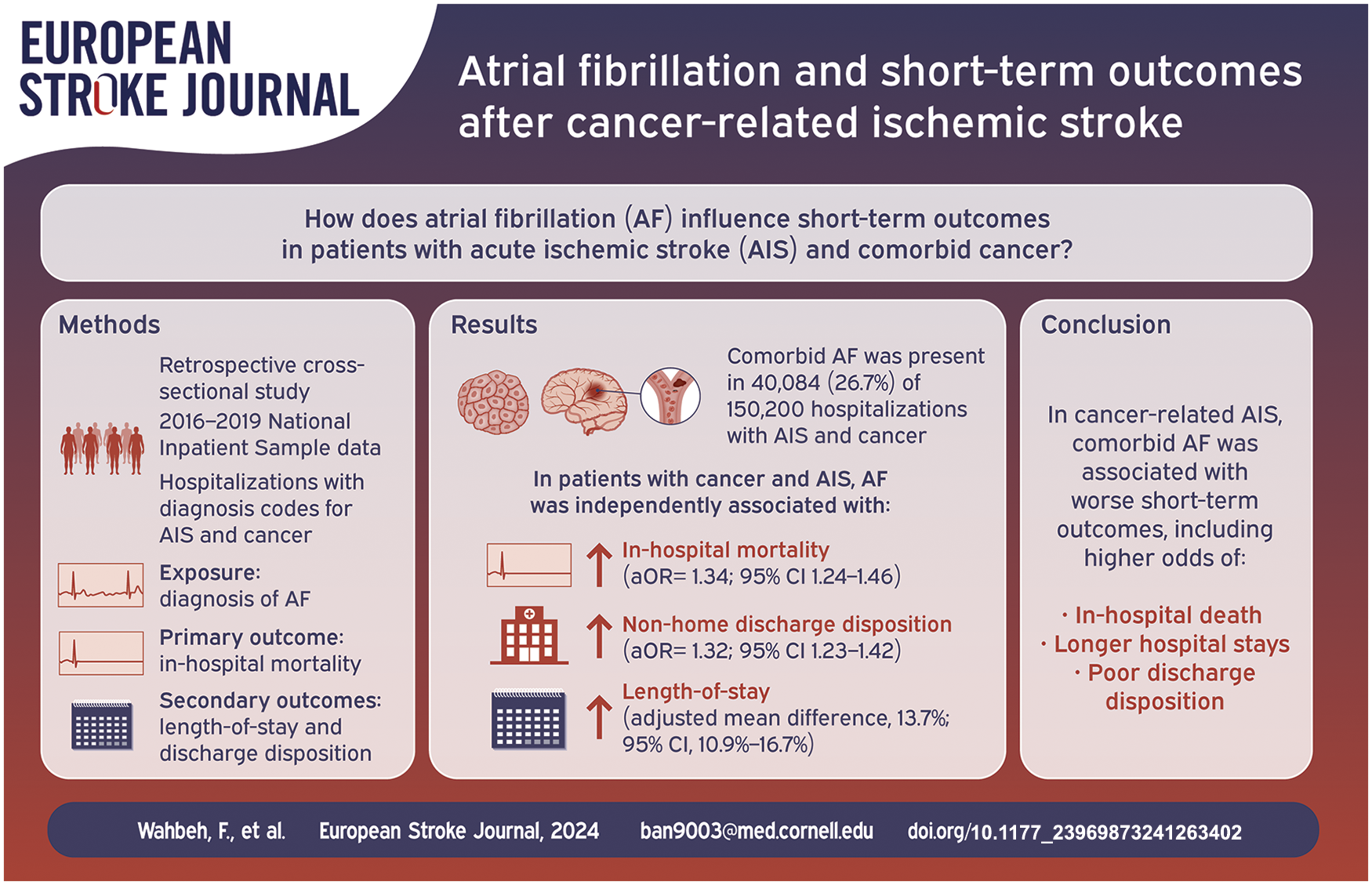
Abstract
Introduction:
Atrial fibrillation (AF) and cancer are each associated with worse outcomes in patients with acute ischemic stroke (AIS). Few studies have evaluated the impact of AF on outcomes of cancer-related stroke.
Patients and methods:
We conducted a retrospective cross-sectional study using the 2016–2019 National Inpatient Sample, identifying all hospitalizations with diagnosis codes for cancer and AIS. The primary exposure was a diagnosis of AF. The primary outcome was in-hospital mortality. The secondary outcomes were length-of-stay and discharge to non-home locations. We used multiple logistic and linear regression models, adjusted for age, gender, race-ethnicity, and the Charlson Comorbidity Index, to examine the association between AF and study outcomes.
