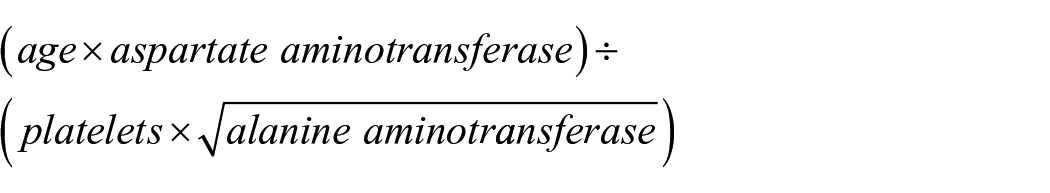Abstract
Background:
Cirrhosis is associated with an increased risk of hemorrhagic stroke. Liver fibrosis, typically a silent condition, is antecedent to cirrhosis. The objective of this study was to test the hypothesis that elevated Fibrosis-4 (FIB-4) index, indicating a high probability of liver fibrosis, is associated with an increased risk of hemorrhagic stroke.
Methods:
We performed a cohort analysis of the prospective United Kingdom Biobank cohort study. Participants 40–69 years old were enrolled between 2007 and 2010 and had available follow-up data until March 1, 2018. We excluded participants with prevalent hemorrhagic stroke or thrombocytopenia. High probability of liver fibrosis was defined as having a value >2.67 of the validated FIB-4 index. The primary outcome was hemorrhagic stroke (intracerebral or subarachnoid hemorrhage), defined based on hospitalization and death registry data. Secondary outcomes were intracerebral and subarachnoid hemorrhage, separately. We used Cox proportional hazards models to evaluate the association of FIB-4 index >2.67 with hemorrhagic stroke while adjusting for potential confounders including hypertension, alcohol use, and antithrombotic use.
Results:
Among 452,994 participants (mean age, 57 years; 54% women), approximately 2% had FIB-4 index >2.67, and 1241 developed hemorrhagic stroke. In adjusted models, FIB-4 index >2.67 was associated with an increased risk of hemorrhagic stroke (HR, 2.0; 95% CI, 1.6–2.6). Results were similar for intracerebral hemorrhage (HR, 2.0; 95% CI, 1.5–2.7) and subarachnoid hemorrhage (HR, 2.2; 95% CI, 1.5–3.5) individually.
Conclusions:
Elevated FIB-4 index was associated with an increased risk of hemorrhagic stroke.
Introduction
The prevalence of chronic liver conditions is increasing steadily, with an estimated 1.5 billion people affected worldwide.1,2 Decompensated cirrhosis, the clinically apparent, end stage manifestation of chronic liver disease, is associated with a variety of well-recognized extrahepatic manifestations, including an increased risk of hemorrhagic stroke.3–7 Liver fibrosis develops in response to chronic liver injury from a variety of causes and is an often subclinical condition that can precede clinically apparent decompensated cirrhosis. 8 While individuals with liver fibrosis commonly have normal liver enzymes and lack clinical signs and symptoms of liver disease,9–11 liver fibrosis may be associated with cerebrovascular disease. Liver fibrosis was associated with expansion of intracerebral hemorrhage volume12,13 and with hemorrhagic transformation of ischemic stroke. 14 However, whether liver fibrosis generally, like its severe form in cirrhosis, is a risk factor for incident hemorrhagic stroke is not known. We therefore sought to test the hypothesis that elevated Fibrosis-4 (FIB-4) index, indicating high probability of liver fibrosis, is associated with an increased risk of hemorrhagic stroke in a large cohort of participants from the United Kingdom Biobank.
Methods
Design
This is a retrospective cohort analysis using prospectively-collected data from the UK Biobank, which is a prospective cohort study that recruited approximately 500,000 participants between 40 and 69 years of age from across the United Kingdom. 15 Baseline assessments occurred from 2007 to 2010 and included detailed health questionnaires, physical measurements, and collection of biological samples for laboratory tests and genotyping. Participants are followed longitudinally, including through linked hospital record and death data. Based on these linked data, the UK Biobank includes time-to-event data for common health outcomes, including hemorrhagic stroke, in addition to death and loss to follow-up. UK Biobank data are subjected to extensive data stewardship measures designed to ensure data integrity. 16
Standard protocols and approvals
The UK Biobank obtained written informed consent from participants. The Weill Cornell Medicine institutional review board certified these analyses of deidentified data as exempt from review. The data that support the findings of this study are made available in an anonymized format to qualified investigators upon application to the UK Biobank (https://www.ukbiobank.ac.uk/). Analytic methods will be made available upon reasonable request. We followed the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.
Population
From among the approximately 500,000 participants in the UK Biobank, we included those who attended a baseline assessment visit and excluded participants with prevalent hemorrhagic stroke or missing exposure variable data. People with prevalent hemorrhagic stroke were excluded because our objective was to maintain temporality between the baseline FIB-4 measurement and future hemorrhagic stroke. We also excluded participants with possible acute hepatitis (aspartate aminotransferase or alanine aminotransferase ⩾250 IU/L) 17 or thrombocytopenia (<100,000/mL) at baseline.
