Abstract
Introduction:
Different serum lipid and lipid-lowering agents are reported to be related to the occurrence of intracerebral aneurysm (IA). However, the causal relationship between them requires further investigation.
Patients and methods:
Mendelian randomization (MR) analysis was performed on IA and its subtypes by using instrumental variants associated with six serum lipids, 249 lipid metabolic traits, and 10 lipid-lowering agents that were extracted from the largest genome-wide association study. Phenome-wide MR analyses were conducted to identify potential phenotypes associated with significant lipid-lowering agents.
Results:
After multiple comparison adjustments (p < 0.0083), genetically proxied triglyceride (TG) (odds ratio [OR] 1.25, 95% confidence interval [CI] 1.07–1.47, p = 0.005) and high-density lipoprotein cholesterol (HDL-C) levels (OR 0.93, 95% CI 0.89–0.98, p = 0.008) showed causal relationships with the risk of IA. Four lipid metabolic traits showed a causal relationship with the risk of IA (p < 0.0002). As confirmed by drug target MR, the causal relationship between the HMGCR target and IA, HMGCR target and subarachnoid hemorrhage (SAH), ANGPTL3 target and SAH, CETP target, and SAH remained statistically significant after multiple adjustments (p < 0.005). Additionally, phenome-wide MR did not identify other diseases linked to the significant lipid-lowering agent (p < 6.39 × 10−5).
Discussion and conclusion:
This study not only supports that serum lipids (TG and HDL-C) are associated with IA but also confirms the positive effect and absence of safety concerns of intervening HMGCR, ANGPTL3, and CETP targets in IA and its subtypes, opening new avenues for IA treatment.

Introduction
Intracranial aneurysm (IA) is an abnormal partial dilatation of the cerebral blood vessels common in the general population, with an estimated global prevalence of 3.2%. 1 IA most commonly occurs between 40 and 50 years old and appears more common among women. 1 Most IAs are unruptured and remain silent throughout the lifetime of patients who harbor them. 2 However, the rupture of IA and subsequent subarachnoid hemorrhage (SAH) may cause severe neurological consequences and poor prognosis. 3 Therefore, identifying the risk factors linked to the formation and rupture of IAs is vital as it could provide preventive strategies for IAs.
Biologically, lipids serve many vital functions, including as structural components of cell membranes, energy sources, and signal transduction participants. 4 Based on previous cross-sectional and mechanistic studies, we believe that they impact aneurysm formation and growth. Previous research showed that lipid levels were positively associated with IA generation and rupture, whereas high levels of high-density lipoprotein (HDL-C) and lipid-lowering agents were negatively associated with IA rupture. 5 Moreover, oxidative stress generated by lipid metabolism may increase the formation and rupture of IA by exacerbating inflammation. 6 Frösen 7 advocated that studying diet, exercise, and other lifestyle-modifying lipid levels or associated biomarkers of lipid metabolism may be the way forward. However, evidence from existing studies only showed a relationship between lipid metabolites and IA, and the causality between them remains yet unknown.
Mendelian randomization (MR) is a statistical method to determine causal relationships that has been widely used to assess the causal relationship between biomarkers and various neurological disorders, such as epilepsy, 8 Parkinson’s disease, 9 and stroke. 10 MR analysis could also be used to repurpose licensed drugs by integrating summary statistics from genome-wide association studies (GWAS) and expression quantitative trait locus (eQTL) studies. 11 Life-long exposure could be determined by levels of expression of druggable genes and eQTLs located in their genomic regions, which are considered proxies. 12 Previous studies have potentially detected the causal relationship between different medications and IA risk using the MR method,13,14 demonstrating the feasibility of applying eQTL data in combination with GWAS studies for drug target MR analysis.
In this study, we conducted a comprehensive MR analysis to explore the genetic role of lipids, metabolites, and lipid-lowering targets in IA and its subtypes.
Methods
Study design and ethical approval
Our MR estimation adhered to the STROBE-MR guidelines (Supplemental Table 1). 15 The study design is shown in Figure 1.
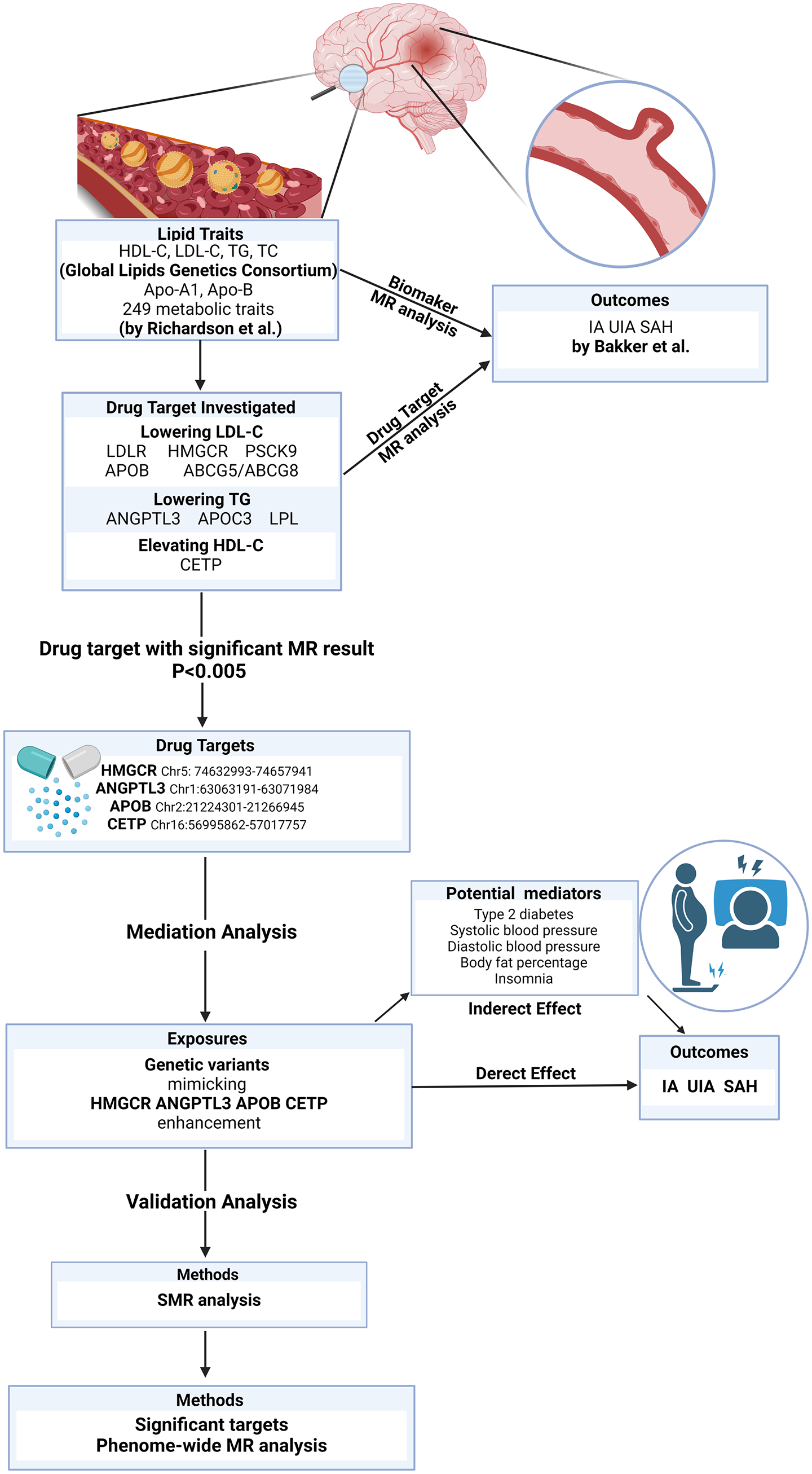
Overview of the study design.