Results:
Among 150,200 hospitalizations with diagnoses of cancer and AIS (mean age 72 years, 53% male), 40,084 (26.7%) included comorbid AF. Compared to hospitalizations without AF, hospitalizations with AF had higher rates of in-hospital mortality (14.8% [95% CI, 14.0%−15.6%] vs 12.1% [95% CI, 11.6%−12.5%]) and non-home discharge disposition (83.5% [95% CI, 82.7%−84.3%] vs 75.1% [95% CI, 74.5%−75.7%]) as well as longer mean length-of-stay (8.4 days [95% CI, 8.2–8.6 days] vs 8.2 days [95% CI, 8.0–8.3 days]). In multivariable analyses, AF remained independently associated with higher odds of in-hospital mortality (adjusted odds ratio [aOR], 1.34; 95% CI, 1.24–1.46), non-home discharge disposition (aOR, 1.32; 95% CI, 1.23–1.42), and longer length-of-stay (adjusted mean difference, 13.7%; 95% CI, 10.9%−16.7%).
Discussion and conclusion:
In cancer-related AIS, comorbid AF is associated with worse short-term outcomes, including higher odds for in-hospital mortality, poor discharge disposition, and longer hospital stays.
Introduction
Atrial fibrillation (AF), the most common cardiac arrhythmia and a major risk factor for acute ischemic stroke (AIS), is projected to affect 6–12 million people in the U.S. by 2050. 1 Notably, AF has been consistently linked to worse outcomes in patients with AIS. These patients experience more severe strokes and higher rates of hemorrhagic transformation, early recurrent stroke, poor functional outcomes, and death.2–4
Cancer-related stroke may be a unique subgroup of stroke with distinct characteristics and underlying mechanisms, and its outcomes appear to be primarily driven by cancer-related factors, rather than stroke-related factors. 5 Cancer is associated with worse outcomes in patients with AIS, including a substantially increased risk of recurrent thromboembolic events and death.6–8 Cancer may also be a risk factor for AF through pro-inflammatory pathways and the effects of cancer treatments.9,10 While previous studies have explored the impact of AF on stroke outcomes, few studies have evaluated the impact of AF on outcomes in patients with cancer-related stroke. 11
In light of this knowledge gap, our study aimed to investigate whether AF is associated with worse outcomes in patients with AIS and comorbid cancer. We hypothesized that in patients with AIS and cancer, AF would be associated with longer hospital stays and higher odds of in-hospital mortality and poor discharge disposition. Understanding the impact of AF in this patient population is crucial as it could lead to improved management and treatment strategies, ultimately enhancing patient outcomes.
Methods
Design
We performed a retrospective cross-sectional study using data from the National Inpatient Sample (NIS). The NIS, 12 developed for the Healthcare Cost and Utilization Project (HCUP), is a publicly available all-payer survey of hospitalizations across the U.S. The NIS 12 provides nationally representative estimates pertaining to outcomes, utilization, cost, and quality. NIS data from 2016 to 2019 was used for this study. All analyses were conducted according to HCUP data use agreements. As the NIS is publicly available and deidentified, our analysis was certified as exempt from institutional review board review. The data from this study can be obtained through direct application to HCUP, while the analytical code can be accessed by request of the corresponding author. We adhered to STROBE (Strengthening the Reporting of Observational studies in Epidemiology) guidelines in the reporting of this study.
Population
We used diagnoses codes from the International Classification of Diseases (ICD)-10 system, as performed in prior relevant studies.13,14 We extracted all records of hospitalizations with diagnoses of AIS (ICD-10 codes I63.0-I63.9) and cancer (ICD-10 codes C00−C96). The latter included all malignant solid, hematological, and primary central nervous system cancers. Localized skin cancers (e.g. squamous cell and basal cell carcinomas) were excluded. The ICD-10 diagnosis code I63 has been previously shown to have a positive predictive value of >92% for AIS compared to electronic health record review. Claims-based diagnoses of cancer have been validated to have a positive predictive value of 80%–90% when compared to cancer registry data.
Measurements
The primary exposure of interest was a diagnosis of AF, including all forms of AF, identified through ICD-10 codes I48.0-I48.92. The primary study outcome was in-hospital mortality. The secondary outcomes were length-of-stay (LoS) and discharge disposition. A tertiary outcome was mechanical ventilation, which is a marker of increased stroke severity and poor outcomes after stroke. 15
The NIS 16 provides hospital LoS and vital status upon hospital discharge as standard variables. For discharge disposition, a favorable outcome was defined as routine discharge to home, while the following were considered unfavorable outcomes: discharge to an acute rehabilitation hospital, discharge to a skilled nursing facility, discharge to hospice, or in-hospital death. We excluded hospitalizations with missing or unknown discharge disposition data.