Measurements
The exposure variable was probable liver fibrosis, as measured by the FIB-4 index. We calculated the FIB-4 index according to its formula, with age in years, aminotransferases in U/L, and platelet count in 109/L 18 :
We classified participants with FIB-4 index >2.67 as having high probability of advanced liver fibrosis.18,19 The FIB-4 index has been widely validated, including specifically in the general population.20–24 Our approach of evaluating risk of liver fibrosis in the general population, irrespective of underlying etiology, is consistent with other efforts to understand the role of liver fibrosis broadly in human disease.25–28 This approach accounts for the real-world overlap between chronic liver conditions such as alcoholic and presumed nonalcoholic fatty liver disease. 29
The primary outcome was incident hemorrhagic stroke, a composite of intracerebral or subarachnoid hemorrhage. Intracerebral and subarachnoid hemorrhage were also assessed individually as secondary outcomes. The UK Biobank dataset contains time-to-event data for hemorrhagic stroke derived using algorithms 30 based on hospital records and mortality register data and standard International Classification of Diseases versions 9 and 10 diagnosis codes for these conditions. 31
Covariates were demographics, comorbidities, and antithrombotic medication use. Demographics were age, sex, and self-reported ethnicity. The UK Biobank provides an “ethnicity” variable based on which participants were categorized as Asian, Black, other/multiple, and White, reflecting UK demographics. Hypertension was defined as a self-reported diagnosis of hypertension, systolic blood pressure ⩾130 mmHg, diastolic blood pressure ⩾80 mmHg, or use of anti-hypertensive medications. 32 Diabetes mellitus was defined as a self-reported diagnosis of diabetes, hemoglobin A1c ⩾6.5%, or use of diabetes medications. 33 Dyslipidemia was defined as use of lipid-lowering medications or total cholesterol ⩾200 mg/dL. 34 Body mass index was categorized using standard cutoffs for normal (18.5–24.9 kg/m2), underweight (<18.5 kg/m2), overweight (25–29.9 kg/m2), and obesity (⩾30 kg/m2). Tobacco use was categorized as never-smoker versus smoker (current or former). Alcohol use was categorized based on consumption frequency as non-drinker, less than once weekly, 1–4 times per week, and daily or almost daily. Patients reporting any prevalent chronic liver condition, including cirrhosis, alcoholic liver disease, hepatitis and non-infectious liver disease during their nurse-led interview were categorized as having a clinically known liver condition. For the purposes of interaction term testing by APOE genotype, given the possible relevance of APOE genotype for intracerebral hemorrhage,35–37 the APOE gene was directly genotyped for UK Biobank participants, and we used standard definitions to ascertain APOE genotypes based on rs429358 and rs7412 alleles.38,39 We defined baseline antithrombotic medication use as use of any antiplatelet or anticoagulant medication, ascertained from nurse-led medication inventories taken for all participants in the UK Biobank. All variables used for calculation of the exposure variable and covariates were ascertained from the baseline visit in which subjects were initially enrolled in the UK Biobank study.
Statistical analyses
Survival analysis was used to estimate cumulative risks, with observations censored at the time of death, loss to follow-up, or at the end of available follow-up in our dataset (March 1, 2018). The primary statistical analysis entailed use of Cox proportional hazards models to compare the risk of hemorrhagic stroke in participants with FIB-4 index >2.67 and ⩽2.67 by calculating cause-specific hazard ratios. Proportionality of hazards was confirmed by visual inspection of survival and log-log plots. We specified the following models. Model 1 was unadjusted. Model 2 was adjusted for age, sex, and ethnicity. Model 3 was additionally adjusted for hypertension, diabetes, dyslipidemia, body mass index category, smoking status, alcohol consumption frequency, and antithrombotic medication use.
We performed several sensitivity analyses. First, we excluded all participants with any known chronic liver condition at baseline to evaluate the association between elevated risk of subclinical liver fibrosis and incident hemorrhagic stroke. Second, we excluded participants taking any antithrombotic medication. Third, we excluded participants with platelet count <150,000 per microliter, a more conservative cut-off than used for the primary analysis study population. Lower platelet counts mathematically increase FIB-4 scores; this sensitivity analysis was intended to evaluate the stability of our results after more stringently excluding participants with elevated FIB-4 scores due to any level of thrombocytopenia. In addition, due to observed differences between participants with FIB-4 index >2.67 and ⩽2.67 in multiple baseline characteristics including age, which is a component of the exposure variable, we performed confirmatory matched-cohort analyses. Participants with FIB-4 index >2.67 and ⩽2.67 were 1:1 matched on age (exact match), sex, hypertension, and antithrombotic medication using greedy nearest-neighbor matching. Conditional Cox proportional hazards regression models were then used to estimate hazard ratios for the primary and secondary outcomes in the matched cohort. Last, given the possible relevance of APOE ε4 and ε2 alleles for hemorrhagic stroke risk,35–37 we performed tests of interaction by APOE genotype for the primary outcome of hemorrhagic stroke in Model 3. The APOE genotype variables were specified as APOE ε4 carriers (⩾1 ε4 alleles), APOE ε4/ε4, APOE ε2 carriers (⩾1 ε2 alleles), and APOE ε2/ε2, all with APOE ε3/ε3 as the reference group. The threshold of statistical significance was α = 0.05. Analyses were performed by NSP and CZ using SAS Version 9.4 (Cary, NC) and R 4.0.5 (R Core Team, Vienna, Austria).