No ethical approval was required for the present study because all data were based on an online-published GWAS analysis that was publicly available. Approval for all of these studies was obtained from the relevant institutional review committees.
Data source
This study aggregates and analyzes four serum lipids: serum total cholesterol (TC), TG, LDL-C, and HDL-C. 16 The GWAS summary statistics pertaining to individuals of European descent from the Global Lipids Genetics Consortium (GLGC) study were procured for this MR investigation. Apolipoprotein A1 (Apo-A1) and apolipoprotein B (Apo-B) were extracted from the UK biobank. 17 The summary level statistic of circulating metabolism traits was obtained from a study conducted by Richardson et al. 18 For the outcome, we used the summary statistics of the largest GWAS of IA. 19 Supplemental Table 2 provides more details of the GWAS summary statistics.
Identification of IVs
The selection of instrumental variables (IVs) was based on the three assumptions of MR analysis. Among serum lipids and circulating metabolic traits, all SNPs selected as IVs met with genome-wide significance (p < 5 × 10−8). Then, we performed linkage disequilibrium (LD) clumping to identify independent SNPs with a threshold of r2 < 0.001, kb = 10,000, using the 1000 Genomics Project Phase 3 (EUR) as the reference panel. During the harmonization of SNP-exposure and SNP-outcome effects, we excluded SNPs characterized by mismatched alleles, palindromic sequences, and those harboring missing values. SNPs with F-statistics, calculated using the formula F = beta2/se2, below the threshold of 10, were excluded to remove weak instrument bias. 20
In drug target MR, we selected commonly used and novel lipid-lowering drugs and then classified them into four main lipid modulations based on their primary pharmacological functions (Supplemental Table 3). The lipid-lowering effect of these drugs was predicted using SNPs under a more lenient LD threshold of r2 < 0.30, kb = 100, and then served as proxies for the drug targets. 21 Of note, the selection of IVs for each drug target was based on the corresponding summary statistics of serum lipids. When significant lipid-lowering targets were identified, we further extracted the IVs for these drug targets based on statistics of other serum lipids or lipoproteins to investigate whether they could exhibit protective effects through multiple lipid-modifying pathways. Next, we utilized publicly accessible eQTL data from the Genotype-Tissue Expression project (GTEx-V8) and eQTLgen to further investigate the association between the significant drug targets and IA. The analysis was directed toward tissues where the target genes were recognized for their elevated expression levels. IVs extracted from cis-eQTLs should satisfy rigorous criteria (p < 5 × 10−8, r2 < 0.1).
Statistical analysis
The inverse-variance weighted (IVW) approach was utilized to estimate the causal association of genetically proxied serum lipids, metabolisms, and lipid-lowering targets on IA, UIA, and SAH. We also used three complementary methods (i.e. MR-Egger regression, weight mode, and weight median) to infer causality. The odds ratio (OR) reflects the causality of serum lipids and IA and presents an increased risk of binary outcomes (aneurysms) per SD increase in lipid levels. To validate our selection of effective IVs that proxied targeted genes, we conducted a positive control analysis by using GWAS data of coronary artery disease (CAD) as the outcome. 22 Furthermore, we applied the summary-based MR (SMR) approach to validate the result of drug target MR, using summary statistics of cis-eQTL from GTEx-V8 and eQTLgen.23,24 The heterogeneity in dependent instruments (HEIDI) test was also conducted to assess the presence of linkage in the observed association, and a HEIDI p-value of <0.05 suggested that the observed association might have originated from two distinct genetic variants in high LD with each other. 25
To identify whether the observed lipid-lowering targets are related to IA risk directly, we evaluated the relationship between lipid-lowering targets and previously determined risk factors for IA such as type 2 diabetes, 26 body fat percentage, systolic blood pressure (SBP), diastolic blood pressure (DBP), 27 and insomnia, 28 via two-step MR analysis, which may be potential mediators. In two-step MR, the impact of lipid-lowering targets on IA after accounting for potential mediators is denoted as the direct effect, while the intermediary effect from potential mediators is considered the indirect effect.