We also collected data on age, sex, race-ethnicity, history of hypertension, history of tobacco use, and the Charlson et al.17,18 comorbidity index. The Charlson comorbidity index, a validated composite marker of overall burden of disease, encompasses the following conditions: myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic pulmonary disease, rheumatologic disease, peptic ulcer disease, hemiplegia, chronic kidney disease, solid and hematological malignancies, HIV infection, liver disease, and diabetes.
Analyses
Descriptive statistics were used to examine patient characteristics and to report proportions, means (standard deviation [SD]), and medians (interquartile range [IQR]), as appropriate. Continuous variables were compared with Student’s t-test or the Wilcoxon rank sum test, while categorical variables were compared with the chi-square test or Fisher’s exact test, depending on data distribution. Sampling weights provided by the NIS were used to determine U.S. national estimates for study outcomes. Multiple logistic regression models were employed to analyze the associations between AF and the outcomes of in-hospital mortality, discharge disposition, and mechanical ventilation. Multivariable log-linear regression models were used to analyze the association between AF and LoS. All models were adjusted for age, sex, race-ethnicity, and the Charlson comorbidity index.
Various secondary analyses were performed. First, subgroup analyses were performed according to broad cancer type, categorized as solid tumor, hematological, or primary central nervous system cancers; and cancer types considered high risk for thromboembolism according to the Khorana score (i.e. cancers originating from the pancreas, stomach, lung, gynecologic system, bladder, testicles, or lymphoma). Second, subgroup analyses were performed among men versus women and White versus non-White race-ethnicities. Third, a sensitivity analysis, restricted to patients with recorded NIH stroke scale (NIHSS) values, compared the initial NIHSS, a measure of stroke severity, between study groups. Fourth, in separate sensitivity analyses, we limited the cohort to hospitalizations in which (A) patients were treated with intravenous thrombolysis, (B) patients with ICD-10 diagnosis codes for cerebral vein thrombosis (I63.6) were excluded, (C) NIHSS scores were available, or (D) patients had an ICD-10 diagnosis code specific to embolic stroke (I63.1, I63.4).