Results
Participant characteristics
From among 502,567 participants in the UK Biobank, we included 452,994 participants after applying study exclusions (Figure 1). The mean age of participants was 56.5 (SD, 8.1) years, and 245,453 (54%) were women. Overall, 72% had hypertension, 6% had diabetes, and 16% had antithrombotic use. Clinically known liver conditions were uncommon, reported by 2578 (0.6%) participants. The overall median FIB-4 score was 1.23 (IQR, 0.97–1.56), falling in the indeterminate range. However, 8787 (1.94%; 95% CI, 1.90–1.98) participants had a FIB-4 index >2.67. Participants with FIB-4 index >2.67 were older, more likely men, had a higher prevalence of hypertension and diabetes, and more often reported daily alcohol consumption (Table 1).
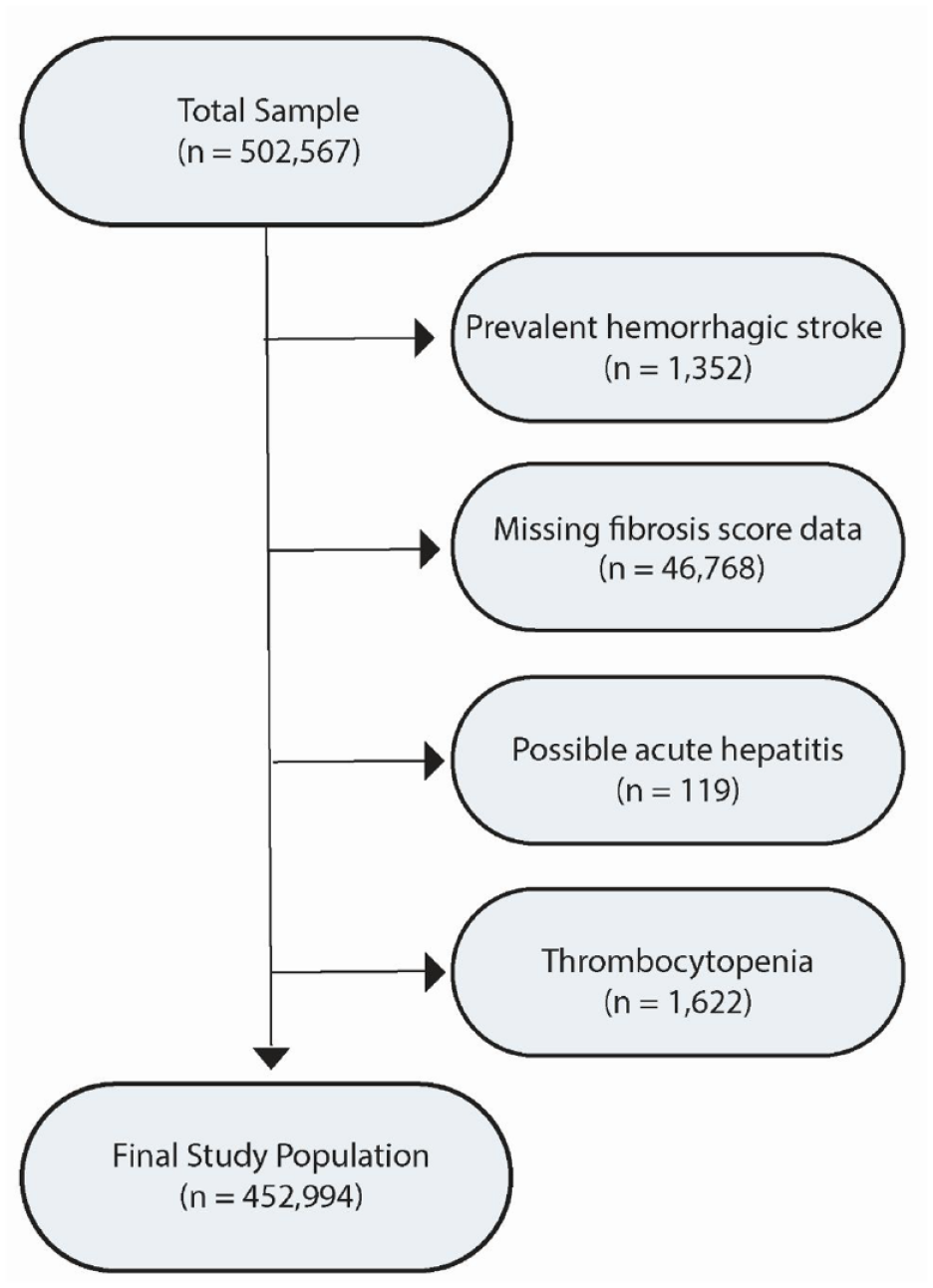
Participant selection flow diagram. From among participants in the UK Biobank, we excluded participants with missing available liver fibrosis score data, possible acute hepatitis (aspartate or alanine aminotransferase ⩾250 IU/L), thrombocytopenia (platelet count <100,000/mL), and prevalent hemorrhagic stroke. Some participants had multiple reasons for exclusion.
Baseline characteristics a of participants in the UK Biobank, stratified by FIB-4 index.