We applied Bonferroni correction for different levels of exposure. For serum lipids, we set p < 0.0083 (0.05/6) as indicative of statistical significance after Bonferroni correction, while for lipid metabolites, a p < 0.0002 (0.05/249) indicates statistical significance. For drug-target MR analysis, we adopted p < 0.005 (0.05/10) as indicative of statistical significance. When IVW exhibited a p-value over the determined threshold and <0.05 after multiple corrections, suggestive evidence for potential causal association was considered. 29
Sensitivity analysis
Several sensitivity analyses, including the MR-Egger intercept, Cochran’s Q test, MR-PRESSO, leave-one-out test, and funnel plot, were undertaken to validate the robustness of MR estimates. Cochran’s Q test was employed to identify potential heterogeneity derived from horizontal pleiotropy or other biases. p < 0.05 was considered indicative of low heterogeneity. 30 MR-Egger intercept and MR-PRESSO were undertaken to examine the presence of horizontal pleiotropy.31,32
Phenome-wide MR
We used effective IVs that exposed drug targets to perform phenome-wide MR on disease. We summarized data from the UK Biobank to assess the potential adverse effects of significant drug targets. Of these disease phenotypes with cases over 500 were selected for further phenome-wide MR estimation, and the summary statistics were downloaded from the SAIGE GWAS website (https://www.leelabsg.org/resources). 33 A previously published study illustrates A detailed description of the methods and parameters.33,34
All data analysis was conducted by TwoSampleMR, RadialMR, Mendel MR, and MR-PRESSO package. SMR analysis was conducted in the SMR software tool using R software (version 4.3).
Results
TSMR for serum lipids and IA risk
The IVs for serum lipids, including HDL-C, LDL-C, TG, TC, Apo-A1, and Apo-B, are shown in Supplemental Tables 4–9. All IVs exhibited F-statistics over 10, indicating no weak IV bias.
Under the threshold of p < 0.0083 after Bonferroni correction, for IA, genetically proxied TG levels were positively associated with the risk of IA (OR 1.25, 95% CI 1.07–1.47, p = 0.005), and genetically proxied HDL levels demonstrated negative association with the risk of IA (OR 0.93, 95% CI 0.89–0.98, p = 0.008). For SAH, a suggestive significant difference was detected between the TG level and the SAH risk (OR 1.23, 95% CI 1.02–1.48, p = 0.03). No causal association was seen between the serum lipids and UIA (Figure 2).
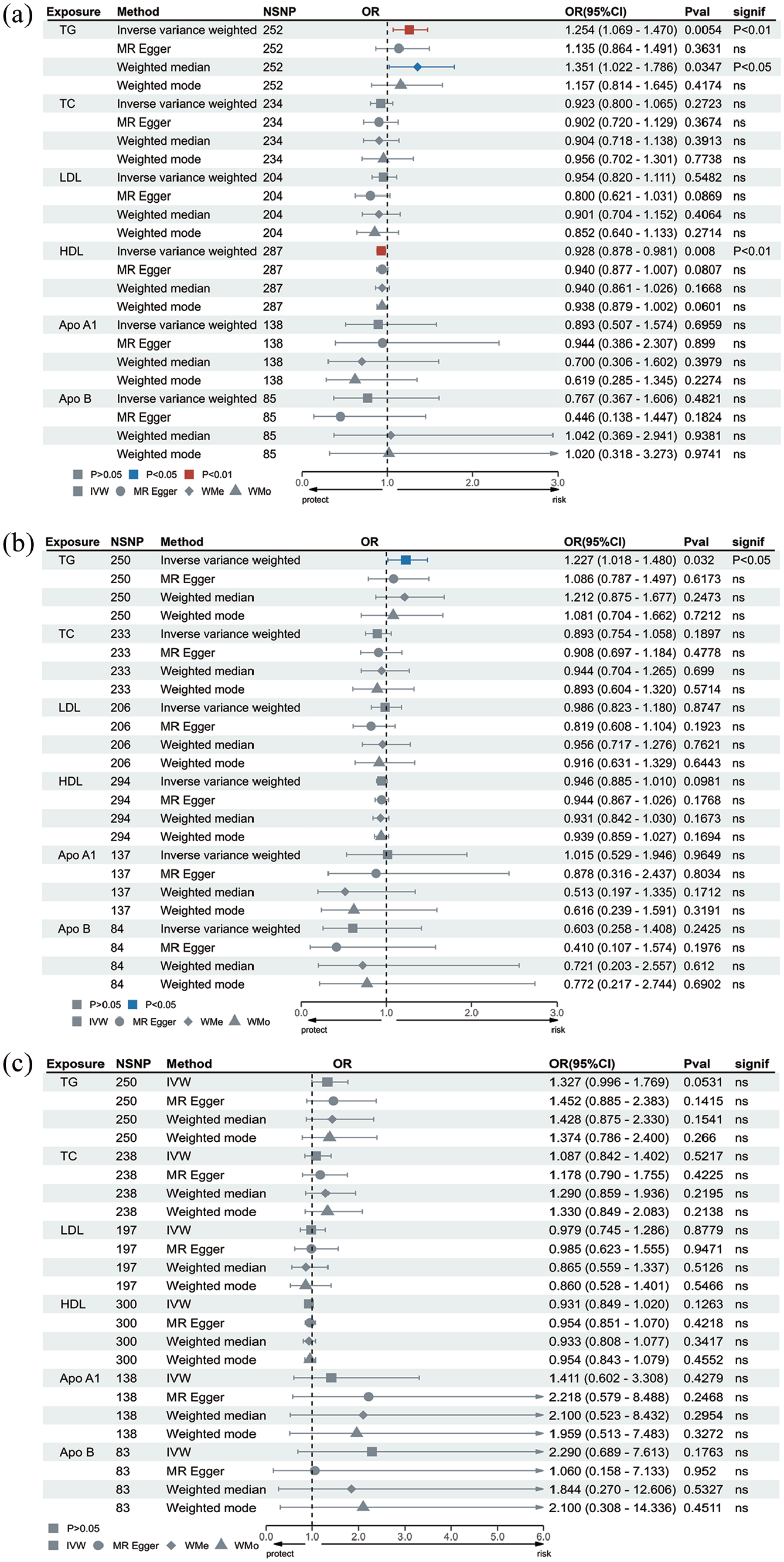
MR result of serum lipid traits on aneurysm.