Statistical analyses were performed by C.Z. using Stata, version 15.1. An alpha error of less than 0.05 was considered statistically significant. C.Z. and H.K. had full access to all the data used in the study and take responsibility for its integrity and the data analysis.
Results
Clinical characteristics
This analysis included 150,200 total hospitalizations with diagnosis codes for cancer and AIS. The mean age was 71.5 years (SD, 12.2); 52.9% were men, and 109,875 (73.2%) were White (Table 1). Regarding broad cancer type, 117,875 (78.5%) of hospitalizations had solid tumor cancers, 30,590 (20.4%) had hematological cancers, and 4890 (3.3%) had primary central nervous system cancers. AF was identified in 40,084 hospitalizations, accounting for 26.7% of all hospitalizations with cancer and AIS.
Cohort characteristics, stratified by the presence of atrial fibrillation.
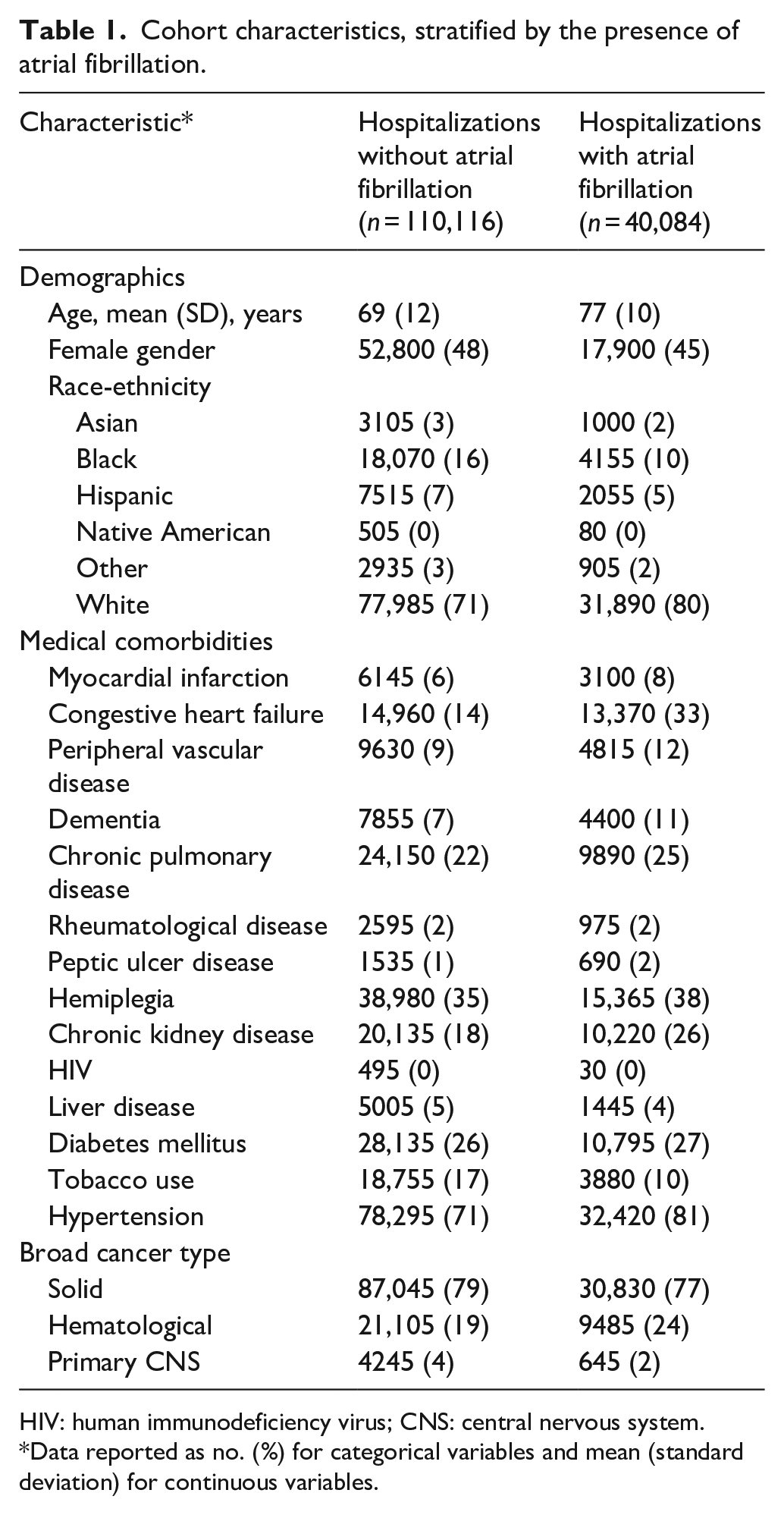
HIV: human immunodeficiency virus; CNS: central nervous system.
Data reported as no. (%) for categorical variables and mean (standard deviation) for continuous variables.
Compared to hospitalizations without AF, hospitalizations with AF included patients who were on average older, more often male and White, had less tobacco use, and had higher rates of cardiovascular disorders, dementia, and hemiplegia. Intravenous thrombolysis was administered in 2440 (6.1%) hospitalizations with AF, compared to 5380 (4.9%) hospitalizations without AF.
Primary outcome analyses
Among all hospitalizations with both AIS and cancer, in-hospital mortality occurred in 19,230 hospitalizations (12.8%; 95% CI, 12.4%−13.2%). In-hospital mortality occurred in 14.8% (95% CI, 14.0%−15.6%) of hospitalizations with comorbid AF, compared to 12.1% (95% CI, 11.6%−12.5%) of hospitalizations without AF (odds ratio [OR], 1.27; 95% CI, 1.18–1.37; p < 0.001) (Figure 1(a), Table 2). After controlling for demographics and comorbidities, comorbid AF remained significantly associated with in-hospital death (adjusted OR, 1.34; 95% CI, 1.24–1.46).