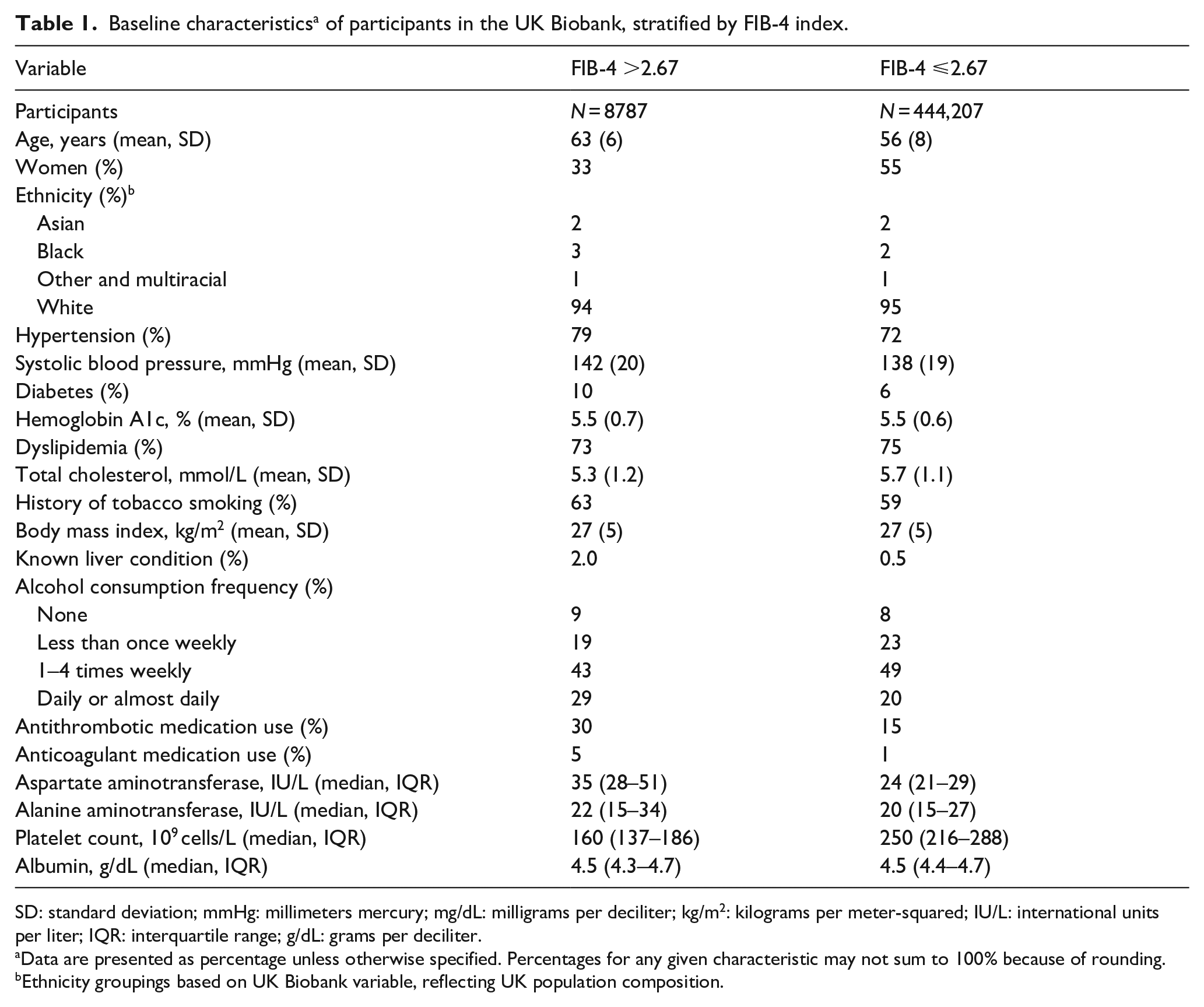
SD: standard deviation; mmHg: millimeters mercury; mg/dL: milligrams per deciliter; kg/m2: kilograms per meter-squared; IU/L: international units per liter; IQR: interquartile range; g/dL: grams per deciliter.
Data are presented as percentage unless otherwise specified. Percentages for any given characteristic may not sum to 100% because of rounding.
Ethnicity groupings based on UK Biobank variable, reflecting UK population composition.
Primary outcome analyses
During a median follow-up of 9.0 (IQR, 8.3–9.7) years, there were 1241 incident hemorrhagic strokes (719 intracerebral hemorrhage, 522 subarachnoid hemorrhage). The rate of hemorrhagic stroke per 1000 person-years was 0.89 (95% CI, 0.70–1.14) in participants with FIB-4 index >2.67 and 0.30 (95% CI, 0.28–0.31) in those with FIB-4 index ⩽2.67 (Figure 2). In an unadjusted Cox proportional hazards model, participants with FIB-4 index >2.67 had an increased risk of incident hemorrhagic stroke compared to participants with FIB-4 index ⩽2.67 (hazard ratio (HR), 3.0; 95% CI, 2.4–3.9). This association remained significant in adjusted models, including when adjusting for demographics and shared risk factors (HR, 2.0; 95% CI, 1.6–2.6) (Table 2). Our findings were unchanged in three separate sensitivity analyses after excluding participants with any clinically known liver conditions at baseline (HR, 2.0; 95% CI, 1.6–2.6), baseline antithrombotic use (HR, 1.8; 95% CI, 1.3–2.6), and platelet count <150,000 per microliter (HR, 2.1; 95% CI, 1.6–2.9) (Table 2). Last, findings remained significant in a complementary matched cohort analysis that matched participants with FIB-4 index >2.67 and ⩽2.67 on age, sex, hypertension, and antithrombotic use (HR, 2.6; 95% CI, 1.6–4.6). There were no statistically significant interactions to suggest interaction by APOE genotype (all p values ⩽0.20).