TSMR for lipid metabolites and IA risk
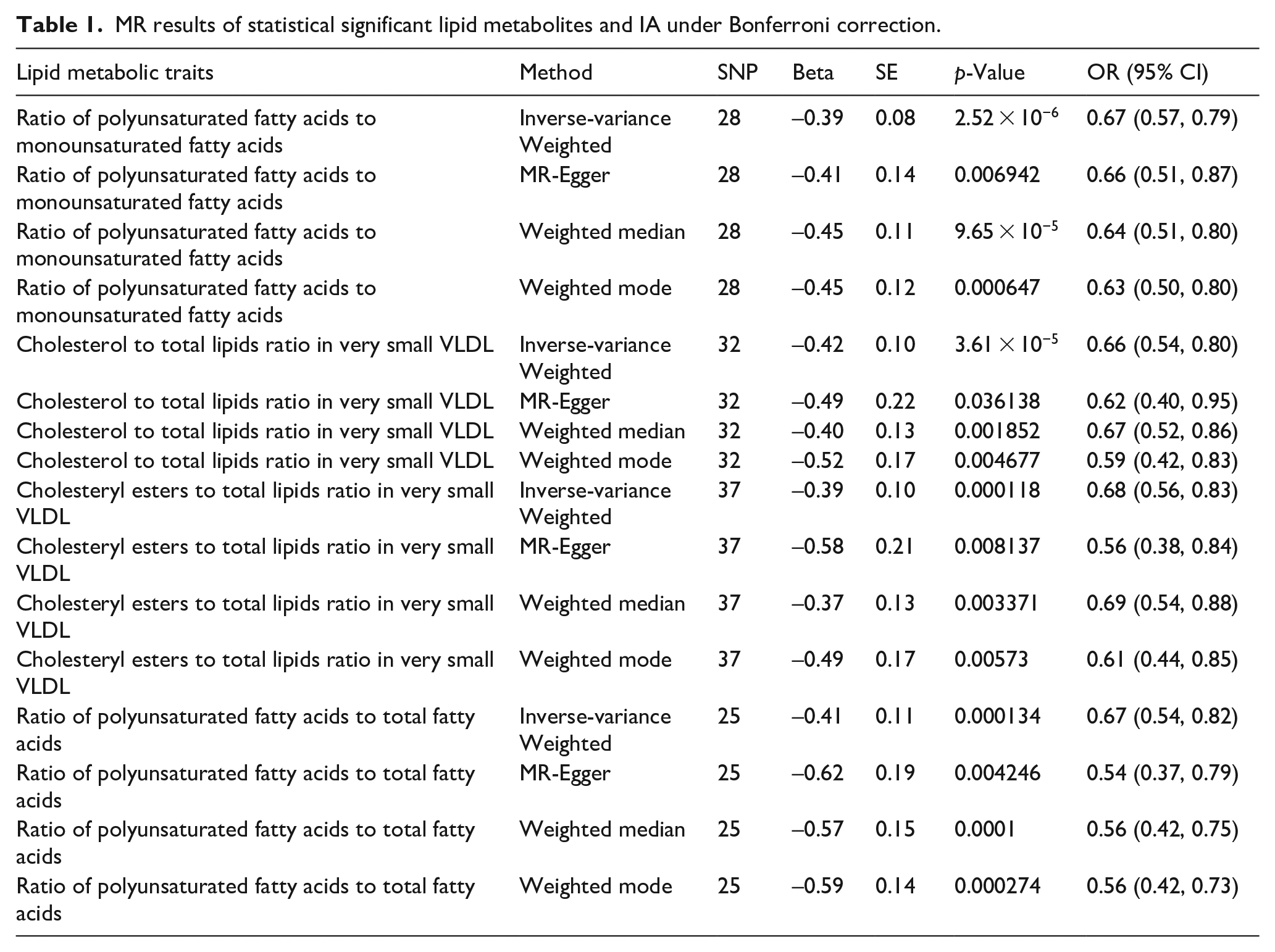
The IVs for lipid metabolites are presented in Supplemental Tables 10–12. The MR estimates of metabolic traits and IA risk are given in Figure 3 and Table 1. After Bonferroni correction (p < 0.0002), ratio of polyunsaturated fatty acids to monounsaturated fatty acids (OR 0.67, 95% CI 0.57–0.79, p = 2.52 × 10−6), cholesterol to total lipids ratio in very small VLDL (OR 0.66, 95% CI 0.54–0.80, p = 3.61 × 10−5), cholesteryl esters to total lipids ratio in very small VLDL (OR 0.68, 95% CI 0.56–0.83, p = 0.0001), and ratio of polyunsaturated fatty acids to total fatty acids (OR 0.67, 95% CI 0.54–0.82, p = 0.0001) showed a significant causal relationship with IA. However, no genetically proxied metabolic traits were related to the risk of SAH and UIA after Bonferroni correction (Supplemental Figures 1 and 2).