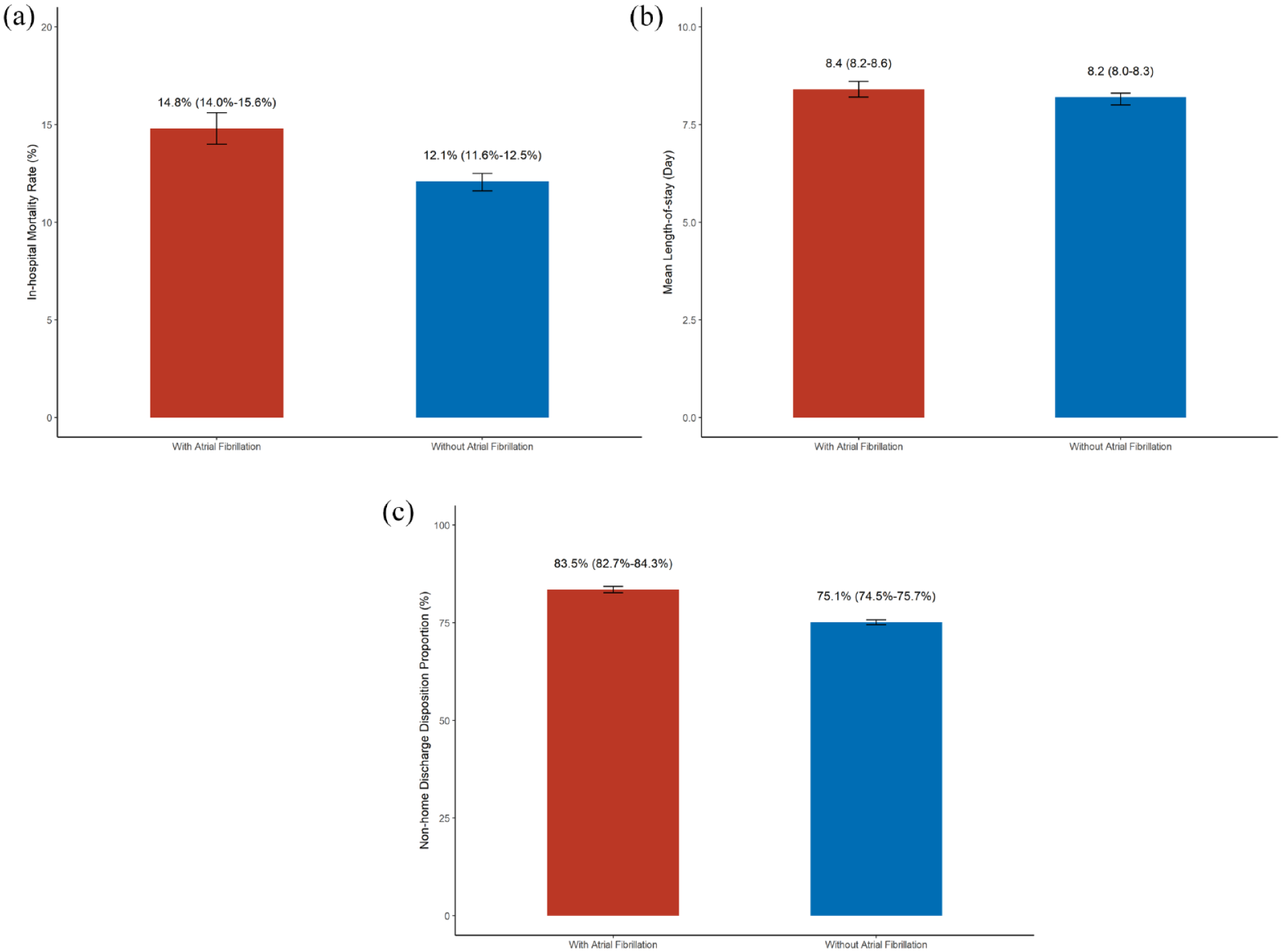
Clinical outcomes among hospitalizations with cancer and acute ischemic stratified by the presence of atrial fibrillation. Bar graphs demonstrating differences in clinical outcomes among hospitalizations with cancer and acute ischemic stroke according to the presence of comorbid atrial fibrillation. Patients with diagnoses of atrial fibrillation are depicted in red, while patients without diagnoses of atrial fibrillation are depicted in blue. 95% confidence intervals are displayed through brackets. (a) In-hospital mortality rate. (b) Mean length-of-stay. (c) Non-home discharge disposition proportion.
Short-term clinical outcomes, stratified by the presence of atrial fibrillation.