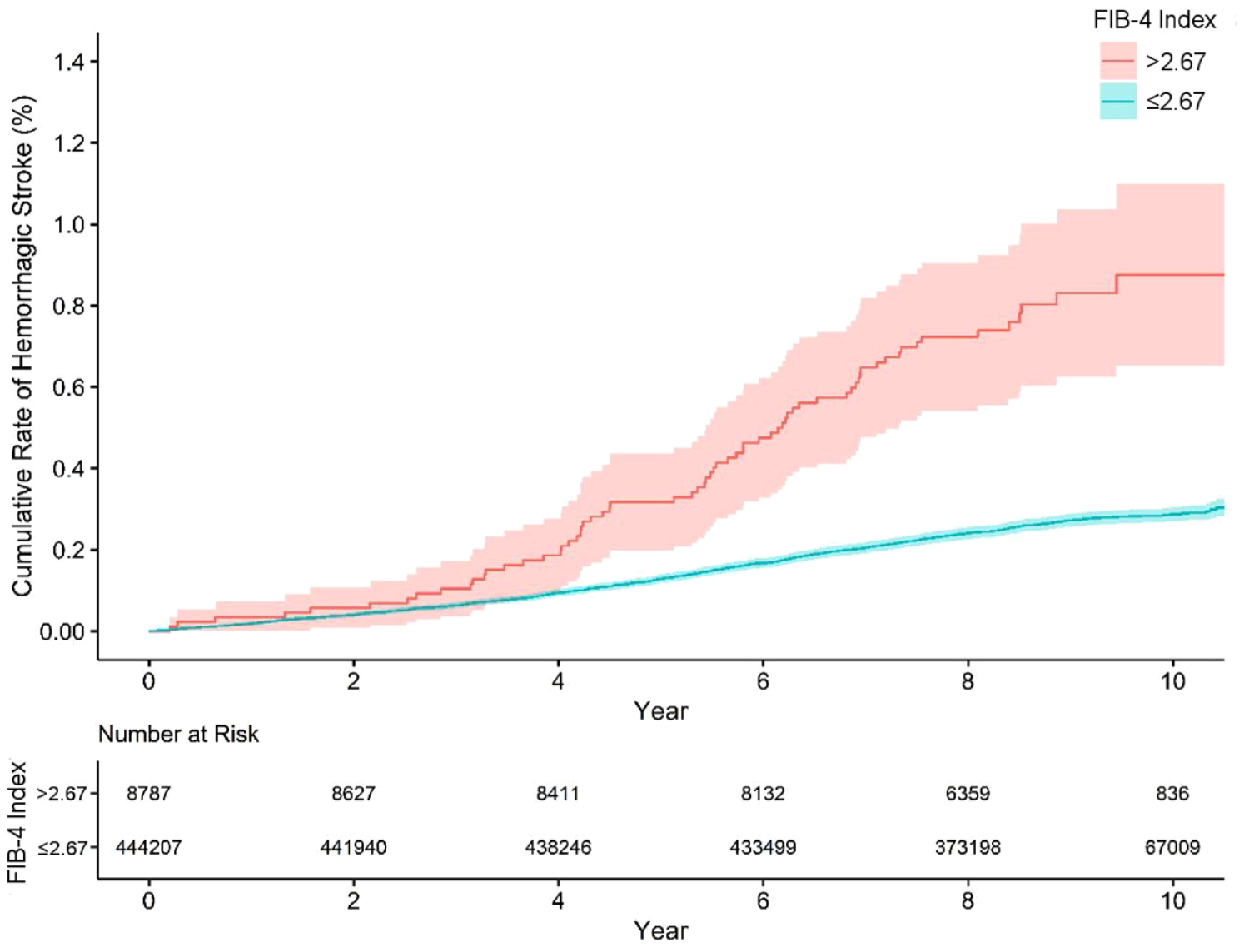
Cumulative risk of hemorrhagic stroke, stratified by FIB-4 index. In this analysis of data from the UK Biobank prospective cohort study, participants with FIB-4 Index >2.67 had an increased risk of hemorrhagic stroke.
Association a of FIB-4 index >2.67 with hemorrhagic stroke in the UK Biobank.