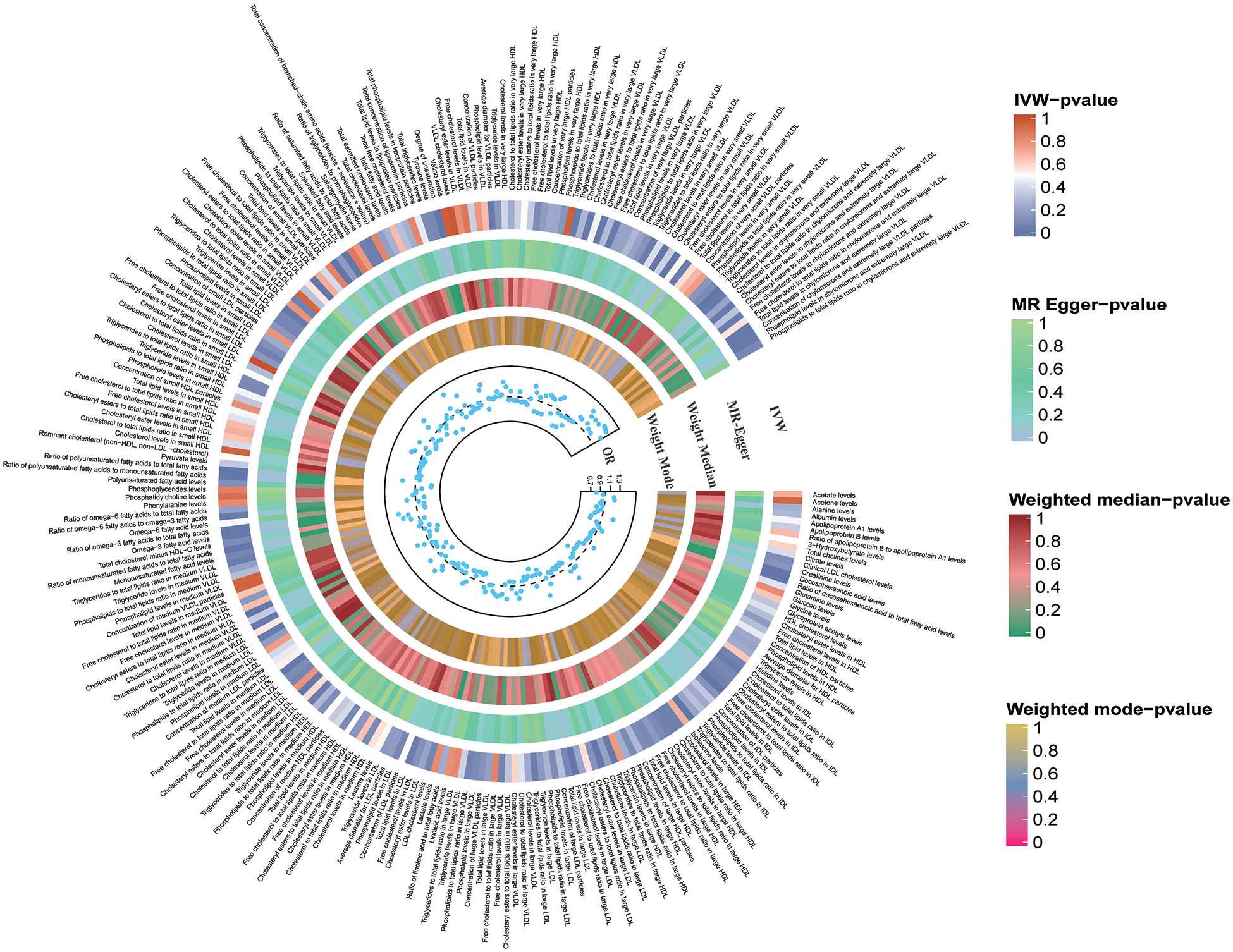
MR result of lipid metabolites and intracerebral aneurysm.
MR results of statistical significant lipid metabolites and IA under Bonferroni correction.
Drug target MR for lipid-lowering targets and IA risk
Information on genetic variants for lipid-lowering targets is shown in Supplemental Table 13. As illustrated in Figure 4 and Supplemental Table 14, genetically proxied HDL-C level modified by CETP (OR 0.86, 95% CI 0.77–0.97, p = 0.01), LDL-C level modified by APOB (OR 0.75, 95% CI 0.59–0.95, p = 0.02) and HMGCR (OR 0.51, 95% CI 0.36–0.72, p = 0.0001) demonstrated negative association with IA risk. For SAH, genetically proxied HDL-C level modified by CETP (OR 0.83, 95% CI 0.73–0.94, p = 0.004), LDL-C level modified by HMGCR (OR 0.51, 95% CI 0.35–0.75, p = 0.0005), and TG level modified by ANGPTL3 (OR 0.48, 95% CI 0.31–0.75, p = 0.001) demonstrated negative association with SAH risk. For UIA, genetically proxied LDL-C level modified by APOB (OR 0.58, 95% CI 0.38–0.88, p = 0.01) and HMCGR (OR 0.55, 95% CI 0.32–0.94, p = 0.03) they demonstrated negative associations with the UIA risk; while a positive association was observed between the genetically proxied TG level modified by ANGPTL3 and UIA (OR 2.56, 95% CI 1.28–5.12, p = 0.007). After Bonferroni correction, LDL-C level modified by HMGCR in IA and SAH, TG level modified by ANGPTL3 in SAH, and HDL-C modified by CETP in SAH remained statistically significant (p < 0.005). In addition, the positive control analysis revealed noteworthy associations between genetically predicted drug targets and CAD, indicating the effectiveness of selected IVs, which was consistent with previous studies18,35 (Supplemental Figure 3). Furthermore, HMGCR, CETP, and ANGPTL3 exhibited multiple lipid-modifying effects since causal effects of gene targets extracted from other serum lipid and lipoproteins on IA and its subtypes risks were identified (Supplemental Table 15 and Supplemental Figure 4).