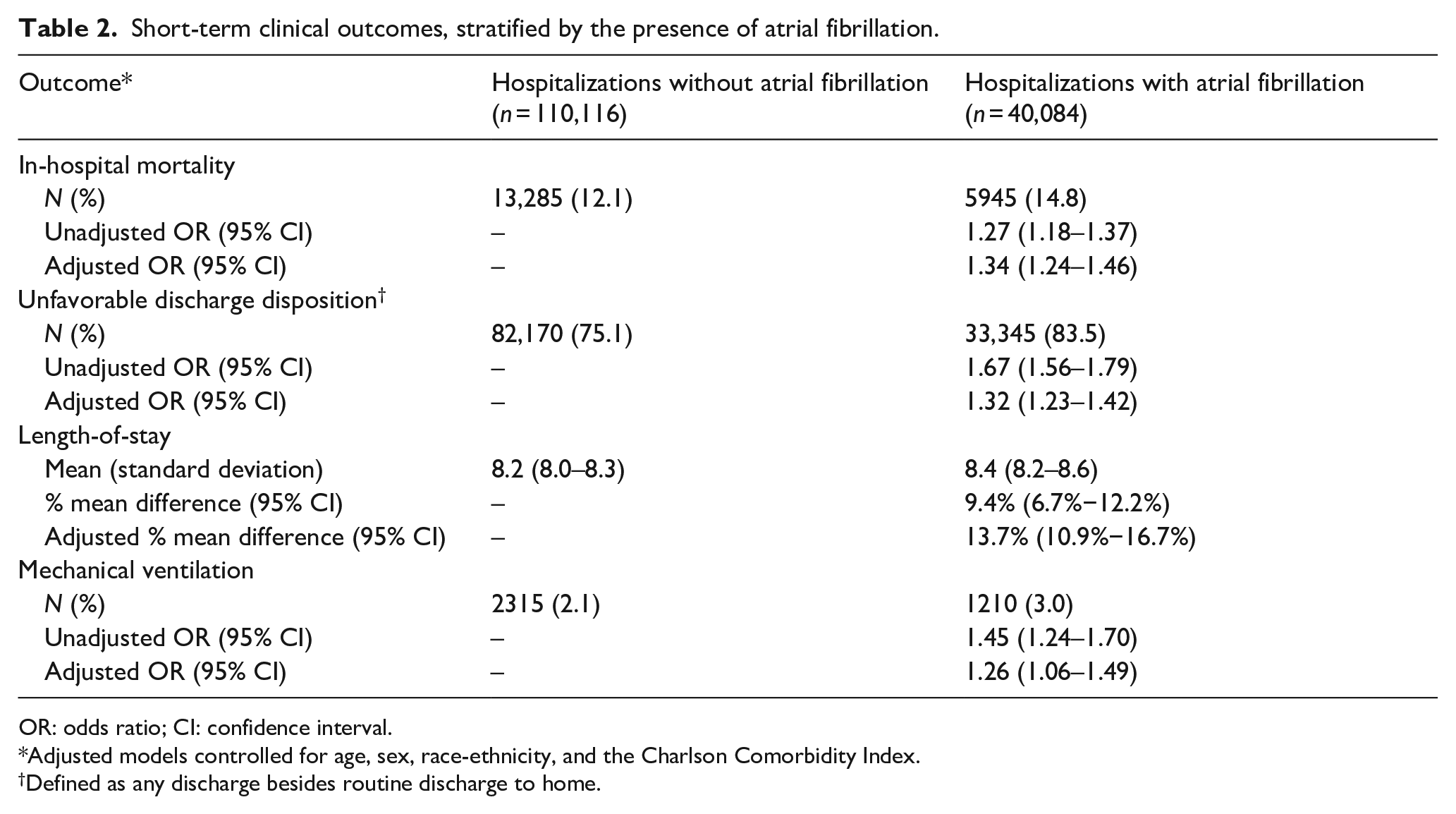
OR: odds ratio; CI: confidence interval.
Adjusted models controlled for age, sex, race-ethnicity, and the Charlson Comorbidity Index.
Defined as any discharge besides routine discharge to home.
Secondary and tertiary outcome analyses
The mean LoS across all hospitalizations was 8.2 days (SD, 10.2). For hospitalizations with AF, the mean LoS was 8.4 days (SD, 9.0), while hospitalizations without AF had a mean LoS of 8.2 days (SD, 10.7) (p = 0.031). In multivariable analysis, the presence of AF, when compared to no AF, was associated with an estimated 13.7% longer hospital stay (95% CI, 10.9%−16.7%) (Figure 1(b)).
Among all hospitalizations, 115,515 (77.4%) had an unfavorable discharge disposition. Unfavorable discharge disposition occurred in 83.5% (95% CI, 82.7%−84.3%) of hospitalizations with AF, compared to 75.1% (95% CI, 74.5%−75.7%) of hospitalizations without AF (OR, 1.67; 95% CI, 1.56–1.79) (Figure 1(c)). The association between AF and unfavorable discharge disposition persisted after controlling for potential confounders (adjusted OR, 1.32; 95% CI, 1.23–1.42).
Mechanical ventilation was administered in 3525 (2.4%) of the overall cohort, including 1210 (3.0%) hospitalizations with AF and 2315 (2.1%) hospitalizations without AF (OR, 1.45; 95% CI, 1.24–1.70). In multivariable analysis, hospitalizations with AF remained significantly more likely to require mechanical ventilation (adjusted OR, 1.26; 95% CI, 1.06–1.49).