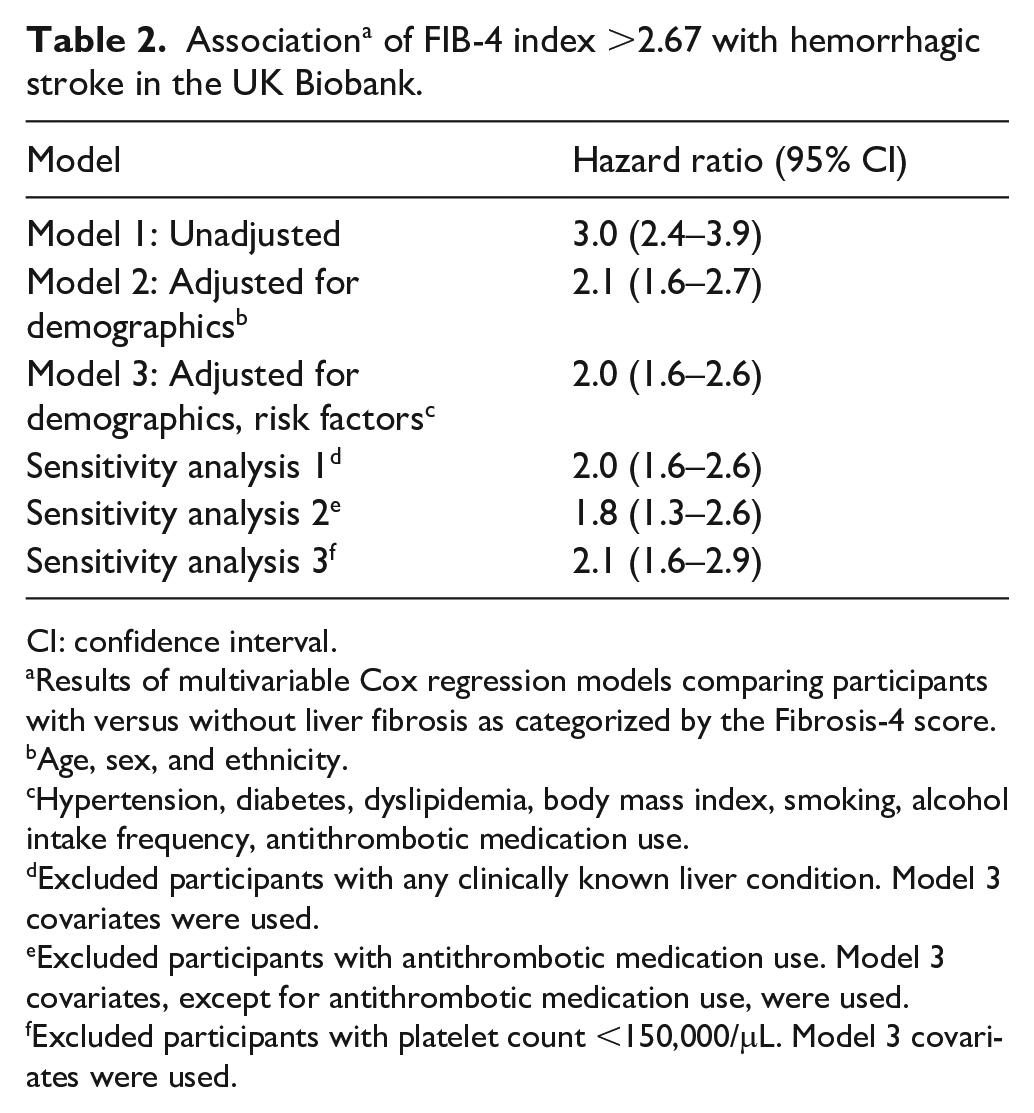
CI: confidence interval.
Results of multivariable Cox regression models comparing participants with versus without liver fibrosis as categorized by the Fibrosis-4 score.
Age, sex, and ethnicity.
Hypertension, diabetes, dyslipidemia, body mass index, smoking, alcohol intake frequency, antithrombotic medication use.
Excluded participants with any clinically known liver condition. Model 3 covariates were used.
Excluded participants with antithrombotic medication use. Model 3 covariates, except for antithrombotic medication use, were used.
Excluded participants with platelet count <150,000/μL. Model 3 covariates were used.
Secondary outcome analyses
Secondary outcomes were intracerebral hemorrhage and subarachnoid hemorrhage, assessed separately. The rate of intracerebral hemorrhage per 1000 person-years was 0.69 (95% CI, 0.52–0.91) in participants with FIB-4 index >2.67 and 0.19 (95% CI, 0.18–0.21) in those with FIB-4 index ⩽2.67. After adjusting for demographics and shared risk factors, FIB-4 index >2.67 was associated with an increased risk of intracerebral hemorrhage (HR, 2.0; 95% CI, 1.5–2.7). The rate of subarachnoid hemorrhage per 1000 person-years was 0.32 (95% CI, 0.21–0.49) in participants with FIB-4 index >2.67 and 0.13 (95% CI, 0.12–0.14) in those with FIB-4 index ⩽2.67. After adjusting for demographics and shared risk factors, FIB-4 index >2.67 was associated with an increased risk of subarachnoid hemorrhage (HR, 2.2; 95% CI, 1.5–3.5). The associations remained significant in matched cohort analyses for intracerebral hemorrhage (HR, 3.3; 95% CI, 1.8–6.0) and subarachnoid hemorrhage (HR, 2.6; 95% CI, 1.2–5.9). Characteristics of the matched cohort appear in Table 3.
Baseline characteristics a of participants with FIB-4 index >2.67 and ⩽2.67 matched on a 1:1 basis by age, sex, hypertension, and antithrombotic medication use.