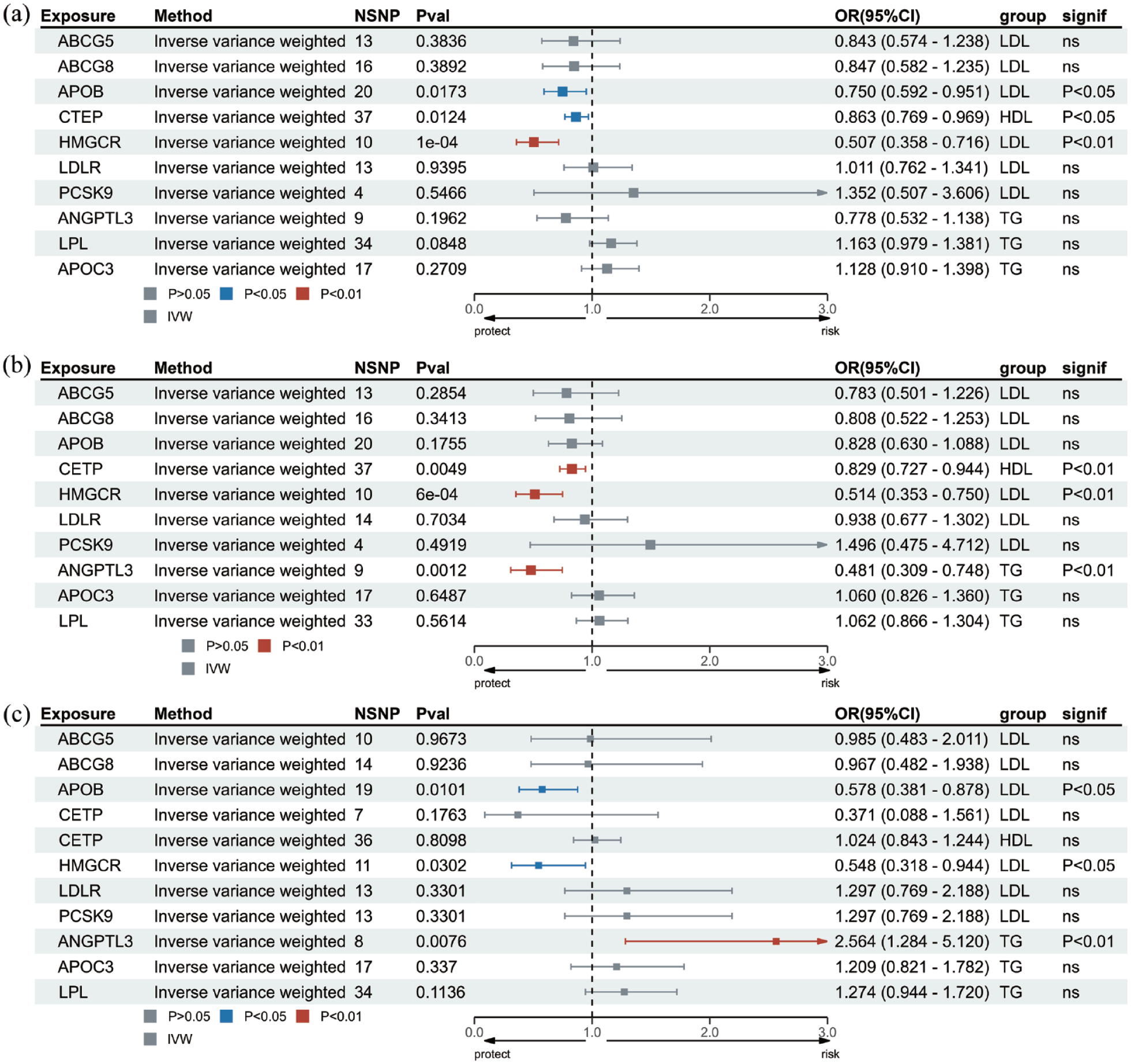
MR result of lipid-lowering targets and aneurysm.
Sensitivity analysis
The outcomes of sensitivity analyses are presented in Supplemental Tables 16–18. According to Cochran’s Q test results, no heterogeneity was found, and no outlier was removed in the MR results with statistical significance except for Cholesteryl esters to total lipids ratio in very small VLDL (Supplemental Table 17). We then used the Radial MR approach to identify potential SNPs that caused horizontal pleiotropy (Supplemental Figure 9). In addition, each method demonstrated the same slope direction in scatter plots, suggesting the robustness of MR estimation (Supplemental Figures 5, 7, 12, 16, and 20). The funnel plot found no bias because the estimated values were symmetrically distributed (Supplemental Figures 6, 8, 13, 17, and 21). The forest plots showed no significant SNPs that dominated MR results (Supplemental Figures 9, 14, 18, and 22). In the leave-one-out test, there was no change in the risk of aneurysm as genetically predicted, proving that the MR results were robust and not driven by a single specific SNP (Supplemental Figures 10, 15, 19, and 23).
Mediation analysis
Among the tested mediators, SBP and DBP were identified as potential mediators in the causal relationship between HMGCR and IA, and similar results were also observed between HMGCR, SAH, and UIA (Supplemental Table 19 and Supplemental Figures 24–26). Body fat percentage, SBP, and DBP were mediators between APOB and IA, but only SBP mediated the relationship between APOB and UIA. In CETP, SBP, and DBP were potential mediators between the CETP and IA, while SBP and DBP also mediated the relationship between CETP and SAH (Figure 5).
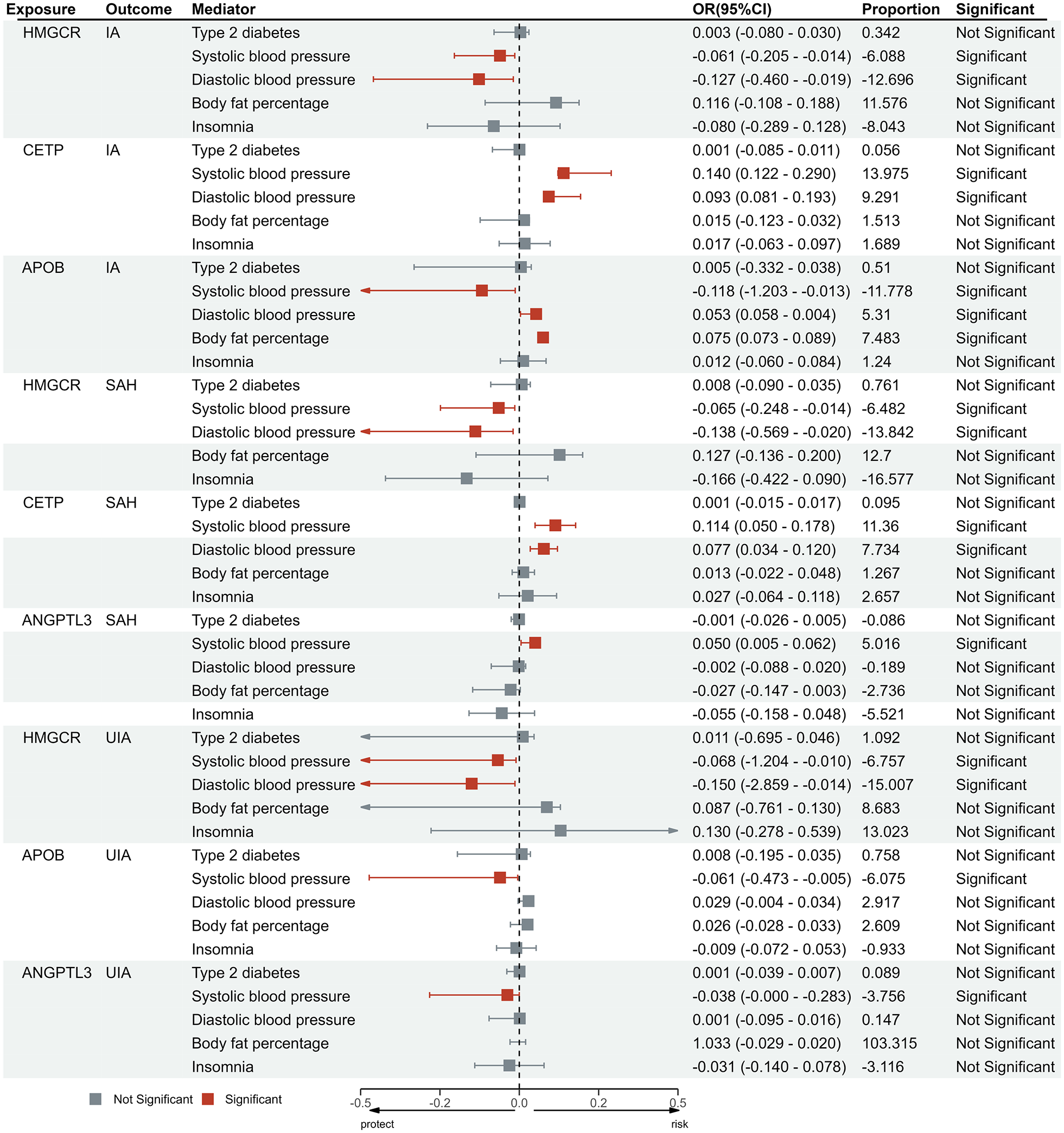
Intermediary effect of potential mediators between the significant lipid-lowering targets and aneurysm.