Subgroup and sensitivity analyses
Our findings remained consistent across subgroup analyses based on demographics (Supplemental Table 1) and cancer type (Supplemental Table 2), including cancer types considered high risk for thromboembolism. Among 34,485 hospitalizations with recorded NIHSS scores (23.0% of all hospitalizations), the median initial scores were 5 (IQR, 2–13) among patients with AF versus 4 (IQR, 2–10) among patients without AF (p < 0.001). When restricting the cohort to hospitalizations with intravenous thrombolysis treatment, hospitalizations excluding codes for cerebral vein thrombosis, hospitalizations with available NIHSS scores, or hospitalizations with codes for embolic stroke, the associations between comorbid AF and study outcomes were similar to those of the primary analyses (Supplemental Tables 3–6).
Discussion
In a retrospective cross-sectional study of 150,200 U.S. hospitalizations with cancer and AIS, presence of AF was independently associated with an increased odds for in-hospital death, unfavorable discharge disposition, and need for mechanical ventilation. Length-of-stay was also longer in hospitalizations with AF; however, the mean difference between groups was only 0.2 days, limiting clinical relevance. These study findings were robust across subgroups stratified by demographics and cancer types, and when the population was restricted to patients with embolic stroke or those treated with intravenous thrombolysis.
The relationship between AF and adverse outcomes in patients with cancer-related ischemic stroke is likely multifaceted. First, both AF and cancer independently increase the risk of AIS, by promoting thromboembolism, with AF acting through an irregular heart rhythm and underlying atrial myopathy and cancer through inflammation and cancer-related coagulopathy.19–21 Second, both AF and cancer are independently associated with worse outcomes in patients with AIS, and with AF this is partly due to higher stroke severity and more large vessel occlusions.2–4,6–8,22–24 Third, shared risk factors between AF and cancer, such as advanced age, hypertension, diabetes, and smoking create a synergistic effect, worsening outcomes in patients with cancer-related stroke who also have AF.25–28 Fourth, cancer treatments, including chemotherapy and immunotherapy, have cardiotoxic effects that may exacerbate the arrhythmia associated with AF and increase thromboembolism risk. 9
Several studies have explored the relationships between AF and either stroke or cancer independently, but few have delved into the direct impact of AF on outcomes in patients with stroke and comorbid cancer.29–31 Some studies did encompass all three conditions but focused on aspects other than outcomes or prognosis. For instance, studies by Aspberg et al and Elbadawi et al. assessed the impact of cancer on the incidence of ischemic stroke in AF patients, while other studies examined the impact of AF on the incidence of ischemic stroke in cancer patients.32–35 A study of 272 patients with cancer reported that comorbid AF increased the risk of stroke or transient ischemic attack nearly eight-fold compared to no AF. 33 Other studies have focused on the use of oral anticoagulants in patients with AF and stroke, with and without concomitant cancer. 36 It is important to note that none of these studies directly addressed the impact of AF on outcomes in patients with stroke and comorbid cancer.
Our study builds on previous research by Nam et al. 37 which investigated the impact of AF and intracranial atherosclerosis on the prognosis of ischemic stroke in 116 patients with active cancer and AIS at a single center in Korea. In this analysis, patients with comorbid AF, compared to those without AF, were numerically but not significantly more likely to experience early neurological deterioration (OR, 3.26; 95% CI, 0.95–11.22; p = 0.06) and unfavorable functional outcomes measured by modified Rankin Scale scores ⩾3 (26% vs 13%, p = 0.09). 37 Our findings are consistent with this study, underscoring the association between AF and poor outcomes in patients with ischemic stroke and comorbid cancer. However, in contrast to the Nam et al. study, ours benefited from a substantially larger sample size thereby providing sufficient power to demonstrate a statistically significant impact of AF on clinical outcomes. Additionally, our study evaluated a broader set of outcome measures to more fully evaluate the impact of AF on cancer-related AIS outcomes. Lastly, our study used a multicenter design with a heterogeneous patient population, thereby enhancing the generalizability of our findings.
The impact of AF on stroke outcomes may be different in patients with cancer than in patients without cancer. Among patients with AIS recorded in the Dijon Stroke Registry from 2014–2017, the conferred ORs for death with AF were 2.8 in univariate analysis and 1.9 in multivariable analysis adjusting for demographics and comorbidities. 24 The corresponding ORs between AF and higher modified Rankin Scale scores were 2.4 and 1.7, respectively. 24 In the Japanese Multicenter Stroke Investigators’ Collaboration Registry, 45% of patients with AIS and AF were discharged home versus 62% of patients with AIS and no AF, an absolute risk difference of 17%. 3 In comparison, among patients with cancer and AIS, we found that AF conferred an adjusted OR of 1.34 for death and an absolute risk decrease of 8% for discharge to home. While the lower conferred risks in our study could be due to differences between patient populations, analytical techniques, or chance, it is also possible that AF has less influence on stroke outcomes in patients with cancer than in those without cancer and that cancer-related prognostic factors play a larger role in determining stroke outcomes than AF does.