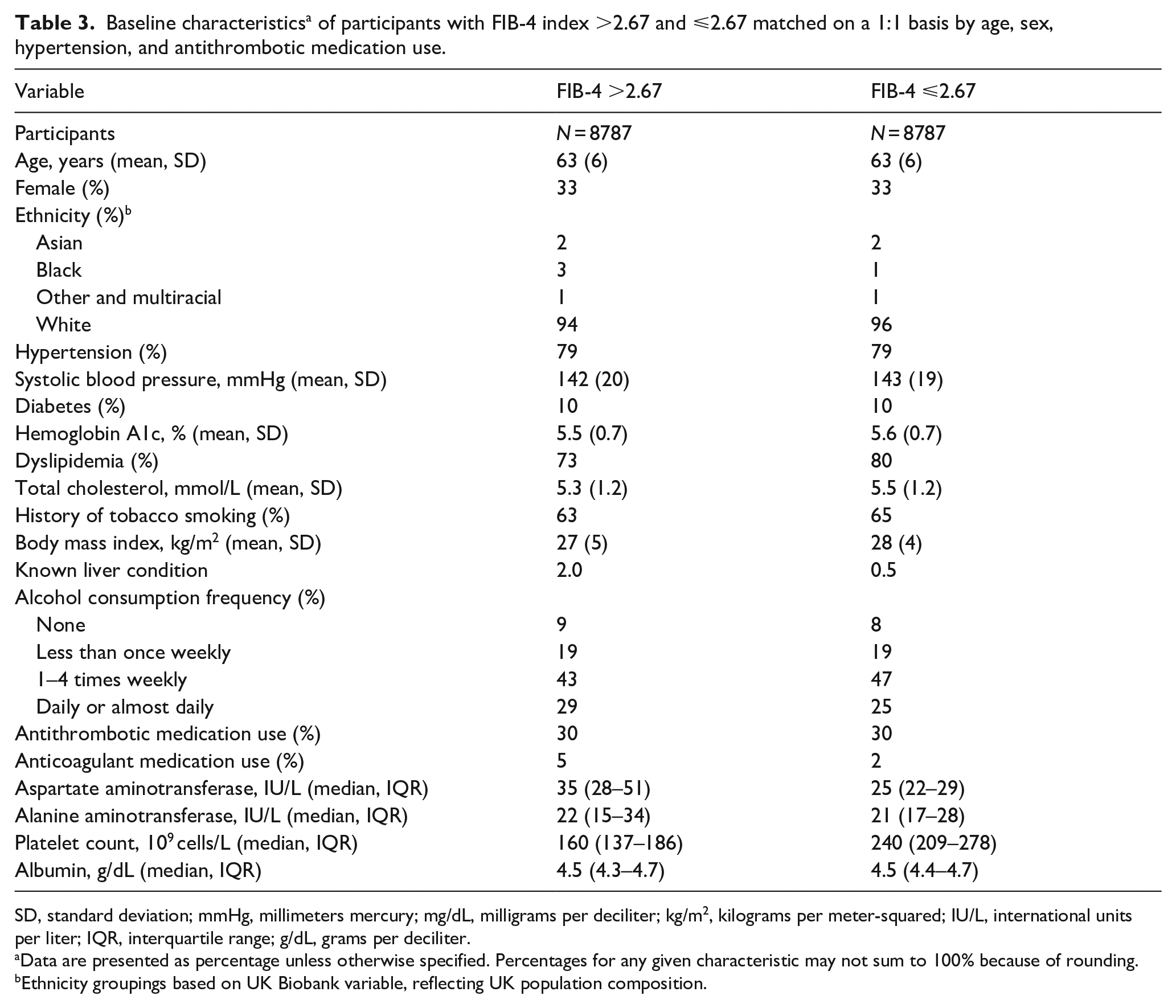
SD, standard deviation; mmHg, millimeters mercury; mg/dL, milligrams per deciliter; kg/m2, kilograms per meter-squared; IU/L, international units per liter; IQR, interquartile range; g/dL, grams per deciliter.
Data are presented as percentage unless otherwise specified. Percentages for any given characteristic may not sum to 100% because of rounding.
Ethnicity groupings based on UK Biobank variable, reflecting UK population composition.
Discussion
In this analysis of adults in the UK Biobank cohort study, participants with FIB-4 index >2.67 had a two-fold increased risk of hemorrhagic stroke, including both intracerebral and subarachnoid hemorrhage. This finding was robust across multiple sensitivity analyses.
Prior studies have shown a consistent association of cirrhosis with an increased risk of hemorrhagic stroke.3–7 In contrast to decompensated cirrhosis, which is often clinically apparent in terms of physical examination findings and laboratory tests, liver fibrosis is often subclinical.8–11 In this analysis, we found an association of FIB-4 index >2.67, indicating a high probability of liver fibrosis, with hemorrhagic stroke even after excluding participants with clinically known chronic liver disease. Mild thrombocytopenia can reflect chronic liver disease 40 ; however, our results were consistent when specifically excluding participants with any thrombocytopenia (<150,000/μL) from the study population. Thus, these results represent a novel finding that probable liver fibrosis, despite being frequently subclinical, is an independent risk factor for hemorrhagic stroke. An important implication of these results is that strategies to improve primary prevention of liver disease may subsequently improve primary prevention of hemorrhagic stroke as well.