Gene target expression and IA risk
SMR analysis investigated the association of lipid-lowering target expression and IA, which provided additional evidence. High APOB and CETP expression was associated with a higher risk of IA and SAH, and high HMGCR and ANGPTL3 expression was associated with a lower risk of IA, SAH, and UIA, suggesting that CETP and APOB inhibition and HMGCR and ANGPTL3 activation had a protective effect against IA and its subtypes (Supplemental Table 20). SMR analysis on these targets and serum lipids also confirmed the multiple lipid-modifying regulations among these lipid-lowering targets (Supplemental Table 21).
Phenome-wide MR analysis of significant lipid-lowering targets
A broader phenome-wide MR was performed to screen 783 non-IA phenotypes in the UK Biobank that may have been associated with these significant lipid-lowering targets (Supplemental Table 22). After Bonferroni correction (p < 0.05/783), no other disease phenotypes may be linked to APOB, HMGCR, ANGPTL3, and CETP levels (Supplemental Table 23–26).
Discussion
This genetic-based study provided evidence of the association of serum lipids, metabolisms, and lipid-lowering targets in IA risk. Initially, we observed that genetically proxied level of TG was associated with IA risk. Four lipid metabolic traits were identified to have a causal association with IA risk after Bonferroni correction. Next, we further investigated the genetically determined lipid-lowering targets and IA risk. Four drug targets were identified (HMGCR, APOB, CETP, and ANGPTL3) to have statistical or suggestive significance with the risk of IA and its subtypes. In addition, we also observed the mediator role of SBP and DBP-related traits in the causal association between lipid-lowering targets and IA. Finally, phenome-wide MR did not identify any significant phenotypes that were associated with significant lipid-lowering targets, indicating no potential safety concerns when utilizing these targets. 34
Despite the correlation between serum lipids and cardiovascular diseases, the relation with IA and its subtypes remains controversial, with studies reporting significant or insignificant associations. In our MR analysis, an increased TG level was associated with the risk of IA and SAH, consistent with the TSMR study conducted by Karhunen et al. 36 An epidemiology investigation concluded that a decreased level of TG was associated with the risk of SAH among women; while Bonaventure et al. suggested a strong correlation between increased levels of TG and hemorrhagic stroke among male patients.37,38 Regarding cholesterols, our MR results suggested a negative association between HDL-C and IA risk, consistent with the viewpoint that low HDL-C levels contributed to the growth of IA. 39 Extensive evidence supports the atheroprotective role of HDL-C in the cardiovascular system, manifested through anti-inflammatory, antithrombotic, and antioxidative effects. 39 Elevated levels of HDL-C have been shown to correlate with diminished atherosclerotic changes, reduced growth of IA, and a lower incidence of CAD during the early stages of IA formation. 40 Among lipid metabolic traits, we found that the cholesterol to total lipids ratio in very small VLDL was associated with the risk of IA after Bonferroni correction, indicating the influence of cholesterol particles on IA. Additionally, the effect of several metabolites reaching suggestive significance on IA was acknowledged by previous studies; Abekura et al. 41 confirmed that n-3 polyunsaturated fatty acids, eicosapentaenoic acid, and docosahexaenoic acid could prevent the progression of IA in rats by inhibiting macrophage infiltration, which was a sign of inflammatory response in lesions. Eicosapentaenoic acid could also inhibit symptomatic cerebral vasospasm after SAH, which improved patient prognosis after IA rupture. 42
This study unveiled that HMGCR (statins) was inversely related to the risk of IA and its subtypes, indicating the protective role of statin use in IA patients. Clinical trials reported decreased circulating inflammatory biomarkers and aneurysmal wall enhancement (AWE) after using atorvastatin for 6 months, suggesting its potential to alleviate aneurysmal wall inflammation.43,44 Statins were also associated with a reduced risk of UIA growth. 45 However, the effect of statin on IA rupture was controversial. Tada et al. 46 concluded a bidirectional effect of statin that could inhibit IA growth and promote IA rupture. At the same time, Yoshimura et al. 47 found an inverse association between statin use and IA rupture. Our MR estimates support a positive preventive role of statins in the growth and rupture of IA, which was also supported by the theory that statins exhibited an inhibitory effect on IA rupture through the anti-NF-KB impact. 48 For other lipid-lowering targets, our study acknowledged the relationship between APOB and the risk of IA and UIA. Apo-B-100 inhibitor, mipomersen, has been approved to treat familial hypercholesterolemia by the US Food and Drug Administration. 49 However, its effect on IA has not yet been explored. An experimental study concluded that an Apo-B-100-related peptide could prevent aortic aneurysm formation by reducing macrophage infiltration in the aorta. 50 Peng et al. found that the Apo-B level was an independent predictor of AWE in UIA. Further investigation is required to determine whether inhibition of Apo-B through mipomersen can influence AWE. 51 We found that CETP inhibitors could reduce IA and SAH risk through multiple lipid-modifying pathways. Substantial evidence supports the protective role of CETP in abdominal aortic aneurysms and coronary events through modulating HDL-C levels. 52 ANGPTL3 is an endogenous inhibitor of endothelial lipase that could reduce TG concentrations and LDL-C through negative regulation of LPL. 35 Previous studies have shown that ANGPTL3 inhibition was related to a lower risk of atherosclerotic cardiovascular disease. 53 Notably, our study found a bidirectional causal effect of ANGPTL3 in UIA and SAH, indicating that ANGPTL3 may have a different influence on the growth and rupture of IA. Studies reporting the application of CETP and ANGPTL3 inhibitors in IA are limited, and more studies investigating lipid-lowering targets in IA are warranted. Considering the firmly established risk factors of IA, they may serve as potential mediators underlying the impact of these targets on IA risk. According to mediation analysis, SBP and DBP may be potential mediators between the lipid-lowering targets and IA, UIA, and SAH. In an RCT conducted by Yoshida et al., 54 greater SBP was observed in the statins group, indicating the potential influence of statin use on SBP. However, the role of blood pressure and other lipid-lowering targets and IA has not yet been validated.