Our study has limitations. First, the cross-sectional design restricted our ability to infer causal associations. Second, the reliance on claims data to define the patient population and determine outcomes introduced the possibility of measurement error and ascertainment bias. We attempted to mitigate this concern by using validated diagnostic codes used in prior studies. Third, we were unable to determine the type and chronicity of AF, whether patients were previously or subsequently anticoagulated for AF or had prior cerebrovascular events, and the mechanism of index stroke. Nor was it possible to determine the stage, activity, or treatments of the underlying cancers. Fourth, due to the known incomplete documentation of NIHSS scores within ICD-10 codes in the U.S., NIHSS scores were only available for 23% of hospitalizations in our cohort, thereby limiting our sensitivity analysis comparing stroke severity between patients with and without AF. Fifth, we were unable to adjust for frailty or other unmeasured factors that could have confounded the association between AF and study outcomes. Sixth, we used the NIS to conduct this study and this dataset is limited by its inability to conclusively distinguish acute from chronic conditions due to its reliance on claims-based diagnoses, the lack of patient linkage across hospitalizations so that some patients contribute multiple hospitalizations during a study period, the restriction to a 20% sample of U.S. hospitalizations with multiple changes in the sampling design used since its construction, and the lack of data on outpatient encounters or observation-only stays so that diseases and procedures that are predominantly diagnosed in ambulatory settings are underrepresented. The NIS also lacks data on premorbid residential status. Additionally, we were unable to assess granular outcomes, such as the modified Rankin Scale score at discharge or at 3-months, the presence of symptomatic hemorrhagic transformation, and the cause of death. Similarly, we lacked data on D-dimer levels and other laboratory biomarkers for cancer-related stroke.
We found that comorbid AF in cancer-related ischemic stroke is associated with higher odds for in-hospital mortality, unfavorable discharge disposition, and longer hospital stay. These findings emphasize the importance of early detection and management of AF in patients with cancer, especially as prior data have indicated that cancer patients with AF are anticoagulated less frequently than noncancer patients with AF. 36 A proactive treatment approach that carefully balances the benefits of stroke prevention with its bleeding risks, considering the interactions between cancer therapies and anticoagulation, is essential for patients with cancer-related stroke and comorbid AF. This approach requires close collaboration between neurologists, oncologists, and cardiologists to optimize patient care. Future prospective studies are warranted to uncover the mechanisms through which AF worsens outcomes in patients with ischemic stroke and comorbid cancer.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241263402 – Supplemental material for Atrial fibrillation and short-term outcomes after cancer-related ischemic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873241263402 for Atrial fibrillation and short-term outcomes after cancer-related ischemic stroke by Farah Wahbeh, Cenai Zhang, Morin Beyeler, Jed H Kaiser, Vanessa Liao, Anokhi Pawar, Hooman Kamel and Babak B Navi in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241263402 – Supplemental material for Atrial fibrillation and short-term outcomes after cancer-related ischemic stroke
Supplemental material, sj-docx-2-eso-10.1177_23969873241263402 for Atrial fibrillation and short-term outcomes after cancer-related ischemic stroke by Farah Wahbeh, Cenai Zhang, Morin Beyeler, Jed H Kaiser, Vanessa Liao, Anokhi Pawar, Hooman Kamel and Babak B Navi in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Navi has received personal fees for serving on an adjudication committee for MindRhythm Inc and for serving as a neurology expert witness. Dr. Kamel reports serving as a PI for the ARCADIA trial (NIH/NINDS U01NS095869), which received in-kind study drug from the BMS-Pfizer Alliance for Eliquis® and ancillary study support from Roche Diagnostics; other funding from NIH (R01HL144541, R01NS123576, U01NS106513); serving as Deputy Editor for JAMA Neurology; serving on clinical trial steering/executive committees for Medtronic, Janssen, and Javelin Medical; serving on endpoint adjudication committees for AstraZeneca, Novo Nordisk, and Boehringer Ingelheim; and household ownership interests in TETMedical, Spectrum Plastics Group, and Burke Porter Group. The other authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Ethical approval
Our analysis was certified as exempt from institutional review board review.
Guarantor
Dr. Navi.
Contributorship
FW and BN researched literature and conceived the study. FW, JK, VL, AP, HK, and BN gained ethical approval. CZ performed data analysis. FW and BN wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final submitted version.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