We found an association of elevated FIB-4 index with hemorrhagic stroke after adjusting for risk factors, and additionally in confirmatory analyses in which we matched participants based on key hemorrhagic stroke risk factors including hypertension and antithrombotic medication use, suggesting that such risk factors alone do not explain our findings. Additional processes related to chronic liver disease may be responsible, such as endothelial dysfunction, subclinical coagulopathy, and vascular inflammation, which have been described in people with a variety of clinically apparent liver conditions.41–44 Liver fibrosis has also been linked to brain imaging markers of cerebral microvascular disease, including white matter hyperintensity volume and cerebral microbleeds,45–47 both of which are linked to an increased risk of hemorrhagic stroke. 48 Future work is required to understand whether these processes account for the association of liver fibrosis and hemorrhagic stroke.
The key strengths of this analysis are the large sample size for the primary analysis and consistency of results across analytical approaches. The findings should be interpreted considering several limitations. First, the FIB-4 index is a surrogate marker and its use introduces the possibility of misclassification. For example, participants with mild liver fibrosis may be misclassified as having no fibrosis, and the index may be less reliable among elderly subjects. 19 Future studies should use more direct liver measures such as transient elastography or magnetic resonance elastography, although the feasibility of this is limited in cohorts of this size. Second, similarly, there is a risk of misclassification of the primary outcome, hemorrhagic stroke. Though the UK Biobank’s algorithm for ascertainment of primary stroke cases has been validated to have high positive predictive value,31,49 occasional inclusion of hemorrhage secondary to trauma, mass lesion, or other causes may occur. 50 Third, the UK Biobank study population has evidence of a healthy participant bias. 51 In this analysis, the prevalence of probable liver fibrosis was approximately 2%, which is lower than other population-based estimates, for example 5% in the United States. 52 Additionally, the UK Biobank cohort is approximately 95% White, limiting generalizability to other race and ethnic groups. Furthermore, lifestyle patterns specific to the UK Biobank cohort regarding diet, smoking, and alcohol consumption may impact the results and limit generalizability to other geographic areas. Future studies should confirm our findings in more diverse populations, and among populations with a greater burden of comorbidities. Fourth, we did not have neuroimaging data; future studies should leverage datasets with more granular hematoma characteristics, such as lobar versus non-lobar location. Fifth, although we adjusted for relevant confounders, there may be unmeasured confounding that accounts for the observed association between probable liver fibrosis and risk of hemorrhagic stroke. In particular, we did not have data on multiple variables that could affect this association, such as chronic kidney disease, atrial fibrillation, diet, and quantity of alcohol consumption.
Conclusion
Probable liver fibrosis, defined by FIB-4 index >2.67, was associated with an increased risk of hemorrhagic stroke. Further research is required to understand the mechanistic basis of this association.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241259561 – Supplemental material for Association between elevated fibrosis-4 index of liver fibrosis and risk of hemorrhagic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873241259561 for Association between elevated fibrosis-4 index of liver fibrosis and risk of hemorrhagic stroke by Neal S. Parikh, Cenai Zhang, Samuel S. Bruce, Santosh B. Murthy, Russell Rosenblatt, Ava L. Liberman, Vanessa Liao, Jed H. Kaiser, Babak B. Navi, Costantino Iadecola and Hooman Kamel in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Parikh and Navi have received personal compensation for medicolegal consulting on stroke. Dr. Parikh is now employed by Alnylam Pharmaceuticals, Inc.; his contribution to this work occurred while employed at Weill Cornell Medicine and does not reflect the views of his current employer. Dr. Murthy reports research support from the NIH outside the submitted work, and has received personal compensation for medicolegal consulting on neurological disorders and stroke. Dr. Bruce reports research support from the NIH (KL2-TR-2385) outside the submitted work. Dr. Liberman reports research support from the NIH outside the submitted work. Dr. Kamel serves as a PI for the NIH-funded ARCADIA trial (NINDS U01NS095869) which receives in-kind study drug from the BMS-Pfizer Alliance for Eliquis and ancillary study support from Roche Diagnostics, serves as Deputy Editor for JAMA Neurology, serves as a steering committee member of Medtronic’s Stroke AF trial, serves on a trial executive committee for Janssen, and serves on an endpoint adjudication committee for a trial of empagliflozin for Boehringer-Ingelheim. Dr. Iadecola serves on the Scientific Advisory Board of Broadview Ventures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Parikh was supported by the NIH/NIA (K23 AG073524) and the Florence Gould Endowment for Discovery in Stroke.
Ethical approval
The Weill Cornell Medicine institutional review board certified these analyses of deidentified data as exempt from review.
Informed consent
The UK Biobank obtained written informed consent from participants.
Guarantor
Dr. Parikh.
Contributorship
Study conception and acquisition of data: NSP and HK. Data analysis: NSP and CZ. Drafting of the manuscript: NSP. Critical review and editing the manuscript for important intellectual content: NSP, CZ, SSB, SBM, RR, ALL, VL, JHK, BBN, CI, and HK. Supervision: HK. All authors made a substantial contribution to the concept or design of the work; or acquisition, analysis or interpretation of data; drafted the article or revised it critically for important intellectual content; approved the version to be published; and have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.