This study has some limitations. First, our study population was limited to Europeans; hence, our results may not apply to other ancestries. Second, the genetic variants employed in this study indicated the sustained influence of lipid level fluctuations on IA risk. This perspective may not parallel the immediate effects of lipid-lowering agents. 21 Therefore, clinical trials are warranted to validate these observational results. Third, this study focused on the intended impact without estimating the potential off-target effects. 55 Fourth, although we applied SMR analysis combined with drug target eQTL to validate the result of drug target MR, different tissues may provide inconsistent results. 21 Most of these associations displayed similar trends, although some did not reach statistical significance, suggesting that the limited sample size of eQTL data may influence these findings. 21
Conclusion
This study supported that serum lipids (TG and HDL-C) were associated with IA, identified four IA-related lipid metabolic traits, and confirmed the effect of intervening HMGCR, ANGPTL3, and CETP targets in IA and its subtypes. In addition, SBP and DBP-related traits may be potential mediators between the causal association of lipid-lowering targets and IA; no safety concern was identified among these significant targets. However, more research is required to understand the potential mechanisms further, and the effects of these novel lipid-lowering targets in IA treatment need to be assessed in future clinical trials.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241265019 – Supplemental material for Novel insights into causal effects of serum lipids, lipid metabolites, and lipid-modifying targets on the risk of intracerebral aneurysm
Supplemental material, sj-docx-1-eso-10.1177_23969873241265019 for Novel insights into causal effects of serum lipids, lipid metabolites, and lipid-modifying targets on the risk of intracerebral aneurysm by Youjia Qiu, Bingyi Song, Ziqian Yin, Menghan Wang, Yuchen Tao, Minjia Xie, Aojie Duan, Zhouqing Chen, Ke Si and Zhong Wang in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241265019 – Supplemental material for Novel insights into causal effects of serum lipids, lipid metabolites, and lipid-modifying targets on the risk of intracerebral aneurysm
Supplemental material, sj-docx-2-eso-10.1177_23969873241265019 for Novel insights into causal effects of serum lipids, lipid metabolites, and lipid-modifying targets on the risk of intracerebral aneurysm by Youjia Qiu, Bingyi Song, Ziqian Yin, Menghan Wang, Yuchen Tao, Minjia Xie, Aojie Duan, Zhouqing Chen, Ke Si and Zhong Wang in European Stroke Journal
Supplemental Material
sj-xlsx-3-eso-10.1177_23969873241265019 – Supplemental material for Novel insights into causal effects of serum lipids, lipid metabolites, and lipid-modifying targets on the risk of intracerebral aneurysm
Supplemental material, sj-xlsx-3-eso-10.1177_23969873241265019 for Novel insights into causal effects of serum lipids, lipid metabolites, and lipid-modifying targets on the risk of intracerebral aneurysm by Youjia Qiu, Bingyi Song, Ziqian Yin, Menghan Wang, Yuchen Tao, Minjia Xie, Aojie Duan, Zhouqing Chen, Ke Si and Zhong Wang in European Stroke Journal
Footnotes
Acknowledgements
We thank the International Stroke Genetics Consortium (ISGC) (![]() ) Intracranial Aneurysm working group for providing the GWAS summary statistics of IA, which can be accessed through the ISGC Cerebrovascular Disease Knowledge Portal. We also thank the International Stroke Genetics Consortium (GLGC) and Richardson TG et al. for providing publicly available serum lipids and lipid metabolic summary statistics, respectively.
) Intracranial Aneurysm working group for providing the GWAS summary statistics of IA, which can be accessed through the ISGC Cerebrovascular Disease Knowledge Portal. We also thank the International Stroke Genetics Consortium (GLGC) and Richardson TG et al. for providing publicly available serum lipids and lipid metabolic summary statistics, respectively.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 82171309).
Ethical approval and informed consent
Not applicable.
Guarantor
Professor Zhong Wang take full responsibility for the integrity of the data presented in this manuscript.
Contributorship
Conceptualization and supervision: KS, ZQC, and ZW; Investigation and writing original draft: YJQ, BYS, and ZQY; Data curation, software, and formal analysis: MHW and YCT; Methodology and visualization: MJX and AJD; Review and editing: KS, ZW, and ZQC. Validation: YJQ, BYS, KS ZW, and ZQC. Resources and Funding acquisition: ZW. All authors confirmed that they had full access to all the data in the study and approved the final submitted manuscript. All authors have final responsibility for the decision to submit for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
