke Association (AHA/ASA) guidelines 20 suggest that, despite uncertain benefits, EVT may be reasonable for selected BAO patients within 6 h from stroke onset, without specifying an NIHSS score cutoff. In contrast, the recent guidelines from the European Stroke Organization (ESO) 21 on the management of BAO place significant emphasis on evaluating data for patients presenting with NIHSS < 10. Based on observational data and clinical trials, the ESO guidelines state that there is no evidence to recommend EVT over best medical therapy for BAO patients with NIHSS < 10. Our findings align with this recommendation, showing no significant difference in efficacy between EVT ± IVT and IVT alone. Additionally, our study further highlights a differential treatment effect of reperfusion therapy based on symptom severity: we found that IVT alone was independently associated with higher rates of 90-day mRS 0–2 and 0–3 compared to EVT ± IVT in early-treated patients with NIHSS 0–5. Potential procedure-related complications and reperfusion injury may partially explain these outcomes, making the risk-benefit ratio of EVT unfavorable in cases with very mild symptoms. The uncertain number of patients undergoing EVT due to early clinical deterioration warrants a cautious interpretation of our results, which require further validation.
To the best of our knowledge, this is the largest study on BAO in a Western population specifically designed to shed light on acute reperfusion treatments in patients presenting with mild symptoms, defined with different NIHSS score cutoffs. Our study has several limitations: this is an observational and register-based study, with a proportion of missing and non-available data, including a proportion of 90-day mRS, mTICI score and neuro-imaging variables (i.e. site of BA occlusion, pc-ASPECTs 22 ). We also acknowledge the lack of precise data concerning the etiology of BAO for a significant percentage of patients, including intra-cranial atherosclerosis which is known to have a high incidence in the Asian population that makes up the cohorts of the BAOCHE and ATTENTION trials. Despite these limitations, our selection-bias-analysis is reassuring and we further attempted to address shortcomings by using the Doubly Robust approach with rigorous PSM and multivariable logistic regression analyses. In our study, patient matching was conducted based on onset-to-treatment times, rather than using specific metrics such as onset-to-IVT or onset-to-EVT. This approach was selected to facilitate a consistent comparison across all patients, irrespective of the treatment they received. This led to a notable difference in onset-to-EVT times when comparing the entire non-matched cohort to the matched group, as observed in Table 2. While this matching strategy may introduce certain biases, particularly in the context of time-related variables, our analysis indicates that these do not significantly alter the study’s primary outcomes. Indeed, despite shorter onset-to-EVT times in the matched group, patients receiving only IVT exhibited comparatively better outcomes. The presence of residual confounding remains a concern, particularly due to the insufficient data on patients requiring EVT following IVT because of early clinical deterioration. This study limitation is further compounded by the unpredictable nature of clinical deterioration, which can significantly affect treatment timing. Deterioration can occur either immediately after the initial evaluation or substantially later, thereby introducing a high degree of variability in the timing of EVT intervention that current analyses on available time-based data cannot adequately address. Observational studies like ours are not intended to advocate for one therapeutic approach over another; rather, our results strongly indicate the need for clinical trials on mild BAO. At present, a comprehensive patient evaluation with a case-by-case decision that also considers elements not available in our study, such as occlusion location and pc-ASPECTS, is essential. Given the limitations of the classic NIHSS score in cases of posterior circulation stroke, the widespread use of the posterior-NIHSS 23 (which has demonstrated higher prognostic accuracy than NIHSS for identifying posterior circulation stroke patients with NIHSS < 10) would be desirable in the future. While awaiting randomized data on mild-presenting BAO, observational data like ours are crucial to guide stroke neurologists in everyday clinical practice and to inform the design of future clinical trials.
Conclusion
Our study found that in patients with basilar artery occlusion and mild symptoms, defined as NIHSS less than 10, there were no significant differences in outcomes between intravenous thrombolysis alone and endovascular treatment with or without thrombolysis. However, in those with very mild symptoms, defined as NIHSS less than 5, intravenous thrombolysis alone was associated with better outcomes compared to endovascular approaches. Despite the challenges posed by the infrequent presentation of these cases, randomized trials are needed to identify the optimal reperfusion therapy for patients with BAO and mild symptoms.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241272517 – Supplemental material for Reperfusion treatment in basilar artery occlusion presenting with mild symptoms
Supplemental material, sj-docx-1-eso-10.1177_23969873241272517 for Reperfusion treatment in basilar artery occlusion presenting with mild symptoms by Ghil Schwarz, Angelo Cascio Rizzo, Marius Matusevicius, Tiago Moreira, Aleksandras Vilionskis, Andrea Naldi, Nicolas Martinez-Majander, Guido Bigliardi, Danilo Toni, Christine Roffe, Elio Clemente Agostoni and Niaz Ahmed in European Stroke Journal
Footnotes
Acknowledgements
NA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approvals,registrations,and informed consent
Requirements for ethical approval and patient consent for participation in the SITS International Thrombectomy Register differed among participating countries. Ethical approval and patient consent were obtained in countries that required this, while other countries approved the register for conduct as an anonymized audit. The SITS Register was approved by the Research Ethics Committee in Stockholm, Sweden any subsequent amendment by the Swedish National Ethics Review Authority.
Guarantor
GS
Contributorship
GS led the study’s conception, design, data analysis, and was primarily responsible for writing the manuscript and preparing tables and figures. NA, MM, CR, DT, ECA and ACR were involved in study conception and design. TM, AV, AN, NMM, GB contributed on refining study design and data acquisition. All authors participated in editing and approving the final manuscript.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request, subject to a favorable opinion from the SITS Steering Committee.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
